Introduction
It is well known that ascorbic acid (AsA) has
anticancer effects due to its antioxidant activity, and the results
of epidemiological surveys have demonstrated that AsA intake is
inversely correlated with carcinogenicity (1). Almost 30 years ago, the study by Cameron
and Pauling was the first to demonstrate the antitumor effect of
AsA and this finding was applied to a clinical setting (2). A few years earlier, the effectiveness of
combining cancer treatment with AsA was reported, but the
anticancer activity of AsA was not confirmed in clinical tests at
the Mayo Clinic (3).
Recent studies have shown that high plasma
concentrations of AsA can only be achieved when AsA is administered
intravenously or intraperitoneally as the rate of absorption from
the gut is limited with oral administration (4). The plasma concentrations of AsA in humans
following high-dose intravenous AsA is ~200 times higher in
comparison to oral administration (5).
Therefore, we hypothesized that the therapeutic use of AsA in
combination with radiation may reduce therapy-related side effects
and increase the antitumor effects (6). It has been reported that pharmacological
ascorbate sensitized cells to the effects of ionizing radiation
(7,8).
Ionizing radiation is known to damage numerous cell
organelles through the production of free radicals (9). The hydroxyl (OH) radical (·OH) is
considered to have a major role in radiation-induced cell damage
and induces apoptotic cell death through activation of a
pro-apoptotic pathway (10). However,
AsA is a known scavenger of reactive radical species, and it is
possible that AsA may reduce the anticancer effect of radiation.
The aim of the present study was to examine the association between
the scavenged activity of AsA and the biological anticancer effect
of OH radicals generated by X-ray irradiation.
Materials and methods
Cell culture, extracellular AsA
treatment and X-ray irradiation
Human promyelocytic leukemia HL60 cells were
purchased from the RIKEN Bioresource Center (Tsukuba, Japan). The
cells were grown in 100-mm culture dishes containing Roswell Park
Memorial Institute (RPMI)-1640 medium (Life Technologies; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10%
fetal bovine serum at 37°C in a 5% CO2 atmosphere. The
cells were passaged every third day, and the density in the culture
was ≤1×106 cells/ml. The cell concentration was adjusted
to 5×105 cells/ml prior to AsA treatment and X-ray
irradiation. The AsA solution was titrated with NaOH at pH 7.4.
HL60 (5×105 cells/ml) in 50-mm culture dishes were
irradiated 1 h after adjusting each experimental AsA dose. The
radiation was administered with Sofron BST 1505 CX (Sofron X-ray
Industry Corporation, Ltd., Tokyo, Japan) under the following
conditions: Tube voltage, 80 kVp (effective 62.3 kV); tube current,
5 mA; focus surface distance, 30 cm; dose rate, 16 Gy/h; filter,
1-mm aluminum; and temperature, room temperature.
Electron spin resonance (ESR)
measurement
OH radicals generated from irradiated
phosphate-buffered saline (PBS) were measured by ESR (JES-FR80;
JEOL, Tokyo, Japan). The spin-trap technique was used with
5,5-dimethyl-1-pyrroline-N-oxide (DMPO) (Labotec Corp., Ltd.,
Tokyo, Japan). The PBS sample (200 µl) contained 150 µl of PBS with
or without AsA, 35 µl of diethylenetriaminepentaacetic acid
(Labotec Corp. Ltd.) and 15 µl of DMPO. Radicals generated in this
sample by X-ray irradiation under the aforementioned condition were
measured within 1 min of irradiation. The ESR spectrometer
(JES-FR80) was used under the following conditions: Frequency,
9.475 GHz; power, 4 mW; center field, 334.5 mT; sweep width, ±10.0
mT; modulation width, 5×0.1 m; receiver gain, 0.4×100; time
constant, 0.1 sec; sweep time, 2.0 min; and temperature, room
temperature. The relative signal intensity (RSI) of OH radicals was
defined as the ratio of the peak height of OH radicals and a
manganese oxide standard marker on the ESR spectra. Each sample was
measured three to five times and the results are shown as average
values of RSI.
Cell viability assay and quantitative
detection of apoptosis
The cells were stained with 2% trypan blue for 1 min
and counted per 1 mm3 in each experiment to estimate the
number of unstained cells within 2 min. The number of unstained
cells was considered to be the number of viable cells.
