Introduction
Idiopathic nephrotic syndrome is one of the most
common kidney diseases found in children, with a prevalence of 1–3
out of 100,000 in those younger than 16 years of age (1). Clinically, nephrotic syndrome is
characterized by massive proteinuria, edema, hypoalbuminemia and
hyperlipidemia with heterogeneous histological features ranging
from minimal change disease (MCD), focal segmental
glomerulosclerosis and mesangial proliferative glomerulonephritis
(MPGN) (1). MCD is the most common
pathological form of pediatric nephrotic syndrome (1). The epidemiological data from the
International Study of Kidney Disease in Children (ISKDC) have
shown that approximately 93% of patients with MCD respond well to
steroids, with a relapse rate as high as 80–90% (2). Long-term steroid treatment and
immunosuppressants, such as calcineurin inhibitors, have been
successful in the management of these patients but result in severe
side effects (3).
Monoclonal antibody, rituximab (RTX), is a 35-kDa
protein highly expressed in B-lymphocytes from early to late B-cell
stages, that inhibits CD20-mediated B-cell proliferation and
differentiation (4). RTX was first
introduced for the treatment of B-cell non-Hodgkin's lymphoma and
was subsequently given to patients with autoimmune diseases, such
as rheumatoid arthritis, lupus erythematosus, and immune
complex-mediated glomerulonephritis (5). Previous findings showed that RTX is
effective in maintaining remission in refractory steroid-dependent
cases (6–10). Increased successful inductions of
remission following RTX treatment in steroid-dependent nephrotic
syndrome (SDNS) or frequently relapsing nehprotic syndrome (FRNS)
than in steroid-resistant nephrotic syndrome were previously
reported (6–7). Anecdotal studies have indicated the
induction of RTX in MCD is superior to focal segmental
glomerulosclerosis (8). In addition,
it was recently demonstrated that the median relapse-free period
was longer in the RTX group than the placebo group in patients with
SDNS or FRNS (9). However, the
patients received RTX four times, and 42% of the patients had
severe side effects (9). Thus, whether
a single dose of RTX for the patients with SDNS or
steroid-resistance nephrotic syndrome with fewer side effects of
RTX remained to be determined. Fujinaga et al showed that a
single dose of RTX allowed for a decreased dose of steroids in the
management of MCD (10).
The aim of the present study was to investigate the
efficiency and side effects of a single dose of RTX in the
management of pediatric SDNS with a histological diagnosis of
MCD.
Materials and methods
Subjects
Patients with SDNS who were hospitalized in the
Children's Hospital of Jiaotong University from October 2011 to
December 2015 were enrolled in the study. The patients were
diagnosed with SDNS according to the Kidney Disease Improving
Global Outcomes (KDIGO) guidelines and ISKDC criteria with
biopsy-proven MCD (11,12). All the patients had frequent relapses
despite combined therapy with steroids and mycophenolate mofetil,
cyclophosphamide or calcineurin inhibitors, tacrolimus (FK506) and
cyclosporine A (CsA).
The patients with SDNS were excluded if they: i) Had
no renal biopsy; ii) had renal biopsy but the pathology of renal
was not MCD; and iii) were not administered with other
immunosuppressants.
Diagnoses and outcomes of all the patients were
according to the KDIGO guidelines and criteria (11): i) For nephrotic syndrome: Edema, urine
protein/creatinine ratio (uPCR) ≥2,000 mg/g (≥300 mg/mmol), or ≥300
mg/dl, or 3+ protein on the urine dipstick,
hypoalbuminaemia ≤2.5 g/dl; ii) complete remission: uPCR <200
mg/g (<20 mg/mmol) or <1+ of protein on the urine
dipstick for 3 consecutive days; iii) partial remission:
proteinuria reduction of ≥50% from the presenting value and
absolute uPCR between 200 and 2,000 mg/g (20–200 mg/mmol); iv) no
remission: failure to reduce urine protein excretion by 50% from
baseline or persistent excretion uPCR 2,000 mg/g (200 mg/mmol); and
v) relapse: uPCR ≥2,000 mg/g (≥200 mg/mmol) or ≥3+
protein on urine dipstick for 3 consecutive days.
