Introduction
Cystitis glandularis (CG) is an unusual
proliferative disorder of the urinary bladder, which is
characterized by transitional cells that have undergone glandular
metaplasia (1). CG was first
described by Morgagni et al (2) in 1761, however, the natural history of
CG in clinical practice remained unknown until recently (2,3). In
1950, a study by Immergut and Cottler (4) implicated CG in the development of
adenocarcinoma of the bladder (4).
Since then, CG has occasionally been proposed as a precursor of
adenocarcinoma by various studies (5–14). The
intestinal subtype of CG and diffuse lesions have been described as
premalignant (15–18); however, certain authors have
considered CG to be a chronic and quiescent histologic lesion
without any clinical significance (19). Furthermore, Smith et al
(20) found no evidence that CG
increased the future risk of malignancy after follow-up for ~2.6
years. Furthermore, Corica et al (21) considered that intestinal metaplasia
was not a marked risk factor for bladder cancer.
Due to a lack of definitive evidence regarding the
potential premalignant nature of CG and the issues associated with
determining an accurate prognosis, the present retrospective study
demonstrates 166 cases of CG with follow-up periods ranging between
0.5 and 17 years.
Patients and methods
Patients with CG
A retrospective review of 166 cases of CG between
1994 and 2010 was performed at the Tumor Hospital of Guangxi
Medical University (Nanning, China), Nanfang Hospital of Southern
Medical University (Guangzhou, China) and Guangzhou First People’s
Hospital (Guangzhou, China). All cases were identified as either
typical or intestinal type CG by surgical pathology, as reviewed by
two pathologists. Clinical information was obtained from the
patients’ clinical charts. The patients included in the current
study were diagnosed using biopsy or surgical specimens, with the
majority of patients possessing data from the two. The association
between intestinal and typical CG, and bladder carcinoma was
evaluated. Patients provided written informed consent. The
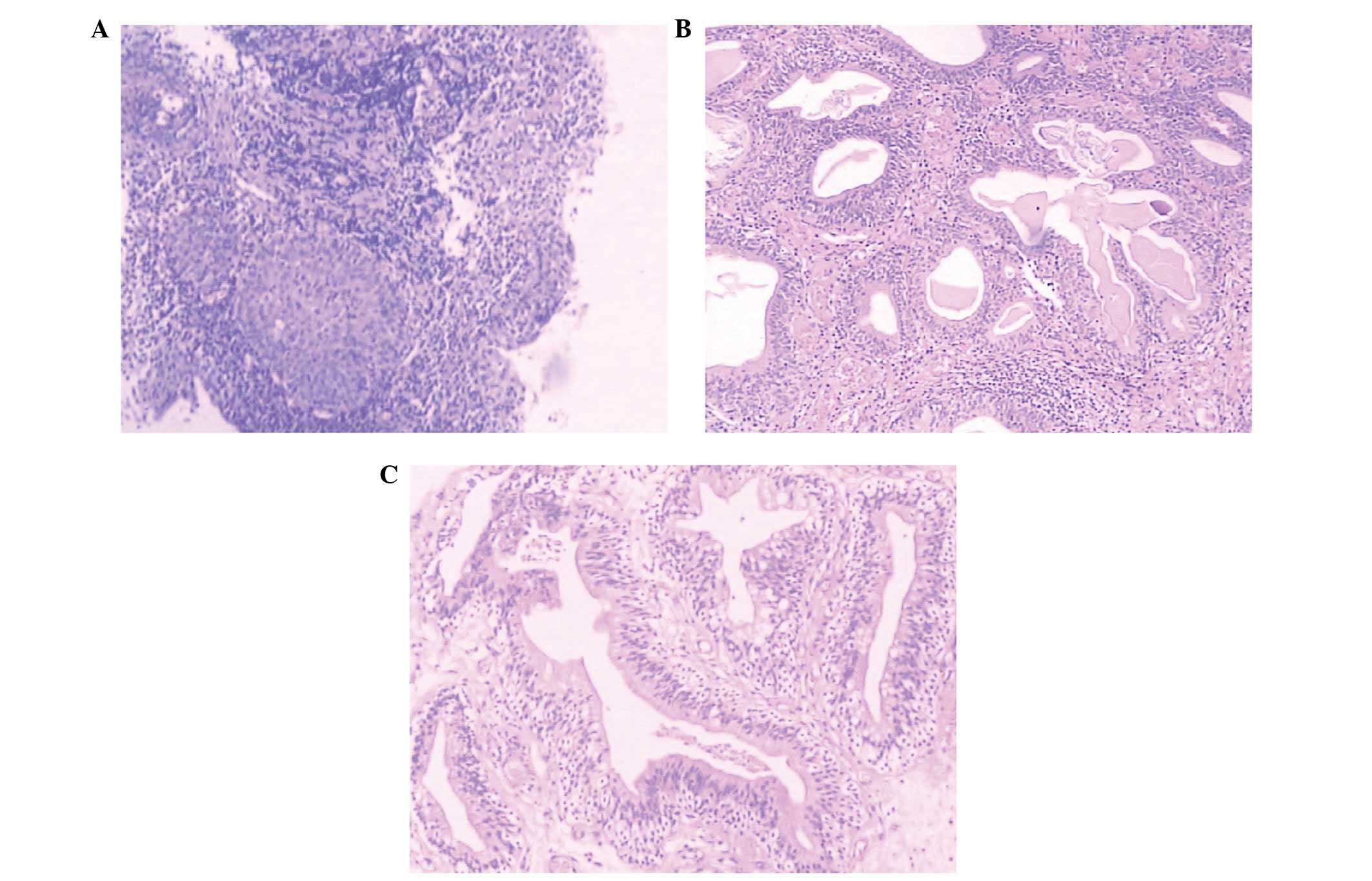
glandular structures, which are formed by nests of columnar