Apoptosis was detected using a cellular DNA
fragmentation enzyme-linked immunosorbent assay kit (Roche
Diagnostics, Basel, Switzerland). The cells were labeled with
bromodeoxyuridine (BrdU) (Roche Diagnostics) on the day prior to
treatment. Following AsA treatment and/or 4 Gy X-ray irradiation,
the cells were cultured for 6 h. After this incubation,
1.5×106 cells were collected and treated with a cell
solution (0.05% Triton-X and 650 µM EDTA) (Research Organics,
Cleveland, OH, USA) at 4°C for 30 min. The cells were subsequently
centrifuged at 4°C and 12,000 × g for 30 min at 4°C, and 160 µl of
the supernatant was used as a sample. This sample was plated on a
microtiter plate coated with an anti-BrdU antibody (Roche
Diagnostics), and color development caused by the
tetramethylbenzidine (Roche Diagnostics) substrate reaction was
measured using a spectrophotometer (450 nm).
Analysis of intracellular AsA
concentration
The OxiSelect™Ascorbic Acid Assay kit (FRASC) was
purchased from Cell Biolabs, Inc. (San Diego, CA, USA). Cells
(1.0×106 cells/ml) were lysed by multiple freeze-thaw
cycles in four volumes of cold 1X assay buffer. The samples and AsA
standard were added to 96-well plate wells. Deionized water (−AO)
or diluted 1X ascorbate oxidase (+AO) was added to each well. The
reaction reagent was added to all the wells, and the absorbance of
each well was measured at a wavelength of 540 nm using a microplate
reader (10043; Bio Rad, Tokyo, Japan). The AsA concentration was
calculated from a standard curve created from the measured
absorbance.
Statistical analysis
All the data are expressed as the mean ± standard
deviation of ≥3 independent experiments. Statistical comparisons
between groups were performed by an analysis of variance and
Student's t-test, where P<0.05 was considered to indicate a
statistically significant difference.
Results
ESR spectra
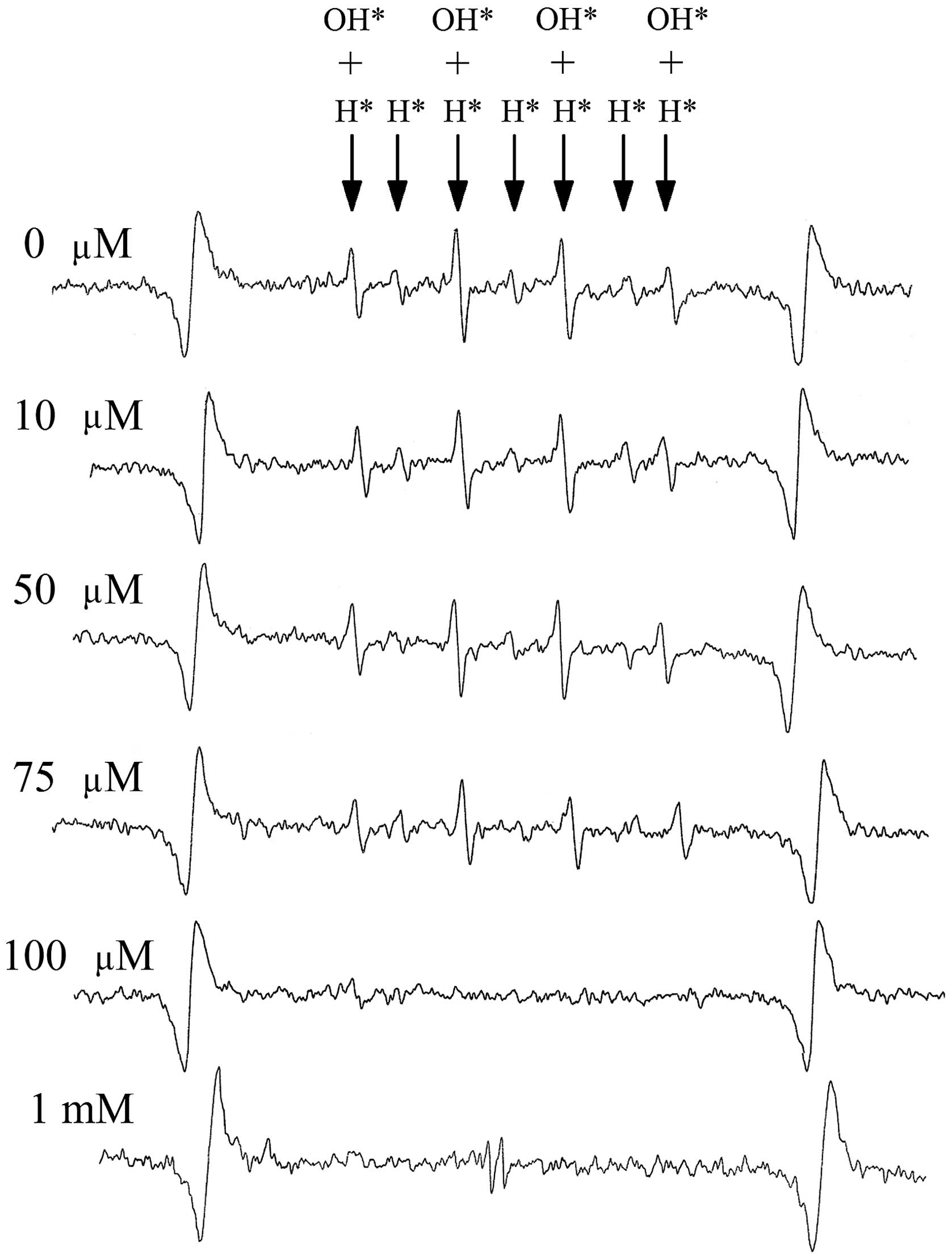
The ESR spectra of PBS following irradiating with a
4 Gy X-ray are shown in Fig. 1. The
spectra without AsA treatment shows the typical wave type of
radicals generated by X-ray irradiation. There were two types,
DMPO-OH (αN = αH = 1.49 mT) and DMPO-H (αN = 1.64, αH = 2.25 mT).
