Introduction
As strong inhibitors of topoisomerase 1, the
derivatives of camptothecin have been thoroughly investigated by
cancer researchers and clinical oncologists. Two compounds of the
camptothecin family, irinotecan and topotecan, have been widely
used by clinicians to treat various types of cancer (1–6).
Several additional new camptothecin derivatives have been
synthesized and tested pre-clinically and clinically. In our
laboratory, various camptothecin were synthesized esters for
biological evaluation (7–10). Crystalline
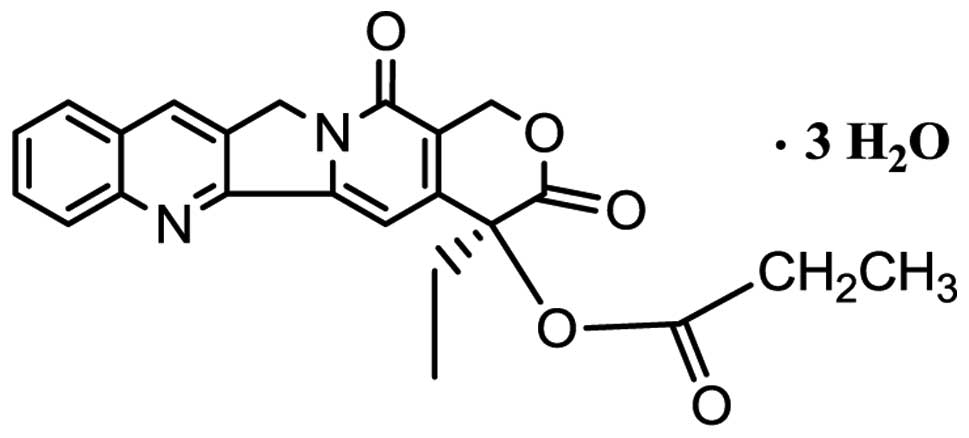
camptothecin-20-O-propionate hydrate (CZ48) was prepared using an
acid-catalyzed method with a high reaction yield (11). The structure of this agent is shown
in Fig. 1. This compound has been
tested against 29 human tumor xenografts grown in our nude mouse
system thus far. Initially, in a previous study, we reported the
results against the first 19 of these xenografts (12). However, further study of these
xenografts has yielded 29 tumors in total. Besides one kidney tumor
(28/29) with a 97% response rate, CZ48 has shown promising
anticancer activity against almost every type of human tumor tested
in our nude mouse system. The toxicity of this agent in mice has
been found to be minimal.
From the above investigations, the effective dose
(ED) with which tumor growth is inhibited >50% was found to vary
widely from tumor to tumor. For instance, human breast xenograft
(CLO) was treated with only 100 mg/kg/day to achieve complete
growth inhibition in nude mice, whereas the human colon McCN
requires a dose as high as 1000 kg/mg/day to achieve an 88.2%
inhibition (12). Sensitivity of
the tumors to treatment by CZ48 were relatively different. The
difference in dose from the most to the least sensitive tumor for
the 29 tumors tested thus far is 20-fold. CZ48 itself is not
active. An enzymatic cleavage of the side ester chain of CZ48 to
release the molecule of camptothecin activates the process of
killing cancer cells (13,14). The esterase activity of each tumor
is the determining factor for the local concentration of the active
metabolite camptothecin.
The doses of a chemotherapeutic agent required for a
particular treatment are calculated based on the pharmacological
and toxicological data of the agent in patients obtained from
clinical trials. The theoretical formula guiding the dosing of an
anticancer agent is not currently available, and at present the
correlation between the sensitivity of an agent to treatment and
the type of tumor remains to be determined. To understand why the
sensitivity of treatment with CZ48 varies widely from one tumor
type to another, we used nude mice carrying human tumors as the
animal model in order to determine the local concentrations of the
parental CZ48 and its active metabolite corresponding metabolite
camptothecin (CPT) in the tumors and major organ tissues. The assay
used in this study was duplicable and consistent for the
measurements. The aim of this study was to determine whether the
data obtained in this study may be used for ongoing and future
clinical studies.
Materials and methods
Chemicals
High-performance liquid chromatography (HPLC)-grade
acetic acid, dimethyl sulfoxide (DMSO), acetonitrile,
dichloromethane and diethyl ether were obtained from Sigma-Aldrich
(St. Louis, MO, USA). Chromatographic-grade water was produced by a
Millipore Milli-Q system (Billerica, MA, USA). CZ48 and CZ44
(internal standard for HPLC quantification) were synthesized
in-house as previously described (7). Camptothecin, with a purity of 99%, was
purchased from China and used according to the manufacturer’s
instructions.