Patients with SDNS were defined as having at least
two relapses during steroid tapering every other day or within 14
days after discontinuation according to the ISKDC criteria
(12). RTX induced B-lymphocyte
depletion was defined as a CD20 count <0.5% of the total
lymphocyte count (9,13). RTX efficiency was defined as complete
remission lasting for ≥6 months after RTX until the end of
follow-up without the administration of steroids or other
immunosuppressants.
Protocol
A written consent form was obtained from all the
patients for the indication and side effects of RTX. A single dose
of RTX (375 mg/m2) was administered. Phenergan and
dexamethasone were administered as premedications prior to
infusion. After RTX, the patients only taking steroids decreased
rapidly after complete remission was achieved, and use of steroids
ceased in 1–3 months depending on the various doses of
steroids.
Follow-up
Follow-up on patients occurred at day 3, week 2,
months 1, 3 and 6, and one year after RTX infusion. Laboratory data
including complete blood count, urinalysis, liver and kidney
function tests, immunoglobulin levels, and lymphocyte fractionation
were obtained prior to RTX infusion and at follow-up visits. All
the patients were followed-up for ≥6 months.
Methods
Blood was collected in an ETDA-anticoagulation tube
and tested immediately for CD20 (CD20 L27 and CD45-PC5; BD
Biosciences, Franklin Lakes, NJ, USA) and lymphocyte fractionation
(BD Multitest™ IMK kit; BD Biosciences) using a flow cytometer (BD
FACSCalibur™; BD Biosciences). The blood count was tested using an
automatic blood cell analyzer. Urinalysis was tested using a urine
analyzer. Liver and kidney function tests were tested using a
automatic biochemistry analyzer. Immunoglobulin was tested using
the immune transmission turbidity method.
Statistical analysis
Data were presented as mean ± standard deviation.
Comparisons before and following RTX infusion were carried out
using the Wilcoxon matched-pairs signed rank test. The correlation
between different indices was studied using the Spearman's
correlation analysis. P<0.05 was considered statistically
significant.
Results
Patient characteristics
Nineteen patients (10 males and 9 females) with MCD,
with an age range of 5–17 years (mean, 8.21±2.99 years) were
included in the present study. The maintenance dose of steroids was
6.82–32.1 mg/m2/day (13.7±7.12 mg/m2/day).
The course of disease prior to RTX was 18–108 months (52.7±27.1
months). The patient follow-up was 1–50 months (28.1±16.6 months).
Pretreatment features of the patients are listed in Table I. Prior to RTX infusion, all the
patients relapsed more than two times with steroids and
immunosupressants.
 | Table I.Clinical characteristics of patients
prior to RTX infusion. |
Table I.
Clinical characteristics of patients
prior to RTX infusion.
