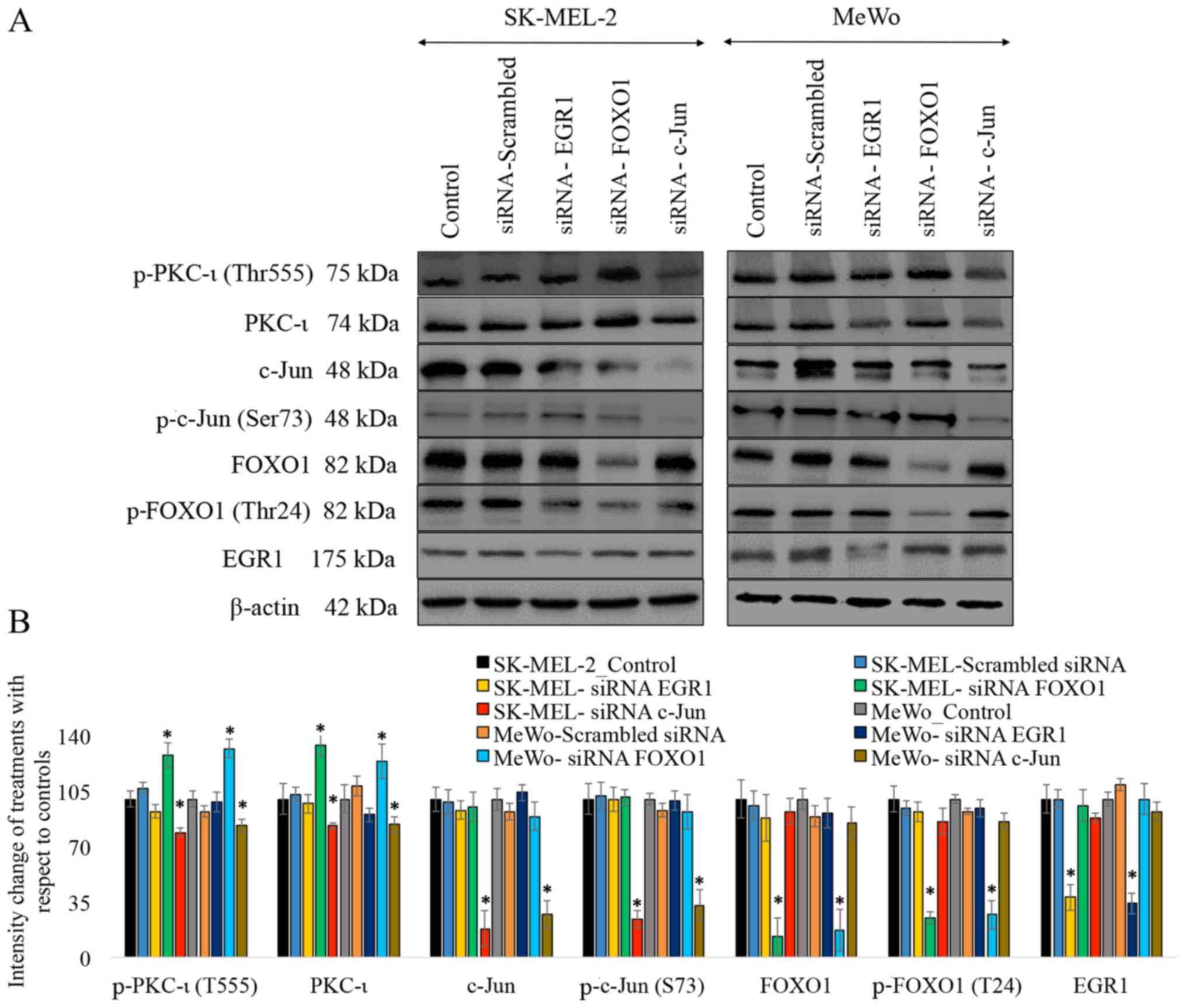
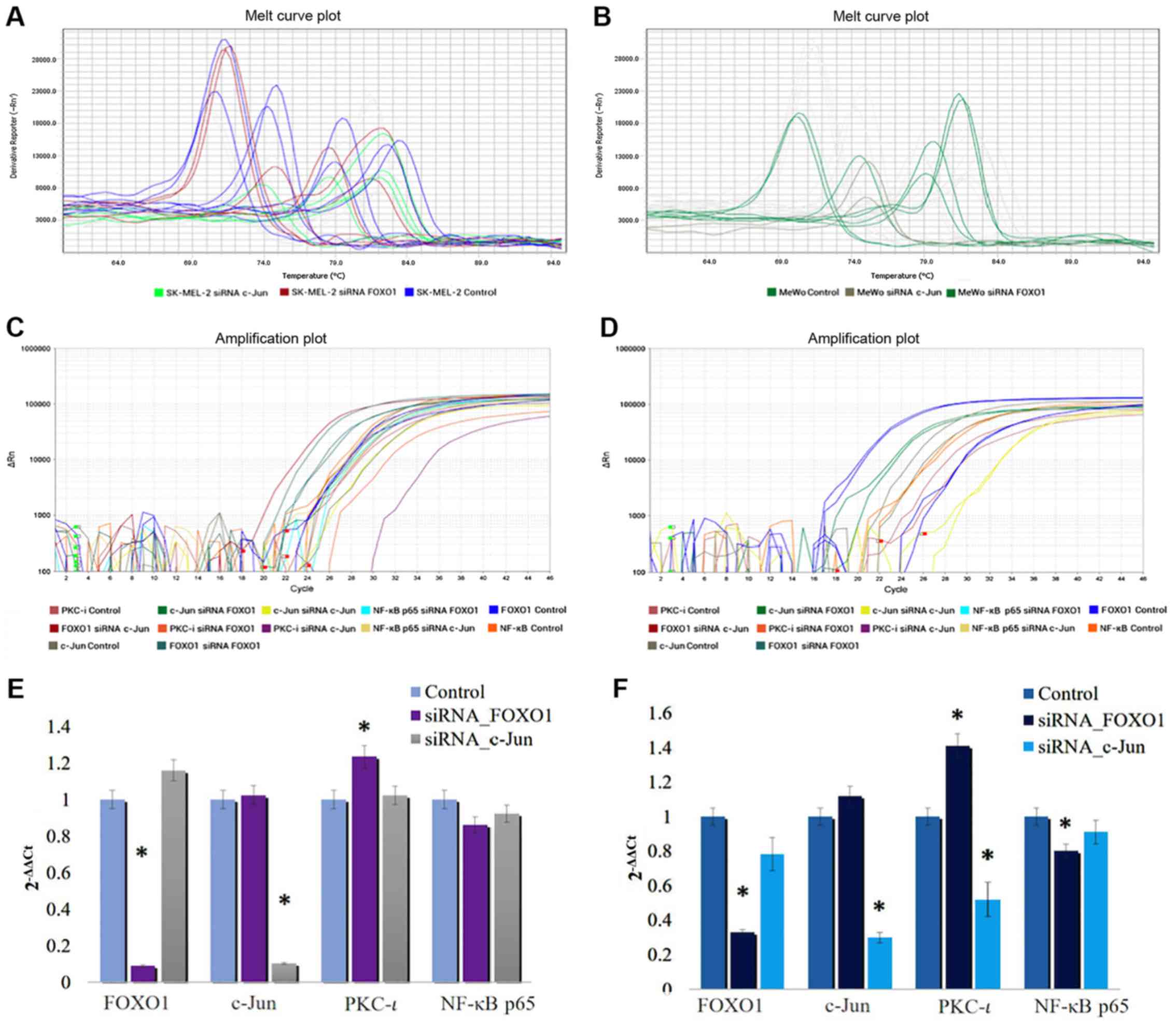
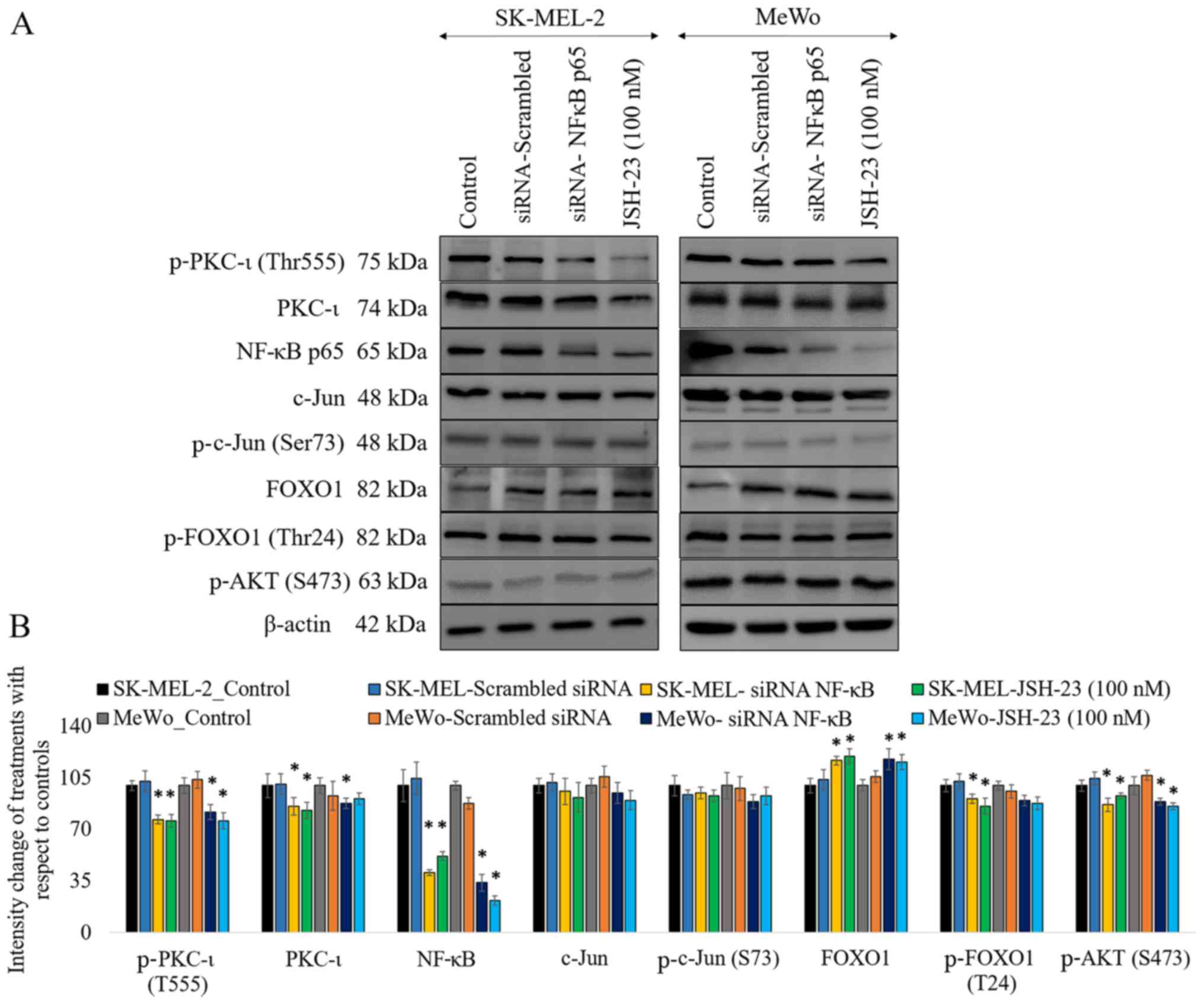
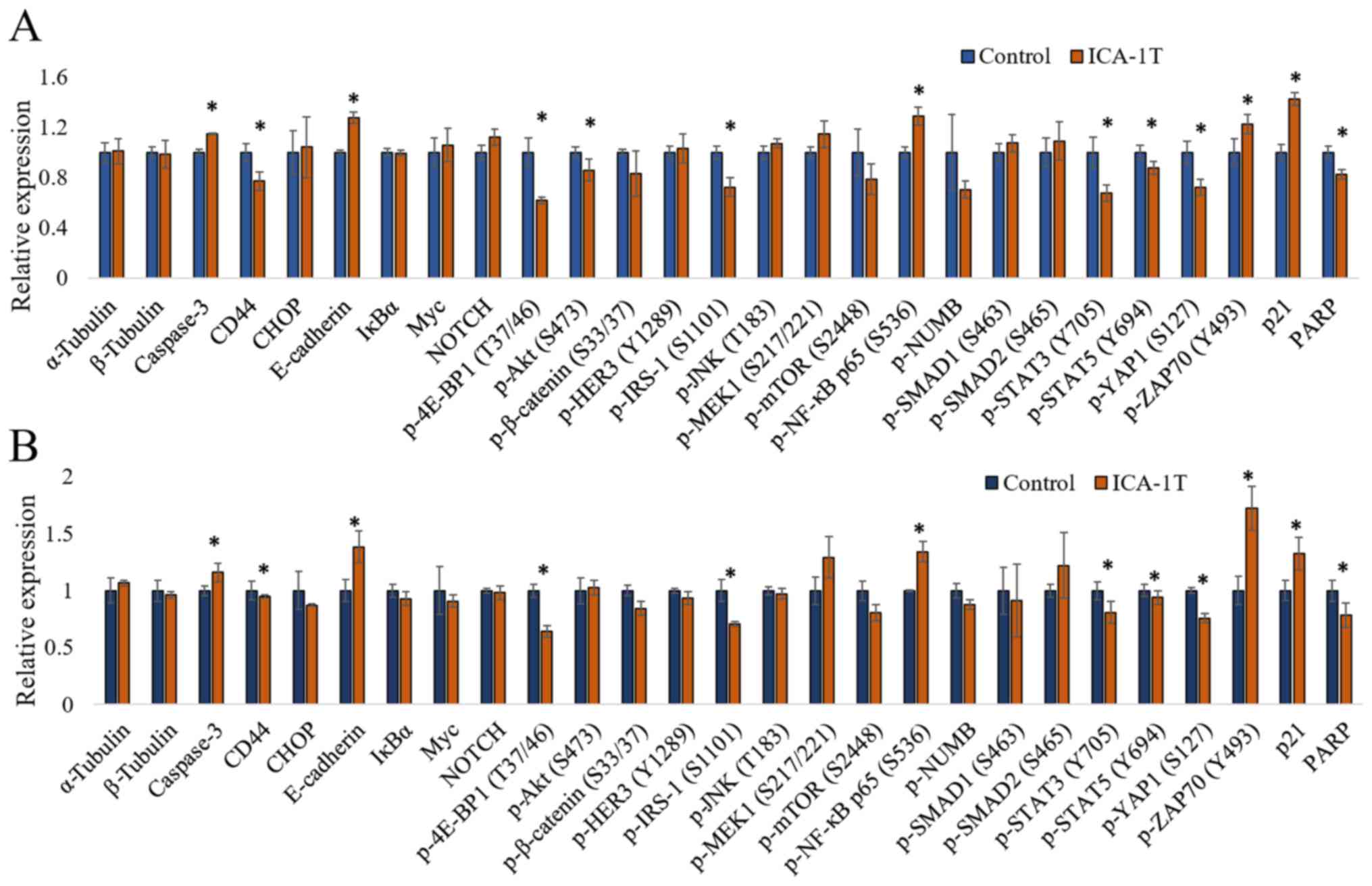
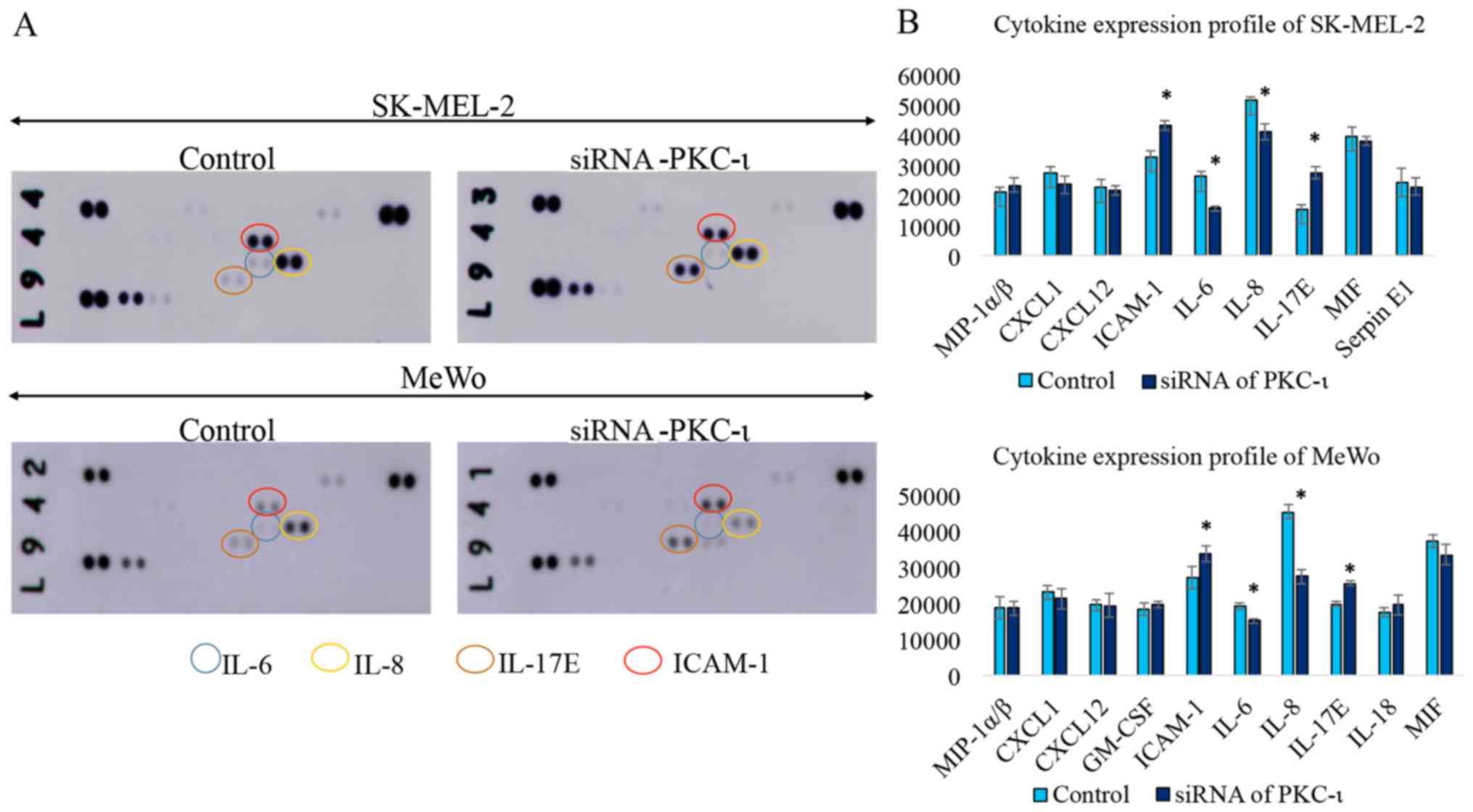
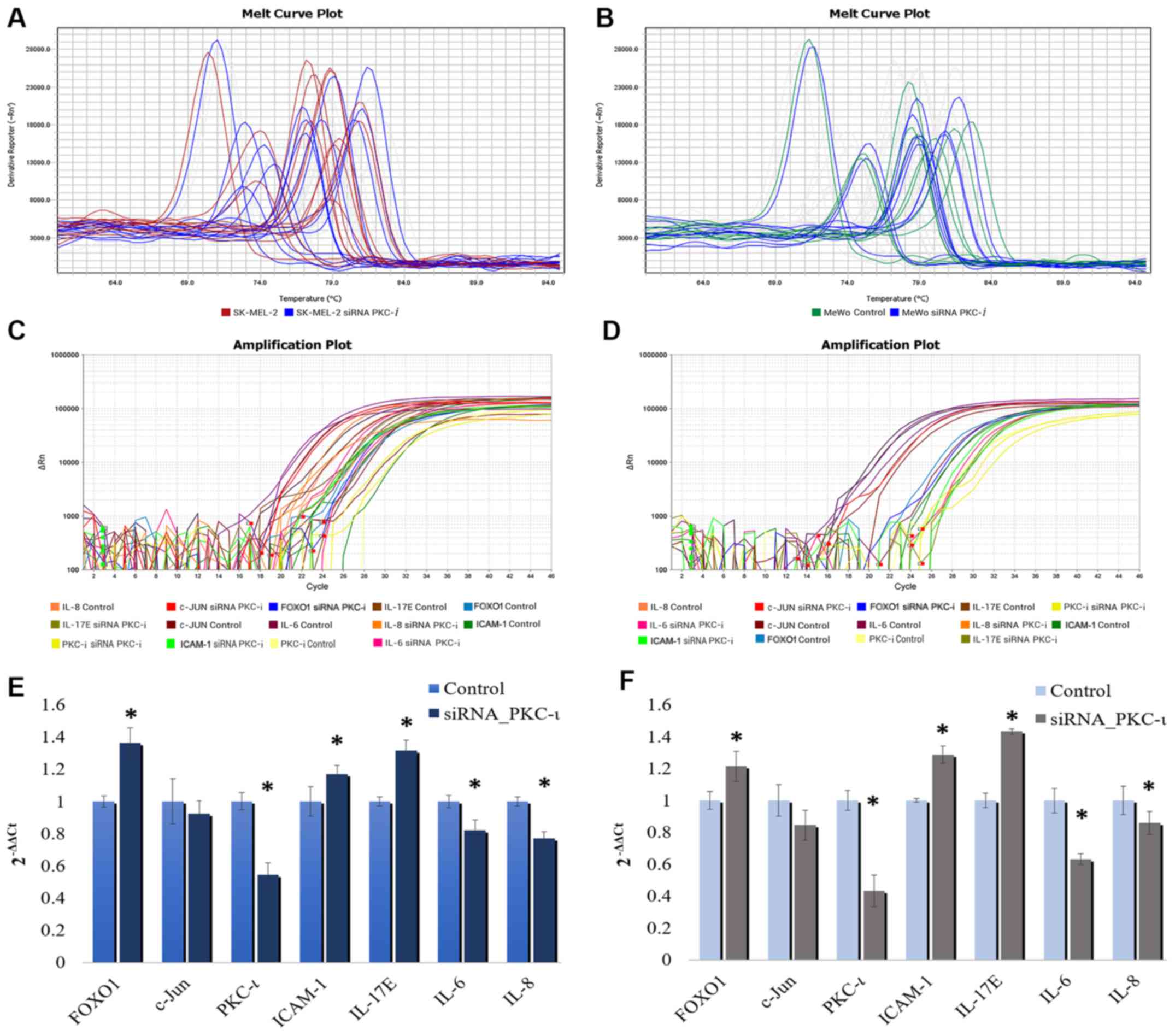
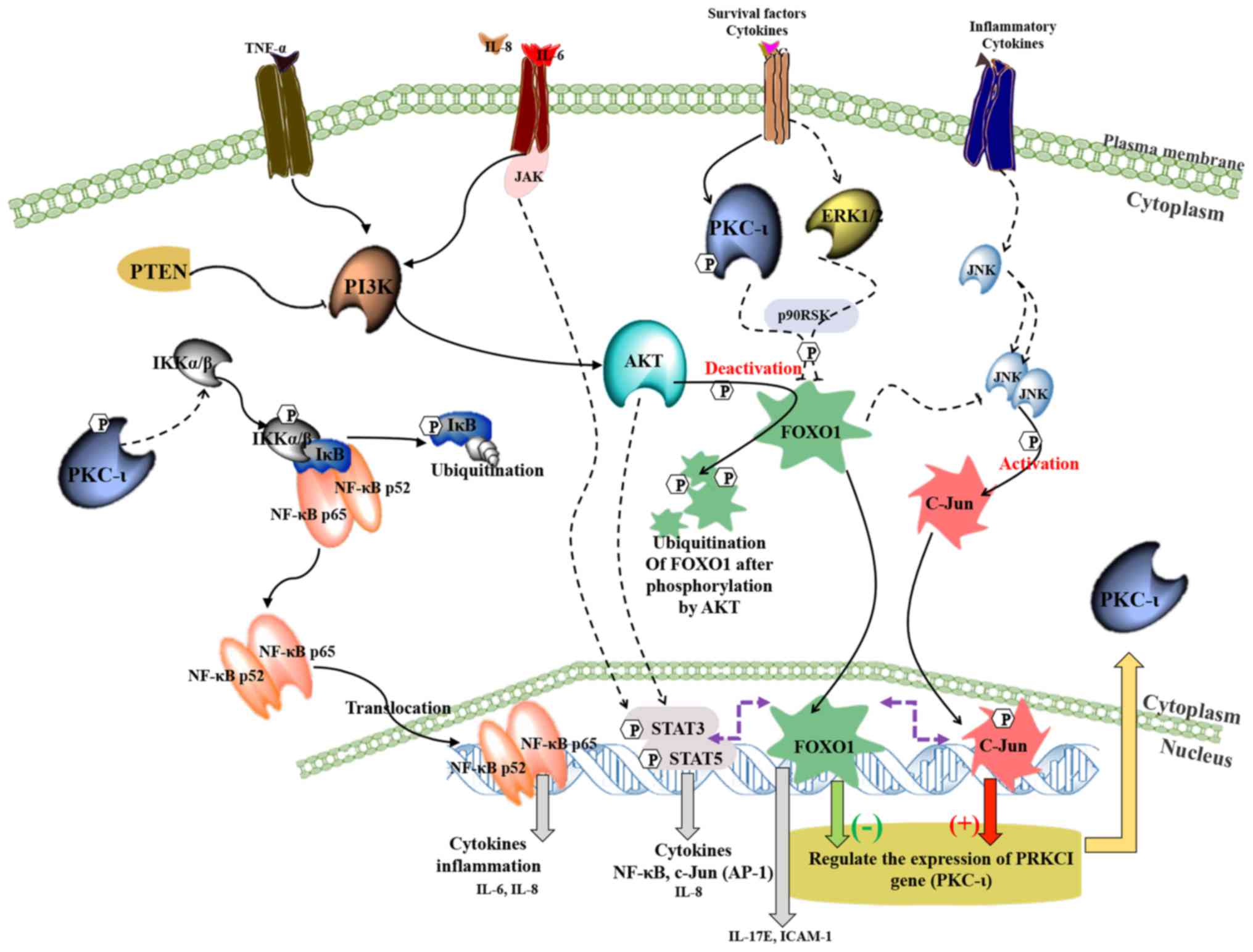
|
1
|
Cronin KA, Lake AJ, Scott S, Sherman RL,
Noone AM, Howlader N, Henley SJ, Anderson RN, Firth AU, Ma J, et
al: Annual Report to the Nation on the Status of Cancer, part I:
National cancer statistics. Cancer. 124:2785–2800. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Nazarian R, Shi H, Wang Q, Kong X, Koya
RC, Lee H, Chen Z, Lee MK, Attar N, Sazegar H, et al: Melanomas
acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS
