Introduction
The new coronavirus, termed COVID-19, emerged in
Wuhan, China in late 2019. Although the newly emerged virus has a
mutation in the sequence of the spike protein, its binding affinity
for the angiotensin-converting enzyme 2 (ACE2) receptor is
identical to that of the severe acute respiratory syndrome
(SARS-CoV)-1(1). The cytokine storm
is as a group of inflammatory responses, which includes interleukin
(IL)-I, IL-2, IL-6, IL-10 and interferon (IFN)-γ (2), is a serious complication of COVID-19
infection (3-6).
Different types of vaccines are being developed to
assist in limiting the spread of the virus, and reduce mortality
rates going forward, some of which have been approved by the
regulatory bodies of several countries, and are being widely
distributed. Various companies are currently developing a vaccine
introducing mRNA to produce viral proteins, specifically the spike
protein, by the host cells. A more stable DNA vaccine is another
option to prevent infection with SARS-CoV-2 using adenovirus
plasmids encoding the SARS-CoV-2 spike protein (7,8). Another
alternative is to use other viral proteins, usually by recombinant
DNA. The inactivated whole SARS-CoV-2 vaccine is also a candidate
being assessed in preclinical trials. However, using a SARS-CoV-2
live attenuated vaccine carries potential risks, such as the
reactivation or the virulence of SARS-CoV-2 in immunocompromised
patients (9).
IFNs were named initially due to their role in
interfering with viral infections. Influenza-infected chick cells
mounted antiviral resistance states by producing secreted
cytokines, which were later termed IFNs (10). IFNs are cytokines that are implicated
in antiviral responses, immune induction and regulating cell
division (11). The gene expression
of type I IFN is primarily regulated at the transcriptional stage,
and in the absence of stimulators, such as double-stranded RNA,
IFNs are not translated. The lack of the IFN-β gene through gene
knockout makes mice vulnerable to infection with viruses, such as
vaccinia virus and blocks the IFN-α response (12).
To induce the innate immune response during viral
infection, it is essential to stimulate the IFN response. The
absence of IFN regulatory factor (IRF)3 or defective IRF7 function
decreases the gene expression of IFN-α/β, making mice more
sensitive to viral infection (13).
IRF3 modulates the innate antiviral response that is triggered by
the invading virus. IRF3 is primarily modified by
hyperphosphorylation when the virus begins replication (14).
IRF3 and IRF7 are the most common regulators of
IFN-β. They replace IRF2, serving a key role in type I IFN
responses (15-17).
IRF3 and IRF7 have specific binding properties that allow them to
bind to the type I IFN promoters, and their ratio to the bound
elements modulates the IFN type I response during viral infection
(18). However, IRF3 degradation has
been reported to repress IFN-β rather than the activation of the
transcriptional repressors. The lack of transcriptionally active
IRF3 abolishes the activation of IFN-β to the Sendai virus in mouse
embryonic fibroblasts (19).
Upon activation, IRF3 molecules translocate to the
nucleus after phosphorylation and bind to the ornithine
cyclodeaminase or P300 to form complexes in the IFN sensitive
response element region (20). Most
RNA viruses elicit a type I IFN response in toll-like receptors
(TLRs) or independent mechanisms (cytosolic recognition system)
through retinoic acid-inducible genes (RIG-1), which sense the
viral RNA molecules (21). Moreover,
RIG-1 is central to the stimulation of the type I IFN response to
RNA virus infection via activation of IRF3 through kinases in
fibroblasts and dendritic cells (22). Synthetic or natural dsRNAs are
differentially recognised by RIG-1 and melanoma
differentiation-associated protein 5 (MAD5), as the former induces
production of IFNs to paramyxoviruses, the influenza virus and
Japanese encephalitis virus, whereas picornavirus is detected by
MAD5(23). As a coronavirus model,
the mouse hepatitis virus antagonises the type I IFN through the
Nucleocapsid protein (24). Previous
outbreaks with SARS and MERS revealed that the virus does not
increase the expression of IFN-β or its promoter activity.
Therefore, treatments with recombinant interferons were used to
boost the effects of antiviral drugs (25,26).
