Introduction
Listeria (L.) monocytogenes is a
gram-positive, non-sporulating, facultatively anaerobic bacillus
that is transmitted to humans through ingestion of foods
contaminated with high-bacterial concentrations (1-3).
Listeriosis is a relatively rare disease (0.1 to 10
cases per 1 million individuals per year according to WHO) with a
mortality rate of 20-30%, especially for those who developed an
invasive disease (4).
Invasive Listeriosis typically affects
immunocompromised patients, pregnant women, the elderly, and
neonates (5). It causes several
clinical syndromes, of which meningitis, meningoencephalitis, and
sepsis are the most challenging to deal with (6,7).
Listeria meningitis should be suspected in children <3 years of
age and adults >50 years of age who show meningeal symptoms
without clear etiology (8).
Ampicillin plays a key role in Listeria treatment,
and it is often combined with gentamicin which may be a suitable
adjuvant due to its favorable pharmacodynamic synergistic action
with penicillin (9). Optimal
treatment durations have not been assessed; however, ampicillin
should be administered for at least 21 days and gentamicin can be
discontinued after 1 week of treatment, taking into consideration
the patients' clinical conditions (9). Conversely, gentamicin administration
may be prolonged for up to 3 weeks, although the renal function and
plasma concentrations of gentamicin should be monitored in such a
case (9,10).
Although there are limited data and inconclusive
evidence, adjunctive dexamethasone administration is discouraged,
as it has been associated with unfavorable outcomes or no
beneficial effects; however, other studies tend to reconsider
dexamethasone therapy due to its effects on inflammation and edema
(11).
The present report describes five cases of L.
monocytogenes meningitis affecting two previously healthy
immunocompetent and three immunocompromised adult patients treated
according to the international guidelines. All the patients
described received a low dose of dexamethasone.
To discuss the appropriate clinical and therapeutic
management of these patients and to argue the possible role of
dexamethasone administration in Listeria meningitis, a brief
literature review including only documented adult cases (≥18 years
old) treated with an antibiotic and dexamethasone combination was
also performed.
Case reports
Consent
The patients signed written informed consent forms.
Within the consent form, it was specified that data collected
during the diagnostic and therapeutic process can be anonymously
used for scientific purposes.
Patient 1
This patient was a 69-year-old healthy Italian man
with 5 days of fever, nausea, and loss of appetite, followed by a
progressive headache and impaired mental status. His past medical
history was unremarkable; he did not take any medications.
On admission, the patient was febrile (39˚C), poorly
responsive to vocal and painful stimuli, and was in a state of
stupor [Glasgow Coma Scale (GCS)] (8). Clinical examination showed neck
stiffness along with positive Kernig's sign. Babinski's sign was
negative.
Blood tests revealed an elevated white blood cell
(WBC) count (18,800/mm3, 89.8% neutrophils), mild anemia
(Hb 11.8 g/dl), normal platelet count (160,000/mm3), as
well as elevated C-reactive-protein (CRP) levels (35.7 mg/dl) and
procalcitonin (PCT) levels (3 ng/dl). Renal and liver function were
normal. The HIV test was negative.
