Introduction
In osteoarthritis (OA), biomechanical forces
constantly damage avascular chondrocytes and change their phenotype
with intensive production of pro-inflammatory cytokines (1). Increased concentrations of
proinflammatory mediators IL-1β, IL-18, IL-15, IL-21, IL-27, IFN-γ,
TNF-α, and tolerogenic IL-10 were found in the synovial fluid
and/or peripheral blood of OA patients compared to that in controls
(1,2). This is consistent with the hypothesis
that locally produced inflammatory substances, supported by
synovial dendritic cells and polarized macrophages, leak from OA
joints into the circulation and induce a systemic immune response
mediated by activated lymphocytes during periods of painful OA
(3). IL-15(4), IL-21(5) and IL-27(6) act via the Janus kinase/signal
transducer and activator of transcription (JAK/STAT) pathway, and
are crucial factors in the natural killer (NK) cell-associated
innate immune response (7). IL-1β
and TNF-α act alongside NF-κB (8),
which exhibits upregulated expression levels in OA joints, and
promotes cellular interaction, proliferation and cytotoxicity
(1).
Granulysin (GNLY), a constitutive cytotoxic mediator
in NK cells, is upregulated in T cells in a pro-inflammatory
environment (9,10). After direct stimulation of effector
cells in close contact with targets, the cytotoxic GNLY form (9
kDa) is released into the immunological synapse, where it enters
the target cells through the perforin pores (10), and kills human cells by inducing
apoptosis and DNA fragmentation (9,11). The
15 kDa form of GNLY is released from activated cells spontaneously
without direct effector-target cell contact and shows regulatory
and chemotactic properties in the dendritic cells (12), but is not cytotoxic (13). To the best of our knowledge, no data
exist regarding the expression and cytotoxic/apoptotic mechanisms
mediated by GNLY in the peripheral blood of the OA patients, given
the mildly pro-inflammatory environment (14-16).
We analyzed GNLY, IFN-γ and IL-4 expression in peripheral blood
lymphocytes (PBLs), GNLY-mediated cytotoxicity of NK cells at the
effector K-562 target cell, and GNLY serum concentration in the OA
patients.
Materials and methods
Patients
A total of 40 women with knee OA and 40 control
individuals without knee OA were enrolled in a prospective study in
the Thallassotherapia-Opatija Hospital, Opatija, Croatia, between
the 1st of July 2018 and the 15th of December 2020. The study was
approved by the Ethical Committee of the Hospital
‘Thallassotherapia-Opatija’ (approval no. 01-000-00-450/2018) and
all experimental procedures were performed according to the
‘Ethical Principles for Medical Research Involving Human Subjects’
outlined by the World Medical Association Declaration of Helsinki
(17). The median age and age range
of the OA group and controls were 64 years old (43-75) and 62 years
old (43-73), respectively.
Examinees were evaluated using the 1986 American
College of Rheumatology clinical classification criteria (18). Exclusion criteria for both groups
were: Infectious and autoimmune diseases, bone marrow and/or
lymphatic system disorders, immune deficiency, women of
reproductive age, uncontrolled blood glucose level (plasma glucose
>11 mmol/l), uncontrolled hypertension (systolic >180 mmHg or
diastolic >100 mmHg), chronic liver and/or renal failure, high
risk of heart failure as per the New York Heart Association
classification (19) III and IV,
and injury to organs, blood transfusions, and/or a malignant
disease within 5 years. Pain intensity was assessed using the
visual analog scale from 0 to 100 mm, as judged by the patients.
Radiological staging of OA in the knee was assessed using the
Kellgren-Lawrence Scale (20). The
clinical and laboratory characteristics of OA patients and controls
are shown in Table I. Out of the 40
OA patients, hypercholesterolemia was present in all patients
(100%), and glucose intolerance in 11 patients (27.5%); they were
controlled with a strict diet (without drug therapy). All controls
without knee OA had hypercholesterolemia and glucose intolerance
was present in 10 (25%) out of the 40 women, which were regulated
only with diet. Arterial hypertension was present in 33 OA patients
(82.5%) and 32 controls (80%); it was controlled with β-blockers
and/or angiotensin-converting enzyme inhibitors.
 | Table IDemographic, clinical and laboratory
characteristics of the OA patients and controls. |
Table I
Demographic, clinical and laboratory
characteristics of the OA patients and controls.
| Demographic
characteristics | OA patients | Controls |
|---|
| Age, years (age
range) | 64 (43-75) | 62 (43-73) |
| Clinical
characteristicsa | | |
|
Body mass
index, kg/m2 | 25 (23-29) | 26 (22-27) |
|
Duration of
diagnosis, years | 6 (3.5-12) | - |
|
Kellgren-Lawrence
Scale, grade | 2 (1-3) | - |
|
Medial knee
compartment involvement, n=40 | 40 | - |
|
Medial and
lateral knee compartment involvement, n=40 | 14 | - |
|
Morning
stiffness, min | 18 (10-20) | - |
|
Visual
analog scale for pain, 1-100 mm | 40 (20-50) | - |
|
Systolic
blood pressure, mmHg | 145 (130-145) | 140 (135-145) |
|
Diastolic
blood pressure, mmHg | 85 (80-105) | 88 (85-92) |
| Laboratory
characteristicsa | | |
|
High
sensitivity C-reactive protein, mg/l | 2.1 (1.8-3) | 2.0 (1.5-2.9) |
|
Erythrocytes,
x1012/l | 4.6 (4.5-4.8) | 4.6 (4.4-4.8) |
|
Hemoglobin,
g/l | 133 (132-147) | 135 (131-144) |
|
Leukocytes,
x109/l | 6.4 (6.2-6.6) | 6.9 (5.2-7.1) |
|
Thrombocytes,
x109/l | 264 (223-283) | 280 (222-325) |
|
Alanine
aminotransferase, IU/l | 35 (28-38) | 26 (17-30) |
|
Aspartate
aminotransferase, IU/l | 20 (20-22) | 18 (15-22) |
|
γ-glutamyl
transferase, IU/l | 21(16-27) | 17 (16-22) |
|
Urate,
mmol/l | 285 (232-308) | 233 (204-271) |
|
Creatinine,
mmol/l | 86 (78.5-91.5) | 75 (68-82) |
|
Glucose,
mmol/l | 5.5 (5.1-6.4) | 6.2 (5.1-6.4) |
|
Low density
lipoprotein-cholesterol, mmol/l | 3.9 (3.6-4.9) | 4.01 (3.1-5.2) |
|
Granulysin,
ng/ml | <0.3 | <0.3 |
Peripheral venous blood (10 ml) was drawn from the
patients and controls according to standard procedures; routine
laboratory tests (erythrocytes, hemoglobin, leukocytes,
thrombocytes, alanine aminotransferase, aspartate aminotransferase,
γ-glutamyl transferase, creatinine, urate and high sensitivity
C-reactive protein) were performed using the Hematology analyzer
XS-1000i (Sysmex, Kobe, Japan) and Biochemical analyzer Dimension
Xpand (Siemens Healthcare Diagnostics, Newark, DE, USA).