Growth of human tumors in mice as
xenografts
A tumor xenograft growing in a nude mouse (size, ∼1
cm3) was surgically removed under sterile conditions,
finely minced with iridectomy scissors and suspended in a cell
culture medium at a ratio of 1:10, v/v. One-tenth to one quarter of
1 ml of this suspension, containing ∼50 mg of wet-weight tumor
mince, was subcutaneously inoculated into the upper half of the
dorsal thorax of the mouse. Groups of female, non-inbred Swiss nude
mice,(17 groups, 4 animals in each group) with a weight of ∼25 g,
were used.
Tissue sample preparation
Human tumors PC3, MIA, SW48, CLO, DOY, MUR, HT29,
SQU and SU86 were individually grown in each group of mice as
xenografts. Tumors growing in animals were monitored daily and
measured using a caliper two times per week. Treatment with 1000
mg/kg/day CZ48 was initiated when the tumor reached a volume of ∼80
mm3, i.e., well-vascularized and measurable. CZ48 was
finely suspended in cottonseed oil. Each group of animals was
treated once a day by gavage with the above dose. The animals were
sacrificed at the pre-determined time points, and tumor and major
organ tissue samples were then collected surgically. Following a
series of processing, the tissue samples were extracted a number of
times and the extracts obtained from each animal were combined and
frozen until analysis.
Measurement of CZ48 and CPT
Animal extracts were analyzed using HPLC. The
detailed procedure for analyzing the concentrations of CZ48 and CPT
was described in a previous study (15). Experiments were performed by
following the protocol approved by the Institutional Animal Care
and Use Committee.
Results and Discussion
To obtain the time point when the steady-state
concentrations (or saturation) of CZ48 and CPT in tumor tissues was
reached, 3 groups of nude mice carrying DOY lung carcinoma
xenografts were treated with 1000 mg/kg/day of CZ48 by gavage, for
2, 4 and 6 days, respectively. At each time point, a group of
animals was sacrificed and tumor tissues were collected and
processed for analysis (Table
I).
 | Table IAccumulation of CPT and CZ48 in tumor
tissues of nude mice lung (DOY) human xenografts after daily oral
treatment with 1000 mg/kg CZ48 for 2, 4 and 6 days. |
Table I
Accumulation of CPT and CZ48 in tumor
tissues of nude mice lung (DOY) human xenografts after daily oral
treatment with 1000 mg/kg CZ48 for 2, 4 and 6 days.
| Day | CZ48 (ng/mg tissue) ±
SD | CPT (ng/mg tissue) ±
SD |
|---|
| 2 | 218.15±77.90 | 35.97±4.10 |
| 4 | 218.17±73.06 | 31.31±4.12 |
| 6 | 191.66±60.76 | 27.86±3.61 |
To evaluate the sensitivity of tumors to CZ48
treatment, 9 groups of the tumor-carrying mice were treated with
1000 mg/kg of CZ48 daily for 5 days. The animals were then
sacrificed and their tumor tissues were collected and extracted.
The local concentration of CZ48 and its active metabolite CPT in
tumors were determined using an established HPLC procedure
(Table II). Measurement of the
biodistributions of CZ48 in major organs of mice was also
performed. The local concentrations of CZ48 and its metabolite CPT
in the brain, heart, kidney, liver, lung and spleen, are shown in
Table III.
 | Table IIAccumulation of CPT and CZ48 in tumor
tissues of nude mice carrying human xenografts following oral
treatment with 1000 mg/kg/day for 5 days. |
Table II
Accumulation of CPT and CZ48 in tumor
tissues of nude mice carrying human xenografts following oral
treatment with 1000 mg/kg/day for 5 days.
| Tumor line | CPT (ng/mg tissue) ±
SD | CZ48 (ng/mg tissue) ±
SD | Ratio of
CPT/CZ48 |
|---|
| Breast-CLO | 12.60±3.87 | 122.51±7.92 | 0.10 |
| Breast-MUR | 14.32±2.49 | 78.16±16.84 | 0.18 |
| Colon-HT29 | 18.30±5.80 | 45.44±3.36 | 0.40 |
| Colon-SQU | 20.16±1.78 | 50.51±9.26 | 0.40 |
| Colon-SW48 | 10.30±2.92 | 82.85±6.37 | 0.12 |
| Lung-DOY | 29.58±7.47 | 121.16±10.73 | 0.24 |
| Pancreatic-MIA | 13.35±1.46 | 72.66±3.54 | 0.18 |
| Pancreatic-SU86 | 9.53±3.96 | 126.14±5.15 | 0.08 |
| Prostate-PC3 | 24.17±6.82 | 34.01±6.20 | 0.71 |
 | Table IIIBiodistribution of CZ48 and its
metabolite CPT in major organs. |
Table III
Biodistribution of CZ48 and its
metabolite CPT in major organs.