epithelial cells within the lamina propria of the bladder, were
classed into a typical CG group (Fig.
1A and B). The cells of the intestinal CG group exhibited
similar glandular structures, however, the simple columnar
epithelial cells were replaced by mucin-secreting goblet cells
(15–18) (Fig.
1C).
Statistical analysis
Student’s t-test was used to compare the
distribution of age and gender. Statistical analyses were performed
using SPSS software (version 13; SPSS, Inc., Chicago, IL, USA).
Data are presented as the mean ± standard error of mean. All
P-values were two-tailed and P<0.05 was considered to indicate a
statistically significant difference.
Results
A total of 166 patients were identified for
inclusion in the present study, who demonstrated typical (n=155) or
intestinal (n=11) CG. The patient age ranged from 9 to 86 years and
there was no statistically significant difference identified
between the two groups with regard to the distribution of age or
gender (Table I), although the age
range was greater in the typical CG group.
 | Table IAge and gender distribution of
intestinal and typical CG. |
Table I
Age and gender distribution of
intestinal and typical CG.
| Diagnosis | Male (n) | Female (n) | t-value | P-value | Minimum age
(years) | Maximum age
(years) | Mean age (years) | t-value | P-value |
|---|
| CG | | | 1.045 | 0.177 | | | | 0.954 | 0.341 |
| Typical | 73 | 82 | | | 9 | 86 | 51.1±16.1 | | |
| Intestinal | 8 | 3 | | | 34 | 68 | 46.4±12.7 | | |
A documented follow-up was available for 114/166
(68.7%) patients and ranged from 0.5 to 17 years (median, 4.22
years; mean, 3.38 years). Six of the 155 (3.9%) patients with
typical CG succumbed due to a previous or concurrent cancer.
Follow-up was available for 9/11 (81.8%) patients
with intestinal CG. Of these patient, 8/11 (72.7%) were in complete
remission following a transurethral fulguration and 1/11 (9.1%)
complained of urgency and dysuria 9 months following a
transurethral fulguration and intravesical instillation; two
patients were lost to follow-up. The follow-up period ranged from
0.7 to 4.5 years (median, 2.67 years; mean, 2.82 years).