Isolation of peripheral blood
mononuclear cells (PBMCs)
PBMCs were isolated by gradient density
centrifugation (Lymphoprep solution, Nycomed Pharma; 600 x g, 20
min at 4˚C) (21). PBMCs were
collected and washed (400 x g, 10 min at 4˚C) in RPMI-1640 medium
(Invitrogen; Thermo Fisher Scientific, Inc.), re-suspended in
tissue culture medium [RPMI-1640 supplemented with L-glutamate (2
mM), penicillin (1x105 IU/l), streptomycin sulphate
(0.05 g/l), and 10% FBS; Gibco; Thermo Fisher Scientific, Inc.],
and counted. The cell viability exceeded 98% as measured using
propidium iodide (PI; 0.5 µg/ml/1x106 cells; 5 min at
22-25˚C; MerckMillipore) and a FACSCalibur flow cytometer with
CellQuest Pro software version 6.0 (both from Becton
Dickinson).
Antigen detection by flow
cytometry
The simultaneous detection of surface and
intracellular antigens was performed as described previously
(21,22). Primary antibodies, mouse IgG1
anti-human GNLY (cat. no. D-185-3; RC8 clone; MBL International) or
isotype-matched IgG1 (cat. no. bd554121; MOPC-21 clone; BD
Pharmingen) were added to cell samples (1 µg/106 cells,
30 min, 4˚C), which were previously washed (400 x g, 10 min at 4˚C)
in FACS buffer (140 mm NaCl, 1.9 mm KH2PO4,
16.5 mm Na2HPO4, 3.75 mm KCl (all from
Kemika), 0.96 mm Na2-EDTA (Fluka), 1.5 mm
NaN3 (Difco), fixed (4% paraformaldehyde, 10 min at
22-25˚C), permeabilized in saponin buffer (0.1% saponin, Sigma,
Poole, Dorset, USA), 2% fetal calf serum in PBS
(NaHPO4x12H2O 33.9 mM, NaCl 136.8 mM,
KH2PO4 3 mM of distilled water; all from
Kemika), 0.05% BSA (MerckMillipore), and incubated with 10%
heat-inactivated fetal calf serum for 20 min at 22-25˚C. The cells
were washed twice in saponin buffer (400 x g, 10 min at 4˚C) and
secondary goat anti-mouse polyclonal antibodies conjugated with
fluorescein isothiocyanate (FITC; cat. no. bd554001; BD Pharmingen,
1 µg/1x106 cells) were added for 30 min at 4˚C. After
two washes in saponin buffer, the cell membranes were restored
using 1 ml FACS buffer (5 min, at 22-25˚C). Cell surface labeling
was performed using a combination of phycoerythrin (PE)-conjugated
anti-CD3 (cat. no. bd555332, UCHT-1 clone; BD Pharmingen; 20
µl/1x106 cells) and PE-CyChrome5 (PE-Cy5)-conjugated
anti-CD56 (cat. no. bd569104, B159 clone; BD Pharmingen; 20
µl/1x106 cells) mAbs (30 min, 4˚C). Samples of PBMCs
(1x106/ml) were incubated with phorbol-myristate-acetate
(10 ng/ml), ionomycin (1 µM), and monensin (3 µM) (all from
MerckMillipore) for 5 h at 37˚C in a humidified incubator supplied
with 5% CO2 and intracellularly labeled for IFN-γ or
IL-4 in lymphocyte subsets using PE-Cy5-conjugated anti-CD56 (20
µl/1x106 cells), FITC-conjugated anti-CD3 (cat. no.
bd561808, UCHT1 clone, 20 µl/1x106 cells); and
PE-conjugated IFN-γ (cat. no. bd554701, B27 clone, 0.25
µg/1x106 cells) or PE-conjugated IL-4 (cat. no.
bd551774, 8D4-8 clone; 20 µl/1x106 cells) (all from BD
Pharmingen). Mouse IgG1 (MOPC-21 clone) conjugated with FITC (cat.
no. bd556649), PE-Cy5 (cat. no. bd555750), or PE (cat. no.
bd556650) (all 20 µl/1x106 cells; all from BD
Pharmingen) were used as isotype controls All the incubations were
performed at 4˚C for 30 min. Samples were resuspended in 2%
paraformaldehyde, and processed immediately using flow cytometry,
and analyzed using WinMDI 2.9 (The Scripps Research
Institute).
Immunocytochemistry
Cytospins were prepared from freshly isolated PBMCs
suspended in PBS/0.05% BSA to reach a final concentration of
8x105 cells/ml. Subsequently, 100 µl of the suspension
was centrifuged (Cytospin centrifuge; Shandon Inc.; 210 x g, 5 min
at 4˚C) onto a glass microscope slide (Marienfeld microscope; Paul
Marienfeld GmbH & Co.). The cytospin samples were air-dried (1
h) and subsequently fixed in pure acetone (5 min at 22-25˚C;
Kemika). Labeling of GNLY was performed using the immunoperoxidase
staining method with an LSAB™-HRP kit (Dako; Agilent Technologies,
Inc.). Briefly, sections preincubated (20 min) with
LSAB™-HRP kit blocking solution were incubated (30 min)
with anti-human GNLY mAb (RC8 clone) or mouse IgG1 (R312-Mouse IgG1
clone, Sigma Aldrich) diluted 1:100 in PBS/0.05% BSA. Incubation
(10 min) with biotinylated anti-mouse secondary antibodies was
followed by incubation with streptavidin for 10 min (both from
LSAB™-HRP kit). The reaction was developed using
aminoethyl carbazole (MerckMillipore); nuclei were counterstained
with hematoxylin (Gill III; MerckMillipore). All incubations were
performed at 22-25˚C; samples were washed twice in PBS for 5 min
between labeling steps. Subsequently, slides were mounted with
Entellan (MerckMillipore) and imaged using an Olympus BX51
microscope with an Olympus DP71 camera and CellA software, v3.0
(magnification, x1,000) (Olympus Corporation).
Confocal microscopy
Confocal microscopy was performed as described by
Donaldson (19). Samples were fixed
with 4% paraformaldehyde in PBS (10 min at 22-25˚C), permeabilized
with Triton X-100 (Rohm and Haas Corporate Headquarters, 7 min),
and incubated in 20% AB serum (Developed in-house) in PBS (45 min
at 22-25˚C). The samples were then incubated with a combination of
anti-human GNLY mAb (dilution 1:100, D185-3 clone) and rat IgG2a
anti-lysosomal-associated membrane protein (LAMP)-1 mAb (dilution
1:100, 1D4B clone, BD Pharmingen) in PBS/1% BSA at 4˚C overnight.
