Introduction
Genetically defined molecular subtypes of clinical
breast cancer facilitate the accurate prediction of disease
progression and the rational selection of targeted therapeutic
options (1). The expression of
human epidermal growth factor receptor-2 (HER-2) on a background of
estrogen receptor-α (ER-α) and progesterone receptor (PR)
positivity or negativity dictates distinct therapeutic options.
These conventional therapeutic options include HER-2 selective
antibodies and dual specific small molecule inhibitors with or
without selective estrogen receptor modulators and aromatase
inhibitors (2-4).
These treatment options are frequently associated with systemic
toxicity, acquired tumor resistance and the emergence of
drug-resistant cancer stem cells, favoring the progression of
therapy-resistant disease (5).
These clinical limitations emphasize the need for the development
and molecular characterization of reliable cancer stem cell models,
and for the identification of testable alternative agents that
display manageable systemic toxicity and exhibit stem cell targeted
therapeutic efficacy.
Naturally occurring phytochemicals and herbal
extracts possessing minimal systemic toxicity may represent
testable alternatives to conventional chemoendocrine therapy for
clinical breast cancer (6,7).
Published evidence on the present model for HER-2 enriched breast
cancer has demonstrated potent anti-proliferative and pro-apoptotic
effects of several mechanistically distinct naturally occurring
compounds, including phytoalexins (8), isoflavones (9-11),
vitamin A derivatives (12,13) and phenolic terpenoids (14,15). This documented evidence for the
susceptibility of the present model raises a possibility that
natural products may also be effective as testable alternatives for
cancer stem cell targeted therapeutic efficacy.
The cancer stem cell-enriched population derived
from the primary tumor is characterized by the presence of a
tumor-initiating drug-resistant phenotype (16,17). The expression of the stem
cell-specific molecular markers, octamer binding transcription
factor-4 (Oct-4), sex determining region Y-box-2 (SOX-2) and the
DNA binding homeobox nuclear transcription factor, NANOG, are
associated with compromised differentiation and overall survival in
HER-2-positive clinical breast cancer (18). Drug-resistant stem cell models
have been developed from the luminal A, HER-2-enriched and
triple-negative molecular subtypes for clinical breast cancer, and
the drug-resistant stem cell phenotypes in these models exhibit an
upregulated cellular expression of select nuclear transcription
factors, including Oct-4, NANOG and c-Myc (19-21).
Thus, the expression status of transcription factors represents
quantitative molecular markers in the developed stem cell
models.
The present study utilized a cellular model of human
mammary epithelial cells that exhibits tumorigenic transformation
due to the targeted expression of the HER-2 oncogene. These
tumorigenic cells lack the expression of estrogen and progesterone
receptors. Thus, the expression of HER-2 on an ER-α- negative and
PR-negative background provides a model for HER-2-enriched breast
cancer. The experiments carried out in this study were designed to:
i) Develop a drug-resistant stem cell model for HER-2-enriched
breast cancer; and ii) examine the stem cell targeted therapeutic
efficacy of select natural agents.
Materials and methods
The experimental model used in the current study is
described below.
Cells and cell culture
The 184-B5/HER tumorigenic cell line is derived from
parental 184-B5 cells that were stably transfected with the HER-2
oncogene (22). The 184-B5/HER
cell line was obtained from Professor C.W. Welsch (Michigan State
University, East Lansing, MI, USA). This cell line was grown in
DME-F12 medium supplemented with 10 ng/ml EGF, 0.5 µg/ml
hydrocortisone, 10 µg/ml transferrin, 10 µg/ml insulin and 5 µg/ml
of gentamycin (all from Sigma-Aldrich, St. Louis, MO, USA). The
184-B5/HER cells were routinely maintained in the presence of 200
µg/ml G418 (Geneticin; Sigma-Aldrich) to eliminate the expression
of spontaneous revertants. The cell cultures were maintained at
37˚C in humidified atmosphere of 95% air: 5% CO2, and
were sub-cultured at 80% confluency.