| Organ | CPT (ng/mg tissue) ±
SD | CZ48 (ng/mg tissue) ±
SD | Ratio of
CPT/CZ48 |
|---|
| Brain | 15.98±17.17 | 92.91±56.90 | 0.17 |
| Heart | 19.37±6.41 | 141.32±50.67 | 0.14 |
| Kidney | 193.70±131.62 | 337.13±11.63 | 0.57 |
| Liver | 129.52±38.62 | 486.31±142.57 | 0.27 |
| Lung | 32.27±16.01 | 87.65±25.02 | 0.37 |
| Spleen | 37.29±1.61 | 178.69±54.13 | 0.21 |
To compare the concentrations of CZ48 and CPT in
tumor tissues with those in plasma, a group of mice carrying LIE
pancreatic carcinoma was treated with 100 mg/kg/day for two weeks
and then sacrificed for analysis (Table
IV).
 | Table IVComparison of concentration levels of
CZ48 and CPT between plasma and tumor tissues of mice following
treatment with effective doses of CZ48. |
Table IV
Comparison of concentration levels of
CZ48 and CPT between plasma and tumor tissues of mice following
treatment with effective doses of CZ48.
| Group | Organ | CPT (ng/mg plasma) ±
SD | CZ48 (ng/mg tissue) ±
SD | CPT/CZ48 |
|---|
| Pancrea-LIE | Plasma | 4.49±1.59 | 61.77±26.94 | 0.07 |
| Tumor | 15.29±6.27 | 119.75±38.26 | 0.13 |
Previously, we reported the results obtained against
the first panel of 19 xenografts (12). ED is commonly defined as the amount
of drug required to completely inhibit the growth of tumors in
animals. The EDs required for these 9 human xenografts grown in
nude mice in this study ranged from 100 to 1000 mg/kg/day. The
correlation between the tumor lines and the EDs is shown in
Table V. The sensitivity of a
particular tumor type to chemotherapy was also assessed by the
amount of agent used; thus, the lower the dose of an agent required
to inhibi tumor growth, the more sensitive the tumor is to the
therapy. Different types of tumors may have different sensitivities
to the same agent. The quantitative correlation of the relationship
between the types of tumors and their required EDs for a particular
agent is difficult to achieve due to the various number of factors
involved. However, the reason for which EDs of CZ48 required for
these 9 human xenografts varied from 100 to 1000 mg/kg/day may be
understood better by studying the biodistribution of this agent in
tumor tissues and major organs. Therefore, 9 tumor lines,
breast-CLO, breast-MUR, colon-HT29, colon-SQU, colon-SW48,
lung-DOY, pancreatic-MIA, prostate-PC3 and pancreatic-SU86, were
selected for the biodistribution investigations in this study.
 | Table VThe EDs required for 9 human
xenografts and the calculated local CPT and CZ48 concentrations
according to the conversion ratios listed in Table II. |
Table V
The EDs required for 9 human
xenografts and the calculated local CPT and CZ48 concentrations
according to the conversion ratios listed in Table II.
| Lines | CLO | MUR | HT29 | SQU | SW48 | DOY | MIA | PC3 | SU86 |
|---|
| EDs (mg/kg) (ref.
12) | 100 | 300 | 300 | 300 | 200 | 200 | 1000 | 1000 | 1000 |
| CZ48 (ng/mg
tissue)a | 12.25 | 23.45 | 13.63 | 15.15 | 16.57 | 24.23 | 72.66 | 34.01 | 126.14 |
| CPT (ng/mg
tissue)b | 2.20 | 4.22 | 5.45 | 6.06 | 1.99 | 5.82 | 13.08 | 24.15 | 10.08 |
To establish the time point of drug saturation for
the biodistribution studies, the tumor tissues of groups of animals
were collected at 3 time points of 2, 4 and 6 days, respectively.
No time point was selected prior to day 2 considering that any
shorter duration was probably insufficient for CZ48 to saturate the
mouse body. The results showed that the concentration of CZ48 in
tumor tissues of the mice carrying DOY reached the steady state
(i.e., saturation) after 2 days of treatment and thus, any time
points after 2 days were acceptable for the biodistribution studies
(Table I). We chose 5 days for the
duration of treatment, ensuring sufficient drug-saturation and a
convenient work schedule. A total of 9 human tumors were
xenotransplanted into 9 groups of nude mice, with 4 mice per tumor
group. After reaching 80 mm3 in tumor size, treatment
with 1000 mg/kg/day of CZ48 for each group was initiated and
continued for 5 days. The animals were sacrificed 1 h following the
fifth administration and their tumor tissues were collected and
processed for analysis.
