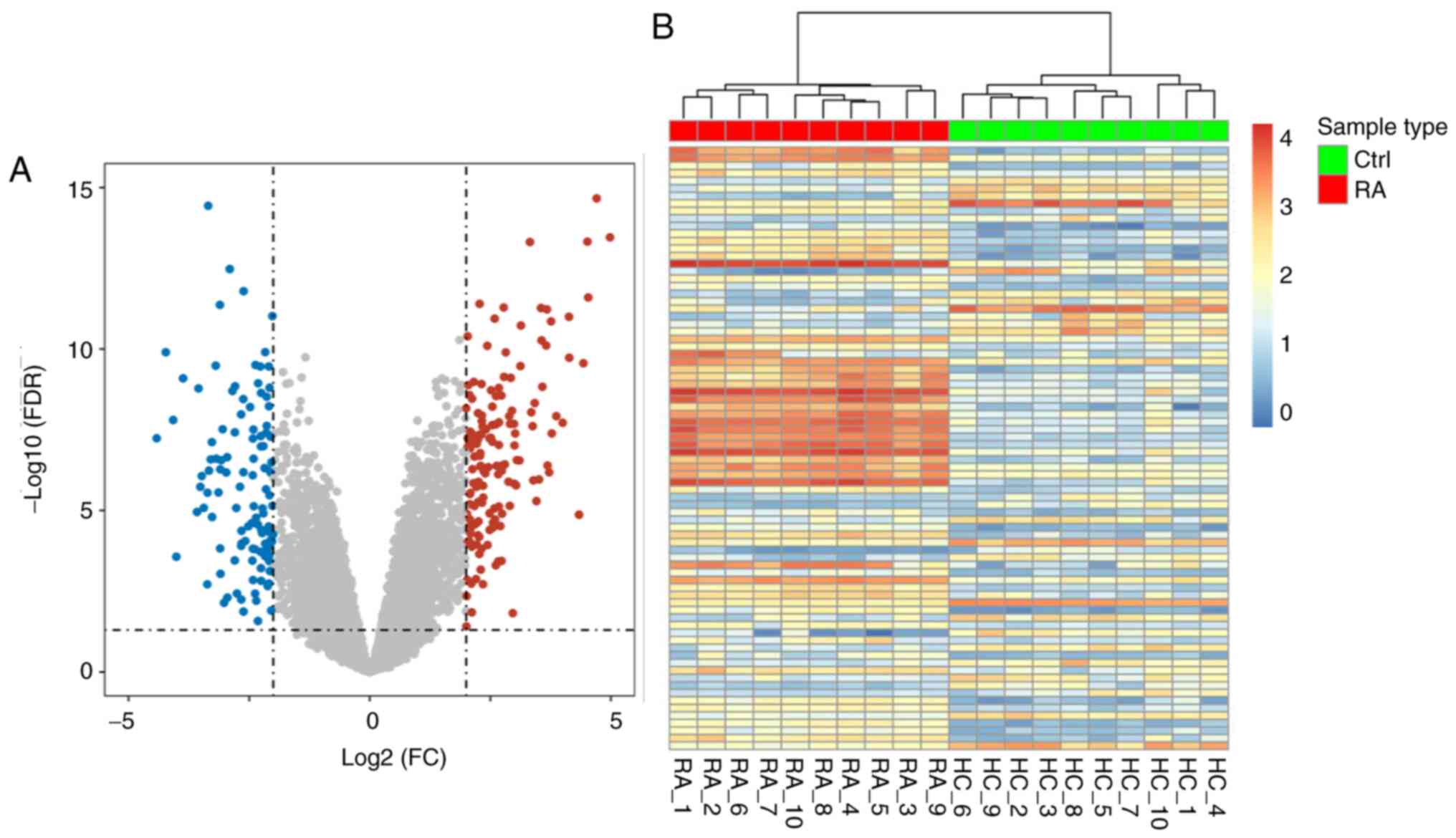
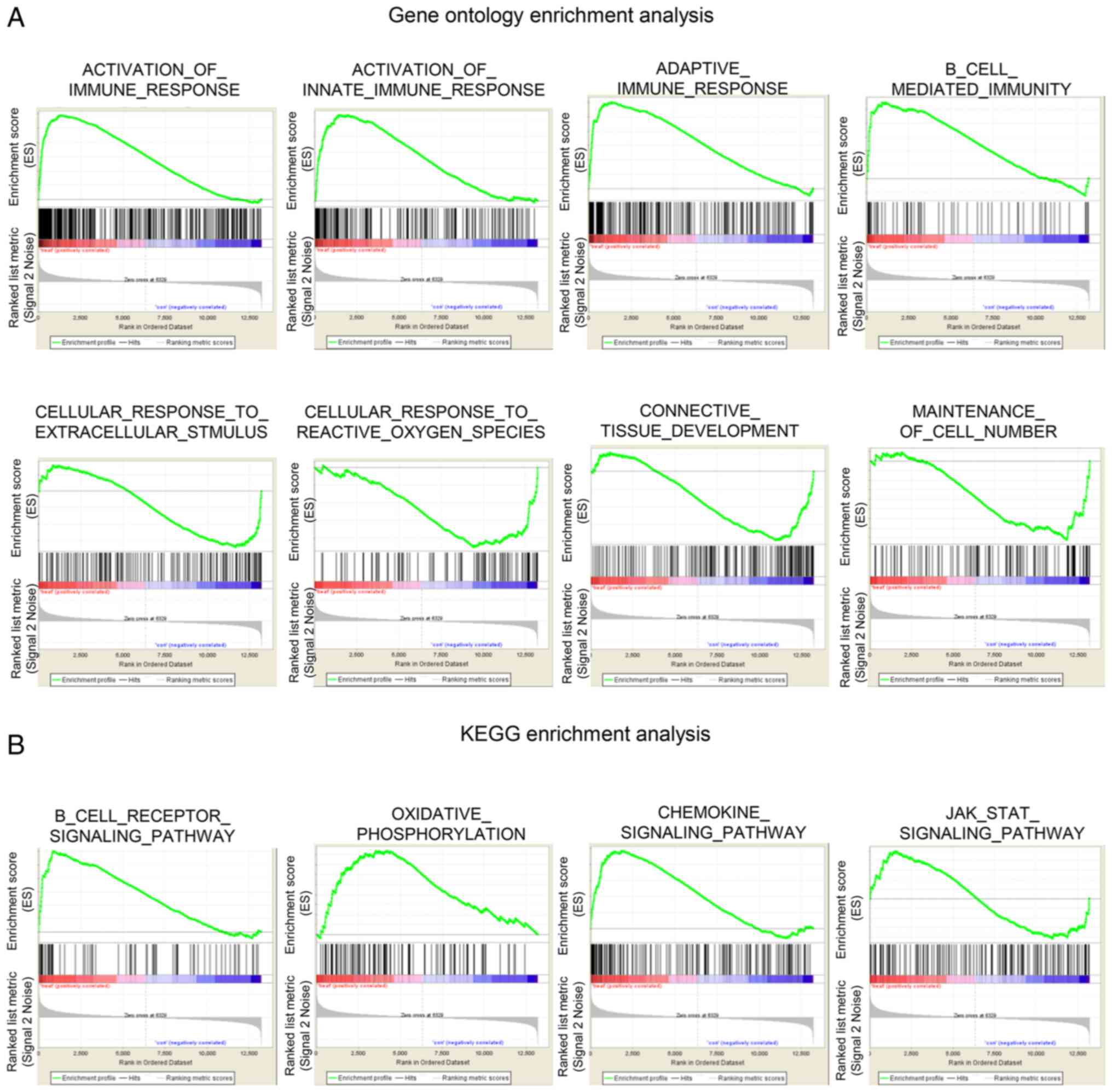
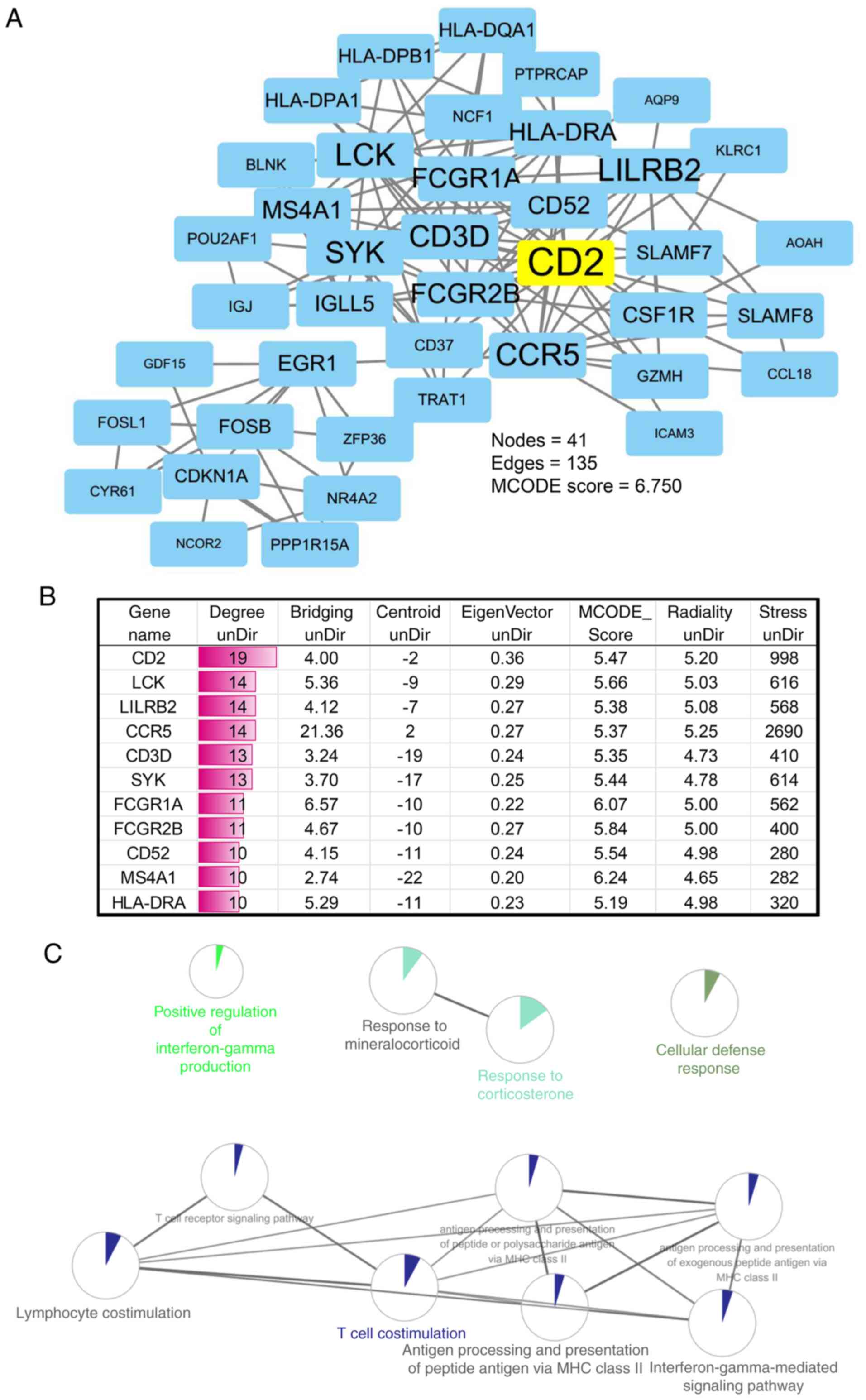
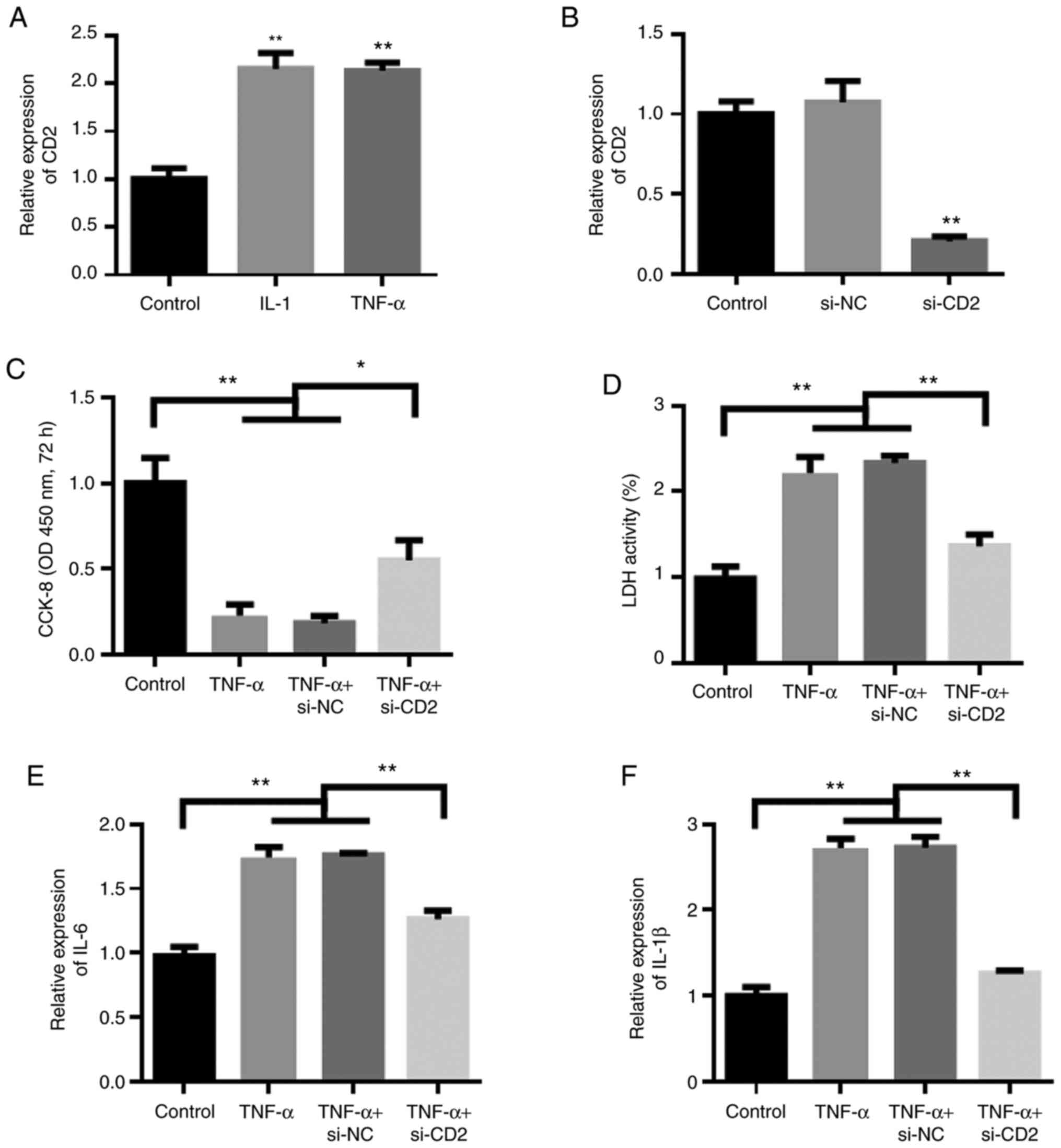
|
1
|
Falconer J, Murphy AN, Young SP, Clark AR,
Tiziani S, Guma M and Buckley CD: Review: Synovial Cell Metabolism
and Chronic Inflammation in Rheumatoid Arthritis. Arthritis
Rheumatol. 70:984–999. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Firestein GS and McInnes IB:
Immunopathogenesis of Rheumatoid Arthritis. Immunity. 46:183–196.
2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kim SY, Schneeweiss S, Liu J, Daniel GW,
Chang CL, Garneau K and Solomon DH: Risk of osteoporotic fracture
in a large population-based cohort of patients with rheumatoid
arthritis. Arthritis Res Ther. 12(R154)2010.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Källberg H, Ding B, Padyukov L, Bengtsson
C, Rönnelid J, Klareskog L and Alfredsson L: EIRA Study Group.