Test agents
The small molecule inhibitor, lapatanib (LAP), and
all trans-retinoic acid (ATRA) were purchased from Sigma-Aldrich.
Carnosol (CSOL) was provided by Nestle Research Center (Lausanne,
Switzerland). The stock solutions of LAP, ATRA and CSOL were
prepared in dimethyl sulfoxide (DMSO; Sigma-Aldrich) at the
concentrations of 10 mM. These stock solutions were serially
diluted in the culture medium to obtain the final concentrations of
10 µM LAP, 2 µM ATRA and 5 µM CSOL. These final concentrations
represented the pre-determined IC90 (maximally
cytostatic) concentrations for the 184-B5/HER cells. The treatment
for the tumor spheroid assay was of 14 days duration and that for
the cellular immuno-fluorescence assays was of 3 day duration.
LAP-resistant (LAP-R) phenotype
To isolate the LAP-R phenotype, parental 184-B5/HER
cells were selected in the presence of 10 µM LAP for approximately
6 months and the surviving cell population was expanded and
maintained in the presence of LAP for at least 5 passages prior to
the experiments.
Antibodies
The human reactive FITC-conjugated antibodies
anti-HER-2 (cat. no. sc-81528), epidermal growth factor receptor
(EGFR; cat. no. sc-373746) and IgG (cat. no. SC 2339) were
purchased from Santa Cruz Biotechnologies (Santa Cruz, CA, USA).
CD44 (cat. no. ab27285) was purchased from Abcam (Cambridge, MA,
USA). NANOG (cat. no. AP 1486c) and Oct-4 (cat. no. AP 2046a) were
purchased from Abgent, Inc. (San Diego, CA, USA). These antibodies
were used following the recommended dilutions provided in the
technical protocols from the vendors.
Tumor spheroid assay
For this assay, the LAP-R cells were seeded at a
density of 100 cells per well in ultra-low adhesion 6-well plates.
(Corning/Costar, Corning, NY, USA) in serum-free DME-F12 medium.
The culture medium was supplemented with 20 ng/ml EGF, 10 ng/ml
basal fibroblast growth factor (FGF), 5 µg/ml insulin, 1 ng/ml
hydrocortisone and 4 µg/ml heparin sodium (all form Sigma-Aldrich),
1% B27 and 10 ng/ml leukaemia inhibitory factor (LIF; Thermo Fisher
Scientific, Inc., Waltham, MA, USA). The cultures were maintained
at 37˚C in humidified atmosphere of 95% air: 5% CO2 and
the spheroids formed at day 14 post-seeding were counted at
magnification, x10 under an inverted light microscope. The data
were expressed as number of spheroids (mean ± SD; n=24) per
treatment group from quadruplicate experiments.
Cellular immunofluorescence assay
This cellular immuno-fluorescence assay monitored
antibody positivity in cells stained with FITC-conjugated
antibodies following the previously published optimized protocol
(10,12). Briefly, the cell suspension fixed
in 0.25% paraformaldehyde (Polysciences, Warrington, PA, USA), was
incubated with 0.1% Triton X-100 (Sigma-Aldrich) on ice for 3 min
to permeabilize the cell membrane. The permeabilized cells were
washed twice with phosphate-buffered saline (PBS; pH 7.4;
Sigma-Aldrich), and stained with appropriate FITC-conjugated
antibodies according to the protocol provided by the supplier. The
stained cells were monitored for the cellular expression of
individual antibody by fluorescence-assisted cell sorting using a
flow cytometer. Cells stained with isotype FITC-conjugated IgG
represented the negative control. The experimental data were
corrected for the fluorescence due to FITC IgG staining, and the
status of cellular expression was presented as log mean
fluorescence units (FU) per 104 fluorescent events from
quadruplicate experiments.