After washing, cells were labeled with secondary antibodies, Alexa
Fluor 594-conjugated donkey anti-mouse (IgG H+L polyclonal; cat.
no. A32744; Invitrogen; Thermo Fisher Scientific, Inc., dilution
1:500) and Alexa Fluor 488 goat anti-rat (IgG H+L polyclonal; cat.
no. A11006; Invitrogen; Thermo Fisher Scientific, Inc., dilution
1:300) in PBS for 1 h at 22-25˚C, washed, and mounted with Mowiol
(MerckMillipore). All washing steps were performed in PBS (three
washes, 3 min each). Images were taken using a confocal imaging
system (magnification x600; Olympus Fluoview FV3000) with CellA
software. The cells were randomly chosen and demonstrated a
well-resolved pattern.
Isolation of NK cells
Freshly isolated PBLs were obtained from the
non-adherent fraction of PBMCs after 45 min incubation in a tissue
culture Petri dish (60x15 mm; Techno Plastic Products). NK cells
were purified by negative magnetic cell separation using the NK
Cell Isolation Kit (cat. no. 130-092-657, Miltenyi Biotec, GmbH).
The NK Cell Biotin-Antibody Cocktail (10 µl) and 40 µl ice-cold
filtered FACS buffer were added to 1x107 pelleted PBLs,
mixed thoroughly, and incubated for 10 min at 4˚C. Subsequently, a
mixture of 30 µl FACS buffer and 20 µl NK Cell MicroBead Cocktail
was added per 1x107 total cells and refrigerated for an
additional 15 min (4˚C). The suspension was washed (300 x g, 10 min
at 4˚C) and the supernatant was aspirated and replaced with 500 µl
FACS buffer. The cell suspension was loaded onto a prewashed
medium-sized column and placed in the magnetic field of the
VarioMACS separator (both from Miltenyi Biotec, GmbH). Unlabeled
cells that passed through the column were collected as NK cells.
The purity and viability of the NK cells were >95%, as estimated
using flow cytometry.
K cytotoxicity assay
NK cell cytotoxicity was estimated by assessing
early apoptosis of K-562 cells (23). Human erythroleukemia K-562 cells
(Department of Physiology and Immunology, Faculty of Medicine,
University of Rijeka, Croatia) were labeled with PKH26 (5 min at
22-25˚C) red lipophilic dye (PKH 26 Red Fluorescent Cell Linker
Kit; MerckMillipore). The samples of effector NK and target K-562
cells, at different ratios in a total volume of 200 µl, were
incubated in tissue culture medium for 18 h at 37˚C with 5%
CO2, and labeled with FITC-conjugated Annexin V (BD
Pharmingen) (5 µg/105 cells, for 15 min at 22-25˚C in
the dark). PI, at a final concentration of 5 µg/ml, was added to
the samples 15-20 min before performing FACS using the FACSCalibur.
NK cell samples were untreated, pre-treated with mouse IgG2b
anti-human perforin (cat. no. NBP1-45774, δG9 clone; 1
µg/1x105 NK cells; Novus Biologicals), anti-human GNLY
(cat. no. D185-3, RC8 clone; MBL International, 1
µg/1x105 NK cells) or a combination of anti-perforin and
anti-GNLY mAbs for 30 min at 4˚C The subset of PKH26 labeled K-562
cells (stained red), detected as PI-negative and FITC-Annexin
V-positive, were considered early apoptotic cells (23). The results are expressed as the
difference in the percentage of early apoptotic K-562 cells in
pretreated samples at a particular effector to target cell ratio,
minus the percentage of apoptotic K-562 cells cultured in the
medium only.
ELISA
Serum GNLY concentrations were measured using the
LEGEND MAX™ Human Granulysin ELISA Kit (cat. no. 438007;
BioLegend, Inc.). The sera of OA patients and controls were
prepared from peripheral venous blood samples (3 ml/sample), which
were taken by venipuncture in plastic Serum Separator Tubes, 3.5 ml
(Greiner Bio-One GmbH), allowed to clot for 20 min at 22-25˚C, and
centrifuged (1,000 x g, 10 min, at 4˚C). The supernatant (serum)
was collected in Cryo.S, 2 ml, round bottom, screw-cap vial
(Greiner Bio-One GmbH) and stored at -80˚C until required for
ELISA. Assays were performed following the manufacturer's
instructions, and the absorbance was measured using an MRX
Revelation microplate reader (Dynex Technologies Inc.).
Statistical analysis
Data are presented as the median (25-75th
percentile). The difference between two groups was assessed using
the non-parametric Mann-Whitney U test (TIBCO Statistica, version
13.4.0.14). Differences between multiple groups were calculated
using a Kruskal-Wallis non-parametric test and a post hoc Dunn's
test using MedCalc Statistical version 20.011 (MedCalc Software
Ltd.). P<0.05 was considered to indicate a statistically
significant difference.
Results
Clinical and laboratory
characteristics of the patients
Table I compares the
clinical and laboratory characteristics of the group of
postmenopausal women with primary OA and controls. The median OA
patients' BMI (25 and 75th percentiles) of 25 kg/m2
(23-29 kg/m2) was comparable to that of the control
group (26; 22-27 kg/m2), as were the values for systolic
and diastolic blood pressure and glucose and LDL-cholesterol
concentrations. Routine hematological parameters (erythrocytes,
hemoglobin, leukocytes, thrombocytes), liver parameters (alanine
aminotransferase, aspartate aminotransferase, γ-glutamyl
transferase), creatinine, urate and high sensitivity C-reactive
protein levels did not differ between the groups. The patients were
diagnosed with primary OA of the knee 6 (3.5-12) years ago. At the
time of participation in the study, OA patients suffered from a
pain intensity of 40/100 (20/100-50/100) mm and morning stiffness
lasting 18 (10-20) min. Radiological staging of the disease in the
affected joint was 2 (1-3)
according to the Kellgren-Lawrence Scale. The medial knee
compartment was affected in all 40 patients with OA, whereas both
compartments (medial and lateral) were affected in 14/40 OA
patients (35%).
