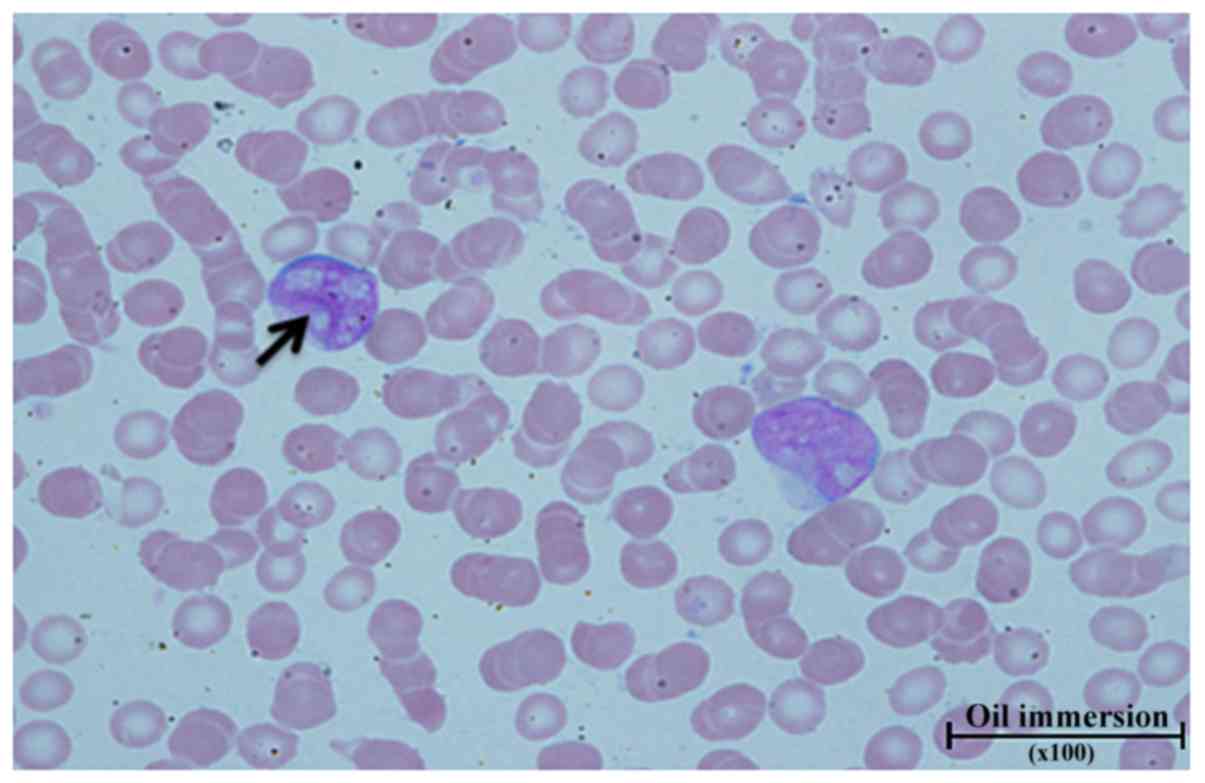
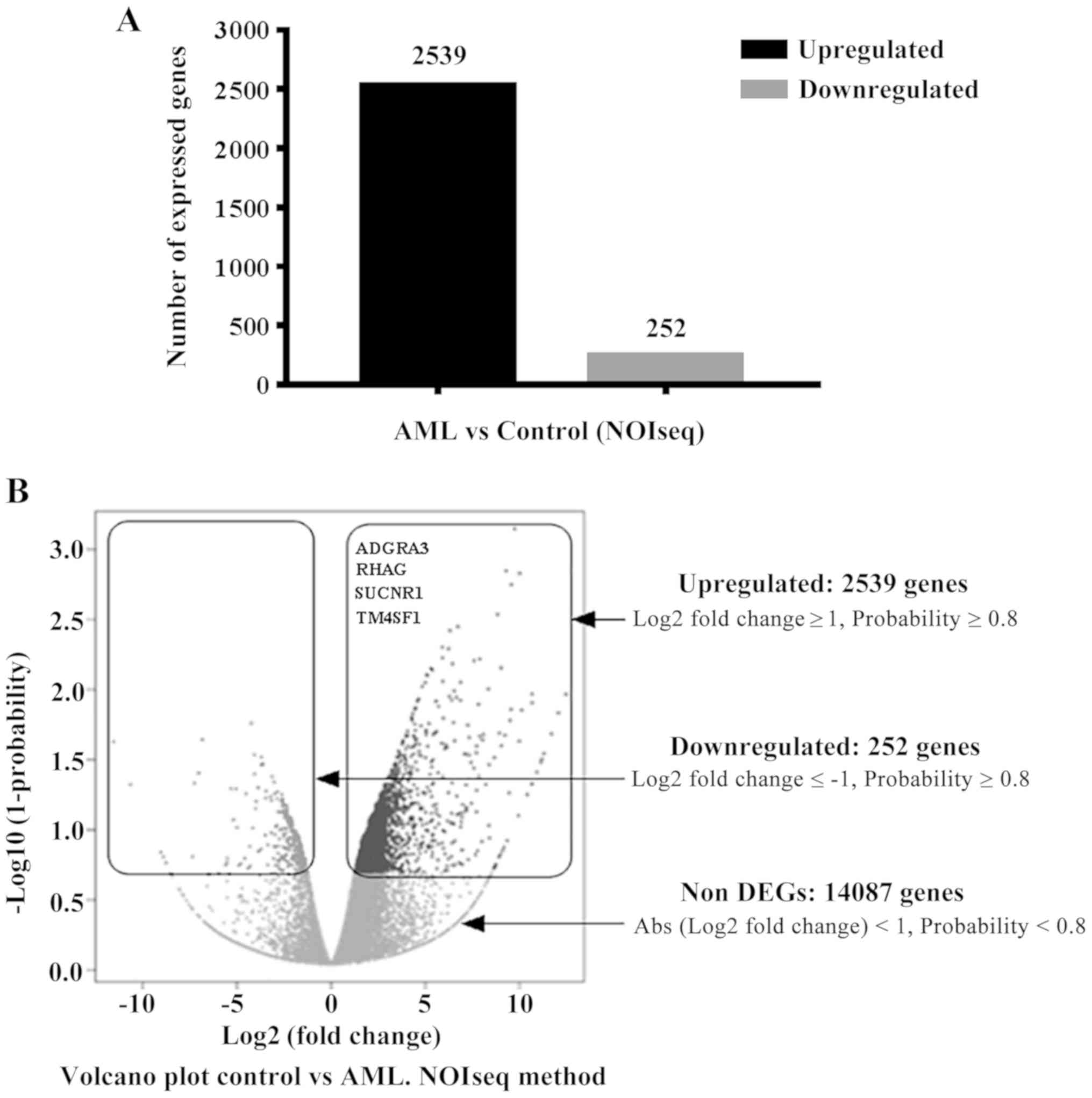
|
1
|
Hasserjian RP: Acute myeloid leukemia:
Advances in diagnosis and classification. Int J Lab Hematol.
35:358–366. 2013.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Bennett JM, Catovsky D, Daniel MT,
Flandrin G, Galton DA, Gralnick HR and Sultan C: Proposals for the
classification of the acute leukaemias. French-American-British
(FAB) co-operative group. Br J Haematol. 33:451–458.
1976.PubMed/NCBI View Article : Google Scholar
|
|
3
|
DiNardo CD, Garcia-Manero G, Pierce S,
Nazha A, Bueso-Ramos C, Jabbour E, Ravandi F, Cortes J and
Kantarjian H: Interactions and relevance of blast percentage and
treatment strategy among younger and older patients with acute
myeloid leukemia (AML) and myelodysplastic syndrome (MDS). Am J
Hematol. 91:227–232. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Döhner H, Estey E, Grimwade D, Amadori S,
Appelbaum FR, Büchner T, Dombret H, Ebert BL, Fenaux P, Larson RA,
et al: Diagnosis and management of AML in adults: 2017 ELN
recommendations from an international expert panel. Blood.
129:424–447. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lagunas-Rangel FA, Chávez-Valencia V,
Gómez-Guijosa MÁ and Cortes-Penagos C: Acute myeloid
leukemia-genetic alterations and their clinical prognosis. Int J
Hematol Oncol Stem Cell Res. 11:328–339. 2017.PubMed/NCBI
|
|
6
|
Martelli MP, Sportoletti P, Tiacci E,
Martelli MF and Falini B: Mutational landscape of AML with normal
cytogenetics: Biological and clinical implications. Blood Rev.