Statistical analysis
Experiments using cellular immunofluorescence assay
were performed in quadruplicate per treatment group. Experiments
using tumor spheroid assay were performed in quadruplicate per
treatment group. The data were presented as the means ± SD and were
analyzed for statistical significance between the control and
experimental groups by a two-sample Student's t-test. Data
comparing multiple treatment groups were analyzed by one-way
analysis of variance (ANOVA) and Dunnett's multiple range test with
a threshold of α=0.05. All statistical analyses were performed
using Microsoft Excel 2013 XLSTAT-Base software. Values of
P#x003C;0.05 were considered to indicate statistically significant
differences.
Results
Inhibition of human epidermal growth
factor receptors by LAP
The experiment was designed to examine the efficacy
of LAP on the parental 184-B5/HER cells by monitoring the status of
the cellular expression of p-EGFR and p-HER-2 (Table I). LAP treatment induced a 57.1
and a 78.2% inhibition in the cellular expression of p-EGFR and
p-HER-2, respectively, relative to the cells treated with the
solvent DMSO.
 | Table IInhibition of human epidermal growth
factor receptors. |
Table I
Inhibition of human epidermal growth
factor receptors.
| | | Receptor expression
(Log mean FU)a |
| Treatment | Concentration | p-EGFR | p-HER-2 |
| DMSO | 0.1% | 11.9±1.4b | 51.5±6.2d |
| LAP | 10 µM | 5.1±1.8c | 11.2±2.3e |
Status of stem cell markers in LAP-R
cells
The experiment was designed to monitor the tumor
spheroid number and the status of the cellular expression of the
stem cell markers, CD44, NANOG and Oct-4, in LAP-sensitive (LAP-S)
and LAP-R phenotypes. The results are presented in Table II. The LAP-R phenotype exhibited
a 2.3-fold increase in tumor spheroid number, a 4.2-fold increase
in CD44, a 4.4-fold increase in NANOG and a 1.9-fold increase in
Oct-4 expression, respectively, relative to that in the LAP-S
phenotype.
 | Table IIStem cell marker status in LAP-R
cells. |
Table II
Stem cell marker status in LAP-R
cells.
| Phenotype | Tumor spheroid
numbera | CD44b | NANOGb | Oct-4b |
| LAP-S | 4.5±1.3c | 4.0±0.7e | 2.2±0.6g | 4.8±0.3i |
| LAP-R | 14.8±2.8d | 20.8±1.9f | 11.8±1.2h | 14.2±1.5j |
Experimental modulation of stem cell
markers in LAP-R cells
The experiment that was designed to examine the
effects of ATRA and CSOL on the number of tumor spheroids on the
LAP-R phenotype is presented in Fig.
1A. Treatment of the spheroid cultures with 2 µM ATRA and 5 µM
CSOL resulted in a 69.1 and 80.3% decrease in the spheroid number
relative to that in the cultures treated with the solvent DMSO. The
effects of ATRA and CSOL on the levels of additional stem
cell-specific markers, CD44, NANOG and Oct-4, as regards the LAP-R
phenotype were also examined and the results are presented in
Fig. 1B. The extent of inhibition
of the cellular expression of these markers in response to a
treatment with 2 µM ATRA ranged from 71.7 to 81.9%. Treatment with
5 µM CSOL resulted in an inhibition range of 69.0 to 84.4%.