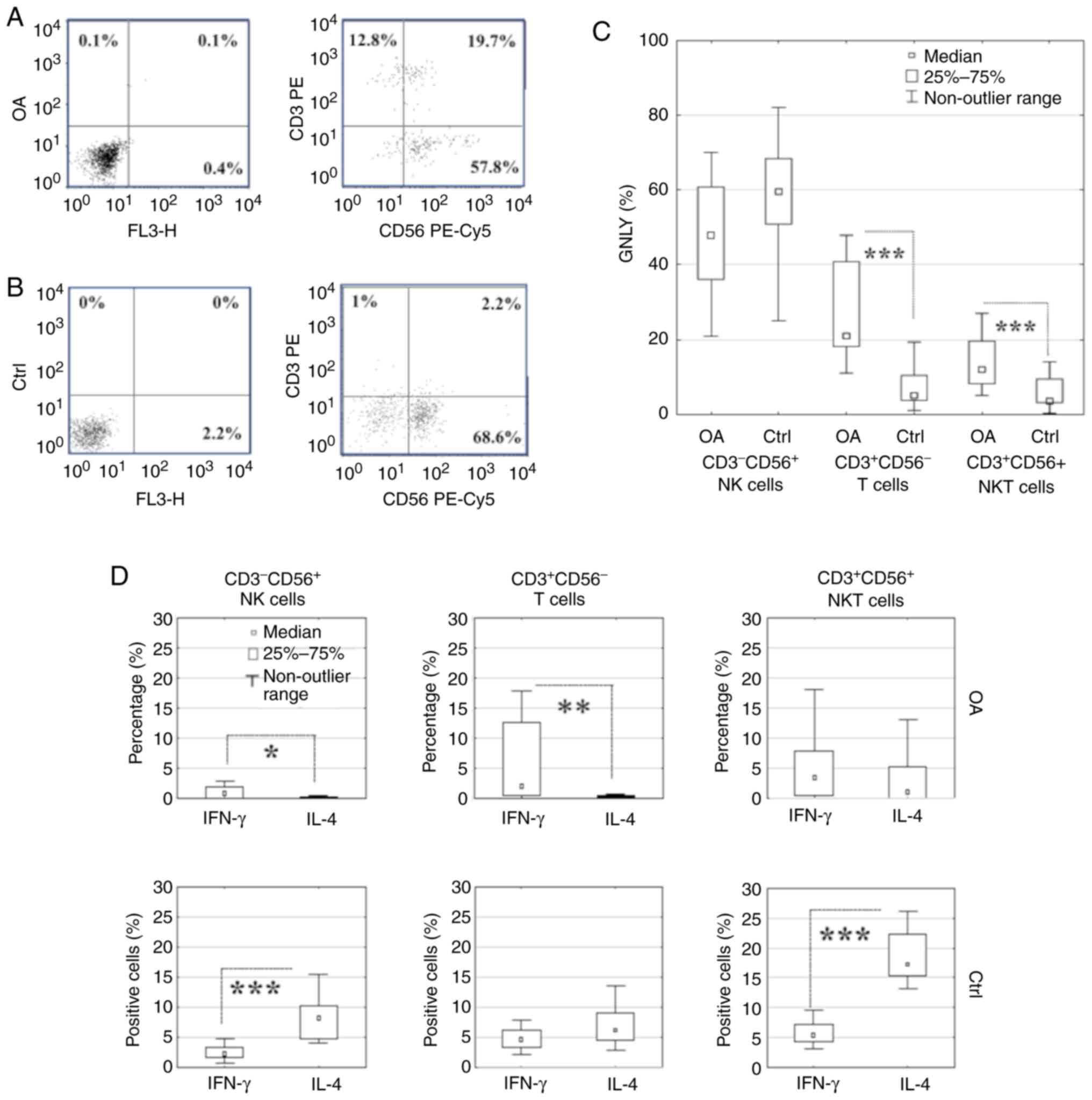
GNLY expression
Fig. 1 illustrates
GNLY expression in PBLs. All PBLs were observed to be within the
lymphocyte gate region (R)1 in the forward scatter/side scatter dot
plot in the OA patients (Fig. 1A)
and controls (Fig. 1B). The dot
plot of OA patients revealed 8.9% of GNLY+ cells gated
in R2 compared to the IgG1 isotype control (0.2%), visualized in
the histogram (Fig. 1A); 3.2%
GNLY+ cells were found in the controls when compared
with the isotype control (0.9%) (Fig.
1B). Immunoperoxidase staining was used to detect higher levels
of GNLY in the OA patients compared with the controls, observed as
red-labeled granules (Fig. 1C).
Confocal microscopy provided better insight into the colocalization
of GNLY (red fluorescence) and LAMP-1, a marker of exocytosis
(green fluorescence) in OA patients (Fig. 1D) and controls (Fig. 1E). The colocalization of GNLY and
LAMP-1 exhibited yellow fluorescence, resulting from the
overlapping of green and red fluorescence in the cytotoxic effector
cells of controls, particularly in OA patients, where it
accumulated on one half of the cell, forming a signet ring shape.
The individual values for each of the four independent experiments
per group (bar samples S1-S4) are shown in the charts (Fig. 1D and E). The median percentage (25-75th
percentiles) of GNLY localized inside LAMP-1+ granules
was 37.2% (35.4-38.5%) in the OA patients and did not significantly
differ compared to the controls [51% (48-51.4%)] (Mann-Whitney U
test). However, the intensity of GNLY labeling was significantly
higher in OA patients than in controls (P=0.000001), as measured by
flow cytometry (Fig. 1F). The
frequency of GNLY+ PBLs, assessed by flow cytometry, was
also higher [7.5% (2.8-9%)] in the OA patients compared with the
controls [1.7% (0.3-3.9%)] (Fig.
1G). In the OA patients and controls, the GNLY serum
concentrations, determined by ELISA were <0.3 ng/ml (Table I).