However, there were unclear waveforms for the OH and H radicals in
PBS with 100 µM (0.1 mM) AsA. With 1 mM AsA, the ascorbate radical
in the middle of the ESR spectrum of the OH and H radicals
disappeared.
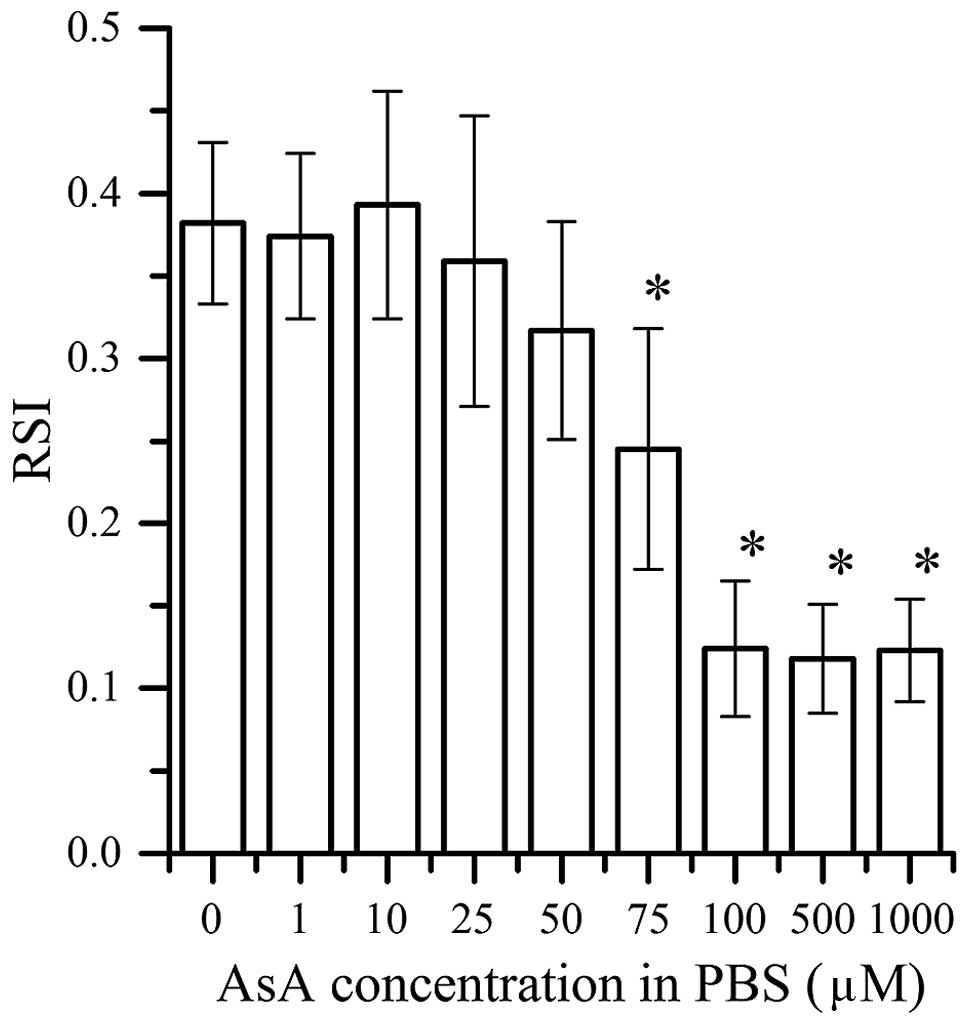
RSIs of OH radicals
Fig. 2 shows the RSIs
of the OH radicals generated from the 4 Gy X-ray irradiation with
various concentrations of AsA in PBS. The RSIs of the OH radicals
were similar for the AsA values <50 µM AsA, suggesting that OH
radicals were not scavenged at concentrations <50 µM AsA. The
RSIs of the OH radicals decreased with 75 µM AsA with similarly low
RSIs at concentrations of 100 µM (0.1 mM) AsA.