Infection with respiratory viruses activates the TLR signalling
pathways, and eventually leads to the induction of the type I IFN
response. The SARS-CoV-2 infection stimulates the TLR downstream
pathway to produce mature-IL-1β. An increase in IL-1β causes lung
fibrosis and fever. The virus is more highly infectious in adults
than children, which may be explained by the high expression levels
of aryl hydrocarbon receptors in children compared with the
relatively lower expression levels in adults (27).
The present study evaluated the gene expression of
IFN and IRF3 in COVID-19-infected patients compared with the
control, suggesting a mechanism for the induction of IFNs, and
highlighting IFNs as a therapeutic option for treating COVID-19
patients in clinical trials.
Materials and methods
Sample collection, RNA extraction and
reverse transcription quantitative PCR
RNA samples were collected from 30 patients
suspected of infection with COVID-19 between February and April
2020 at the Public Health Laboratory in Basrah, Iraq. The age range
of the patients was 25-55 years old, whereas that of the
non-COVID-19 infected individuals was 28-60 years old. The infected
patients included 8 females and 12 males whereas the non-infected
individuals consisted of were 3 females and 7 males.
Infection was diagnosed using a LightMix SarbecoV
E-gene plus EAV control (cat. no. 40-0776-96). The control samples
were negative for COVID-19 and were diagnosed with either the
common cold or influenza. The present study was approved by the
Public Health Department, Basrah Health Directorate (approval no.
F112020). All patients provided signed consent to participate in
the present study.
The RNA from nasal swabs (total RNA) was extracted
using an easy spin™ total RNA extraction kit (Intron; cat. no.
17221) according to the manufacturer's protocol, and used to
evaluate the gene expression of IFN-β and IRF3 in COVID-19-infected
or uninfected samples using gene-specific primers. An intron
HiSenScript™ (RH-)RT-PCR PreMix transcription kit was used to
reverse transcribe the RNA, according to the manufacturer's
protocol. Quantitative PCR was performed using SYBR-Green
MasterMix, according to the manufacturer's protocol (Bioneer; cat.
no. K-6210). The reaction consisted of a mixture of 10 µl
SYBR-Green, 3 µl cDNA template, 1 µl each of both the forward and
reverse primers against IFN-β (forward, CAACTTGCTTGGATTCCTACAAAG
and reverse primer, TATTCAAGCCTCCCATTCAATTG); IRF3 (forward,
CGGAAAGAAGTGTTGCGGTTAG and reverse primer, TTTGCCATTGGTGTCAGGAGAG);
and β-actin (forward, CCTGGCACCCAGCACAAT and reverse primer,
GCCGATCCACACGGAGATCT), and 5 µl free deionised diethylpyrocarbonate
D.W. to a final volume of 20 µl. The sequences of the primers are
based on previous studies (28,29).
The thermocycling conditions were: Initial
denaturation at 94˚C for 5 min; followed by 40 cycles of 15 sec of
denaturation at 95˚C, annealing at 58˚C for 30 sec, and an
extension step at 72˚C for 45 sec; with a final extension at 72˚C
for 5 min. The products were subjected to dissociation curve
analysis. Using the 2-∆∆Cq method for analysis of mRNA
expression, data were normalised to β-actin, which was used as a
housekeeping gene (30).
Statistical analysis
Data are presented as the mean ± standard deviation
of three technical repeats per patient. All data were analysed
using a Student's t-test. Statistical analysis was performed using
Microsoft Excel (Office 365; Microsoft Corporation). P<0.05 was
considered to indicate a statistically significant difference.