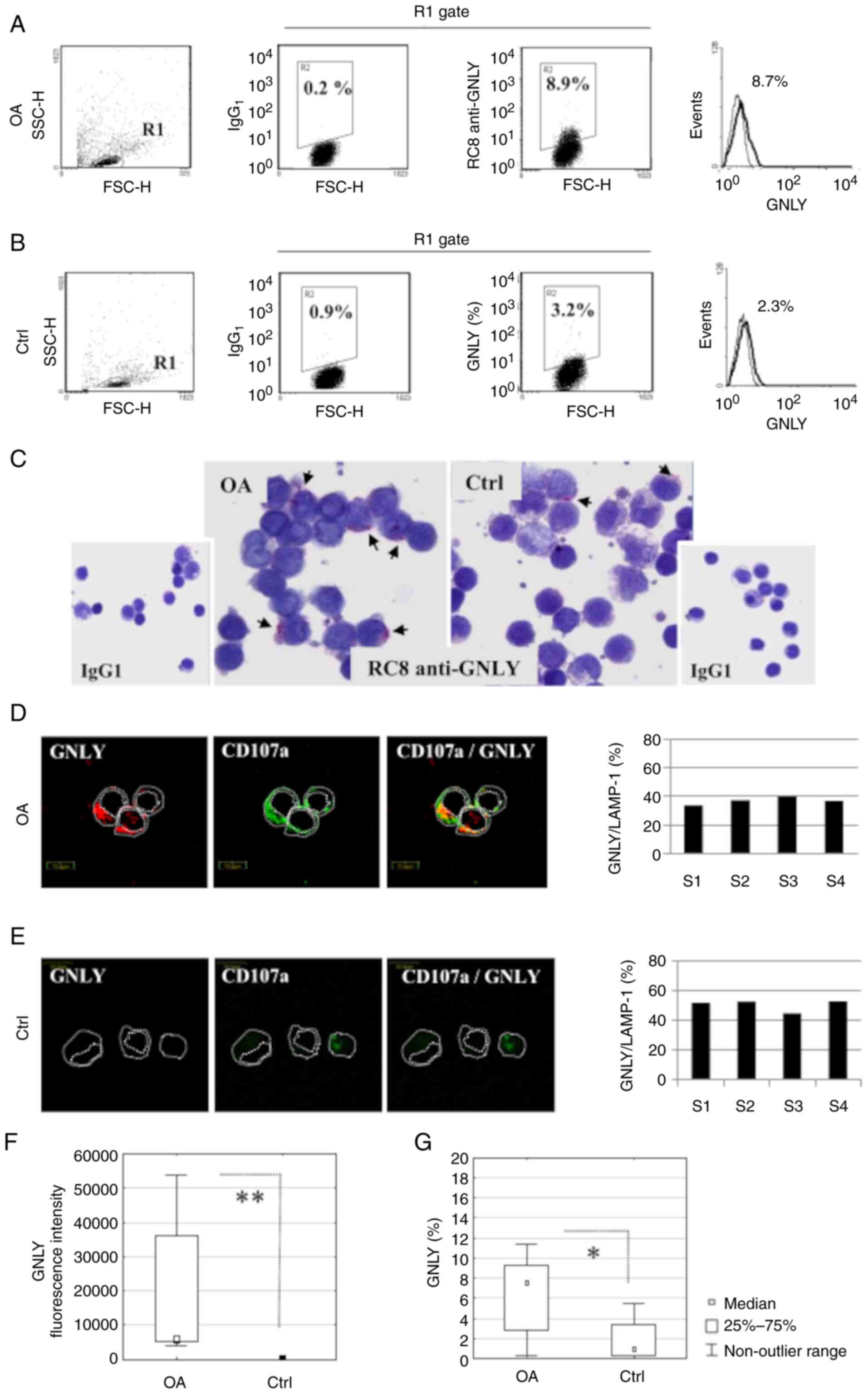 | Figure 1GNLY expression in the peripheral
blood lymphocytes of OA patients and controls. Representative
examples of dot plots and histograms showing the results from flow
cytometry analyses of the R1 lymphocyte gate for R2
GNLY+ cells in the (A) OA patients and (B) controls in
comparison to that of the isotype control (n=18). (C) Images show
immunohistochemically labeled GNLY in lymphocytes (red color,
indicated with arrows) compared with the isotype control. One of
three independent samples is shown. Magnification, x1,000. Confocal
laser microscopy of GNLY (red fluorescence) and LAMP-1 (green
fluorescence) in lymphocytes from (D) OA patients and (E) controls.
The colocalization of GNLY and LAMP-1 exhibited yellow
fluorescence. The original magnification of the images are 900 x 8.
Scale bar, 10 µm. The percentage of GNLY and LAMP-1 colocalization
is shown for each of the four independent experiments/samples. (F)
The intensities of GNLY fluorescence measured using confocal
microscopy in 12 cells from OA patients and controls. (G)
Percentage of GNLY+ lymphocytes in OA patients and
controls measured using flow cytometry (n=18). *P=0.003,
**P=0.000001 (Mann-Whitney U test). GNLY, granulysin;
OA, osteoarthritis; R, region; LAMP-1, lysosomal-associated
membrane protein-1; FSC-H, forward scatter-height; SSC-H, side
scatter-height. |
Cytotoxic potential of
GNLY+ effector cells
The proportion of GNLY-expressing
CD3-CD56+ NK cells,
CD3+CD56- T cells and
CD3+CD56+ NKT subsets were significantly
increased in the OA patients compared with the controls (P=0.01,
P=0.03 and P=0.003, respectively; Fig.
2A). Dot plots illustrate the analysis of PBL subsets from OA
patients (Fig. 2B) and controls
(Fig. 2C) and show the frequency of
CD3-CD56+ NK (10 vs. 8.6%),
CD3+CD56+ NKT (7 vs. 5.8%), and
CD3+CD56- T cells (64.6 vs. 73.4%), with
respect to isotype control. These values correspond to the median
(25-75th percentiles) of the percentage of NK cells [8.3%
(6.8-10%)], T cells [73.2% (70.4-76.5%)] and NKT cells [2.9%
(2.1-3.9%)] in OA patients, and these did not significantly differ
from the values of NK cells [10.3% (7.2-14.5%)], T cells [70.6%
(67.2-75%)], and NKT cells [3.5% (2.3-5.4%)] in the controls.
Histograms included in Fig. 2B and
C illustrate the frequency of
GNLY-expressing PBL subsets in the OA patients and controls,
respectively. The histograms were calculated based on the
difference between the percentages of the GNLY+ cells
minus the percentage obtained in the isotype-matched control.
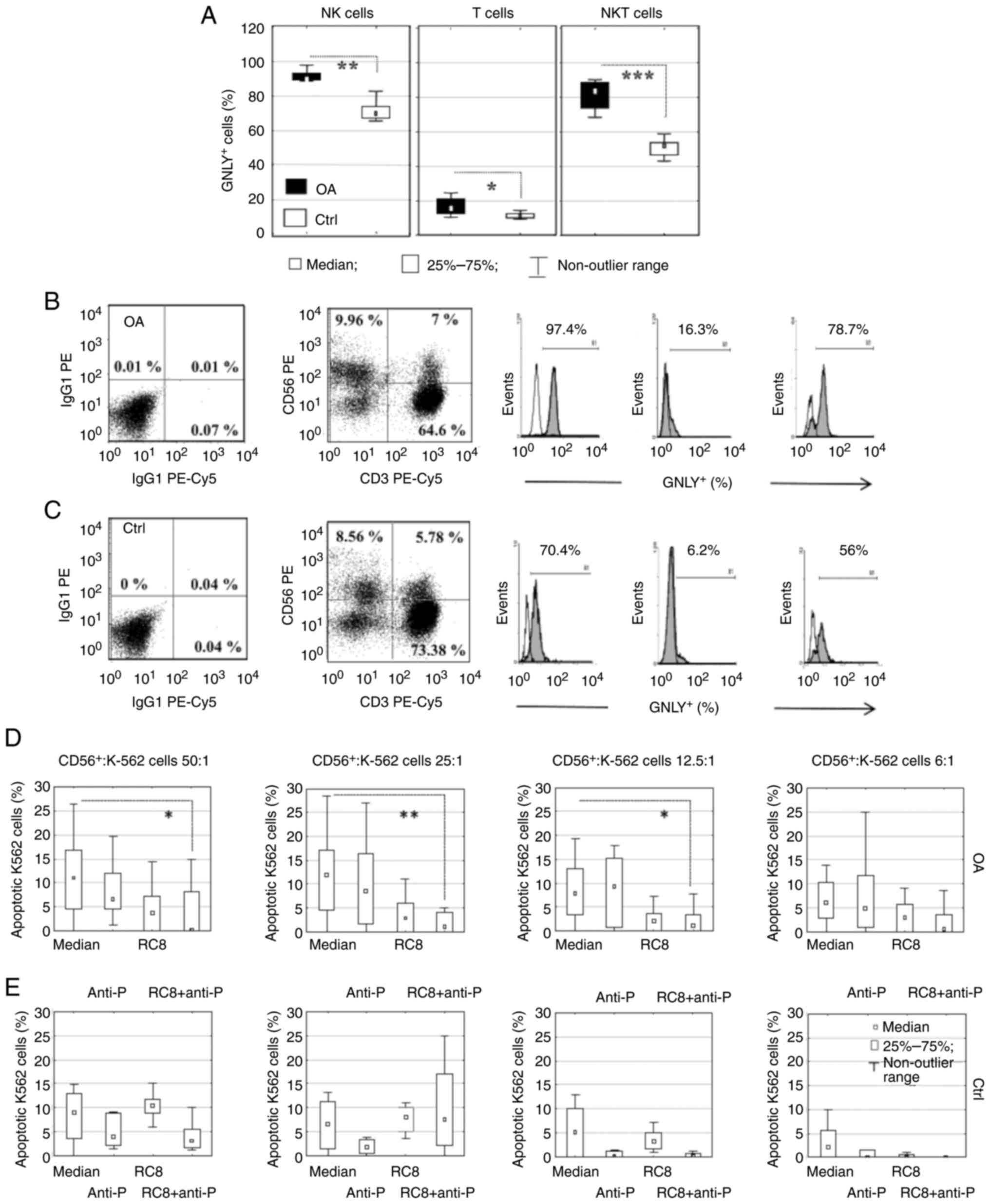 | Figure 2Proportion of GNLY+ cells in the
peripheral blood subpopulations and analysis of GNLY mediated
apoptosis in NK cells. (A) Frequency of GNLY in
CD3-CD56+ NK cells,
CD3+CD56- T cells and
CD3+CD56+ NKT cells between OA patients and
controls. *P=0.03, **P=0.01,
***P=0.003 (Mann-Whitney U test) in 18 experiments.
Quadrants in dot plots illustrate the analysis of lymphocyte
subpopulations from (B) OA patients and (C) controls pertaining to
CD3/CD56 labeling or isotype control, respectively.
GNLY+ cells within lymphocyte subpopulations are shown
as histograms for the (B) OA patients and (C) controls. Bars in the
charts depict the median values of early apoptosis of K-562 cells
cultured with NK cells from (D) OA patients or (E) control at the
indicated ratios in the medium alone, with an anti-perforin mAb,
with an anti-GNLY RC8 mAb, or with a combination of anti-perforin
and anti-GNLY RC8 mAbs (n=5-6 per group). *P=0.01,
**P=0.006 (Kruskal-Wallis and post-hoc Dunn's test).
GNLY, granulysin; OA, osteoarthritis; anti-P, anti-perforin; PE,
phycoerythrin. |
Early apoptosis did not differ significantly between
the OA patients and controls when obtained cells were cultured in
medium only [Fig. 2D and E, Mann-Whitney U test for each, effector:
target ratio (E:T)]. However, the analysis of molecular mechanisms
involved in the NK cell-mediated killing of K-562 targets revealed
GNLY-mediated early apoptosis only in the OA patients (Fig. 2D). The combination of anti-GNLY and
anti-perforin mAbs almost completely abolished early apoptosis in
cells, at E:T cell ratios of 50:1 (P=0.01), 25:1 (P=0.006) and