| Patient no. | Gender | Age (years) | Years since
onset | Immunosuppressant
before RTX | Relapse
rate/year | Prednisone dose
before RTX infusion (mg/kg/day) | Dependent-steroid
dosage (mg/m2/day) | Urine protein |
|---|
| 1 | F | 6 | 4 | MMF, CTX, FK506 | 3 | 0.5 | 17.5 | +++ |
| 2 | M | 6 | 4.5 | MMF, CTX | 4 | 0.4 | 10.5 | − |
| 3 | F | 11 | 8 | MMF, CTX, FK506 | 5 | 1.0 | 30.0 | ++ |
| 4 | M | 17 | 6 | MMF, CTX, CsA | 4 | 0.28 | 9.55 | ++ |
| 5 | M | 11 | 9 | MMF, CTX | 3 | 0.22 | 7.4 | +++ |
| 6 | F | 8 | 3 | CsA | 3 | 0.23 | 8.45 | + |
| 7 | M | 9 | 7.5 | CsA | 2 | 0.43 | 13.5 | +++ |
| 8 | F | 9 | 6 | MMF | 2 | 0.22 | 6.82 | +++ |
| 9 | M | 8 | 3.5 | CsA | 3 | 0.35 | 12.1 | +++ |
| 10 | F | 7 | 5 | MMF, CTX, FK506 | 4 | 0.77 | 14.4 | +++ |
| 11 | F | 6 | 3 | CsA | 2 |
0.78 | 8.89 | ++ |
| 12 | M | 5 | 4 | MMF, CsA | 3 |
0.69 | 8.77 | +++ |
| 13 | M | 5 | 2 | FK506 | 2 |
1.25 | 32.1 | +++ |
| 14 | M | 7 | 4 | CsA | 2 |
0.35 | 11.5 | − |
| 15 | M | 10 | 2.5 | MMF, FK506 | 4 |
0.41 | 18.2 | +++ |
| 16 | F | 6.5 | 5 | FK506, MMF | 3 |
1.46 | 9.15 | ++ |
| 17 | F | 6.5 | 4.5 | MMF, CsA | 3 |
0.57 | 13.0 | +++ |
| 18 | F | 6 | 2.5 | CTX | 2 |
0.78 | 19.5 | ++ |
| 19 | M | 12 | 10 | FK506 | 2 |
0.39 | 8.93 | − |
RTX infusion
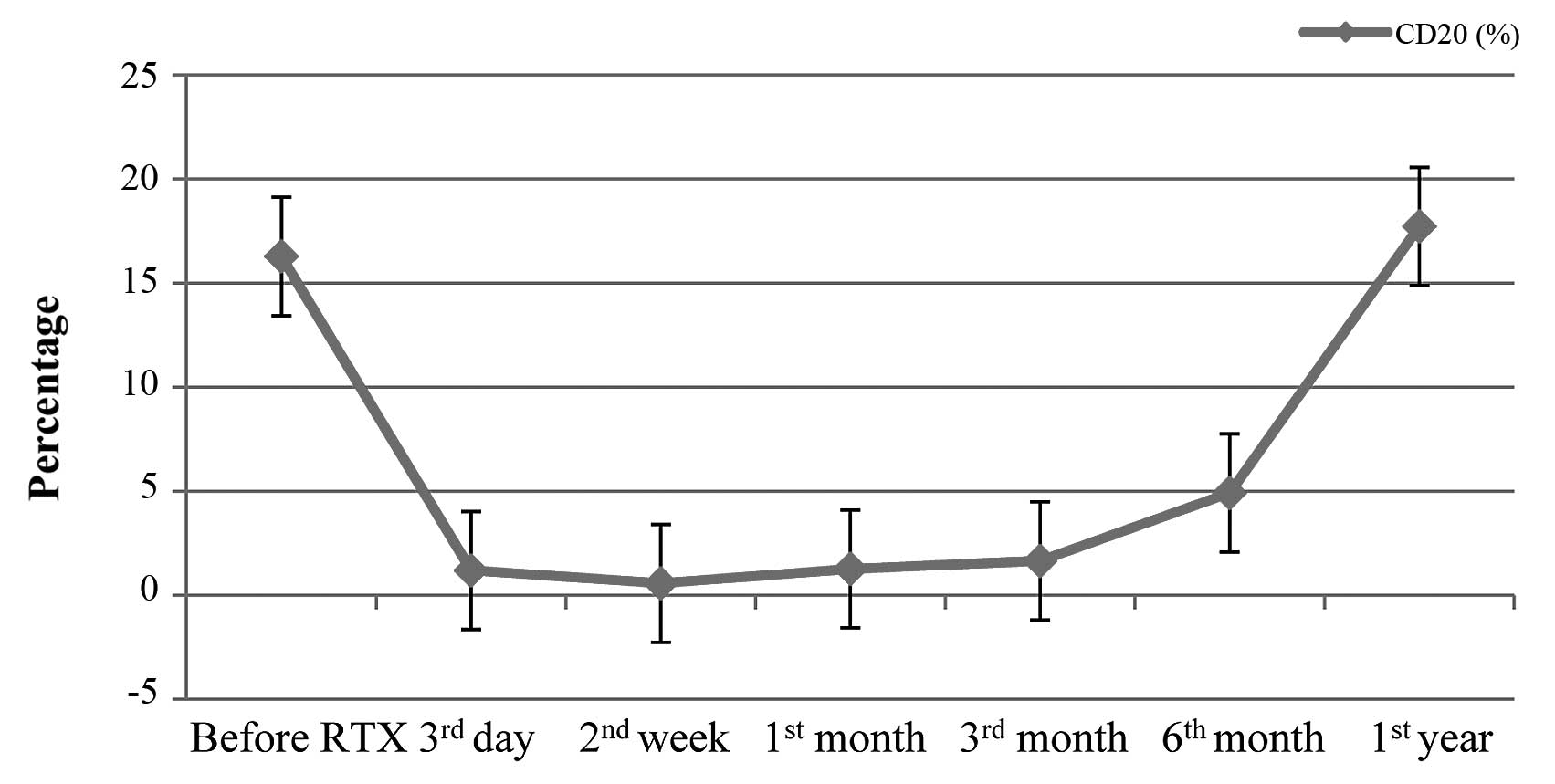
Following RTX infusion, B-cell depletion
(CD20<0.5%) was achieved and lasted for 1–6 months (mean,
2.92±1.57 months). The variability of B-cells is shown in Fig. 1. To the end of follow-up, 10 patients
remained in complete remission and did not relapse following a