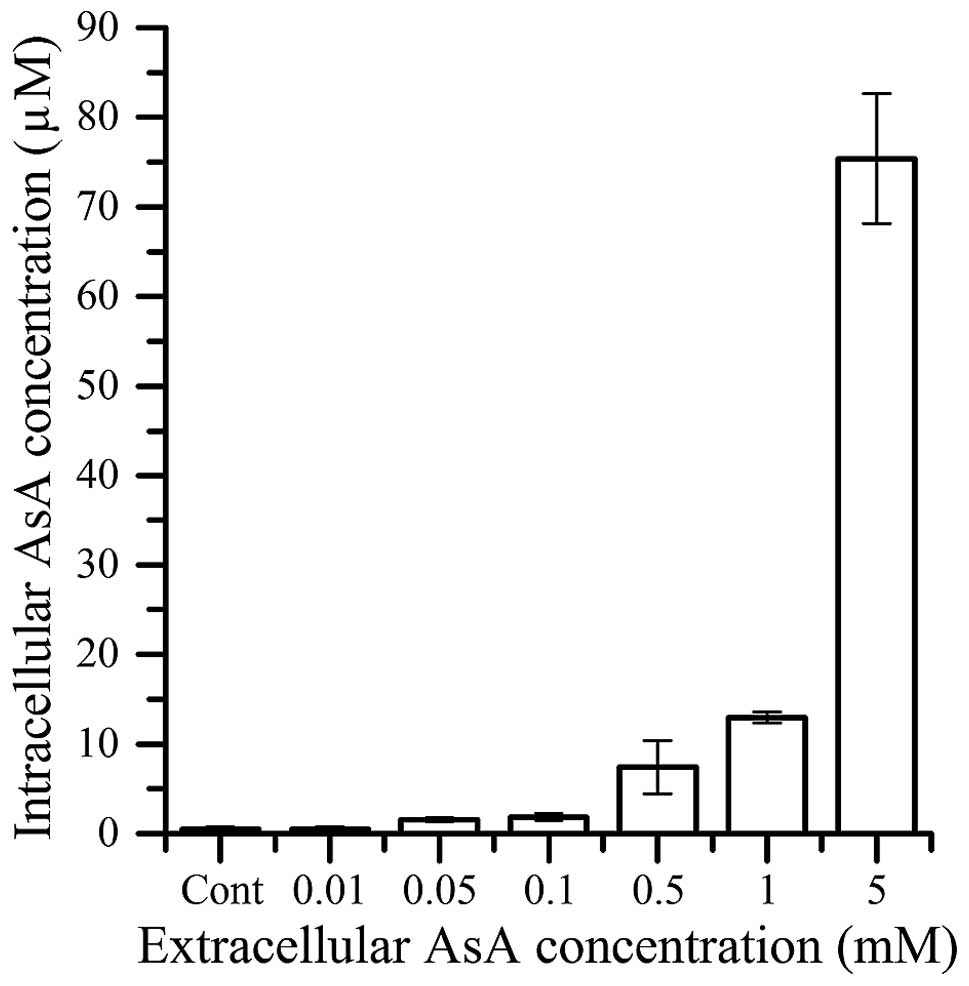
Intracellular AsA concentration
The intracellular AsA concentrations in HL60 cells 1
h after extracellular AsA treatment are shown in Fig. 3. The intracellular concentration of AsA
was <20 µM after treatment with 1 mM AsA. However, the
intracellular AsA concentration was 75 µM after the 5 mM AsA
extracellular treatment.