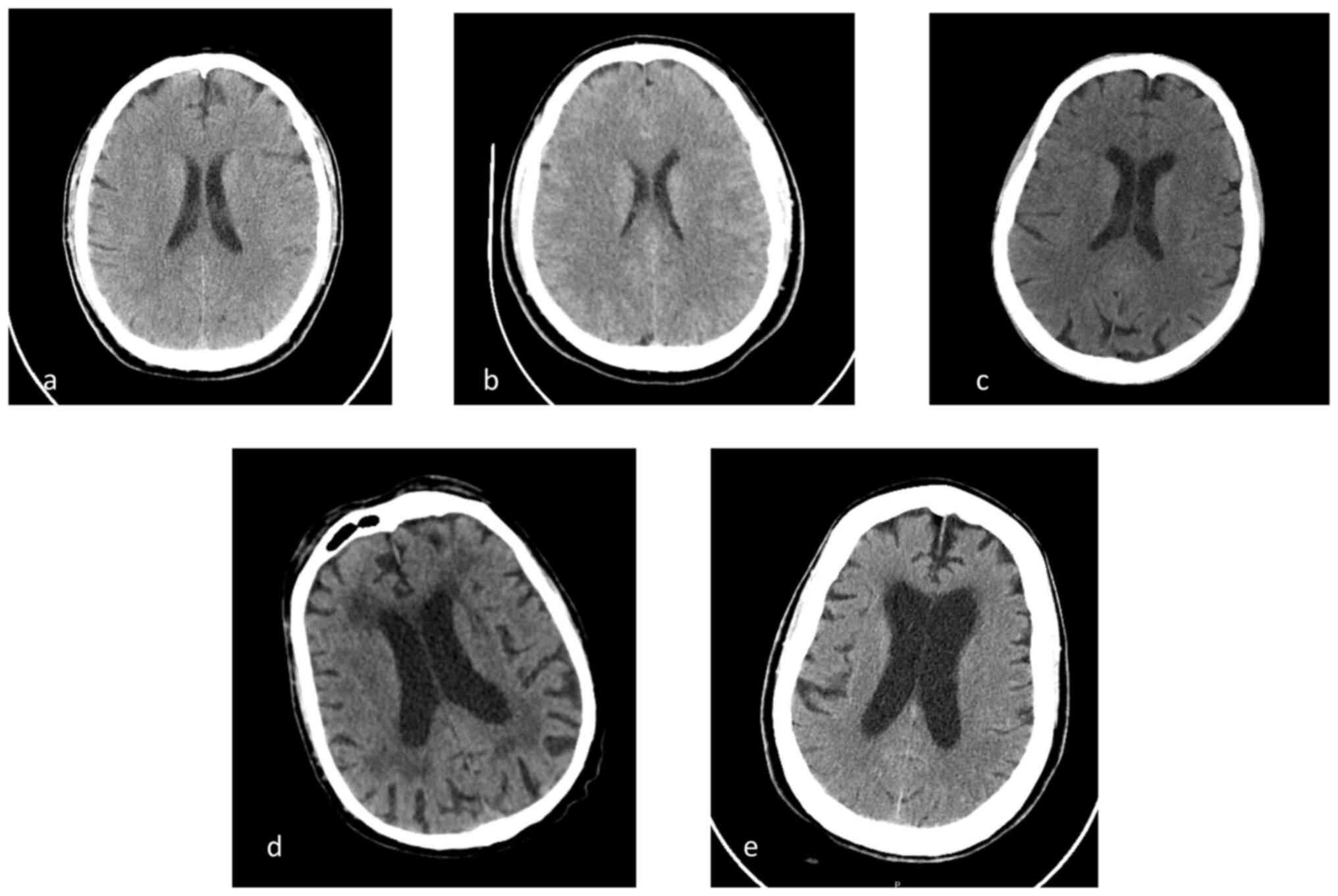
A head CT scan (SOMATOM® Definition Flash
scanner, Siemens AG) showed no abnormalities (Fig. 1A). Cerebrospinal fluid (CSF)
examination revealed high protein levels (4,175 mg/l; normal range
150-450 mg/l), high lactate dehydrogenase levels (193 IU/l),
reduced glucose levels (18 mg/dl), and hypercellularity (152
cells/mcl mostly lymphocytes; normal range <5 cells/mcl).