single dose of RTX infusion for 4–50 months (mean, 30.1±12.6
months) despite recovery of the B-cell (CD20) count. During steroid
tapering, 9 patients relapsed within 2–21 months (mean, 7.67±6.26
months) of RTX infusion, and 4 of them relapsed after steroids were
ceased for several months. However, the maintenance dose of
steroids and relapse rate within 12 months were less than those
prior to RTX treatment (T=0, P<0.05; T=95, P<0.05). No
correlation between relapses and B-cell count (r=−0.194, P>0.05)
was observed. Details of the 9 patients who relapsed are shown in
Table II.
 | Table II.Patient relapse following RTX
infusion. |
Table II.
Patient relapse following RTX
infusion.
|
| Patient no. |
|---|
|
|
|
|---|
| Relapse | 2 | 3 | 4 | 5 | 7 | 9 | 10 | 15 | 16 |
|---|
| Relapse-free
duration (months) | 2 | 4 | 10 | 21 | 5 | 3 | 14 | 6 | 4 |
| Cause of
relapse | Infection | No | Infection | No | No | No | Infection | No | Infection |
| Steroid dosage on
relapse (mg/kg/day) | 5.26 | 1.88 | 3.05 | No | No | 2.02 | No | No | 0.13 |
| Time to second
relapse (months) | 19 | 19 | 20 | – | – | 9 | – | – | – |
| Cause of
relapse | No | No | Rabies vaccine | – | – | Infection | – | – | – |
| Steroid dosage on
relapse (mg/kg/day) | 3.95 | 7.52 | No | – | – | 4.05 | – | – | – |
The efficacy of a single induction of RTX was 47.4%
(9/19). Details of patients after RTX are presented in Table III.
 | Table III.Clinical characeristics of patients
following RTX infusion. |
Table III.
