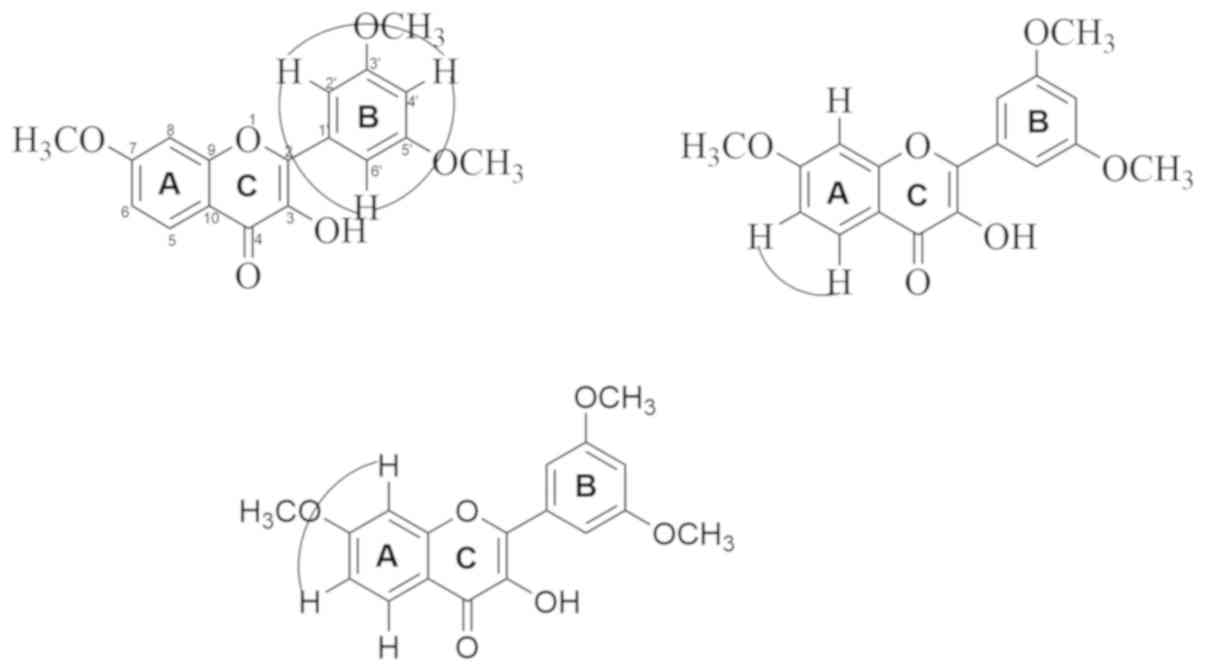
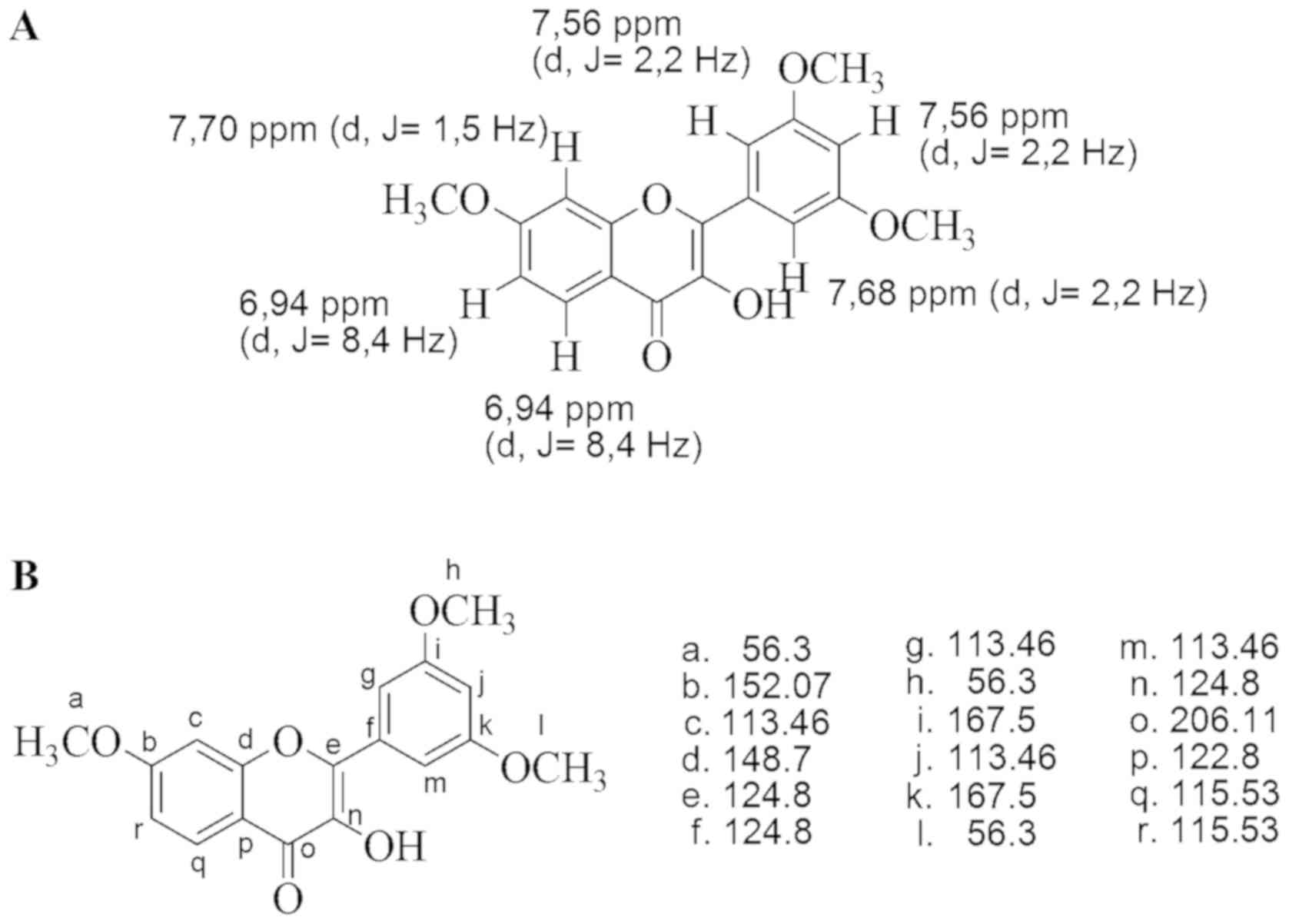
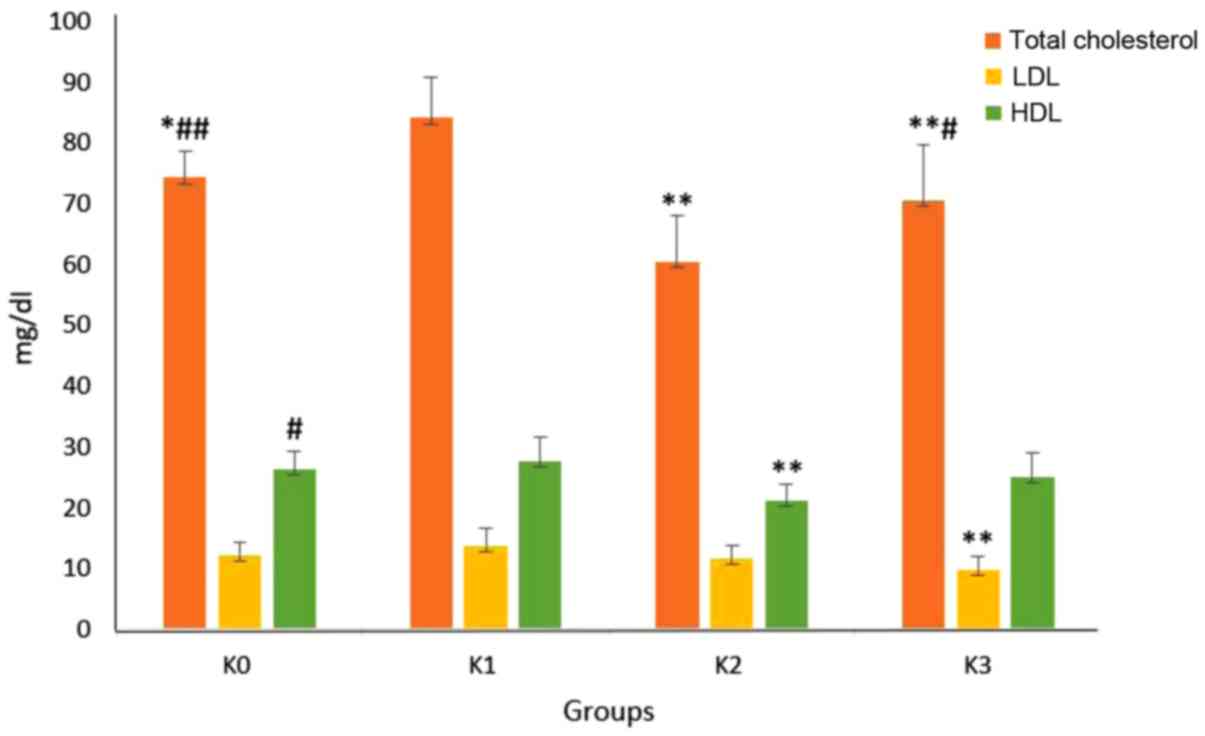
|
1
|
Hidayat S and Rachmadiyanto AN:
Utilization of Alang-Alang (Imperata cylindrica (L.)
Raeusch.) as Traditional Medicine in Indonesian Archipelago. Proc
1st Satreps Conf. 1:82–89. 2017.
|
|
2
|
Subositi D and Widodo H: Genetic diversity
of Cogon grass (Imperata cylindrica (L.) Beauv) based on the
intersimpel marking of the sequence repeat (ISSR). Jurnal Ilmu-Ilmu
Hayati. 17:115–122. 2018.
|
|
3
|
Khaerunnisa S: Utilization of bioactive
compounds from alang-alang root (Imperata cylindrica) as an
antioxidant, 2009.
|
|
4
|
Lalthanpuii PB, Zarzokimi and