Results
The RNAs of the COVID-19 positive and negative
samples were extracted, reverse transcribed, used as the templates
and mixed with IRF3 or IFN-β specific primers to assess their
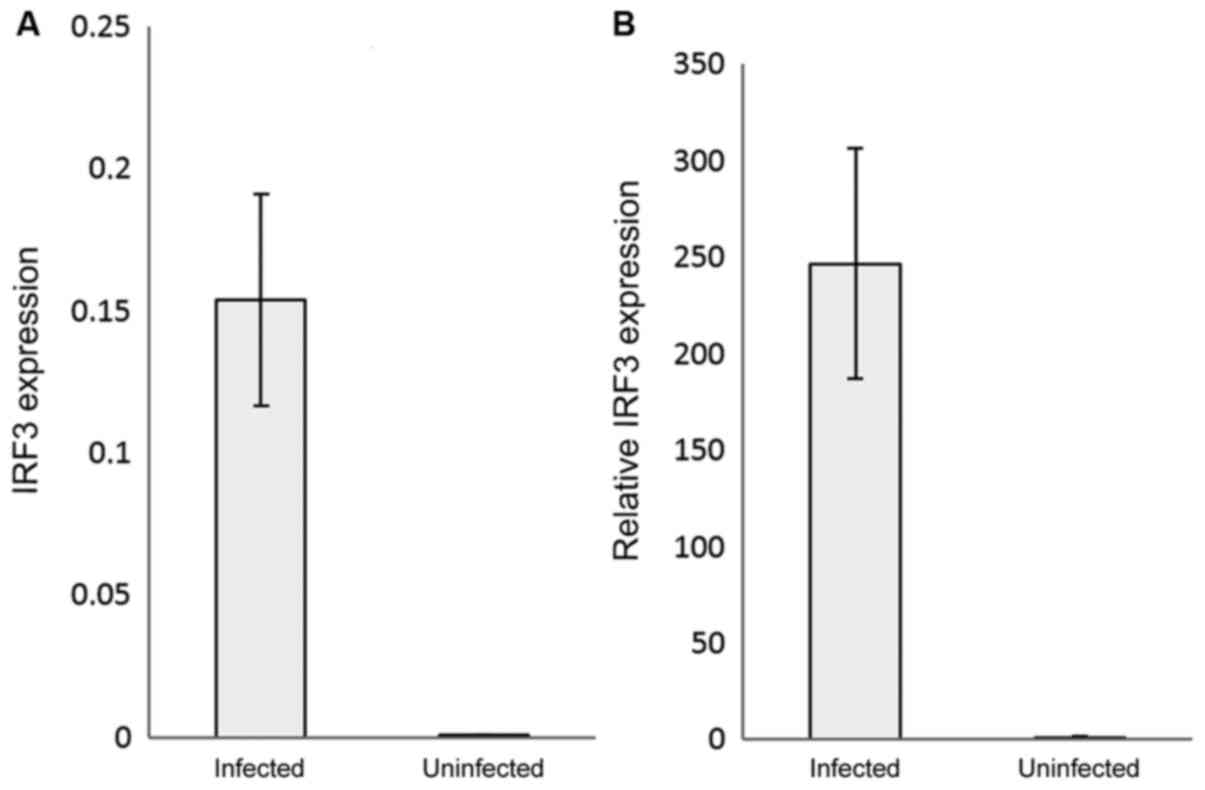
relative expression by qPCR. IRF3 gene expression was significantly
(P<0.05) upregulated in the COVID-19-infected patients by
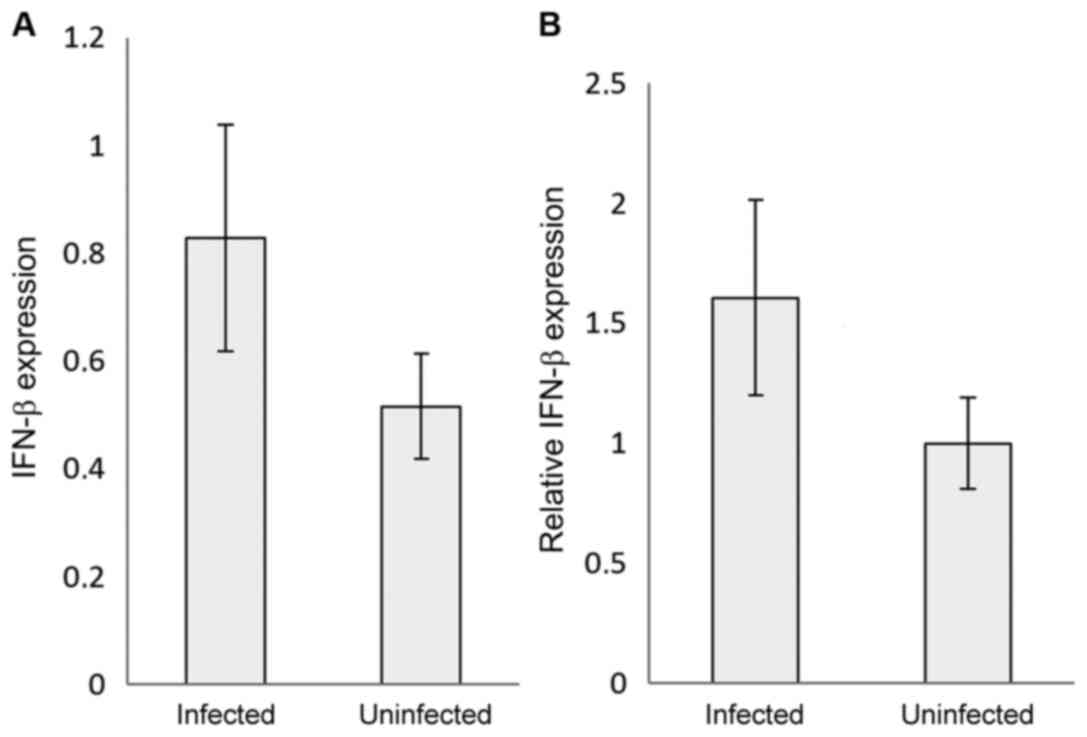
~250-fold compared with the control (uninfected samples) (Fig. 1A and B). Interestingly, IFN-β relative expression
was ~1.5-fold higher in the COVID-19-infected samples compared with
the control samples (P<0.05) (Fig.
2A and B). The 2-∆∆Cq
analysis was used to detect the relative expression after
subtracting the β-actin value from each sample, and the control was
normalised to 1, to express the results as the fold change.
Discussion
Studies have shown that IRF7 is expressed at a very
low level physiologically, and requires activation of a type I
interferon response for its induction (31,32).
Both MERS and SARS trigger a low level of interferon response
(33,34). IRF3 is a key regulator of type I IFN,
which triggers the host response against the invading viruses. IRF3
also implicated in unwanted inflammatory responses and septic shock
response (35-37).
Thus, in the present study, the effects of COVID-19 on an innate
immune response were determined.
The results showed that the gene expression levels
of IRF3 were notably increased by ~250-fold compared with its
expression in the virus-free samples. The increase in the IFN-β
levels were not consistent with the increase in the expression of
its primary regulator. The results agree with a study concerning
coronavirus infection and IFN responses; infection with SARS-CoV
does not induce IFN-β production or its promoter activity (38). A lower IFN response was detected in