Clinical characeristics of patients
following RTX infusion.
| Patient no. | RTX dose | Prednisone dose
(mg/kg/day) | Dependent-steroid
dose (mg/m2/day) | Urine protein | Relapse
rate/year | Follow-up
(months) | Drugs used now
(mg/kg/day) |
|---|
| 1 | 1 | 0 | – | – | 0 | 50 | No |
| 2 | 1 | 0.08 | 3.95 | – | 2 | 50 | Pred 0.08 |
| 3 | 1 | 0.1 | 1.88 | – | 2 | 47 | Pred 0.1 |
| 4 | 1 | 0.09 | 3.05 | – | 2 | 45 | Pred 0.17 |
| 5 | 1 | 0 | – | – | 1 | 41 | No |
| 6 | 1 | 0 | – | – | 0 | 40 | No |
| 7 | 1 | 0 | 13.5 | – | 1 | 37 | Pred 0.5, MMF |
| 8 | 1 | 0 | – | – | 0 | 34 | No |
| 9 | 1 | 0.05 | 2.02 | – | 2 | 34 | Pred 0.02 |
| 10 | 1 | 0 | – | – | 1 | 31 | No |
| 11 | 1 | 0 | – | – | 0 | 30 | No |
| 12 | 1 | 0 | – | – | 0 | 25 | No |
| 13 | 1 | 0 | – | – | 0 | 24 | No |
| 14 | 1 | 0 | – | – | 0 | 22 | No |
| 15 | 1 | 0.29 | 4.46 | – | 1 | 10 | Pred 0.4 |
| 16 | 1 | 0.13 | 1.41 | – | 1 | 7 | Pred 1.04 |
| 17 | 1 | 0 | – | – | 0 | 4 | No |
| 18 | 1 | 0.20 | – | – | 0 | 1 | Pred 0.2 |
| 19 | 1 | 0.20 | – | – | 0 | 1 | Pred 0.2 |
Side effects of RTX
One patient developed a rash during the infusion of
RTX with transient tachycardia (130 bpm) and tachypnea (35
times/min). The patient had no fever or anaphylaxis and the rash
improved by retarding the infusion. Another patient complained of
transient dizziness during the infusion. The remaining patients
tolerated the infusion well without complications.
Studies available on RTX in children
with SDNS or FRNS
There are several studies with regard to RTX
administration in children (6,7,9,10,13–20). The information is presented in Table IV. The noted cases of
steroid-sensitive nephrotic syndrome relapsed more than two times
so were classified as SDNS.
 | Table IV.Studies available on RTX
administration in children with SDNS or FRNS. |
Table IV.
Studies available on RTX
administration in children with SDNS or FRNS.
| Study (author,
year) | No. of cases | Time of RTX | Follow-up | Co-drugs | Efficiency (%) | Refs. |
|---|
| Iijima et
al, 2014 | 24 | 375
mg/m2/time | 53 weeks | Pred | 14/24 (58.3) | (9) |
|
| FRNS/SDNS | Once a week for 4
week |
|
|
|
|
| Sun et al,
2014 | 9 for SSNS | 375
mg/m2/time | 4–16 months | Pred | 5/9 (55.6) | (14) |
|
| 2 for SRNS | One or two
times |
|
|
|
|
| Ravani et
al, 2013 | 46 | 375
mg/m2/time | >3 years | Pred and CsA | 22/46 (48.0) | (6) |
|
| SDNS | One to five
times |
|
|
|
|
| Ito et al,
2013 | 53 | 375
mg/m2/time | 8–51 months | Pred or CsA | 41/53 (77.0) | (15) |
|
| SDNS/FRNS | One or more
times |
|
|
|
|
| Tellier et
al, 2013 | 18 | 375
mg/m2/time | Median 3.2
years | Pred | 8/18 (44.5) | (16) |
|
| SDNS | One to four
times |
|
|
|
|
| Sellier-Leclerc
et al, 2012 | 30 | 375
mg/m2/time | >24 months | Pred or MMF or
CsA | 19/30 (63.3) | (17) |
|
| SDNS | One to four
times |
|
|
|
|
| Kemper et
al, 2012 | 29/37 | 375
mg/m2/time | >24 months | Pred or CI or
MPI | 7/29 (24.1) | (18) |
|
| SDNS | Once a week for 1
to 4 weeks |
|
|
|
|
| Ravani et
al, 2011 | 54 | 375
mg/m2/time | >9 months | Pred and CsA | 27/54 (50.0) | (19) |
|
| SDNS | One or two
times |
|
|
|
|
| Fujinaga et
al, 2010 | 10 | 375
mg/m2/time | 17 months | CsA | 5/10 (50.0 | (10) |
|
| MCD, SDNS | One time |
|
|
|
|
| Sellier-Leclerc
et al, 2010 | 22 | 375
mg/m2/time | 3.2–28.5
months | Pred or CsA | 9/22 (41.0) | (20) |
|
| SDNS | One or two
times |
|
|
|
|
| Prytuła et
al, 2010 | 28 | 375