12.5:1 (P=0.01) (Fig. 2D), but
without an additional effect compared to the cells treated with
anti-GNLY antibody alone. In controls, the addition of anti-GNLY
antibody or the combination of anti-GNLY antibodies with an
anti-perforin mAb did not significantly alter the proportion of
early apoptotic cells (Fig. 2E).
Anti-perforin mAb alone did not affect the early apoptosis of K-562
cells in the OA patients (Fig. 2D)
or controls (Fig. 2E).
Distribution of GNLY and intracellular
cytokine expression in PBL subsets
The distribution of GNLY was analyzed in gated
GNLY+ cells observed in the fluorescence 2/fluorescence
3 dot plots showing CD3/CD56 labeling in OA patients (Fig. 3A) and controls (Fig. 3B) in comparison to their isotype
controls. Considering the samples from OA patients vs. that from
controls, the GNLY distribution percentages were 57.8 vs.
68.6% in CD3-CD56+ NK cells, 12.8 vs.
1% in CD3+CD56- T cells, 19.7 vs. 2.2%
in CD3+CD56+ NKT cells, and negligible in the
isotype controls (Fig. 3A and
B). The T and NKT cells of OA
patients had significantly higher GNLY distribution than that in
the controls (P=0.004; Fig.
3C).
Analysis of intracellular IFN-γ and IL-4 expression
levels revealed differences in the balance between the OA patients
(Fig. 3D) and controls (Fig. 3E). IFN-γ dominated over IL-4 in NK
and T cells of OA patients (P=0.03 and P=0.01, respectively), while
they were codominant in NKT cells (Fig.
3D). In the controls, IL-4 levels were higher than IFN-γ levels
in NK and NKT cells (P=0.004), and it was only marginally increased
in T cells (P=0.07; Fig. 3E).
Discussion
In the present study, it was shown that the
frequency of GNLY+ PBLs was higher in a group of primary
OA patients compared with the control group using an anti-GNLY mAb
of RC8 clone, which binds to the common core epitope of both the 9
and 15 kDa isoforms (24). Due to a
high percentage of GNLY+ cells (~30%) in the peripheral blood of OA
patients and a wide scattering of the results in our pilot study
(3), the OA group and controls in
this investigation were defined more precisely according to the
exclusion criteria mentioned above. Increased expression of GNLY in
NK, T, and NKT cells in the OA patients is consistent with the
slightly dominant systemic, low grade pro-inflammatory immune
response in OA (1,16), and the knowledge that GNLY increases
inflammation (25,26). Here, the dominance of Th1 IFN-γ over
Th2 IL-4 in the NK and T cells he peripheral blood of OA patients
was demonstrated, whereas in controls IL-4 dominated over IFN-γ in
the NK and NKT cell subsets. Correspondingly, Th1 and cytotoxic T
cells producing IFN-γ were found in the synovial fluid and joint
tissues of OA patients (2). IFN-γ
mRNA and protein expression levels in T and NK cell types depends
on the JAK/STAT signaling pathway, which is known to enhance the
immune response (7,15). This corresponds to an increased
intensity of GNLY labeling in PBLs of OA patients, compared with
the negligible fluorescent signals of GNLY in controls. It is known
that mild expression of GNLY in T cells and constitutive expression
of GNLY in innate NK cell-effectors increases following activation
by antigen or NK-cell receptor ligands (10). Immunocytochemistry illustrated a
greater proportion of GNLY+ PBLs in the OA patients compared with
the controls. Moreover, it was shown here for the first time that
the NK cells of OA patients employed GNLY-mediated apoptosis to
kill K-562 cells in vitro, in contrast to the controls,
whose NK cells exhibited low levels of GNLY and thus could not
employ GNLY-mediated cytotoxicity. GNLY acts in concert with
perforin in the realization of K-562 cell apoptosis, as the
anti-GNLY mAb of RC8 clone significantly decreased early apoptosis
in the OA patients only. The perforin pores enable the entry of
GNLY into cells, but perforin does not perform apoptosis by itself
(10,27). The apoptotic process is initiated by
intracellular Ca2+ increase, membrane damage,
mitochondrial reactive oxygen species generation, release of
apoptosis-inducing factor, cytochrome C and activation of caspase
3, which disrupt the normal physiology of targets (10,26).