Smoking is a major preventable risk factor for rheumatoid
arthritis: Estimations of risks after various exposures to
cigarette smoke. Ann Rheum Dis. 70:508–511. 2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Klareskog L, Gregersen PK and Huizinga TW:
Prevention of autoimmune rheumatic disease: State of the art and
future perspectives. Ann Rheum Dis. 69:2062–2066. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Viatte S, Plant D, Bowes J, Lunt M, Eyre
S, Barton A and Worthington J: Genetic markers of rheumatoid
arthritis susceptibility in anti-citrullinated peptide antibody
negative patients. Ann Rheum Dis. 71:1984–1990. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hogan DB: Did Osler suffer from ‘paranoia
antitherapeuticum baltimorensis’? A comparative content analysis of
The Principles and Practice of Medicine and Harrison's Principles
of Internal Medicine, 11th edition. CMAJ. 161:842–845.
1999.PubMed/NCBI
|
|
8
|
McInnes IB and Schett G: Pathogenetic
insights from the treatment of rheumatoid arthritis. Lancet.
389:2328–2337. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Reis-Filho JS and Pusztai L: Gene
expression profiling in breast cancer: Classification,
prognostication, and prediction. Lancet. 378:1812–1823.
2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tan L, Yu JT, Tan MS, Liu QY, Wang HF,
Zhang W, Jiang T and Tan L: Genome-wide serum microRNA expression
profiling identifies serum biomarkers for Alzheimer's disease. J
Alzheimers Dis. 40:1017–1027. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shen Y, Jiang T, Wang R, He S, Guo M, Zuo
J and He D: (5R)-5-Hydroxytriptolide (LLDT-8) inhibits
osteoclastogenesis via RANKL/RANK/OPG signaling pathway. BMC
Complement Altern Med. 15(77)2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chen Y, Zhang L, Ni J, Wang X, Cheng J, Li
Y, Zhen X, Cao T and Jia J: LLDT-8 protects against cerebral
ischemia/reperfusion injury by suppressing post-stroke
inflammation. J Pharmacol Sci. 131:131–137. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ren YX, Zhou R, Tang W, Wang WH, Li YC,
Yang YF and Zuo JP: (5R)-5-hydroxytriptolide (LLDT-8) protects
against bleomycin-induced lung fibrosis in mice. Acta Pharmacol
Sin. 28:518–525. 2007.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Xie ZC, Dang YW, Wei DM, Chen P, Tang RX,
Huang Q, Liu JH and Luo DZ: Clinical significance and prospective
molecular mechanism of MALAT1 in pancreatic cancer exploration: A
comprehensive study based on the GeneChip, GEO, Oncomine, and TCGA
databases. OncoTargets Ther. 10:3991–4005. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Woetzel D, Huber R, Kupfer P, Pohlers D,
Pfaff M, Driesch D, Häupl T, Koczan D, Stiehl P, Guthke R, et al:
Identification of rheumatoid arthritis and osteoarthritis patients
by transcriptome-based rule set generation. Arthritis Res Ther.
16(R84)2014.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Guo S, Liu J, Jiang T, Lee D, Wang R, Zhou
X, Jin Y, Shen Y, Wang Y, Bai F, et al: (5R)-5-Hydroxytriptolide
(LLDT-8) induces substantial epigenetic mediated immune response
network changes in fibroblast-like synoviocytes from rheumatoid
arthritis patients. Sci Rep. 9(11155)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW,
Shi W and Smyth GK: limma powers differential expression analyses
for RNA-sequencing and microarray studies. Nucleic Acids Res.
43:e47. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
R Core Team: A Language and Environment
for Statistical Computing. R Foundation for Statistical Computing,
Vienna, 2013. Available from https://www.R-project.org/.
|
|
19
|
Team R: RStudio: Integrated Development
for R. RStudio, Inc., Boston MA, 2015. Available from URL
http://www.rstudio.com.
|
|
20
|
Xi X, Liu N, Wang Q, Chu Y, Yin Z, Ding Y
and Lu Y: ACT001, a novel PAI-1 inhibitor, exerts synergistic
effects in combination with cisplatin by inhibiting PI3K/AKT
pathway in glioma. Cell Death Dis. 10(757)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Xi X, Chu Y, Liu N, Wang Q, Yin Z, Lu Y
and Chen Y: Joint bioinformatics analysis of underlying potential
functions of hsa-let-7b-5p and core genes in human glioma. J Transl
Med. 17(129)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhong W, Sun B, Gao W, Qin Y, Zhang H,
Huai L, Tang Y, Liang Y, He L, Zhang X, et al: Salvianolic acid A
targeting the transgelin-actin complex to enhance vasoconstriction.