mg/m2/time | 1–16 months | Pred | 10/28 (35.7) | (13) |
|
| SDNS/FRNS | One to five
times |
|
|
|
|
| Kamei et al,
2009 | 12 | 375
mg/m2/time | >12 months | Pred | 3/12 (25.0) | (7) |
|
| SDNS | One time |
|
|
|
|
Discussion
Steroid dependency poses a challenge in the
treatment of MCD in pediatric nephrotic syndrome. Long-term use and
a high dose of steroids and immunosuppressants cause severe side
effects (3). Thus, oral agent-free
treatment is an attractive therapeutic approach for patients with
nephrotic syndrome (21).
The role of B lymphocytes in the pathogenesis of
MCD, a T-cell, has been previously considered to play a key role
(22–26). Previous findings have shown that CD23
increased in patients with nephrotic syndrome (22,23).
Interleukin-13, a cytokine associated with type 2 T-helper cells,
leads to antibody production and allergic reactions caused by
immune responses involving B-cells in patients with nephrotic
syndrome (24,25). B-cells are essential in CD4 T-cell
activation for antigen presenting and providing co-stimulation
signals (26). Tokunaga et al
(4) showed that RTX decreases CD40-
and CD80-expressing cells in activated B-cells in patients with
systemic lupus erythematosus and downregulates CD40L and CD69 on
CD4-positive cells. The successful induction of remission following
RTX treatment in some cases of MCD provides evidence supporting the
hypothesis that the interference of B- and T-cells is a potential
strategy for MCD treatment (8,10).
In our clinical study, patients receiving RTX
treatment presented an outcome of fewer relapses, longer
relapse-free periods and reduced maintenance doses of steroids.
Patients remained relapse-free despite the recovery of B-cell
count, indicating that RTX treatment may regulate the immunity of
patients and induce remission by inhibiting B-cells. B-cell
depletion by RTX may block T-cell activation induced by B-cells or
B-cell-derived factors resulting in a change of T-cell cytokine
production responsible for the development of idiopathic nephrotic
syndrome. B-cell depletion by RTX may also change regulatory T-cell
functions, inducing changes in T- and B-cell cytokine production
(18). Further studies are needed to
confirm this hypothesis.
One to four doses of RTX were used to achieve and
maintain complete remission in children with SDNS or FRNS in most
clinical studies (6,7,9,10,13–20). The actual regimen of RTX may require
adjustment in an individualized manner, depending on the response
and actual clinical condition. In the present study, RTX efficacy
on MCD in children with SDNS was 47.4% (9/19). Other studies showed
efficacy to be between 25 and 77% with several doses of RTX. This
result shows that a single dose of RTX can achieve a comparable
outcome in the management of pediatric MCD, with a favorable
cost-benefit ratio and a decrease in side effects.
In conclusion, RTX is a safe and effective
alternative for MCD forms of SDNS in children. For patients with
severe side effects from steroid treatment or those who cannot
tolerate immunosuppressants, RTX is an effective treatment in the
rapid induction of remission, reducing relapses and steroid
dependence. We recommend a single dose of RTX for children with
steroid-dependent minimal change nephrotic syndrome for the rapid
induction of remission, reduction on long-term steroid dose and
decreasing the number of relapses, and having fewer side effects.