upregulation. Nature. 468:973–977. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ratnayake WS, Apostolatos CA, Apostolatos
AH, Schutte RJ, Huynh MA, Ostrov DA and Acevedo-Duncan M: Oncogenic
PKC-ι activates Vimentin during epithelial-mesenchymal transition
in melanoma; a study based on PKC-ι and PKC-ζ specific inhibitors.
Cell Adhes Migr. 0:1–17. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ratnayake WS, Apostolatos AH, Ostrov DA
and Acevedo-Duncan M: Two novel atypical PKC inhibitors; ACPD and
DNDA effectively mitigate cell proliferation and epithelial to
mesenchymal transition of metastatic melanoma while inducing
apoptosis. Int J Oncol. 51:1370–1382. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ratnayake WS and Acevedo-Duncan M:
Abstract 862: Atypical protein kinase c inhibitors can repress
epithelial to mesenchymal transition (type III) in malignant
melanoma. Cancer Res. 77((Suppl 13)): 862. 2017. View Article : Google Scholar
|
|
6
|
Manning G, Whyte DB, Martinez R, Hunter T
and Sudarsanam S: The protein kinase complement of the human
genome. Science. 298:1912–1934. 2002.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Regala RP, Weems C, Jamieson L, Khoor A,
Edell ES, Lohse CM and Fields AP: Atypical protein kinase C iota is
an oncogene in human non-small cell lung cancer. Cancer Res.
65:8905–8911. 2005.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Murray NR and Fields AP: Atypical protein