Lalchhandama K: Some phytochemical analyses of different extracts
of the cogon grass Imperata cylindrica from Mizoram, India.
Sci Vis. 18:120–124. 2018.
|
|
5
|
Padma R, Parvathy NG, Renjith V and Rahate
KP: Quantitative estimation of tannins, phenols and antioxidant
activity of methanolic extract of Imperata cylindrica. Int J
Res Pharm Sci. 4:73–77. 2013.
|
|
6
|
Anggraeni N, Syamsunarno MRA, Mukarromah
GR, Zada A, Triatin RD, Pamela Y and Dhianawaty D: Low Serum
cholesterol in Mice Pre-treated with Imperata cylindrica L.
after Acute Olive Oil Gavage. KnE Life Sci. 3(460)2017.
|
|
7
|
National Heart Foundation of Australia:
The economic burden of Hypercholesterolaemia. Australia, 2018.
|
|
8
|
Alinde OBL, Esterhuyse AJ and Oguntibeju
OO: Role of reactive oxygen species in the pathogenesis of
cardiovascular disease. Sci Res Essays. 7:4151–4159. 2012.
|
|
9
|
Malekmohammad K, Sewell RDE and
Rafieian-Kopaei M: Antioxidants and atherosclerosis: Mechanistic
aspects. Biomolecules. 9(301)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Panth N, Paudel KR and Parajuli K:
Reactive oxygen species: A key hallmark of cardiovascular disease.
Adv Med. 2016(9152732)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Singh UN, Kumar S and Dhakal S: Study of
oxidative stress in hypercholesterolemia. Int J Contemp Med Res.