Larger samples of randomized controlled clinical trials are
required to justify the definite efficacy and safety of RTX in this
group of patients.
Acknowledgements
The present study was supported by the Chinese
National Natural Science Foundation (grant no. 81370813 awarded to
Professor Wen-Yan Huang) and the Program for Outstanding Subject
Leaders of Shanghai Health Bureau Foundation (grant no. XBR 2011010
awarded to Professor Wen-Yan Huang).
References
|
1
|
McKinney PA, Feltbower RG, Brocklebank JT
and Fitzpatrick MM: Time trends and ethnic patterns of childhood
nephrotic syndrome in Yorkshire, UK. Pediatr Nephrol. 16:1040–1044.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tarshish P, Tobin JN, Bernstein J and
Edelmann CM Jr: Prognostic significance of the early course of
minimal change nephrotic syndrome: report of the International
Study of Kidney Disease in Children. J Am Soc Nephrol. 8:769–776.
1997.PubMed/NCBI
|
|
3
|
Hodson EM, Willis NS and Craig JC:
Non-corticosteroid treatment for nephrotic syndrome in children.
Cochrane Database Syst Rev. 1:CD0022902008.PubMed/NCBI
|
|
4
|
Tokunaga M, Saito K, Kawabata D, Imura Y,
Fujii T, Nakayamada S, Tsujimura S, Nawata M, Iwata S, Azuma T, et
al: Efficacy of rituximab (anti-CD20) for refractory systemic lupus
erythematosus involving the central nervous system. Ann Rheum Dis.
66:470–475. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Iijima K: Rituximab for childhood
refractory nephrotic syndrome. Pediatr Int. 53:617–621. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ravani P, Ponticelli A, Siciliano C,
Fornoni A, Magnasco A, Sica F, Bodria M, Caridi G, Wei C,
Belingheri M, et al: Rituximab is a safe and effective long-term
treatment for children with steroid and calcineurin
inhibitor-dependent idiopathic nephrotic syndrome. Kidney Int.
84:1025–1033. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kamei K, Ito S, Nozu K, Fujinaga S,
Nakayama M, Sako M, Saito M, Yoneko M and Iijima K: Single dose of
rituximab for refractory steroid-dependent nephrotic syndrome in
children. Pediatr Nephrol. 24:1321–1328. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gulati A, Sinha A, Jordan SC, Hari P,
Dinda AK, Sharma S, Srivastava RN, Moudgil A and Bagga A: Efficacy
and safety of treatment with rituximab for difficult
steroid-resistant and -dependent nephrotic syndrome: Multicentric
report. Clin J Am Soc Nephrol. 5:2207–2212. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Iijima K, Sako M, Nozu K, Mori R, Tuchida
N, Kamei K, Miura K, Aya K, Nakanishi K, Ohtomo Y, et al: Rituximab
for Childhood-onset Refractory Nephrotic Syndrome (RCRNS) Study
Group: Rituximab for childhood-onset, complicated, frequently
relapsing nephrotic syndrome or steroid-dependent nephrotic
syndrome: a multicentre, double-blind, randomised,
placebo-controlled trial. Lancet. 384:1273–1281. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fujinaga S, Hirano D, Nishizaki N, Kamei
K, Ito S, Ohtomo Y, Shimizu T and Kaneko K: Single infusion of
rituximab for persistent steroid-dependent minimal-change nephrotic
syndrome after long-term cyclosporine. Pediatr Nephrol. 25:539–544.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kidney Disease Improving Global Outcomes:
KDIGO Clinical Practice Guideline for Glomerulonephritis. Kidney
Int Suppl. 2:139–274. 2012.
|
|
12
|
Schulman SL, Kaiser BA, Polinsky MS,
Srinivasan R and Baluarte HJ: Predicting the response to cytotoxic
therapy for childhood nephrotic syndrome: superiority of response
to corticosteroid therapy over histopathologic patterns. J Pediatr.