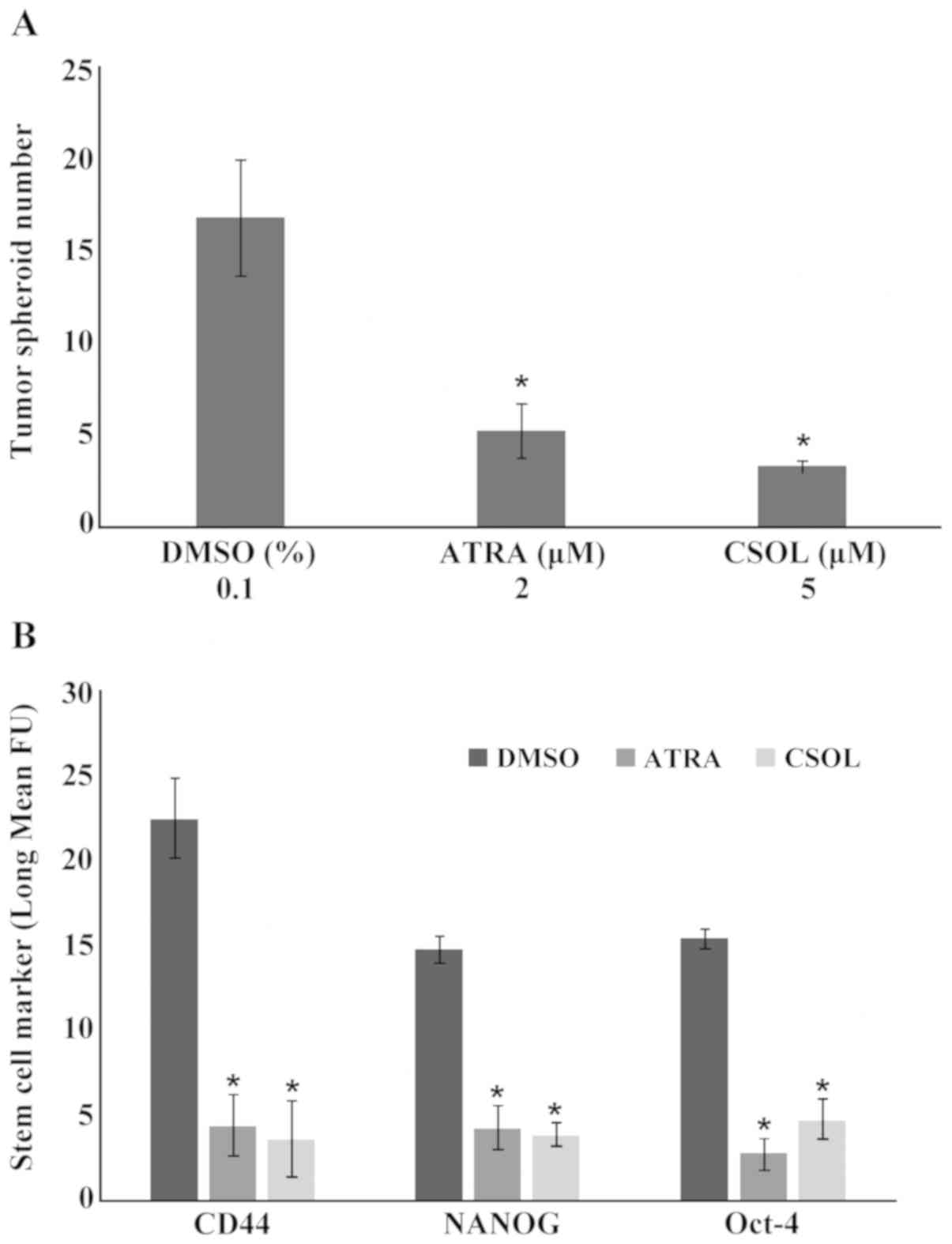 | Figure 1.Experimental modulation of stem cell
markers by ATRA and CSOL in LAP-R stem cells. (A) Tumor spheroids
treated with ATRA and CSOL exhibit the inhibition in the number of
tumor spheroids. Results are presented as the means ± SD, n=4 per
treatment group. Data analyzed by ANOVA and Dunnett's test.
*P#x003C;0.05, ATRA vs. DMSO and CSOL vs. DMSO. (B)
Treatment with ATRA and CSOL downregulated the cellular expression
of CD44, NANOG and Oct-4. Results are presented as the log mean FU
± SD, n=4 per treatment group. Data analyzed by ANOVA and Dunnett's
test. Statistical significance is indicated as follows: for CD44: 2
µM ATRA vs. 0.1% DMSO (*P#x003C;0.05), 5 µM CSOL vs.