4:2454–7379. 2017.
|
|
12
|
Phaniendra A and Latha P: Free radicals:
Properties, sources, targets, and their implication in various free
radicals: Properties, sources, targets, and their implication in
various diseases. Ind J Clin Biochem. 30:11–26. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Das Sarma A, Rahaman Mallick A and Ghosh
A: Free radicals and their role in different clinical conditions:
An overview. Int J Pharma Scie Res. 1:185–192. 2010.
|
|
14
|
Abdul Qadir M, Shahzadi SK, Bashir A,
Munir A and Shahzad S: Evaluation of phenolic compounds and
antioxidant and antimicrobial activities of some common herbs. Int
J Anal Chem. 2017(6)2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Tungmunnithum D, Thongboonyou A, Pholboon
A and Yangsabai A: Flavonoids and other phenolic compounds from
medicinal plants for pharmaceutical and medical aspects: An
overview. Medicines. 5(93)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Rajput B, Golave A, Yadav S and Jadhav JP:
Total phenolic concentrations and antioxidant activities in Drimia
sp. J Herbs Spices Med Plants. 24:28–36. 2018.
|
|
17
|
Sánchez-Rangel Carlos J, Jorge BJ, Heredia
JB, Zevallos LC and Jacobo-Velázquez DA: The Folin-Ciocalteu assay
revisited: Improvement of its speci fi city for total phenolic
content determination. Anal Methods. 5:5990–5999. 2013.
|
|
18
|
Blainski A, Lopes G and de Mello J:
Application and analysis of the folin ciocalteu method for the
determination of the total phenolic content from Limonium
Brasiliense L. Molecules. 18:6852–6864. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Mohamed OS, Said MM, Ali ZY, Atia HA and
Mostafab HS: Improving effect of dietary oat bran supplementation
on oxidative stress induced by hyperlipidemic diet. Researcher.
3:1–10. 2011.
|
|
20
|
National Research Council (US) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals. Guide for the Care and Use of Laboratory Animals. 8th
edition. Washington (DC), National Academies Press (US); 2011.
Available from: https://www.ncbi.nlm.nih.gov/books/NBK54050/
doi: 10.17226/12910.
|
|
21
|
Suratman S, Listyawati S and Sutarno S:
Physical characteristics and NaCl content of urine white male rat
(Rattus norvegicus L.) after orally intakes of cogon grass rhizome
(Imperata cylindrica L.) extract. Biofarmasi J Nat Prod
Biochem. 1:7–12. 2003.
|
|
22
|
Gilbert G and Lickliter R: The various
roles of animal models in understanding human development. Soc Dev.
13:311–325. 2004.
|
|
23
|
Suryaningtyas W, Prasetyo R and Dewi B:
Research and laboratory techniques in experimental animals and
human. Airlangga University Press, Surabaya, 2015.
|
|
24
|
Chow PKH, Ng RTH and Ogden BE: Using
Animal Models in Biomedical Research. World Scientific Publishing,
Singapore, pp 1-308, 2008.
|
|
25
|
Institutional Animal care and use
committee: Anesthesia (Guideline). Available from: https://animal.research.uiowa.edu/iacuc-guidelines-anesthesia.
|
|
26
|
Mohamed DA, Hamed TE and Al-Okbi SY:
Reduction in hypercholesterolemia and risk of cardiovascular
diseases by mixtures of plant food extracts: A study on plasma
lipid profile, oxidative stress and testosterone in rats. Grasas Y
Aceites: 61: 2010 doi: 10.3989/gya.021210.
|
|
27
|
Awaad S, Mohamed N, Maitland D and Soliman
G: Anti-ulcerogenic activity of extract and some isolated
flavonoids from Desmostachia bipinnata (L.) Stapf. Rec Nat Prod.
2(76)2008.
|
|
28
|
Markham K: How to identify flavonoids.
Institut Teknologi Bandung, Bandung, 1988.
|
|
29
|
Krishnaiah D, Devi T, Bono A and Sarbatly
R: Studies on phytochemical constituents of six Malaysian medicinal
plants. J Med Plants Res. 3:067–072. 2009.
|
|
30
|
Babu RH and Savithramma N: Phytochemical
screening of underutilized species of Poaceae. An Int J. 1:947–951.