EBioMedicine. 37:246–258. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhong W, Yang W, Qin Y, Gu W, Xue Y, Tang
Y, Xu H, Wang H, Zhang C, Wang C, et al: 6-Gingerol stabilized the
p-VEGFR2/VE-cadherin/β-catenin/actin complex promotes microvessel
normalization and suppresses tumor progression. J Exp Clin Cancer
Res. 38(285)2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yu G, Wang LG, Han Y and He QY:
clusterProfiler: An R package for comparing biological themes among
gene clusters. OMICS. 16:284–287. 2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2-ΔΔCT method. methods. 25:402–408. 2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Scardoni G, Petterlini M and Laudanna C:
Analyzing biological network parameters with CentiScaPe.
Bioinformatics. 25:2857–2859. 2009.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Bader GD and Hogue CW: An automated method
for finding molecular complexes in large protein interaction
networks. BMC Bioinformatics. 4(2)2003.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Szklarczyk D, Gable AL, Lyon D, Junge A,
Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork
P, et al: STRING v11: Protein-protein association networks with
increased coverage, supporting functional discovery in genome-wide
experimental datasets. Nucleic Acids Res. 47:D607–D613.
2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Bodian DL, Jones EY, Harlos K, Stuart DI
and Davis SJ: Crystal structure of the extracellular region of the
human cell adhesion molecule CD2 at 2.5 A resolution. Structure.
2:755–766. 1994.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhong W, Liu P, Zhang Q, Li D and Lin J:
Structure-based QSAR, molecule design and bioassays of
protease-activated receptor 1 inhibitors. J Biomol Struct Dyn.
35:2853–2867. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
DeLano WL: Pymol: An open-source molecular
graphics tool. CCP4 Newsletter on protein crystallography.
40:82–92. 2002.
|
|
33
|
Reimand J, Isserlin R, Voisin V, Kucera M,
Tannus-Lopes C, Rostamianfar A, Wadi L, Meyer M, Wong J, Xu C, et
al: Pathway enrichment analysis and visualization of omics data
using g:Profiler, GSEA, Cytoscape and EnrichmentMap. Nat Protoc.
14:482–517. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Catrina AI, Joshua V, Klareskog L and
Malmström V: Mechanisms involved in triggering rheumatoid
arthritis. Immunol Rev. 269:162–174. 2016.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Glaab E, Baudot A, Krasnogor N and
Valencia A: TopoGSA: Network topological gene set analysis.
Bioinformatics. 26:1271–1272. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bao R, Huang L, Andrade J, Tan W, Kibbe
WA, Jiang H and Feng G: Review of current methods, applications,
and data management for the bioinformatics analysis of whole exome
sequencing. Cancer Inform. 13 (Suppl 2):67–82. 2014.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yang JJ, Ye Y, Carroll A, Yang W and Lee
HW: Structural biology of the cell adhesion protein CD2:
Alternatively folded states and structure-function relation. Curr
Protein Pept Sci. 2:1–17. 2001.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Watzl C and Long EO: Signal transduction
during activation and inhibition of natural killer cells. Curr
Protoc Immunol. 11(17)2010.PubMed/NCBI View Article : Google Scholar
|
|
39
|
James JR and Vale RD: Biophysical
mechanism of T-cell receptor triggering in a reconstituted system.
Nature. 487:64–69. 2012.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Wong KF, Yuan Y and Luk JM:
Tripterygium wilfordii bioactive compounds as anticancer and
anti-inflammatory agents. Clin Exp Pharmacol Physiol. 39:311–320.
2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Bao J and Dai SM: A Chinese herb
Tripterygium wilfordii Hook F in the treatment of rheumatoid
arthritis: Mechanism, efficacy, and safety. Rheumatol Int.
31:1123–1129. 2011.PubMed/NCBI View Article : Google Scholar
|