0.1% DMSO (*P#x003C;0.05); NANOG: 2 µM ATRA vs. 0.1%
DMSO (*P#x003C;0.05), 5 µM CSOL vs. 0.1% DMSO
(*P#x003C;0.05); Oct-4: 2 µM ATRA vs. 0.1% DMSO
(*P#x003C;0.05), 5 µM CSOL vs. 0.1% DMSO
(*P#x003C;0.05). DMSO, dimethyl sulfoxide; ATRA, all-
trans retinoic acid; CSOL, carnosol; CD44, cluster of
differentiation; NANOG, DNA binding homeobox transcription factor;
Oct-4, octamer binding protein-4; LAP-R, lapatinib LAP-R resistant;
FU, fluorescence units; ANOVA, analysis of variance. |
Discussion
The hormone receptor-positive HER-2-expressing
breast cancer (luminal B) molecular subtype is treated with HER-2
targeted therapy and conventional endocrine therapy that includes
selective estrogen receptors, selective estrogen receptor degraders
and aromatase inhibitors (4,5).
Hormone receptor-negative HER-2-expressing breast cancer is treated
with HER-2 targeted therapy and conventional chemotherapy that
includes anthracyclines and taxanes (2). These long-term treatment options are
associated with the emergence of drug-resistant cancer stem cells,
representing one of the major reasons for the compromised treatment
response (17), and thereby,
emphasizing the importance of identifying novel, less toxic
treatment options for chemo-endocrine therapy-resistant breast
cancer. The present study developed a LAP-R stem cell model, and
validated the model using ATRA and CSOL as naturally occurring test
agents. This approach now facilitates evaluation of growth
inhibitory efficacy of additional natural products.
LAP, a dual function small molecule inhibitor of
human epidermal growth factor receptors operates by inhibiting the
phosphorylation of EGFR and HER-2, as demonstrated in ER-negative,
PR-negative, HER-2-positive human breast carcinoma-derived models
(23,24). Consistent with these data, the
184/B5/HER cells, in response to treatment with LAP, exhibited a
substantial inhibition in the cellular expression of p-EGFR and
p-HER-2.
The emergence of drug-resistant phenotypes, tumor
spheroid formation and the increased expression of nuclear
transcription factors have been demonstrated in stem cell models
for select breast cancer subtypes (19-21).
Furthermore, the nuclear transcription factors, Oct-4, Kruppel-like
factor 4 (Klf-4), SOX-2 and c-Myc, represent essential components
for induced pluripotent cell models developed from adult somatic
cells (25-27).
Thus, these cellular signaling proteins represent stem cell
selective markers. The data on the status of expression of these
markers have demonstrated substantial upregulation in LAP-R cells
in comparison to LAP-S cells. Collectively, these data provide
evidence for the effective development and stringent
characterization of the LAP-R cancer stem cell model.
The vitamin A derivative, ATRA, functions as a
potent anti-proliferative and pro-apoptotic agent in
HER-2-expressing 184-B5/HER cells, by increasing the cellular
expression of the negative growth regulator, retinoic acid
receptor-β (RAR-β), and decreasing the cellular expression of the
anti-apoptotic BCL-2 protein (12). In addition, ATRA has been shown to
inhibit the transcriptional activity of inducible cyclooxygenase-2
(COX-2) via RAR-mediated mechanisms, while CSOL functions via
protein kinase C, ERK1/2, Jun terminal kinase and p38-related
mitogen-activated protein kinase (MAPK)-dependent pathways
(13) to target the
transcriptional activation of inducible COX-2 and of prostaglandin
production in 184-B5/HER cells.