All patients with follow-up data in the intestinal
and typical CG groups showed no evidence of a subsequent carcinoma.
The follow-up procedures included an additional cystoscopy, urine
cytology and a clinical examination.
A concurrent carcinoma of the bladder was identified
in 15/166 (9.0%) patients. These included two cases of squamous
cell carcinoma and one case of sarcoma, all of which were found
prior to or concurrently with CG. Other types of concurrent cancer
included two cases of cervical, one case of breast and three cases
of colon cancer. In one case, CG was identified prior to the
presentation of colon cancer.
Discussion
The natural history of CG in the clinical setting
remains unclear. Shaw et al (22) first described a case of CG with a
gradual transition to adenocarcinoma in 1958. The patient had an
extensive history of chronic urinary tract infection that
eventually progressed to adenocarcinoma. The biopsy sampling was
not conducted in this case due to a large mass that was present at
the time of diagnosis (21).
Hereafter, two cases of a conversion from CG to adenocarcinoma have
been reported (7,23) and it has been proposed that these
findings support that CG should be considered as a precancerous
lesion (7). By contrast, Ito et
al (24) reported the
characteristic development of CG in 40–92% of apparently healthy
bladders in a review of 125 autopsy cases. In these cases, no
evidence of malignancy had been found (24). Furthermore, Smith et al
(20) and Corica et al
(21) found no evidence that CG or
intestinal metaplasia increases the future risk of malignancy.
Although CG has been implicated as a premalignant
condition in >16 case reports (20), in 10/16 the evidence was based on
the high incidence of a coexistence of CG and adenocarcinoma
(20,25). The present study found no evidence
of carcinoma subsequent to typical or intestinal CG with a 0.5 to
17-year follow-up. The results did not support that CG increases
the future risk of malignancy, however, this was only evaluated in
the short term. In addition, repeated cystoscopies over a short
period are not recommended (20).
In conclusion, a limitation of the present study was
that the follow-up period may not have been long enough. The
progression of CG to cancer may be a long-term process, therefore,
definitive evidence is lacking from the present study and a longer
follow-up period is required (1).
Currently, a therapeutic recommendation for CG cannot be proposed,
due to the lack of previous cases reported and due to uncertainty
regarding the etiopathogenesis (26). Therefore, a large-scale,
multi-center follow-up study is required in order to facilitate the
accurate determination of a prognosis for patients with CG.
Acknowledgements
This study was supported by the self-financing
research of the Department of Health, Guangxi Autonomous Region in
2012 (grant no. Z2012349) and in 2013 (grant no. Z2013429).
References
|
1
|
Semins MJ and Schoenberg MP: A case of
florid cystitis glandularis. Nat Clin Pract Urol. 4:341–345.
2007.
|
|
2
|
Morgagni G and Alexander B: The Seats and
Causes of Diseases Investigated by Anatomy. 3. 2nd edition. London,
A. Millar; and T. Cadell, his successor and Johnson and Payne,
London; pp. 8351769
|
|
3
|
Davies G and Castro JE: Cystitis
glandularis. Urology. 10:128–129. 1977.
|
|
4
|
Immergut S and Cottler ZR: Mucin producing
adenocarcinoma of the bladder associated with cystitis follicularis
and glandularis. Urol Cutaneous Rev. 54:531–534. 1950.
|
|
5
|
Nesbit RM: Is cystitis cystica an innocent
or a malefic lesion? J Urol. 75:443–447. 1956.
|
|
6
|
Hochberg DA, Motta J and Brodherson MS:
Cystitis glandularis. Urology. 51:112–113. 1998.
|
|
7
|
Edwards PD, Hurm RA and Jaeschke WH:
Conversion of cystitis glandularis to adenocarcinoma. J Urol.