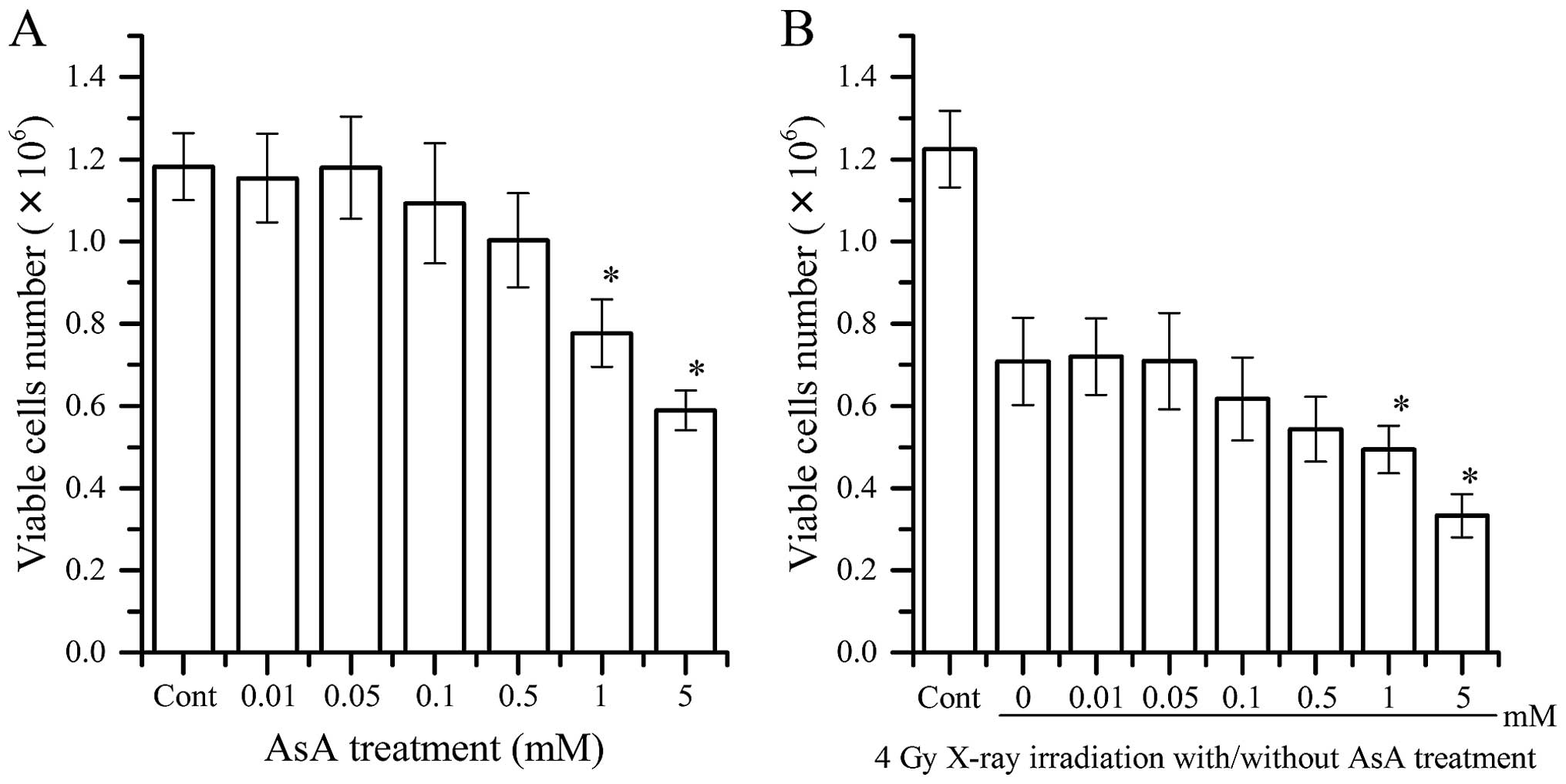
HL60 cell viability
Fig. 4A shows the cell
viability of HL60 cells 24 h after AsA treatment alone. AsA
treatment at concentrations >1 mM AsA decreased the number of
living cells, and no effect was observed for concentrations <1
mM. The cell viability of HL60 cells 24 h after 4 Gy X-ray
irradiation with/without AsA treatment is shown in Fig. 4B. At concentrations >1 mM AsA,
combination AsA treatment and 4 Gy X-ray irradiation decreased the
number of living cells compared to X-ray irradiation alone. The
number of living cells with AsA treatment combined with 4 Gy X-ray
irradiation was not higher than that for cells exposed to X-ray
irradiation alone.