the COVID-19-infected lung tissue compared with SARS, which makes
the former virus more sensitive to treatment with a type I IFN
(22,39). However, in SARS infections, IRF3 is
shown to translocate to the nucleus, independent of nay
phosphorylation, dimerization or binding to cAMP response
element-binding protein (CREB) binding protein. The SARS-CoV virus
may block IRF3 hyperphosphorylation-mediated homodimerization CREB
after transport of IRF3 to the nucleus (38).
Another hypothesis suggests that coronaviruses use
the IFN-inducible transmembrane proteins (IFITM) to enter the cell,
and the IFITM structural motifs required for entry inhibit the
entry of other viruses. The IFITM theory explains how the virus can
invade the lower respiratory tract (40). Coronaviruses, such as SARS, avoid the
inhibitory effects of type I IFNs either through induction of
double-membrane vesicles to physically hide the viral RNA
intermediates from pattern recognition receptors or by expressing
open reading frame (ORF)3, ORF6, ORF7, nucleocapsid protein and
non-structural RNA binding protein 1, which when combined, abolish
the IRF-3-dependent IFN-β pathway (41,42).
Based on the mechanism by which SARS inhibits the
IFN response, recombinant IFNs were used to treat SARS-infected
patients. The treatment of human corona Erasmus medical centre
(HcoV-EMC) human-infected tissues with the type I or III IFN, 1 h
post-infection, decreased the replication of the virus (43). In vitro, treating
SARS-CoV-infected Vero and Caco2 cells with human recombinant IFN-β
inhibited viral replication in the Caco2 cells by ~5 times compared
with the Vero cells (44).
Replication of HcoV-EMC was notably reduced when treated with type
I or type III IFN in the human airway epithelium culture (43,44).