kinase C iota protects human leukemia cells against drug-induced
apoptosis. J Biol Chem. 272:27521–27524. 1997.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Desai S, Pillai P, Win-Piazza H and
Acevedo-Duncan M: PKC-ι promotes glioblastoma cell survival by
phosphorylating and inhibiting BAD through a phosphatidylinositol
3-kinase pathway. Biochim Biophys Acta. 1813:1190–1197.
2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Win HY and Acevedo-Duncan M: Role of
protein kinase C-iota in transformed non-malignant RWPE-1 cells and
androgen-independent prostate carcinoma DU-145 cells. Cell Prolif.
42:182–194. 2009.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Eder AM, Sui X, Rosen DG, Nolden LK, Cheng
KW, Lahad JP, Kango-Singh M, Lu KH, Warneke CL, Atkinson EN, et al:
Atypical PKCiota contributes to poor prognosis through loss of
apical-basal polarity and cyclin E overexpression in ovarian
cancer. Proc Natl Acad Sci USA. 102:12519–12524. 2005.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Apostolatos AH, Ratnayake WS, Win-Piazza
H, Apostolatos CA, Smalley T, Kang L, Salup R, Hill R and
Acevedo-Duncan M: Inhibition of atypical protein kinase C-ι
effectively reduces the malignancy of prostate cancer cells by
downregulating the NF-κB signaling cascade. Int J Oncol.
53:1836–1846. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wu J, Lu M, Li Y, Shang YK, Wang SJ, Meng
Y, Wang Z, Li ZS, Chen H, Chen ZN, et al: Regulation of a
TGF-β1-CD147 self-sustaining network in the differentiation
plasticity of hepatocellular carcinoma cells. Oncogene.
35:5468–5479. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Venter JC, Adams MD, Myers EW, Li PW,
Mural RJ, Sutton GG, Smith HO, Yandell M, Evans CA, Holt RA, et al:
The sequence of the human genome. Science. 291:1304–1351.
2001.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Fagerberg L, Hallström BM, Oksvold P,
Kampf C, Djureinovic D, Odeberg J, Habuka M, Tahmasebpoor S,
Danielsson A, Edlund K, et al: Analysis of the human
tissue-specific expression by genome-wide integration of
transcriptomics and antibody-based proteomics. Mol Cell Proteomics.