Empirical treatment with ceftriaxone 2 g twice daily, acyclovir 10
mg/kg three times daily, and corticosteroids (dexamethasone 4 mg
three times daily) was initiated. Molecular biology [reverse
transcription (RT)-PCR] for Listeria detection was performed using
a FilmArray Meningitis/Encephalitis Panel (ME) CE-IVD according to
the manufacturer's protocol (cat. no. RFIT-PRT-0005, BioFire
Diagnostics, LLC) on the CSF, and the results were positive for
L. monocytogenes, which was confirmed by CSF culture
[performed in 5% blood agar with Brain Heart Infusion (BHI) to
enhance recovery of Listeria isolates with incubation in aerobic
conditions at 37˚C for 24 to 48 h]. Targeted therapy was
established with intravenous (i.v.) ampicillin 3 g every 6 h, and
gentamicin 400 mg i.v. Dexamethasone 4 mg three times a day was
also maintained.
The patient's clinical conditions rapidly improved
with notable amelioration 72 h after targeted therapy initiation.
Overall, the patient was treated for 21 days with antibiotics and
dexamethasone, and the latter was gradually reduced before
discontinuation. After 1 month of hospitalization, the patient was
discharged with no evidence of neurological deficits.
Patient 2
A previously healthy, 39-year-old man was
hospitalized for speech problems and intense dizziness. He reported
a 4-day history of fever, headache, and vomiting, which were
treated with antiemetics and paracetamol.
On admission, the patient was febrile (39.6˚C), with
a normal mental status and with neck stiffness. Other signs of
meningeal irritation were absent. Clinical examination showed no
abnormalities.
Laboratory tests revealed elevated WBC counts
(13,300/mm3, 73.3% neutrophils), high CRP levels (28.91
mg/dl), and elevated transaminase levels [aspartate transferase
(AST) 314 IU/l; alanine transferase (ALT) 116 IU/l]. A head CT scan
was normal (Fig. 1B).
CSF analysis showed high protein levels (675 mg/l),
low glucose levels (29 mg/dl; serum glucose 109 mg/dl), with 502
cells/mcl (mostly lymphocytes). Empiric treatment was established
with ceftriaxone 2 g twice daily, ampicillin 3 g four times daily,
acyclovir 10 mg/kg three times daily, and dexamethasone 4 mg three
times daily. RT-PCR on CSF was positive for L. monocytogenes
and this result was then confirmed by CSF culture.
Ampicillin 12 g daily i.v., and gentamicin 4
mg/kg/day i.v. for 21 days were administered along with
dexamethasone at the same dosage which was gradually tapered. The
patient's clinical conditions improved rapidly, and the fever
disappeared after 48 h of antibiotic treatment. In 34 days, he was
discharged without complications.
Patient 3
A 49-year-old Italian man with a history of
ulcerative colitis presented to the emergency department with
abdominal pain, nausea, an intense headache, and a fever ~40˚C. He
did not have other comorbidities. He took mesalamine and
corticosteroids.
On admission, the patient had intense photophobia,
and neck stiffness along with positive signs of meningeal
irritation (Brudzinski, Kernig). In addition, he was febrile
(39.5°C).
Blood tests revealed an elevated WBC count
(11,000/mmc3, 70% neutrophils), CRP levels (44.2 mg/dl)
and PCT levels (25.32 ng/ml).
Head CT scan was negative (Fig. 1C). CSF analysis showed 1,364
leukocytes/µl with a preponderance of lymphocytes. CSF protein
levels were 420 mg/l, and the glucose level was 32 mg/dl (blood
glucose, 98 mg/dl).
The patient was treated empirically with ceftriaxone
2 g twice daily, acyclovir 10 mg/kg three times daily, vancomycin
15 mg/kg, and dexamethasone 4 mg three times daily.
Gram stains of the CSF showed the presence of
Gram-positive rods. RT-PCR showed the presence of Listeria
monocytogenes, later confirmed by culture.
Antibiotic therapy was switched to ampicillin 12 g
daily i.v. plus gentamicin 4 mg/kg/day i.v. Dexamethasone was
maintained at the same dosage, and was gradually decreased. He was
treated for 4 weeks, with complete recovery of symptoms. After 1
month, the patient was discharged without neurological
sequelae.