Target cells are killed through induction of apoptosis, but also by
necrosis 4 h after the rapid entry of GNLY through perforin pores
(26,27), which meant a proportion of cells
were not included in the Annexin+PI- subset
(gate) used for the detection of early apoptotic targets by flow
cytometry at the end of an 18 h cytotoxicity test (23).
Killing of K-562 targets, which are devoid of HLA
class I antigens, represents a model for in vitro analysis
of NK cell-mediated self-aggression, as damaged tissue cells
downregulate HLA class I antigens and become more susceptible to NK
cell cytotoxicity (28). This
implies that activated NK cells in the circulation may harm
endothelial cells during atherogenesis, independently or in concert
with classical metabolic risk factors and support endothelial
dysfunction underlying cardiovascular diseases (29). It was previously shown that >80%
of OA patients suffer from arterial hypertension and ~30% are
diabetic (30), as reflected in the
OA patients group in this investigation, signifying metabolically
triggered systemic inflammation (31,32).
All patients with OA enrolled in this study, as well as the
controls, had hypercholesterolemia, as a component of the metabolic
syndrome. An intake of particular kinds of fats could affect knee
OA progression independent of weight and induce metabolically
triggered low grade, systemic inflammation (31,33).
We enrolled the non-obese patients with primary OA, as patients
with disease of unknown cause. However, the contribution of
biochemical and metabolic factors, together with the factors that
can be modified by age, heritage and behavior cannot be completely
excluded (34). Additionally, knee
OA affects men and women differently, with women exhibiting
increased disease frequency, severity and disability (35). To avoid sex-based differences and
the possible role of estrogen in granulysin expression (36), only postmenopausal women were
recruited in the OA group and controls in this study. To visualize
GNLY and LAMP-1, a marker of lysosome-related secretory organelles
(37), confocal microscopy was
used. Confocal microscopy confirmed more frequent visualization of
GNLY in PBLs from the OA group compared with the controls, although
the colocalization of GNLY and LAMP-1 was ~40% in both groups. It
indicates the accumulation of cytotoxic 9 kDa GNLY in dense,
exocytic granules, whereas 15 kDa GNLY is not associated with
LAMP-1+ organelles and does not overlap with 9 kDa GNLY
(9). The 15 kDa form of GNLY is
released by activated cells in a protein kinase C-dependent manner
(9) and lacks cytotoxic activity in
eukaryotic cells (13). It
represents the soluble serum GNLY with regulatory functions
(24), which was negligible in the
OA patients and controls.
Based on the results shown herein, it was concluded
that in women with knee OA, GNLY expression in the PBL subsets and
GNLY-mediated apoptosis of K-562 targets were higher than that in
the controls, alongside lymphocyte pro-inflammatory polarization.
However, it would be optimal to broaden the study with a larger
number of patients and different OA phenotypes in both sexes to
reach a more definitive conclusion and/or determine differences
between the sexes.
Acknowledgements
We would like to thank Mrs. Tihana Gržetić Beljak
(Hospital for Medical Rehabilitation of Health and Lung Diseases
and Rheumatism ‘Thalassotherapia-Opatija’ Opatija, Croatia) for
language editing, and Dr Emina Babarović (Department of Pathology,
Faculty of Medicine, University of Rijeka, Rijeka, Croatia) for
assistance with the statistical analysis.
Funding
Funding: This work was supported by funding from the University
of Rijeka, Croatia (grant nos. 13.06.1.1.06, Uni-ri-biomed-18-110,
and Uni-ri-biomed-18-160).
Availability of data and materials
The data sets used and/or analyzed during the
present study are available from the corresponding author on
reasonable request.
Authors' contributions
All authors contributed to the study conception and
design. Material preparation, acquisition of data and analysis were
performed by VD, GL, BĆC, LK, MA, MR, TK and DL. The first draft of
the manuscript was written by GL and DR. All authors revised the
manuscript. All authors have read and approved the final manuscript
(except DR who has unfortunately passed away). VD, GL and BĆC
confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
All participants provided written informed consent.
The study was approved by the Ethical Committees of
Thalassotherapia-Opatija Hospital and the Faculty of Medicine,
University of Rijeka, Rijeka, Croatia (approval no.
01-000-00-450/2018). All experimental procedures were performed
according to the ethical principles for medical research involving
human subjects outlined in the World Medical Association
Declaration of Helsinki.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Koh SM, Chan CK, Teo SH, Singh S, Merican
A, Ng WM, Abbas A and Kamarul T: Elevated plasma and synovial fluid
interleukin-8 and interleukin-18 may be associated with the
pathogenesis of knee osteoarthritis. Knee. 27:26–35.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Woodell-May JE and Sommerfeld SD: Role of
inflammation and the immune system in the progression of
osteoarthritis. J Orthop Res. 38:253–257. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kehler T, Laskarin G, Massari D, Dominovic
M, Persic V, Rosovic I, Laginja J and Rukavina D: Possible role of
granulysin in pathogenesis of osteoarthritis. Med Hypotheses.
85:850–853. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Chenoweth MJ, Mian MF, Barra NG, Alain T,
Sonenberg N, Bramson J, Lichty BD, Richards CD, Ma A and Ashkar AA:
IL-15 can signal via IL-15Rα, JNK, and NF-κB to drive RANTES
production by myeloid cells. J Immunol. 188:4149–4157.
2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zeng R, Spolski R, Casas E, Zhu W, Levy DE
and Leonard WJ: The molecular basis of IL-21-mediated
proliferation. Blood. 109:4135–4142. 2007.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hu X, Chen Q, Sowrirajan B, Bosche M,
Imamichi T and Sherman BT: Genome-wide analyses of MicroRNA
profiling in interleukin-27 treated monocyte-derived human
dendritic cells using deep sequencing: A pilot study. Int J Mol
Sci. 18(925)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Gotthardt D, Trifinopoulos J, Sexl V and
Putz EM: JAK/STAT cytokine signaling at the crossroad of NK cell
development and maturation. Front Immunol. 10(2590)2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wu Y, Lin Z, Yan Z, Wang Z, Fu X and Yu K:
Sinomenine contributes to the inhibition of the inflammatory
response and the improvement of osteoarthritis in mouse-cartilage
cells by acting on the Nrf2/HO-1 and NF-κB signaling pathways. Int
Immunopharmacol. 75(105715)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lettau M, Dietz M, Dohmen K, Leippe M,
Kabelitz D and Janssen O: Granulysin species segregate to different
lysosome-related effector vesicles (LREV) and get mobilized by
either classical or non-classical degranulation. Mol Immunol.