The data in Table II
were obtained by performing the HPLC procedure on tissue samples,
and the results clearly demonstrate that the local concentrations
of CZ48 in tissues of breast-CLO, lung-DOY and pancreatic-SU86 are
much higher compared to the remaining tumor lines. The
concentrations of CPT, the active metabolite, were in the range of
10–20 ng/mg tissue, considering the standard deviations. The data
in Table II also suggest that the
esterase activity varies depending on the tumors. Prostate-PC3 was
found to have the highest esterase activity in this panel of tumors
due to its exhibiting the highest CPT/CZ48 ratio, while the
pancreatic tumor line SU86 showed the least esterase activity with
a CPT/CZ48 ratio of only 0.08. The difference in esterase activity
in the two breast tumor lines, CLO and MUR, is small (0.10 vs.
0.18). Two colon lines, HT29 and SQU, revealed an identical
esterase activity level. Uptake of CZ48 by Br-CLO, lung-DOY and
pan-SU86 were better compared to other lines in the panel (Table II). Prostate-PC3 had the least
uptake and the highest cleavage. The EDs of CZ48 required for the 9
tumor lines listed in Table V range
from 100 to 1000 mg/kg/day. The calculated local concentrations of
CPT relative to EDs range from 1.99 to 24.15 ng/mg tissue. These
results clearly demonstrate that breast-CLO and colon-SW48 tumor
lines are the most sensitive to CZ48 treatment, requiring only 2
ng/mg tissue of the active metabolite to achieve complete
inhibition. Prostate-PC3 and the two pancreatic lines MIA and SU86
were found to be the least sensitive group in this panel, ∼5- to
10-fold less sensitive compared to the group of CLO and SW48,
requiring ≥10 ng/mg tissue of the active CPT for the same level of
inhibition. The four lines, breast-MUR, colon-HT29 and SQU and
lung-DOY, were similar in the level of sensitivity to the
treatment, requiring 4–6 ng/mg tissue of the active CPT to inhibit
tumor growth.
The biodistribution of CZ48 among major organs
including brain, heart, kidney, liver, lung and spleen were also
measured. Liver tissues contained the highest local concentrations
of CZ48 (Table III). The kidney
tissue was also detected to have a high concentration of CZ48.
Kidney esterase(s) showed great ability of transferring CZ48 into
CPT (CPT/CZ48 ratio, 0.57). Lung tissue also demonstrated good
esterase activity with a CPT/CZ48 ratio of 0.37. Thus, physicians
would need to pay attention to potential renal toxicity induced by
high kidney conversion of CZ48 into CPT. Of note, 2 patients,
enrolled in an early phase 1 trial (unpublished data), were found
to have light grades of bladder toxicity. Increased hydration in
these patients solved the problem.
The comparison of concentrations of CZ48 and CPT in
plasma and tumor tissues was performed using the pancreatic-LIE
line. The results indicate that concentrations of CZ48 and CPT for
this tumor line were much higher in tumor tissues compared to those
in plasma (Table IV). CZ48 was
stable in plasma with an extremely low CPT/CZ48 ratio (0.07).
In conclusion, the EDs of CZ48 required to achieve
complete inhibition of tumor growth are influenced by the
properties of each tumor: the sensitivity to CZ48 and the esterase
activity (i.e., cleavability). The ratios of CPT/CZ48 in tumor
tissues range from 0.08 to 0.71, almost 9 times, for the panel of 9
assayed tumor tissues, suggesting that the activity of esterase
varies widely from tumor to tumor. The most sensitive tumors only
need ∼2 ng/mg tissue of the active metabolite to kill cancer cells,
while one prostate and two colon lines required at least 10 ng/mg
tissue of the active CPT to achieve the cell-killing. Major organs,
such as liver and kidney, contain high local concentrations of
CZ48. The kidney esterase(s) has the highest cleavability of
converting CZ48 into CPT. Hydration with water or juices is highly
recommended for patients enrolled in the clinical trials to avoid
urinary tract toxicity when treated with CZ48.
Acknowledgements
The authors would like to thank the
Christus Stehlin Foundation for Cancer Research and the Friends of
the Stehlin Foundation for Cancer Research for their financial
support.