Mechanistically distinct naturally occurring
phytochemicals have also been demonstrated to target cancer stem
cells from multiple organ site cancers. For example, ATRA has been
shown to target gastric cancer stem cells by downregulating the
expression levels of CD44, aldehyde dehydrogenase-1 (ALDH-1), as
well as transcription factors Klf-4 and SOX-2(28). Sulforaphane, present in broccoli,
has been shown to increase drug-mediated cytotoxicity in pancreatic
and prostatic cancer stem cells, and to inhibit tumor spheroid
formation and ALDH-1. In addition, this natural product inhibits
the Notch-1 and nuclear factor-κB (NF-κB) pathways (29). The natural polyphenol, quercetin,
targets pancreatic cancer stem cells by inhibiting tumor spheroid
formation and ALDH-1 expression (30). Furthermore, sulforaphane, acting
synergistically with the pan multi-kinase inhibitor, sorafenib,
abolishes the stem cell characteristics of pancreatic cancer stem
cells by inhibiting tumor spheroid formation, and the ALDH-1 and
NF-κB pathways (31). Recent
evidence has also demonstrated that sulforaphane decreases the
expression of stem cell markers NANOG, ALDH1A1, Wnt3 and Notch,
thereby, inhibiting the growth of triple negative breast cancer
stem-like cells (32), and benzyl
isothiocyanate, present in cruciferous vegetables, inhibits breast
cancer stem-like population via upregulation of the KLF-4-p21CIP1
axis (33). Collectively, these
data provide evidence that the downregulated signaling molecules
may represent molecular targets relevant to stem cell-specific
therapeutic efficacy of natural products. Thus, in the present
study, the significant inhibition of select stem cell markers, such
as tumor spheroids, CD44, NANOG and Oct-4 in LAP-R stem cells
treated with ATRA and CSOL, provides evidence for stem cell
targeting efficacy of these naturally occurring agents.
In conclusion, the outcomes of the present study
provide strong evidence supporting the effective development of a
cancer stem cell model for the HER-2-enriched breast cancer
subtype. Furthermore, experiments with ATRA and CSOL together have
provided valuable mechanistic leads for their stem cell specific
efficacy, and thereby, have validated the present experimental
approach for the evaluation of stem cell-specific mechanistic
efficacy of natural products. However, it needs to be emphasized
that to extend clinical translatability of the present approach,
reliable stem cell models from patient derived xenografts and
organoid cultures from HER-2 targeted therapy resistant breast
cancer are essential. These aspects represent promising future
directions for the clinical application of patient-derived
pre-clinical data (34,35).
Acknowledgements
The author wishes to acknowledge the active
participation of former colleagues and collaborators in the
research program ‘Cellular models for molecular subtypes of
clinical breast cancer: Mechanistic approaches for lead compound
efficacy’.
Funding
The research in the author's laboratory has been
supported in the past by the National Cancer Institute (NCI) FIRST
Award CA 44741, the Department of Defense Breast Cancer Research
Program IDEA Award DAMD-17-94-J-4208, the NCI Contract Research
Master Agreement CN 75029-63, and by philanthropic funds to Strang
Cancer Prevention Center.
Availability of data and materials
The data used and/or analyzed in this study are
available from the corresponding author upon reasonable
request.
Author's contribution
NT contributed towards the study conception,
experimental design, data analysis, data interpretation and
prepared the manuscript for publication.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The author declares that there are no competing
interests.
References
|
1
|
Sørlie T, Perou CM, Tibshirani R, Aas T,
Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey
SS, et al: Gene expression patterns of breast carcinomas
distinguish tumor subclasses with clinical implications. Proc Natl
Acad Sci USA. 98:10869–10874. 2001.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Romond EH, Perez EA, Bryant J, Suman VJ,
Geyer CE Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman
PA, et al: Trastuzumab plus adjuvant chemotherapy for operable
HER2-positive breast cancer. N Engl J Med. 353:1673–1684.
2005.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Baselga J and Swain SM: Novel anticancer
targets: Revisiting ERBB2 and discovering ERBB3. Nat Rev Cancer.
9:463–475. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Johnston SRD and Dowsett M: Aromatase
inhibitors for breast cancer: Lessons from the laboratory. Nat Rev
Cancer. 3:821–831. 2003.PubMed/NCBI View
Article : Google Scholar
|
|
5
|
Musgrove EA and Sutherland RL: Biological
determinants of endocrine resistance in breast cancer. Nat Rev
Cancer. 9:631–643. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Lee KW, Bode AM and Dong Z: Molecular
targets of phytochemicals for cancer prevention. Nat Rev Cancer.