13:397–406. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Win HY and Acevedo-Duncan M: Atypical
protein kinase C phosphorylates IKKalphabeta in transformed
non-malignant and malignant prostate cell survival. Cancer Lett.
270:302–311. 2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Butler AM, Scotti Buzhardt ML, Erdogan E,
Li S, Inman KS, Fields AP and Murray NR: A small molecule inhibitor
of atypical protein kinase C signaling inhibits pancreatic cancer
cell transformed growth and invasion. Oncotarget. 6:15297–15310.
2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wisdom R, Johnson RS and Moore C: c-Jun
regulates cell cycle progression and apoptosis by distinct
mechanisms. EMBO J. 18:188–197. 1999.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Angel P, Hattori K, Smeal T and Karin M:
The jun proto-oncogene is positively autoregulated by its product,
Jun/AP-1. Cell. 55:875–885. 1988.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lopez-Bergami P, Huang C, Goydos JS, Yip
D, Bar-Eli M, Herlyn M, Smalley KS, Mahale A, Eroshkin A, Aaronson
S, et al: Rewired ERK-JNK signaling pathways in melanoma. Cancer
Cell. 11:447–460. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Vogt PK: Fortuitous convergences: The
beginnings of JUN. Nat Rev Cancer. 2:465–469. 2002.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Szabo E, Riffe ME, Steinberg SM, Birrer MJ
and Linnoila RI: Altered cJUN expression: An early event in human
lung carcinogenesis. Cancer Res. 56:305–315. 1996.PubMed/NCBI
|
|
24
|
Vleugel MM, Greijer AE, Bos R, van der
Wall E and van Diest PJ: c-Jun activation is associated with
proliferation and angiogenesis in invasive breast cancer. Hum
Pathol. 37:668–674. 2006.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Behrens A, Sibilia M and Wagner EF:
Amino-terminal phosphorylation of c-Jun regulates stress-induced
apoptosis and cellular proliferation. Nat Genet. 21:326–329.
1999.PubMed/NCBI View
Article : Google Scholar
|
|
26
|
Nateri AS, Spencer-Dene B and Behrens A:
Interaction of phosphorylated c-Jun with TCF4 regulates intestinal
cancer development. Nature. 437:281–285. 2005.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Rena G, Guo S, Cichy SC, Unterman TG and
Cohen P: Phosphorylation of the transcription factor forkhead
family member FKHR by protein kinase B. J Biol Chem.
274:17179–17183. 1999.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Nakae J, Kitamura T, Kitamura Y, Biggs WH
III, Arden KC and Accili D: The forkhead transcription factor Foxo1
regulates adipocyte differentiation. Dev Cell. 4:119–129.
2003.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Matsuzaki H, Daitoku H, Hatta M, Tanaka K
and Fukamizu A: Insulin-induced phosphorylation of FKHR (Foxo1)
targets to proteasomal degradation. Proc Natl Acad Sci USA.
100:11285–11290. 2003.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lu H and Huang H: FOXO1: A potential
target for human diseases. Curr Drug Targets. 12:1235–1244.
2011.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Borkhardt A, Repp R, Haas OA, Leis T,
Harbott J, Kreuder J, Hammermann J, Henn T and Lampert F: Cloning
and characterization of AFX, the gene that fuses to MLL in acute
leukemias with a t(X;11)(q13;q23). Oncogene. 14:195–202.
1997.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Anderson MJ, Viars CS, Czekay S, Cavenee
WK and Arden KC: Cloning and characterization of three human
forkhead genes that comprise an FKHR-like gene subfamily. Genomics.
47:187–199. 1998.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhang X, Tang N, Hadden TJ and Rishi AK:
Akt, FoxO and regulation of apoptosis. Biochim Biophys Acta.
1813:1978–1986. 2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Farhan M, Wang H, Gaur U, Little PJ, Xu J
and Zheng W: FOXO signaling pathways as therapeutic targets in
cancer. Int J Biol Sci. 13:815–827. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fu Z and Tindall DJ: FOXOs, cancer and
regulation of apoptosis. Oncogene. 27:2312–2319. 2008.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhang Y, Zhang L, Sun H, Lv Q, Qiu C, Che
X, Liu Z and Jiang J: Forkhead transcription factor 1 inhibits
endometrial cancer cell proliferation via sterol regulatory
element-binding protein 1. Oncol Lett. 13:731–737. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Hodge DR, Hurt EM and Farrar WL: The role
of IL-6 and STAT3 in inflammation and cancer. Eur J Cancer.
41:2502–2512. 2005.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Yue P and Turkson J: Targeting STAT3 in
cancer: How successful are we? Expert Opin Investig Drugs.
18:45–56. 2009.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Jing N and Tweardy DJ: Targeting Stat3 in
cancer therapy. Anticancer Drugs. 16:601–607. 2005.PubMed/NCBI
|
|
40
|
Page BDG, Khoury H, Laister RC, Fletcher
S, Vellozo M, Manzoli A, Yue P, Turkson J, Minden MD and Gunning
PT: Small molecule STAT5-SH2 domain inhibitors exhibit potent
antileukemia activity. J Med Chem. 55:1047–1055. 2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Pardanani A, Lasho T, Smith G, Burns CJ,
Fantino E and Tefferi A: CYT387, a selective JAK1/JAK2 inhibitor:
In vitro assessment of kinase selectivity and preclinical studies
using cell lines and primary cells from polycythemia vera patients.