107:44–53. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Liu X and Lieberman J: Knocking ‘em Dead:
Pore-forming proteins in immune defense. Annu Rev Immunol.
38:455–485. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Krawczyk PA, Laub M and Kozik P: To kill
but not be killed: Controlling the activity of mammalian
pore-forming proteins. Front Immunol. 11(601405)2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tewary P, Yang D, de la Rosa G, Li Y, Finn
MW, Krensky AM, Clayberger C and Oppenheim JJ: Granulysin activates
antigen-presenting cells through TLR4 and acts as an immune
alarmin. Blood. 116:3465–3474. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Clayberger C, Finn MW, Wang T, Saini R,
Wilson C, Barr VA, Sabatino M, Castiello L, Stroncek D and Krensky
AM: 15 kDa granulysin causes differentiation of monocytes to
dendritic cells but lacks cytotoxic activity. J Immunol.
188:6119–6126. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Robinson WH, Lepus CM, Wang Q, Raghu H,
Mao R, Lindstrom TM and Sokolove J: Low-grade inflammation as a key
mediator of the pathogenesis of osteoarthritis. Nat Rev Rheumatol.
12:580–592. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Griffin TM and Scanzello CR: Innate
inflammation and synovial macrophages in osteoarthritis
pathophysiology. Clin Exp Rheumatol. 37 (Suppl 120):S57–S63.
2019.PubMed/NCBI
|
|
16
|
Berenbaum F and Walker C: Osteoarthritis
and inflammation: A serious disease with overlapping phenotypic
patterns. Postgrad Med. 132:377–384. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
World Medical Association. World medical
association declaration of Helsinki: Ethical principles for medical
research involving human subjects. JAMA. 310:2191–2194.
2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Altman R, Asch E, Bloch D, Bole G,
Borenstein D, Brandt K, Christy W, Cooke TD, Greenwald R, Hochberg
M, et al: Development of criteria for the classification and
reporting of osteoarthritis. Classification of osteoarthritis of
the knee. Diagnostic and therapeutic criteria committee of the
American rheumatism association. Arthritis Rheum. 29:1039–1049.
1986.PubMed/NCBI View Article : Google Scholar
|
|
19
|
American Heart Association: Classes of
heart failure. American Heart Association, Dallas, TX, 2017.
https://www.heart.org/en/health-topics/heart-failure/what-is-heart-failure/classes-of-heart-failure.
Accessed May 31, 2017.
|
|
20
|
Menkes CJ: Radiographic criteria for
classification of osteoarthritis. J Rheumatol Suppl. 27:13–15.
1991.PubMed/NCBI
|
|
21
|
Persic V, Ruzic A, Miletic B, Samsa DT,
Rakic M, Raljevic D, Pejcinovic VP, Eminovic S, Zaputovic L and
Laskarin G: Granulysin expression in lymphocytes that populate the
peripheral blood and the myocardium after an acute coronary event.
Scand J Immunol. 75:231–242. 2012.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Donaldson JG: Immunofluorescence staining.
Curr Protoc Cell Biol. 69:4.3.1–4.3.7. 2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Kabakov AE and Gabai VL: Cell death and
survival assays. Methods Mol Biol. 1709:107–127. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ogawa K, Takamori Y, Suzuki K, Nagasawa M,
Takano S, Kasahara Y, Nakamura Y, Kondo S, Sugamura K, Nakamura M
and Nagata K: Granulysin in human serum as a marker of
cell-mediated immunity. Eur J Immunol. 33:1925–1933.
2003.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Krensky AM and Clayberger C: Biology and
clinical relevance of granulysin. Tissue Antigens. 73:193–198.
2009.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Belizário JE, Neyra JM and Setúbal Destro
Rodrigues MF: When and how NK cell-induced programmed cell death
benefits immunological protection against intracellular pathogen
infection. Innate Immun. 24:452–465. 2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Saini RV, Wilson C, Finn MW, Wang T,
Krensky AM and Clayberger C: Granulysin delivered by cytotoxic
cells damages endoplasmic reticulum and activates caspase-7 in
targeT cells. J Immunol. 186:3497–3504. 2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Biassoni R, Ugolotti E and De Maria A: NK
cell receptors and their interactions with MHC. Curr Pharm Des.
15:3301–3310. 2009.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Laskarin G, Persic V, Kukic SR, Massari D,
Legovic A, Boban M, Miskulin R, Rogoznica M and Kehler T: Can pain
intensity in osteoarthritis joint be indicator of the impairment of
endothelial function? Med Hypotheses. 94:15–19. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zakaria ZF, Bakar AA, Hasmoni HM, Rani FA
and Kadir SA: Health-related quality of life in patients with knee
osteoarthritis attending two primary care clinics in Malaysia: A
cross-sectional study. Asia Pac Fam Med. 8(10)2009.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Courties A, Sellam J and Berenbaum F:
Metabolic syndrome-associated osteoarthritis. Curr Opin Rheumatol.
29:214–222. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Coskun Benlidayi I: Diet in
osteoarthritis. Rheumatol Int. 41:1699–1700. 2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Villalvilla A, Larrañaga-Vera A, Lamuedra
A, Pérez-Baos S, López-Reyes AG, Herrero-Beaumont G and Largo R:
Modulation of the inflammatory process by hypercholesterolemia in
osteoarthritis. Front Med (Lausanne). 7(566250)2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Deveza LA, Melo L, Yamato TP, Mills K,
Ravi V and Hunter DJ: Knee osteoarthritis phenotypes and their
relevance for outcomes: A systematic review. Osteoarthritis
Cartilage. 25:1926–1941. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Laitner MH, Erickson LC and Ortman E:
Understanding the impact of sex and gender in osteoarthritis:
Assessing research gaps and unmet needs. J Womens Health (Larchmt).
30:634–641. 2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Hughes SM, Pandey U, Johnston C, Marrazzo
J, Hladik F and Micks E: Impact of the menstrual cycle and ethinyl
estradiol/etonogestrel contraceptive vaginal ring on granulysin and
other mucosal immune mediators. Am J Reprod Immunol.
86(e13412)2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Alter G, Malenfant JM and Altfeld M:
CD107a as a functional marker for the identification of natural
killer cell activity. J Immunol Methods. 294:15–22. 2004.PubMed/NCBI View Article : Google Scholar
|