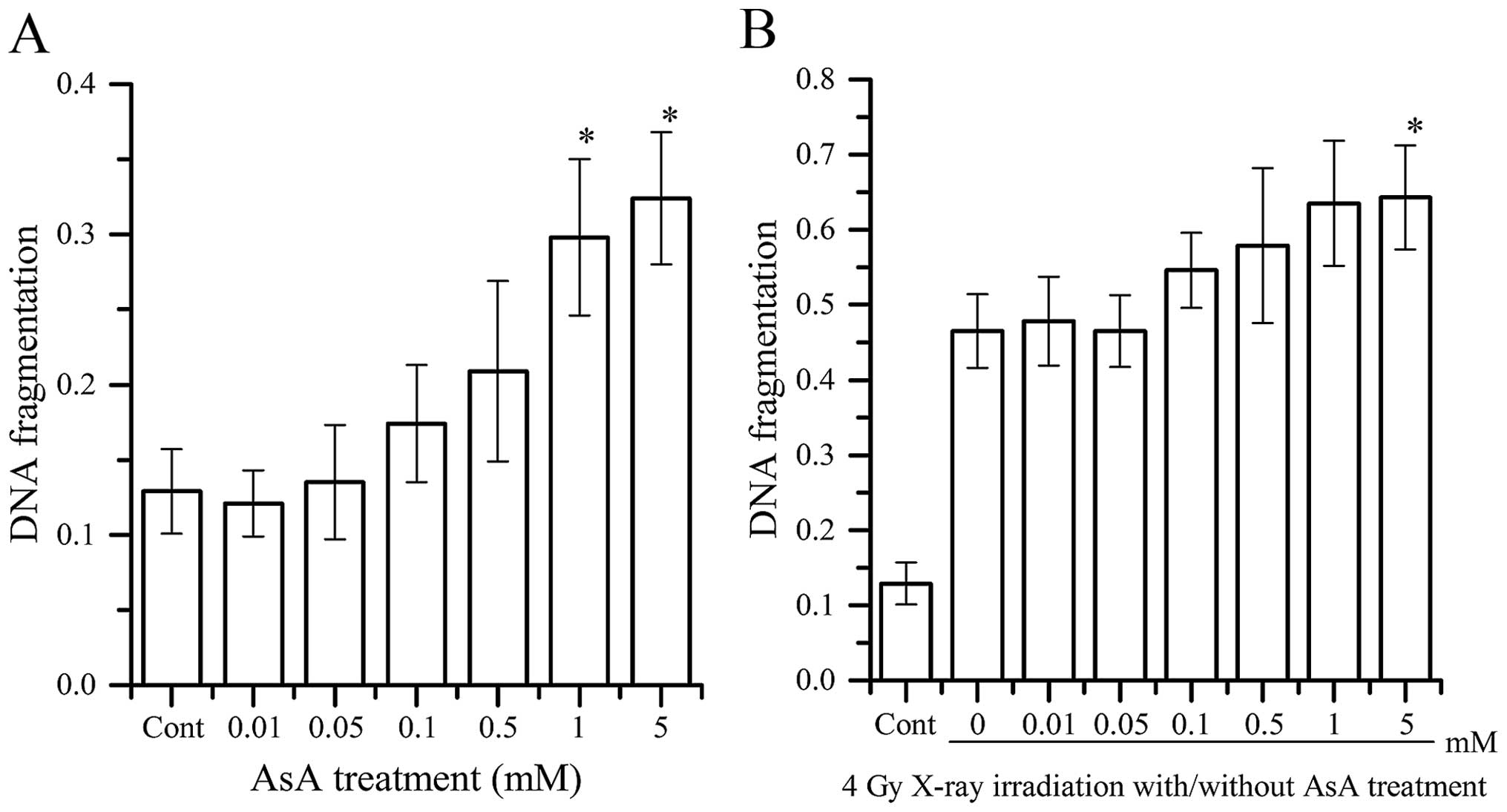
DNA fragmentation
DNA fragmentation following AsA treatment alone and
combination AsA treatment with 4 Gy X-ray irradiation is shown in
Fig. 5. AsA treatment increased DNA
fragmentation at concentrations >1 mM in a
concentration-dependent manner. DNA fragmentation with AsA
treatment concentrations >5 mM in combination with 4 Gy X-ray
irradiation was more than that of 4 Gy X-ray irradiation alone.
When AsA was added to 4 Gy X-ray irradiation, a concentration that
induced lower DNA fragmentation than 4 Gy irradiation alone was not
identified.
Discussion
When ionizing radiation is delivered to living
cells/animals, reactive oxygen species (ROS) such as ·OH, hydrogen
radicals (·H) superoxide (O2·−), and hydrogen peroxide
(H2O2) are produced. Initial generation of
·OH is via direct radiolysis of water. Highly reactive OH cannot
travel long distances, but instead immediately reacts with other
nearby molecules (11). For this
reason, ·OH generated by ionizing irradiation near DNA in cells
have a major role in radiation-induced cell damage. Our previous
study reported the significant association between the amount of
intracellular ·OH and apoptotic cell death (12).