113:996–1001. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Prytuła A, Iijima K, Kamei K, Geary D,
Gottlich E, Majeed A, Taylor M, Marks SD, Tuchman S, Camilla R, et
al: Rituximab in refractory nephrotic syndrome. Pediatr Nephrol.
25:461–468. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun L, Xu H, Shen Q, Cao Q, Rao J, Liu HM,
Fang XY and Zhou LJ: Efficacy of rituximab therapy in children with
refractory nephrotic syndrome: a prospective observational study in
Shanghai. World J Pediatr. 10:59–63. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ito S, Kamei K, Ogura M, Udagawa T,
Fujinaga S, Saito M, Sako M and Iijima K: Survey of rituximab
treatment for childhood-onset refractory nephrotic syndrome.
Pediatr Nephrol. 28:257–264. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tellier S, Brochard K, Garnier A, Bandin
F, Llanas B, Guigonis V, Cailliez M, Pietrement C, Dunand O,
Nathanson S, et al: Long-term outcome of children treated with
rituximab for idiopathic nephrotic syndrome. Pediatr Nephrol.
28:911–918. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sellier-Leclerc AL, Baudouin V, Kwon T,
Macher MA, Guérin V, Lapillonne H, Deschênes G and Ulinski T:
Rituximab in steroid-dependent idiopathic nephrotic syndrome in
childhood - follow-up after CD19 recovery. Nephrol Dial Transplant.
27:1083–1089. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kemper MJ, Gellermann J, Habbig S, Krmar
RT, Dittrich K, Jungraithmayr T, Pape L, Patzer L, Billing H, Weber
L, et al: Long-term follow-up after rituximab for steroid-dependent
idiopathic nephrotic syndrome. Nephrol Dial Transplant.
27:1910–1915. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ravani P, Magnasco A, Edefonti A, Murer L,
Rossi R, Ghio L, Benetti E, Scozzola F, Pasini A, Dallera N, et al:
Short-term effects of rituximab in children with steroid- and
calcineurin-dependent nephrotic syndrome: a randomized controlled
trial. Clin J Am Soc Nephrol. 6:1308–1315. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sellier-Leclerc AL, Macher MA, Loirat C,
Guérin V, Watier H, Peuchmaur M, Baudouin V and Deschênes G:
Rituximab efficiency in children with steroid-dependent nephrotic
syndrome. Pediatr Nephrol. 25:1109–1115. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
El-Reshaid K, Sallam HT, Hakim AA,
Al-Attiyah R and EI-Reshaid K: Rituximab in treatment of idiopathic
glomerulopathy. Saudi J Kidney Dis Transpl. 23:973–978. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Samarghitean C, Väliaho J and Vihinen M:
Online registry of genetic and clinical immunodeficiency diagnostic
laboratories, IDdiagnostics. J Clin Immunol. 24:53–61. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kyrieleis HA, Löwik MM, Pronk I, Cruysberg
HR, Kremer JA, Oyen WJ, van den Heuvel BL, Wetzels JF and
Levtchenko EN: Long-term outcome of biopsy-proven, frequently
relapsing minimal-change nephrotic syndrome in children. Clin J Am
Soc Nephrol. 4:1593–1600. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kemper MJ, Meyer-Jark T, Lilova M and
Müller-Wiefel DE: Combined T- and B-cell activation in childhood
steroid-sensitive nephrotic syndrome. Clin Nephrol. 60:242–247.
2003. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cho BS, Yoon SR, Jang JY, Pyun KH and Lee
CE: Up-regulation of interleukin-4 and CD23/FcepsilonRII in minimal
change nephrotic syndrome. Pediatr Nephrol. 13:199–204. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kimata H, Fujimoto M and Furusho K:
Involvement of interleukin (IL)-13, but not IL-4, in spontaneous
IgE and IgG4 production in nephrotic syndrome. Eur J Immunol.
25:1497–1501. 1995. View Article : Google Scholar : PubMed/NCBI
|