11:211–218. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Ye I, Jia Y, Ji KE, Sanders AJ, Xue K, Ji
J, Mason MD and Jiang WG: Traditional Chinese medicine in the
prevention and treatment of breast cancer and cancer metastasis.
Oncol Lett. 10:1240–1250. 2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Telang NT, Katdare M, Bradlow HL, Osborne
MP and Fishman J: Inhibition of proliferation and modulation of
estradiol metabolism: Novel mechanisms for breast cancer prevention
by the phytochemical indole-3-carbinol. Proc Soc Exp Biol Med.
216:246–252. 1997.PubMed/NCBI
|
|
9
|
Subbaramaiah K, Chung WJ, Michaluart P,
Telang N, Tanabe T, Inoue H, Jang M, Pezzuto JM and Dannenberg AJ:
Resveratrol inhibits cyclooxygenase-2 transcription and activity in
phorbol ester-treated human mammary epithelial cells. J Biol Chem.
273:21875–21882. 1998.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Katdare M, Osborne M and Telang NT: Soy
isoflavone genistein modulates cell cycle progression and induces
apoptosis in HER-2/neu oncogene expressing human breast epithelial
cells. Int J Oncol. 21:809–815. 2002.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Katdare M, Osborne MP and Telang NT: Novel
cell culture models for prevention of human breast cancer (Review).
Int J Oncol. 22:509–515. 2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Jinno H, Steiner MG, Nason-Burchenal K,
Osborne MP and Telang NT: Preventive efficacy of receptor class
selective retinoids on HER-2/neu oncogene expressing preneoplastic
human mammary epithelial cells. Int J Oncol. 21:127–134.
2002.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Subbaramaiah K, Cole PA and Dannenberg AJ:
Retinoids and carnosol suppress cyclooxygenase-2 transcription by
CREB-binding protein/p300-dependent and -independent mechanisms.
Cancer Res. 62:2522–2530. 2002.PubMed/NCBI
|
|
14
|
Subbaramaiah K, Michaluart P, Sporn MB and
Dannenberg AJ: Ursolic acid inhibits cyclooxygenase-2 transcription
in human mammary epithelial cells. Cancer Res. 60:2399–2404.
2000.PubMed/NCBI
|
|
15
|
Telang N: Anti-proliferative and
pro-apoptotic effects of rosemary and constituent terpenoids in a
model for the HER-2-enriched molecular subtype of clinical breast
cancer. Oncol Lett. 16:5489–5497. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Dean M, Fojo T and Bates S: Tumour stem
cells and drug resistance. Nat Rev Cancer. 5:275–284.
2005.PubMed/NCBI View
Article : Google Scholar
|
|
17
|
Patel SA, Ndabahaliye A, Lim PK, Milton R
and Rameshwar P: Challenges in the development of future treatments
for breast cancer stem cells. Breast Cancer (Dove Med Press).
2:1–11. 2010.PubMed/NCBI
|
|
18
|
Yang F, Zhang J and Yang H: OCT4, SOX2,
and NANOG positive expression correlates with poor differentiation,
advanced disease stages, and worse overall survival in HER2+ breast
cancer patients. OncoTargets Ther. 11:7873–7881. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Telang N: Putative cancer-initiating stem
cells in cell culture models for molecular subtypes of clinical
breast cancer. Oncol Lett. 10:3840–3846. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Telang N: Growth inhibitory efficacy of
natural products in a model for triple negative molecular subtype
of clinical breast cancer. Biomed Rep. 7:199–204. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Telang N: Stem cell targeted therapeutic
approaches for molecular subtypes of clinical breast cancer. World
Acad Sci J. 1:20–24. 2019. View Article : Google Scholar
|
|
22
|
Zhai YF, Beittenmiller H, Wang B, Gould
MN, Oakley C, Esselman WJ and Welsch CW: Increased expression of
specific protein tyrosine phosphatases in human breast epithelial
cells neoplastically transformed by the neu oncogene. Cancer Res.