2013.
|
|
31
|
Zhou X, Wang J, Jiang B, Shang J and Zhao
C: A study of extraction process and in vitro antioxidant activity
of total phenols from Rhizoma imperatae. Afr J Traditional
Complementary Alternative Med. 10:175–178. 2013.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Akpakpan A, Ukpong EJ and Willie I:
Phytochemical screening of the ethanol and aqueous extracts of
dicliptera verticillata leaves phytochemical screening of the
ethanol and aqueous extracts of dicliptera verticillata Leaves,
2017.
|
|
33
|
Afrose S, Hossain S, Salma U, Miah AG and
Tsujii H: Dietary karaya saponin and Rhodobacter capsulatus
exert hypocholesterolemic effects by suppression of hepatic
cholesterol synthesis and promotion of bile acid synthesis in
laying hens. Cholesterol. 2010(7)2010.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Shi Y, Guo R, Wang X, Yuan D, Zhang S,
Wang J, Yan X and Wang C: The regulation of alfalfa saponin extract
on key genes involved in hepatic cholesterol metabolism in
hyperlipidemic rats. PLoS One. 9(e88282)2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Vinarova L, Vinarov Z, Atanasov V,
Pantcheva I, Tcholakova S, Denkov N and Stoyanov S: Lowering of
cholesterol bioaccessibility and serum concentrations by saponins:
In vitro and in vivo studies. Food Funct. 6:501–512.
2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kobayashi S: The effect of polyphenols on
hypercholesterolemia through inhibiting the transport and
expression of Niemann-Pick C1-Like 1. Int J Mol Sci.
20(4939)2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Zeka K, Ruparelia K, Arroo R, Budriesi R
and Micucci M: Flavonoids and their metabolites: Prevention in
cardiovascular diseases and diabetes. Diseases. 5(19)2017.
View Article : Google Scholar
|
|
38
|
Millar CL, Duclos Q and Blesso CN: Effects
of dietary flavonoids on reverse cholesterol transport, HDL
metabolism, and HDL function. Adv Nutr. 8:226–239. 2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Caesar LK and Cech NB: Synergy and
antagonism in natural product extracts: When 1 + 1 does not equal
2. Nat Prod Rep. 36:869–888. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Roza JM, Xian-Liu Z and Guthrie N: Effect
of citrus flavonoids and tocotrienols on serum cholesterol levels
in hypercholesterolemic subjects. Altern Ther Health Med. 13:44–48.
2007.PubMed/NCBI
|
|
41
|
Dauqan EMA, Abdullah A, Sani HA and
Selangor B: Lipid profile and antioxidant enzymes in normal and
stressed rat fed with palm olein school of biosciences and
biotechnology, school of chemical sciences and food technology,
faculty of science and technology. Am J Appl Sci. 9:1071–1078.
2012.
|
|
42
|
Hayek T, Ito Y, Azrolan N, Verdery RB,
Aalto-Setmla K, Walsh A and Breslow JL: Dietary fat increases high
density lipoprotein (HDL) levels both by increasing the transport
rates and decreasing the fractional catabolic rates of HDL
cholesterol ester and apolipoprotein (Apo) A-I. Presentation of a
new animal model and mechanistic studies in human Apo A-I
transgenic and control mice. J Clin Invest. 91:1665–1671.
1993.PubMed/NCBI View Article : Google Scholar
|
|
43
|
San Mauro Martín I, Collado Yurrita L,
Cuadrado Cenzual MÁ, Ciudad Cabañas MJ and Mendive Dubourdieu P:
Role of ApoA1 on High-density lipoprotein: An intervention with
plant sterols in patients with hypercholesterolemia. Nutr Hosp.
31:494–499. 2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wallace TC: Anthocyanins in cardiovascular
disease prevention anthocyanins in cardiovascular disease 1. Adv
Nutr. 2:1–7. 2011.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Grassi D, Desideri G and Ferri C:
Flavonoids: Antioxidants against atherosclerosis. Nutrients.
2:889–902. 2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Cassidy A, Bertoia M, Chiuve S, Flint A,
Forman J and Rimm EB: Habitual intake of anthocyanins and
flavanones and risk of cardiovascular disease in men. Am J Clin
Nutr. 104:587–594. 2016.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Vauzour D, Rodriguez-Mateos A, Corona G,
Oruna-Concha MJ and Spencer JPE: Polyphenols and human health:
Prevention of disease and mechanisms of action. Nutrients.
2:1106–1131. 2010.PubMed/NCBI View Article : Google Scholar
|