Leukemia. 23:1441–1445. 2009.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Rani A and Murphy JJ: STAT5 in cancer and
immunity. J Interferon Cytokine Res. 36:226–237. 2016.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Korneev KV, Atretkhany KN, Drutskaya MS,
Grivennikov SI, Kuprash DV and Nedospasov SA: TLR-signaling and
proinflammatory cytokines as drivers of tumorigenesis. Cytokine.
89:127–135. 2017.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Zhang X, Wrzeszczynska MH, Horvath CM and
Darnell JE Jr: Interacting regions in Stat3 and c-Jun that
participate in cooperative transcriptional activation. Mol Cell
Biol. 19:7138–7146. 1999.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Hornsveld M, Dansen TB, Derksen PW and
Burgering BM: Re-evaluating the role of FOXOs in cancer. Semin
Cancer Biol. 50:90–100. 2018.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Sunters A, Madureira PA, Pomeranz KM,
Aubert M, Brosens JJ, Cook SJ, Burgering BM, Coombes RC and Lam EW:
Paclitaxel-induced nuclear translocation of FOXO3a in breast cancer
cells is mediated by c-Jun NH2-terminal kinase and Akt. Cancer Res.
66:212–220. 2006.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Yuan ZL, Guan YJ, Wang L, Wei W, Kane AB
and Chin YE: Central role of the threonine residue within the p+1
loop of receptor tyrosine kinase in STAT3 constitutive
phosphorylation in metastatic cancer cells. Mol Cell Biol.
24:9390–9400. 2004.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Antonicelli F, Lorin J, Kurdykowski S,
Gangloff SC, Le Naour R, Sallenave JM, Hornebeck W, Grange F and
Bernard P: CXCL10 reduces melanoma proliferation and invasiveness
in vitro and in vivo. Br J Dermatol. 164:720–728. 2011.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Zaynagetdinov R, Sherrill TP, Gleaves LA,
McLoed AG, Saxon JA, Habermann AC, Connelly L, Dulek D, Peebles RS
Jr, Fingleton B, et al: Interleukin-5 facilitates lung metastasis
by modulating the immune microenvironment. Cancer Res.
75:1624–1634. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Sun X, Cheng G, Hao M, Zheng J, Zhou X,
Zhang J, Taichman RS, Pienta KJ and Wang J: CXCL12/CXCR4/CXCR7
chemokine axis and cancer progression. Cancer Metastasis Rev.
29:709–722. 2010.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Wightman SC, Uppal A, Pitroda SP, Ganai S,
Burnette B, Stack M, Oshima G, Khan S, Huang X, Posner MC, et al:
Oncogenic CXCL10 signalling drives metastasis development and poor
clinical outcome. Br J Cancer. 113:327–335. 2015.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Ishiguro H, Akimoto K, Nagashima Y, Kojima
Y, Sasaki T, Ishiguro-Imagawa Y, Nakaigawa N, Ohno S, Kubota Y and
Uemura H: aPKClambda/ι promotes growth of prostate cancer cells in
an autocrine manner through transcriptional activation of
interleukin-6. Proc Natl Acad Sci USA. 106:16369–16374.
2009.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Peng H, Chen P, Cai Y, Chen Y, Wu QH, Li
Y, Zhou R and Fang X: Endothelin-1 increases expression of
cyclooxygenase-2 and production of interlukin-8 in hunan pulmonary
epithelial cells. Peptides. 29:419–424. 2008.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Timani KA, Győrffy B, Liu Y, Mohammad KS
and He JJ: Tip110/SART3 regulates IL-8 expression and predicts the
clinical outcomes in melanoma. Mol Cancer. 17(124)2018.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Yang M, Liu J, Piao C, Shao J and Du J:
ICAM-1 suppresses tumor metastasis by inhibiting macrophage M2
polarization through blockade of efferocytosis. Cell Death Dis.
6(e1780)2015.PubMed/NCBI View Article : Google Scholar
|
|
56
|
de Groote ML, Kazemier HG, Huisman C, van
der Gun BT, Faas MM and Rots MG: Upregulation of endogenous ICAM-1
reduces ovarian cancer cell growth in the absence of immune cells.
Int J Cancer. 134:280–290. 2014.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Benatar T, Cao MY, Lee Y, Lightfoot J,
Feng N, Gu X, Lee V, Jin H, Wang M, Wright JA, et al: IL-17E, a
proinflammatory cytokine, has antitumor efficacy against several
tumor types in vivo. Cancer Immunol Immunother. 59:805–817.
2010.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Benatar T, Cao MY, Lee Y, Li H, Feng N, Gu
X, Lee V, Jin H, Wang M, Der S, et al: Virulizin induces production
of IL-17E to enhance antitumor activity by recruitment of
eosinophils into tumors. Cancer Immunol Immunother. 57:1757–1769.
2008.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Wei C, Sirikanjanapong S, Lieberman S,
Delacure M, Martiniuk F, Levis W and Wang BY: Primary mucosal
melanoma arising from the eustachian tube with CTLA-4, IL-17A,
IL-17C, and IL-17E upregulation. Ear Nose Throat J. 92:36–40.
2013.PubMed/NCBI
|