There is renewed interest in the use of high dose
AsA (20 M in plasma) in the treatment of numerous different types
of cancer (13–15). AsA has been shown to selectively kill
cancer cells via the extracellular formation of
H2O2 (16). It
was hypothesized that the presence of a catalyst, such as ferric
ion, within the extracellular matrix of tumors could oxidize AsA to
ascorbate radicals, which could subsequently donate electrons to
oxygen to form O2-. O2− can be converted by
superoxide dismutase to the tumoricidal peroxide ion (5). In the present study, ascorbate radicals
were detected on the ESR spectrum of 1 mM AsA PBS. The number of
living cells decreased, and DNA fragmentation increased at
concentrations >1 mM AsA. These results indicate that at
concentrations >1 mM AsA treatment induces apoptosis and cell
death of HL60 cells. Combination treatment with >1 mM AsA and
X-ray irradiation decreased the number of living cells more than
exposure to cells with X-ray irradiation alone. DNA fragmentation
was also higher in cells treated with combination AsA treatment
>1 mM and X-ray irradiation compared to that in cells treated
with X-ray irradiation alone. The result confirmed that combination
AsA treatment with X-ray irradiation increased apoptosis in HL60
cells.
AsA is present in aqueous compartments (such as
cytosol, plasma and other body fluids) and has several
metabolically important cofactor functions in enzyme reactions,
particularly hydroxylation. AsA has been shown to efficiently
scavenge O2·−, H2O2, hypochlorite,
·OH and peroxyl radicals (17). The
radical scavenging effects of AsA are used to protect normal tissue
from radiation injury (18). However,
no increase in the number of viable HL60 cells following AsA
treatment was observed with irradiation when comparing to
irradiation alone. Therefore, the amount of ·OH scavenged by AsA
was estimated. ·OH generated with 4 Gy X-ray irradiation were
scavenged by AsA at concentrations >75 µM in the ESR analysis.
The result suggests that AsA concentrations >75 µM in the fluid
inside the cell could scavenge ·OH generated with X-ray
irradiation. The actual AsA concentration inside the cell was 75 µM
when cells underwent extracellular treatment with 5 mM AsA.
However, 5 mM AsA significantly induced HL60 cell death even
without irradiation. The result was evidenced in the HL60 cells in
the present study. However, our previous study reported that 5 mM
AsA treatment suppressed the growth of epithelial cancer cells
in vitro (6). It was suggested
that extracellular H2O2 generated with >5
mM AsA mainly damaged the cell membrane due to passing more
cascades than radiation alone (19,20). The
current study revealed that caspase-3, −8 and −9 were activated 2 h
after exposure to H2O2, whereas in irradiated
cells activation of caspase-3 and −9 occurred 4 h after exposure;
caspase-8 activation was not observed (19). These high fluxes of
H2O2 appear to have a limited effect on
normal cells but can be detrimental to certain tumor cells
(13).
In conclusion, treating cancer cells with high
extracellular concentrations of AsA (>5 mM) could achieve an
intracellular AsA concentration level of 75 µM, leading to
scavenging of ·OH generated with X-ray irradiation. However,
treatment with the high extracellular concentrations of AsA alone
directly induced cancer cell death. As aforementioned, the
disappearance of radiation anticancer effects with AsA treatment in
combination with radiotherapy for cancer treatment is not a
concern. Previously, clinical trials on the effects of high-dose
intravenous AsA on patients with cancer have been performed
(21,22). We hypothesize that a clinical trial to
evaluate the high-dose intravenous AsA combined with radiotherapy
in patients with cancer will be launched in the near future.
Acknowledgements
The present study was supported by the Japan Society
for the Promotion of Science (Grants-in-Aid for Scientific
Research, project nos. 21591603 and 24591831).
References
|
1
|
Lu SH, Ohshima H, Fu HM, Tian Y, Li FM,
Blettner M, Wahrendorf J and Bartsch H: Urinary excretion of
N-nitrosamino acids and nitrate by inhabitants of high- and
low-risk areas for esophageal cancer in Northern China: Endogenous
formation of nitrosoproline and its inhibition by vitamin C. Cancer
Res. 46:1485–1491. 1986.PubMed/NCBI
|
|
2
|
Cameron E and Pauling L: Ascorbic acid and
cancer: A review. Proc Natl Acad Sci USA. 75:4538–3542. 1987.