A delay in the induction of the type I IFN response
enables SARS-CoV to replicate efficiently in mice and augments the
accumulation of inflammatory monocyte-macrophages (45). A lack of type I and type III IFN
responses in signal transducer and activator transcription-1
knockout mice resulted in uncontrolled SARS-CoV replication with
both liver and neurological consequences (46). Treatment of MERS-CoV-infected
patients with IFN-α2a improved the survival rates to a maximum of
14 days (43). The type I IFN and
TLR3 agonist were the most effective combined drugs for SARS/MERS
CoV treatment (26).
Regarding COVID-19 infections, a clinical trial
showed that treating hospitalised patients with IFN-α2b, either
alone or in combination with arbidol, shortened the time of
detectable viral presence in upper respiratory infections and
reduced the IL-6 and C-reactive protein levels (47). Addition of IFNs to the national
regime of treating COVID-19 patients reduced the 28-day mortality
rate (48). The antiviral effect was
augmented when Lopinavir-ritonavir was administered to mild or
moderate cases of COVID-19 in combination with IFN-β 1b, and the
enhancing effect was associated with a reduction in symptoms, the
length of stay in hospital and viral shedding (49). In terms of COVID-19 infections and
IFN responses, it was revealed that the reduced type I IFN levels
in the peripheral blood system increased the expression of IL-6 and
tumour necrosis factor (50). A
limited type I IFN response was detected concomitantly with a large
chemokine response, including production of IL-6, in the
transcriptomes of SARS-CoV2 infected cells (51). In contrast, increased type I IFN and
interferon stimulatory gene responses were reported in COVID-19
hospitalised patients. Several factors may underlie these
contradictory results, such as the individual immune systems of
patients, duration between initial infection and when the samples
were obtained, and the severity of the infection (52).
Based on the similarities between the results of the
present study and previous studies regarding the pattern of IFN
responses, it is hypothesized that IFNs may be used as a potential
treatment for management of COVID-19 infections. However, the
present study has some limitations. The data assessed was done so
irrespective of the severity of infections. Additionally, clinical
trials will be required to assess both the safety and efficacy of
IFN in managing COVID-19 infections.
In conclusion, increases in the gene expression of
the key regulator of type I interferon was not shown to be
effective and efficient in mounting an interferon response.
Acknowledgements
Not applicable.
Funding
Funding:No funding was received.
Availability of data and materials
The datasets used and/or analysed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
AAS and MHW completed the RNA extraction and
SARS-CoV-2 diagnosis. AAA-A and ZWA achieved the gene expression of
the target gene and data analysis. The writing of the study was
mainly conducted by ZWA. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Health
Directorate (approval no. F112020) and according to an application
that was made by the authors. All patients provided signed consent
to participate in the present study and gave their written consent
to publish any corresponding data.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Docea AO, Tsatsakis A, Albulescu D,
Cristea O, Zlatian O, Vinceti M, Moschos SA, Tsoukalas D, Goumenou
M, Drakoulis N, et al: A new threat from an old enemy: Re emergence
of coronavirus (Review). Int J Mol Med. 45:1631–1643.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Tsatsakis A, Petrakis D, Nikolouzakis TK,
Docea AO, Calina D, Vinceti M, Goumenou M, Kostoff RN, Mamoulakis
C, Aschner M, et al: COVID-19, an opportunity to reevaluate the
correlation between long-term effects of anthropogenic pollutants
on viral epidemic/pandemic events and prevalence. Food Chem
Toxicol. 141(111418)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Conti P, Caraffa A, Gallenga CE, Ross R,
Kritas SK, Frydas I, Younes A and Ronconi G: Coronavirus-19
(SARS-CoV-2) induces acute severe lung inflammation via IL-1
causing cytokine storm in COVID-19: A promising inhibitory
strategy. J Biol Regul Homeost Agents. 34:1971–1975.
2020.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Shi H, Wang W, Yin J, Ouyang Y, Pang L,
Feng Y, Qiao L, Guo X, Shi H, Jin R, et al: The inhibition of
IL-2/IL-2R gives rise to CD8+ T cell and lymphocyte
decrease through JAK1-STAT5 in critical patients with COVID-19
pneumonia. Cell Death Dis. 11(429)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Del Valle DM, Kim-Schulze S, Huang HH,
Beckmann ND, Nirenberg S, Wang B, Lavin Y, Swartz TH, Madduri D,
Stock A, et al: An inflammatory cytokine signature predicts
COVID-19 severity and survival. Nat Med. 26:1636–1643.