27:13–22. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rubnitz JE, Gibson B and Smith FO: Acute
myeloid leukemia. Hematol Oncol Clin North Am. 24:35–63.
2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Takahashi S: Current findings for
recurring mutations in acute myeloid leukemia. J Hematol Oncol.
4(36)2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Batcha AMN, Bamopoulos SA, Kerbs P, Kumar
A, Jurinovic V, Rothenberg-Thurley M, Ksienzyk B, Philippou-Massier
J, Krebs S, Blum H, et al: Allelic imbalance of recurrently mutated
genes in acute myeloid leukaemia. Sci Rep. 9(11796)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Castle JC, Loewer M, Boegel S, Tadmor AD,
Boisguerin V, de Graaf J, Paret C, Diken M, Kreiter S, Türeci O and
Sahin U: Mutated tumor alleles are expressed according to their DNA
frequency. Sci Rep. 4(4743)2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Vardiman JW, Harris NL and Brunning RD:
The world health organization (WHO) classification of the myeloid
neoplasms. Blood. 100:2292–2302. 2002.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hemsing AL, Hovland R, Tsykunova G and
Reikvam H: Trisomy 8 in acute myeloid leukemia. Expert Rev Hematol.
12:947–958. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Thiele J, Kvasnicka HM, Facchetti F,
Franco V, van der Walt J and Orazi A: European consensus on grading
bone marrow fibrosis and assessment of cellularity. Haematologica.
90:1128–1132. 2005.PubMed/NCBI
|
|
14
|
Mortazavi A, Williams BA, McCue K,
Schaeffer L and Wold B: Mapping and quantifying mammalian
transcriptomes by RNA-Seq. Nat Methods. 5:621–628. 2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wang Z, Gerstein M and Snyder M: RNA-Seq:
A revolutionary tool for transcriptomics. Nat Rev Genet. 10:57–63.
2009.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Li B and Dewey CN: RSEM: Accurate
transcript quantification from RNA-Seq data with or without a
reference genome. BMC Bioinformatics. 12(323)2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Tarazona S, Furió-Tarı P, Nueda MJ, Ferrer
A and Conesa A: NOISeq: Differential expression in RNA-seq 2013.
https://bicoductor.statistik.tu-dortmund.de/packages/3.7/bior/vignettes/NOISwq/inst/doc/NOISeq.pdf.
|
|
18
|
McKenna A, Hanna M, Banks E, Sivachenko A,
Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly
M and DePristo MA: The genome analysis toolkit: A mapreduce
framework for analyzing next-generation DNA sequencing data. Genome
Res. 20:1297–1303. 2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Daver N, Schlenk RF, Russell NH and Levis
MJ: Targeting FLT3 mutations in AML: Review of current knowledge
and evidence. Leukemia. 33:299–312. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Berman JN, Gerbing RB, Alonzo TA, Ho PA,
Miller K, Hurwitz C, Heerema NA, Hirsch B, Raimondi SC, Lange B, et
al: Prevalence and clinical implications of NRAS mutations in
childhood AML: A report from the Children's oncology group.
Leukemia. 25:1039–1042. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Heath EM, Chan SM, Minden MD, Murphy T,
Shlush LI and Schimmer AD: Biological and clinical consequences of
NPM1 mutations in AML. Leukemia. 31:798–807. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Spiess K, Bagger SO, Torz LJ, Jensen KHR,
Walser AL, Kvam JM, Møgelmose ASK, Daugvilaite V, Junnila RK,
Hjortø GM and Rosenkilde MM: Arrestin-independent constitutive
endocytosis of GPR125/ADGRA3. Ann N Y Acad Sci. 1456:186–199.
2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Maiga A, Lemieux S, Pabst C, Lavallée VP,
Bouvier M, Sauvageau G and Hébert J: Transcriptome analysis of G
protein-coupled receptors in distinct genetic subgroups of acute
myeloid leukemia: Identification of potential disease-specific
targets. Blood Cancer J. 6(e431)2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Wu Y, Chen W, Gong L, Ke C, Wang H and Cai
Y: Elevated G-protein receptor 125 (GPR125) expression predicts
good outcomes in colorectal cancer and inhibits wnt/β-catenin
signaling pathway. Med Sci Monitor. 24:6608–6616. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Institute NC: Genomic data commons data
portal. https://portal.gdc.cancer.gov/genes/ENSG00000152990.