View Article : Google Scholar
|
|
3
|
Creagan ET, Moertel CG, O'Fallon JR,
Schutt AJ, O'Connell MJ, Rubin J and Frytak S: Failure of high-dose
vitamin C (ascorbic acid) therapy to benefit patients with advanced
cancer. A controlled trial. N Engl J Med. 301:687–690. 1979.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hoffer LJ, Levine M, Assouline S,
Melnychuk D, Padayatty SJ, Rosadiuk K, Rousseau C, Robitaille L and
Miller WH Jr: Phase I clinical trial of i.v. ascorbic acid in
advanced malignancy. Ann Oncol. 19:1969–1974. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wilson MK, Baguley BC, Wall C, Jameson MB
and Findlay MP: Review of high-dose intravenous vitamin C as an
anticancer agent. Asia Pac J Clin Oncol. 10:22–37. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hosokawa Y, Monzen S, Yoshino H, Terashima
S, Nakano M, Toshima K, Saga R and Kashiwakura I: Effects of X ray
irradiation in combination with ascorbic acid on tumor control. Mol
Med Rep. 12:5449–5454. 2015.PubMed/NCBI
|
|
7
|
Herst PM, Broadley KW, Harper JL and
McConnell MJ: Pharmacological concentrations of ascorbate
radiosensitize glioblastoma multiforme primary cells by increasing
oxidative DNA damage and inhibiting G2/M arrest. Free Radic Biol
Med. 52:1486–1493. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shinozaki K, Hosokawa Y, Hazawa M,
Kashiwakura I, Okumura K, Kaku T and Nakayama E: Ascorbic acid
enhances radiation-induced apoptosis in an HL60 human leukemia cell
line. J Radiat Res (Tokyo). 52:229–237. 2011. View Article : Google Scholar
|
|
9
|
Taneja N, Tjalkens R, Philbert MA and
Rehemtulla A: Irradiation of mitochondria initiates apoptosis in a
cell free system. Oncogene. 20:167–177. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Riley PA: Free radicals in biology:
Oxidative stress and the effects of ionizing radiation. Int J
Radiat Biol. 65:27–33. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Matsumoto K, Ueno M, Nakanishi I and Anzai
K: Density of hydroxyl radicals generated in an aqueous solution by
irradiating carbon-ion beam. Chem Pharm Bull (Tokyo). 63:195–199.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hosokawa Y, Tanaka L, Kaneko M, Sakakura
Y, Tsuruga E, Irie K and Yajima T: Apoptosis induced by generated
OH radicals inside cells after irradiation. Arch Histol Cytol.
65:301–305. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Du J, Cullen JJ and Buettner GR: Ascorbic
acid: Chemistry, biology and the treatment of cancer. Biochim
Biophys Acta. 1826:443–457. 2012.PubMed/NCBI
|
|
14
|
Espey MG, Chen P, Chalmers B, Drisko J,
Sun AY, Levine M and Chen Q: Pharmacologic ascorbate synergizes
with gemcitabine in preclinical models of pancreatic cancer. Free
Radic Biol Med. 50:1610–1619. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mikirova NA, Casciari JJ, Hunninghake RE
and Riordan NH: Intravenous ascorbic acid protocol for cancer
patients:scientific rationale, pharmacology, and clinical
experience. Funct Foods Health Dis. 3:344–366. 2013.
|
|
16
|
Terashima S, Hosokawa Y, Yoshino H,
Yamaguchi M and Nakamura T: Effect of ascorbic acid and
X-irradiation on HL-60 human leukemia cells: The kinetics of
reactive oxygen species. Oncol Rep. 30:2653–2658. 2013.PubMed/NCBI
|
|
17
|
Sies H and Stahl W: Vitamins E and C,
beta-carotene, and other carotenoids as antioxidants. Am J Clin
Nutr. 62:1315S–1321S. 1995.PubMed/NCBI
|
|
18
|
Sato T, Kinoshita M, Yamamoto T, Ito M,
Nishida T, Takeuchi M, Saitoh D, Seki S and Mukai Y: Treatment of
irradiated mice with high-dose ascorbic acid reduced lethality.
PLoS One. 10:e01170202015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hosokawa Y, Sakakura Y, Tanaka L, Okumura
K, Yajima T and Kaneko M: Radiation-induced apoptosis is
independent of caspase-8 but dependent on cytochrome c and the
caspase-9 cascade in human leukemia HL60 cells. J Radiat Res
(Tokyo). 46:293–303. 2005. View Article : Google Scholar
|
|
20
|
Mojić M, Bogdanović Pristov J,
Maksimović-Ivanić D, Jones DR, Stanić M, Mijatović S and Spasojević
I: Extracellular iron diminishes anticancer effects of vitamin C:
An in vitro study. Sci Rep. 4:59552014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Stephenson CM, Levin RD, Spector T and Lis
CG: Phase I clinical trial to evaluate the safety, tolerability,
and pharmacokinetics of high-dose intravenous ascorbic acid in
patients with advanced cancer. Cancer Chemother Pharmacol.
72:139–146. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Monti DA, Mitchell E, Bazzan AJ, Littman
S, Zabrecky G, Yeo CJ, Pillai MV, Newberg AB, Deshmukh S and Levine
M: Phase I evaluation of intravenous ascorbic acid in combination
with gemcitabine and erlotinib in patients with metastatic
pancreatic cancer. PLoS One. 7:e297942012. View Article : Google Scholar : PubMed/NCBI
|