2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Han H, Ma Q, Li C, Liu R, Zhao L, Wang W,
Zhang P, Liu X, Gao G, Liu F, et al: Profiling serum cytokines in
COVID-19 patients reveals IL-6 and IL-10 are disease severity
predictors. Emerg Microbes Infect. 9:1123–1130. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhu FC, Guan XH, Li YH, Huang JY, Jiang T,
Hou LH, Li JX, Yang BF, Wang L, Wang WJ, et al: Immunogenicity and
safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine
in healthy adults aged 18 years or older: A randomised,
double-blind, placebo-controlled, phase 2 trial. Lancet.
396:479–488. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yu J, Tostanoski LH, Peter L, Mercado NB,
McMahan K, Mahrokhian SH, Nkolola JP, Liu J, Li Z, Chandrashekar A,
et al: DNA vaccine protection against SARS-CoV-2 in rhesus
macaques. Science. 369:806–811. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Calina D, Docea AO, Petrakis D, Egorov AM,
Ishmukhametov AA, Gabibov AG, Shtilman MI, Kostoff R, Carvalho F,
Vinceti M, et al: Towards effective COVID-19 vaccines: Updates,
perspectives and challenges (Review). Int J Mol Med. 46:3–16.
2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Isaacs A and Lindenmann J: Virus
interference. I. The interferon. Proc R Soc Lond B Biol Sci.
147:258–267. 1957.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Vilcek J and Sen GC: Interferons and other
cytokines. In: Fields Virology. Fields BN, Knipe DM and Howley PM
(eds). 3rd edition. Lippincott-Raven Publishers, Philadelphia, PA,
pp375-399, 1996.
|
|
12
|
Deonarain R, Alcami A, Alexiou M, Dallman
MJ, Gewert DR and Porter AC: Impaired antiviral response and
alpha/β interferon induction in mice lacking beta interferon. J
Virol. 74:3404–3409. 2000.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sato M, Suemori H, Hata N, Asagiri M,
Ogasawara K, Nakao K, Nakaya T, Katsuki M, Noguchi S, Tanaka N, et
al: Distinct and essential roles of transcription factors IRF-3 and
IRF-7 in response to viruses for IFN-alpha/beta gene induction.
Immunity. 13:539–548. 2000.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Collins SE, Noyce RS and Mossman KL:
Innate cellular response to virus particle entry requires IRF3 but
not virus replication. J Virol. 78:1706–1717. 2004.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Taniguchi T, Ogasawara K, Takaoka A and
Tanaka N: IRF family of transcription factors as regulators of host
defense. Annu Rev Immunol. 19:623–655. 2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Garoufalis E, Kwan I, Lin R, Mustafa A,
Pepin N, ARoulston A, Lacoste J and Hiscott J: Viral induction of
the human beta interferon promoter: modulation of transcription by
NF-kappaB/rel proteins and interferon regulatory factors. J Virol.
68:4707–4715. 1994.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Paun A and Pitha PM: The IRF family,
revisited. Biochimie. 89:744–753. 2007.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Génin P, Vaccaro A and Civas A: The role
of differential expression of human interferon - a genes in
antiviral immunity. Cytokine Growth Factor Rev. 20:283–295.
2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ye J and Maniatis T: Negative regulation
of interferon-beta gene expression during acute and persistent
virus infections. PLoS One. 6(e20681)2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Weaver BK, Kumar KP and Reich NC:
Interferon regulatory factor 3 and CREB-binding protein/p300 are
subunits of double-stranded RNA-activated transcription factor
DRAF1. Mol Cell Biol. 18:1359–1368. 1998.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Takeuchi O and Akira S: MDA5/RIG-I and
virus recognition. Curr Opin Immunol. 20:17–22. 2008.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kato H, Sato S, Yoneyama M, Yamamoto M,
Uematsu S, Matsui K, Tsujimura T, Takeda K, Fujita T, Takeuchi O,
et al: Cell type-specific involvement of RIG-I in antiviral
response. Immunity. 23:19–28. 2005.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Kato H, Takeuchi O, Sato S, Yoneyama M,
Yamamoto M, Matsui K, Uematsu S, Jung A, Kawai T, Ishii KJ, et al:
Differential roles of MDA5 and RIG-I helicases in the recognition
of RNA viruses. Nature. 441:101–105. 2006.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ye Y, Hauns K, Langland JO, Jacobs BL and
Hogue BG: Mouse hepatitis coronavirus A59 nucleocapsid protein is a
type I interferon antagonist. J Virol. 81:2554–2563.