108:568–570. 1972.
|
|
8
|
Bell TE and Wendel RG: Cystitis
glandularis: benign or malignant? J Urol. 100:462–465. 1968.
|
|
9
|
O’Brien AM and Urbanski SJ: Papillary
adenocarcinoma in situ of bladder. J Urol. 134:544–546. 1985.
|
|
10
|
Fariña LA: Re: Pelvic lipomatosis
associated with cystitis glandularis and adenocarcinoma of the
bladder. J Urol. 147:13801992.
|
|
11
|
Sözen S, Gürocak S, Uzüm N, Biri H, Memiş
L and Bozkirli I: The importance of re-evaluation in patients with
cystitis glandularis associated with pelvic lipomatosis: a case
report. Urol Oncol. 22:428–430. 2004.
|
|
12
|
Heyns CF, De Kock ML, Kirsten PH and van
Velden DJ: Pelvic lipomatosis associated with cystitis glandularis
and adenocarcinoma of the bladder. J Urol. 145:364–366. 1991.
|
|
13
|
Thrasher JB, Rajan RR, Perez LM, Humphrey
PA and Anderson EE: Cystitis glandularis. Transition to
adenocarcinoma of the urinary bladder. N C Med J. 55:562–564.
1994.
|
|
14
|
Sauty L, Ravery V, Toublanc M and
Boccon-Gibod L: Florid glandular cystitis: study of 3 cases and
review of the literature. Prog Urol. 8:561–564. 1998.(In
French).
|
|
15
|
Bullock PS, Thoni DE and Murphy WM: The
significance of colonic mucosa (intestinal metaplasia) involving
the urinary tract. Cancer. 59:2086–2090. 1987.
|
|
16
|
Jacobs LB, Brooks JD and Epstein JI:
Differentiation of colonic metaplasia from adenocarcinoma of
urinary bladder. Hum Pathol. 28:1152–1157. 1997.
|
|
17
|
Young RH and Bostwick DG: Florid cystitis
glandularis of intestinal type with mucin extravasation: a mimic of
adenocarcinoma. Am J Surg Pathol. 20:1462–1468. 1996.
|
|
18
|
Bryan RT, Nicholls JH, Harrison RF,
Jankowski JA and Wallace DM: The role of beta-catenin signaling in
the malignant potential of cystitis glandularis. J Urol.
170:1892–1896. 2003.
|
|
19
|
Mitre AI, Silveira CAP, Leite KR and
Piovesan AC: Glandular cystitis: a rare benign condition presenting
as a pseudo-tumor of the bladder. Clinics (Sao Paulo). 62:93–94.
2007.
|
|
20
|
Smith AK, Hansel DE and Jones JS: Role of
cystitis cystica et glandularis and intestinal metaplasia in
development of bladder carcinoma. Urology. 71:915–918. 2008.
|
|
21
|
Corica FA, Husmann DA, Churchill BM and
Young RH: Intestinal metaplasia is not a strong risk factor for
bladder cancer: study of 53 cases with long-term follow-up.
Urology. 50:427–431. 1997.
|
|
22
|
Shaw JL, Gislason GJ and Imbriglia JE:
Transition of cystitis glandularis to primary adenocarcinoma of the
bladder. J Urol. 79:815–822. 1958.
|
|
23
|
Susmano D, Rubenstein AB, Dakin AR and
Lloyd FA: Cystitis glandularis and adenocarcinoma of the bladder.
Urology. 105:671–674. 1971.
|
|
24
|
Ito N, Hirose M, Shirai T, Tsuda H,
Nakanishi K and Fukushima S: Lesions of the urinary bladder
epithelium in 125 autopsy cases. Acta Pathol Jpn. 31:545–557.
1981.
|
|
25
|
Lin JI, Yong HS, Tseng CH, Marsidi PS,
Choy C and Pilloff B: Diffuse cystitis glandularis. Associated with
adenocarcinomatous change. Urology. 15:411–415. 1980.
|
|
26
|
Mitre A, Silveira C, Leite K and Piovesan
A: Glandular cystitis: a rare benign condition presenting as a
pseudo-tumor of the bladder. Clinics. 62:93–94. 2007.
|