53:2272–2278. 1993.PubMed/NCBI
|
|
23
|
Rusnak DW, Lackey K, Affleck K, Wood ER,
Alligood KJ, Rhodes N, Keith BR, Murray DM, Knight WB, Mullin RJ
and Gilmer TM: The effects of the novel, reversible epidermal
growth factor receptor/ErbB-2 tyrosine kinase inhibitor, GW2016, on
the growth of human normal and tumor-derived cell lines in
vitro and in vivo. Mol Cancer Ther. 1:85–94.
2001.PubMed/NCBI
|
|
24
|
Nahta R, Yuan LXH, Du Y and Esteva FJ:
Lapatinib induces apoptosis in trastuzumab-resistant breast cancer
cells: Effects on insulin-like growth factor I signaling. Mol
Cancer Ther. 6:667–674. 2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Takahashi K, Tanabe K, Ohnuki M, Narita M,
Ichisaka T, Tomoda K and Yamanaka S: Induction of pluripotent stem
cells from adult human fibroblasts by defined factors. Cell.
131:861–872. 2007.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Park IH, Zhao R, West JA, Yabuuchi A, Huo
H, Ince TA, Lerou PH, Lensch MW and Daley GQ: Reprogramming of
human somatic cells to pluripotency with defined factors. Nature.
451:141–146. 2008.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Yu J, Hu K, Smuga-Otto K, Tian S, Stewart
R, Slukvin II and Thomson JA: Human induced pluripotent stem cells
free of vector and transgene sequences. Science. 324:797–801.
2009.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Nguyen PH, Giraud J, Staedel C,
Chambonnier L, Dubus P, Chevret E, Bœuf H, Gauthereau X, Rousseau
B, Fevre M, et al: All-trans retinoic acid targets gastric cancer
stem cells and inhibits patient-derived gastric carcinoma tumor
growth. Oncogene. 35:5619–5628. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kallifatidis G, Labsch S, Rausch V,
Mattern J, Gladkich J, Moldenhauer G, Büchler MW, Salnikov AV and
Herr I, Sainikov AV and Herr I: Sulforaphane increases
drug-mediated cytotoxicity toward cancer stem-like cells of
pancreas and prostate. Mol Ther. 19:188–195. 2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zhou W, Kallifatidis G, Baumann B, Rausch
V, Mattern J, Gladkich J, Giese N, Moldenhauer G, Wirth T, Büchler
MW and Herr I: Dietary polyphenol quercetin targets pancreatic
cancer stem cells. Int J Oncol. 37:551–561. 2010.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Rausch V, Liu L, Kallifatidis G, Baumann
B, Mattern J, Gladkich J, Wirth T, Schemmer P, Büchler MW, Zöller
M, et al: Synergistic activity of sorafenib and sulforaphane
abolishes pancreatic cancer stem cell characteristics. Cancer Res.
70:5004–5013. 2010.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Castro NP, Rangel MC, Merchant AS,
MacKinnon G, Cuttitta F, Salomon DA and Kim YS: Sulforaphane
suppresses the growth of triple negative breast cancer stem-like
cells in vitro and in vivo. Cancer Prev Res (Phila).
12:147–158. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kim SH and Singh SV: Role of Kruppel-like
factor 4-p21CIP1 axis in breast cancer stem-like cell
inhibition by benzyl isothiocyanate. Cancer Prev Res (Phila).
12:125–134. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Bruna A, Rueda OM, Greenwood W, Batra AS,
Callari M, Batra RN, Pogrebniak K, Sandoval J, Cassidy JW,
Tufegdzic-Vidakovic A, et al: A biobank of breast cancer explants
with preserved intra-tumor heterogeneity to screen anti-cancer
compounds. Cell. 167:260–274, e22. 2016.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Sachs N, de Ligt J, Kopper O, Gogola E,
Bounova G, Weeber E, Balgobind AV, Wind K, Gracanin A, et al: A
living biobank of breast cancer organoids captures disease
heterogeneity. Cell. 172:373–386, e10. 2018.PubMed/NCBI View Article : Google Scholar
|