2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Huang Z and Tunnacliffe A: Response of
human cells to desiccation: comparison with hyperosmotic stress
response. J Physiol. 558:181–191. 2004.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Strayer DR, Dickey R and Carter WA:
Sensitivity of SARS/MERS CoV to interferons and other drugs based
on achievable serum concentrations in humans. Infect Disord Drug
Targets. 14:37–43. 2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Tsatsakis A, Calina D, Falzone L, Petrakis
D, Mitrut R, Siokas V, Pennisi M, Lanza G, Libra M, Doukas SG, et
al: SARS-CoV-2 pathophysiology and its clinical implications: An
integrative overview of the pharmacotherapeutic management of
COVID-19. Food Chem Toxicol. 146(111769)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Nakamura M, Funami K, Komori A, Yokoyama
T, Aiba Y, Araki A, Takii Y, Ito M, Matsuyama M, Koyabu M, et al:
Increased expression of Toll-like receptor 3 in intrahepatic
biliary epithelial cells at sites of ductular reaction in diseased
livers. Hepatol Int. 2:222–230. 2008.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Liu LM, Tu WJ, Zhu T, Wang XT, Tan ZL,
Zhong H, Gao DY and Liang DY: IRF3 is an important molecule in the
UII/UT system and mediates immune inflammatory injury in acute
liver failure. Oncotarget. 7:49027–49041. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Xu RH, Schuster DM, Lee JE, Smith M,
Potter J, Dhariwal G, Rosenthal K, Nathan M, Gerard GF and
Rashtchian A: One-step analysis and quantification of RNA by
RT-PCR: Using high-temperature reverse transcription. Focus.
22:3–5. 2000.
|
|
31
|
Génin P, Morin P and Civas A: Impairment
of interferon-induced IRF-7 gene expression due to inhibition of
ISGF3 formation by trichostatin A. J Virol. 77:7113–7119.
2003.PubMed/NCBI View Article : Google Scholar
|
|
32
|
de Lang A, Baas T, Smits SL, Katze MG,
Osterhaus AD and Haagmans BL: Unraveling the complexities of the
interferon response during SARS-CoV infection. Future Virol.
4:71–78. 2009.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chan RW, Chan MC, Agnihothram S, Chan LL,
Kuok DI, Fong JH, Guan Y, Poon LL, Baric RS, Nicholls JM, et al:
Tropism of and innate immune responses to the novel human
betacoronavirus lineage C virus in human ex vivo respiratory organ
cultures. J Virol. 87:6604–6614. 2013.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Tang BS, Chan KH, Cheng VC, Woo PC, Lau
SK, Lam CC, Chan TL, Wu AK, Hung IF, Leung SY, et al: Comparative
host gene transcription by microarray analysis early after
infection of the Huh7 cell line by severe acute respiratory
syndrome coronavirus and human coronavirus 229E. J Virol.
79:6180–6193. 2005.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Au WC, Moore PA, Lowther W, Juang YT and
Pitha PM: Identification of a member of the interferon regulatory
factor family that binds to the interferon-stimulated response
element and activates expression of interferon-induced genes. Proc
Natl Acad Sci USA. 92:11657–11661. 1995.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Atwan Z, Wright J, Woodman A and Leppard
KN: Promyelocytic leukemia protein isoform II inhibits infection by
human adenovirus type 5 through effects on HSP70 and the interferon
response. J Gen Virol. 97:1955–1967. 2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Walker WE, Bozzi AT and Goldstein DR: IRF3
contributes to sepsis pathogenesis in the mouse cecal ligation and
puncture model. J Leukoc Biol. 92:1261–1268. 2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Spiegel M, Pichlmair A, Martínez-Sobrido
L, Cros J, García-Sastre A, Haller O and Weber F: Inhibition of
Beta interferon induction by severe acute respiratory syndrome
coronavirus suggests a two-step model for activation of interferon
regulatory factor 3. J Virol. 79:2079–2086. 2005.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Loukugamage KG, Hage A, de Vries M,
Valero-Jimenez AM, Schindewolf C, Dittmann M, Rajsbaum R and
Menachery VD: SARS-CoV-2 sensitive to type I interferon
pretreatment. bioRxiv: Apr 9, 2020 (Epub ahead of print). doi:
10.1101/2020.03.07.982264.