References
|
1
|
ten Bokkel Huinink W, Gore M, Carmichael
J, Gordon A, Malfetano J, Hudson I, Broom C, Scarabelli C, Davidson
N, Spanczynski M, Bolis G, Maimstrom H, Coleman R, Fields C and
Heron F: Topotecan versus paclitaxel for the treatment of recurrent
epithelial ovarian cancer. J Clin Oncol. 15:2183–2193.
1997.PubMed/NCBI
|
|
2
|
Schiller JH, Adak S, Cella D, DeVore RF
III and Johnson DH: Topotecan versus observation after cisplatin
plus etoposide in extensive-stage small-cell lung cancer: E7593-a
phase III trial of the Eastern Cooperative Oncology Group. J Clin
Oncol. 19:2114–2122. 2001.
|
|
3
|
von Pawel J, Schiller JH, Shepherd FA,
Fields SZ, Kleisbauer JP, Chrysson NG, Stewart DJ, Clark PI, Palmer
MC, Depierre A, Carmichael J, Krebs JB, Ross G, Lane SR and Gralla
R: Topotecan versus cyclophosphamide, doxorubicin, and vincristine
for the treatment of recurrent small-cell lung cancer. J Clin
Oncol. 17:658–667. 1999.PubMed/NCBI
|
|
4
|
Pizzolato JF and Saltz LB: The
camptothecins. Lancet. 361:2235–2242. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Saltz LB, Cox JV, Blanke C, Rosen LS,
Fehrenbacher L, Moore MJ, Maroun JA, Ackland SP, Locker PK, Pirotta
N, Elfring GL and Miller LL: Irinotecan plus fluorouracil and
leucovorin for metastatic colorectal cancer. N Engl J Med.
343:905–914. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Douillard JY, Cunningham D, Roth AD,
Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J,
Alakl M, Gruia G, Awad L and Rougier P: Irinotecan combined with
fluorouracil compared with fluorouracil alone as first-line
treatment for metastatic colorectal cancer: a multicenter
randomized trial. Lancet. 355:1041–1047. 2000. View Article : Google Scholar
|
|
7
|
Cao Z, Harris N, Kozielski A, Vardeman D,
Stehlin J and Giovanella B: Alkyl esters of camptothecin and
9-nitrocamptothecin: Synthesis, in vitro pharmacokinetics,
toxicity, and antitumor activity. J Med Chem. 41:31–37. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cao ZS, Mendoza J, Dejesus A and
Giovanella B: Synthesis and anti-tumor activity of alkenyl
camptothecin esters. Acta Pharmacol Sin. 26:235–241. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cao Z, Mendoza J, Dejesus A, Vardeman D
and Giovanella B: Synthesis and antitumor activity of aromatic
camptothecin esters. Int J Mol Med. 21:477–487. 2008.PubMed/NCBI
|
|
10
|
Cao Z, Mendoza J, Kozielski A, Liu X,
Dejesus A, Wang Y, Zhan CG, Vardeman D and Giovanella B: Anticancer
activity of new haloalkyl camptothecin esters against human cancer
cell lines and human tumor xenografts grown in nude mice.
Anticancer Agents Med Chem. 12:818–828. 2012. View Article : Google Scholar
|
|
11
|
Cao Z, Kozielski A, Harris N, Vardeman D
and Giovanella D: Sulfuric acid catalyzed preparation of alkyl and
alkenl camptothecin ester derivatives and antitumor activity
against human xenografts grown in nude mice. Open J Med Chem.
2:10–14. 2012. View Article : Google Scholar
|
|
12
|
Cao Z, Kozielski A, Liu X, Wang Y,
Vardeman D and Giovanella B: Crystalline
camptothecin-20(S)-O-propionate hydrate: a novel anticancer agent
with strong activity against 19 human tumor xenografts. Cancer Res.
69:4742–4749. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cao ZS, Pantazis P, Mendoza J, Early J,
Kozielski A, Harris N and Giovanella B: Structure-activity
relationship of alkyl 9-nitrocamptothecin esters. Acta Pharmacol
Sin. 24:109–119. 2003.PubMed/NCBI
|
|
14
|
Cao Z, Pantazis P, Mendoza J, Early J,
Kozieslki A, Harris N, Vardeman D, Liehr J, Stehlin J and
Giovanella B: Structure-activity relationship of alkyl camptothecin
esters. Annu N Y Acad Sci. 992:122–135. 2000.
|
|
15
|
Liu X, Wang Y, Vardeman D, Cao Z and
Giovanella B: Development and validation of a reverse-phase HPLC
with fluorescence detector method for simultaneous determination of
CZ48 and its active metabolite camptothecin in mouse plasma. J
Chromatogr B Analyt Technol Biomed Life Sci. 867:84–89. 2008.
View Article : Google Scholar
|