J 2020.
|
|
26
|
Institute NC: Genomic data commons data
portal. https://portal.gdc.cancer.gov/genes/ENSG00000198829.
J 2020.
|
|
27
|
Louer EMM, Lores-Motta L, Ion AM, Den
Hollander AI and Deen PMT: Single nucleotide polymorphism
rs13079080 is associated with differential regulation of the
succinate receptor 1 (SUCNR1) gene by miRNA-4470. RNA Biol.
16:1547–1554. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Mu X, Zhao T, Xu C, Shi W, Geng B, Shen J,
Zhang C, Pan J, Yang J, Hu S, et al: Oncometabolite succinate
promotes angiogenesis by upregulating VEGF expression through
GPR91-mediated STAT3 and ERK activation. Oncotarget. 8:13174–13185.
2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Thutkawkorapin J, Picelli S, Kontham V,
Liu T, Nilsson D and Lindblom A: Exome sequencing in one family
with gastric- and rectal cancer. BMC Genet. 17(41)2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Macaulay IC, Tijssen MR, Thijssen-Timmer
DC, Gusnanto A, Steward M, Burns P, Langford CF, Ellis PD,
Dudbridge F, Zwaginga JJ, et al: Comparative gene expression
profiling of in vitro differentiated megakaryocytes and
erythroblasts identifies novel activatory and inhibitory platelet
membrane proteins. Blood. 109:3260–3269. 2007.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Ariza AC, Deen PMT and Robben JH: The
succinate receptor as a novel therapeutic target for oxidative and
metabolic stress-related conditions. Front Endocrinol (Lausanne).
3(22)2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hakak Y, Lehmann-Bruinsma K, Phillips S,
Le T, Liaw C, Connolly DT and Behan DP: The role of the GPR91
ligand succinate in hematopoiesis. J Leukoc Biol. 85:837–843.
2009.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chen H, Pan T, Liu P, Wang P and Xu S:
Baihu Jia Guizhi decoction improves rheumatoid arthritis
inflammation by regulating succinate/SUCNR1 metabolic signaling
pathway. Evid Based Complement Alternat Med.
2019(3258572)2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Bardella C, Pollard PJ and Tomlinson I:
SDH mutations in cancer. Biochim Biophys Acta. 1807:1432–1443.
2011.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Ricketts C, Woodward ER, Killick P, Morris
MR, Astuti D, Latif F and Maher ER: Germline SDHB mutations and
familial renal cell carcinoma. J Natl Cancer Inst. 100:1260–1262.
2008.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Baysal BE, Ferrell RE, Willett-Brozick JE,
Lawrence EC, Myssiorek D, Bosch A, van der Mey A, Taschner PE,
Rubinstein WS, Myers EN, et al: Mutations in SDHD, a mitochondrial
complex II gene, in hereditary paraganglioma. Science. 287:848–851.
2000.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Zhao T, Mu X and You Q: Succinate: An
initiator in tumorigenesis and progression. Oncotarget.
8:53819–53828. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Chen BS, Xu ZX, Xu X, Cai Y, Han YL, Wang
J, Xia SH, Hu H, Wei F, Wu M and Wang MR: RhCG is downregulated in
oesophageal squamous cell carcinomas, but expressed in multiple
squamous epithelia. Eur J Cancer. 38:1927–1936. 2002.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ye L, Pu C, Tang J, Wang Y, Wang C, Qiu Z,
Xiang T, Zhang Y and Peng W: Transmembrane-4 L-six family member-1
(TM4SF1) promotes non-small cell lung cancer proliferation,
invasion and chemo-resistance through regulating the
DDR1/Akt/ERK-mTOR axis. Respir Res. 20(106)2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Yang JC, Zhang Y, He SJ, Li MM, Cai XL,
Wang H, Xu LM and Cao J: TM4SF1 promotes metastasis of pancreatic
cancer via regulating the expression of DDR1. Sci Rep.
7(45895)2017.PubMed/NCBI View Article : Google Scholar
|