|
|
40
|
Zhao X, Guo F, Liu F, Cuconati A, Chang J,
Block TM and Guo JT: Interferon induction of IFITM proteins
promotes infection by human coronavirus OC43. Proc Natl Acad Sci
USA. 111:6756–6761. 2014.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Snijder EJ, van der Meer Y,
Zevenhoven-Dobbe J, Onderwater JJ, van der Meulen J, Koerten HK and
Mommaas AM: Ultrastructure and origin of membrane vesicles
associated with the severe acute respiratory syndrome coronavirus
replication complex. J Virol. 80:5927–5940. 2006.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Stertz S, Reichelt M, Spiegel M, Kuri T,
Martinez-Sobrido L, Garcia-Sastre A, Weber F and Koch G: The
intracellular sites of early replication and budding of
SARS-coronaviru. Virology. 361:304–315. 2007.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Cinatl J, Morgenstern B, Bauer G, Chandra
P, Rabenau H and Doerr HW: Treatment of SARS with human
interferons. Lancet. 362:293–294. 2003.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Mahlakõiv T, Ritz D, Mordstein M, DeDiego
ML, Enjuanes L, Müller MA, Drosten C and Staeheli P: Combined
action of type I and type III interferon restricts initial
replication of severe acute respiratory syndrome coronavirus in the
lung but fails to inhibit systemic virus spread. J Gen Virol.
93:2601–2605. 2012.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Channappanavar R, Fehr AR, Vijay R, Mack
M, Zhao J, Meyerholz DK and Perlman S: Dysregulated type I
interferon and inflammatory monocyte-macrophage responses cause
lethal pneumonia in SARS-CoV-infected mice. Cell Host Microbe.
19:181–193. 2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Zhou Q, Chen V, Shannon CP, Wei XS, Xiang
X, Wang X, Wang ZH, Tebbutt SJ, Kollmann TR and Fish EN:
Interferon-α2b Treatment for COVID-19. Front Immunol.
11(1061)2020.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Davoudi-Monfared E, Rahmani H, Khalili H,
Hajiabdolbaghi M, Salehi M, Abbasian L, Kazemzadeh H and
Yekaninejad MS: A randomized clinical trial of the efficacy and
safety of interferon beta-1a in treatment of severe COVID-19.
Antimicrob Agents Chemother. 64:e01061–e20. 2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Hung IF, Lung KC, Tso EY, Liu R, Chung TW,
Chu MY, Ng YY, Lo J, Chan J, Tam AR, et al: Triple combination of
interferon beta-1b, lopinavir-ritonavir, and ribavirin in the
treatment of patients admitted to hospital with COVID-19: an
open-label, randomised, phase 2 trial. Lancet. 395:1695–1704.
2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Hadjadj J, Yatim N, Barnabei L, Corneau A,
Boussier J, Smith N, Péré H, Charbit B, Bondet V, Chenevier-Gobeaux
C, et al: Impaired type I interferon activity and inflammatory
responses in severe COVID-19 patients. Science. 369:718–724.
2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Blanco-Melo D, Nilsson-Payant BE, Liu WC,
Uhl S, Hoagland D, Møller R, Jordan TX, Oishi K, Panis M, Sachs D,
et al: Imbalanced Host Response to SARS-CoV-2 Drives Development of
COVID-19. Cell. 181:1036–1045.e9. 2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Wilk AJ, Rustagi A, Zhao NQ, Roque J,
Martínez-Colón GJ, McKechnie JL, Ivison GT, Ranganath T, Vergara R,
Hollis T, et al: A single-cell atlas of the peripheral immune
response in patients with severe COVID-19. Nat Med. 26:1070–1076.
2020.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Lee JS and Shin EC: The type I interferon
response in COVID-19: Implications for treatment. Nat Rev Immunol.
20:585–586. 2020.PubMed/NCBI View Article : Google Scholar
|