Patient 4
A 67-year-old man presented to the emergency
department with a worsening frontal headache associated with a high
fever (up to 40˚C), and impaired consciousness (GCS 12). His
medical history included a B-cell lymphoma, treated with rituximab,
and secondary hypoglobulinemia.
On admission, he had marked neck stiffness; Kernig's
and Lasegue's signs were positive. He was febrile (38.4˚C) and
confused.
Blood tests revealed elevated CRP levels (18 mg/dl),
reduced WBC counts (3,500/mm3) with normal formula, mild
anemia (11.3 g/dl), and normal PCT levels. A head CT scan showed no
abnormalities (Fig. 1D).
CSF examination revealed a glucose level of 0.24
mg/dl, a protein level of 1,400 mg/l, and a cell count of 425/mcl
(with a predominance of mononuclear cells). Empirical therapy was
started with vancomycin 15 mg/kg daily plus dexamethasone 4 mg
three times daily.
CSF was positive for L. monocytogenes and
antibiotic therapy was administered with ampicillin 12 g daily i.v.
and gentamicin 4 mg/kg/day i.v. for 21 days. Dexamethasone 4 mg
three times daily i.v. was also prolonged for 21 days and was
gradually reduced. He was discharged 35 days after hospitalization
without complications.
Patient 5
A 72-year-old man with a history of recurrent
pulmonary infections, chronic kidney disease (CKD), and diabetes
was admitted to the emergency department with fever (38.6˚C),
photophobia, and confusion (GCS 11).
Neurological examination revealed neck stiffness
with positive Kernig's and Lasegue's signs. Laboratory data showed
high WBC counts (5,600/mm3, 76% neutrophils), high CRP
levels (36 mg/dl), creatinine 2.1 mg/dl (eGFR with CKD-EPI 31
ml/min), AST 56 IU/l, ALT 41 IU/l, and glucose 176 mg/dl. A head CT
scan was normal (Fig. 1E).
CSF showed 1,125 mononuclear cells/mm3,
glucose 0.66 mg/dl and protein 2,100 mg/dl. Empiric therapy was
started with ceftriaxone, acyclovir, ampicillin, and dexamethasone
4 mg three times daily. The CSF molecular panel was positive for
L. monocytogenes, which was confirmed on CSF culture.
Antimicrobial therapy with ampicillin 3 g four times
daily along with intravenous gentamicin 3 mg/kg/day was initiated.
Due to the impaired kidney function, diminished doses of gentamicin
were administered for 10 days, strictly monitoring creatinine and
eGFR, the levels of which remained constant. Dexamethasone therapy
was maintained for 21 days, although gradually reduced.
Unfortunately, despite appropriate therapy, the
patient's condition deteriorated rapidly, especially his mental
status, and he died on the 11th day after admission.
Discussion
Worldwide, Listeria monocytogenes is the
third leading cause of community-acquired meningitis, accounting
for 5% of all cases (1). Aging,
chronic disease, sepsis, and malignancies represent the most
important predictors of mortality in patients with Listeria
meningitis (12).
Two of the patients reported on in this study were
healthy and immunocompetent men who were not involved in farming or
animal husbandry. We consider as ʻimmunosuppressedʼ every patient
with impaired immunity or an impaired immune response due to
congenital or acquired disease or those patients who took drugs
that can induce an impaired immune status (e.g. chemotherapy,
chronic corticosteroid administration). The third patient was
immunosuppressed due to chronic corticosteroid treatment but
reported no contact with animals or unknown foods prior to
infection. The fourth patient had a history of hematologic disease;
the fifth patient was affected by CKD due to diabetes, which could
be considered an immunosuppressive disease, and reported multiple
respiratory infections previously. None of them had a clear
exposure to foods classically associated with an increased risk of
Listeria infection (1). None of
them were HIV positive, as performing an HIV test is strongly
suggested in the presence of Listeria meningeal infection (13,14),
nor were they HCV or HBV positive (15-17).
It is hypothesized that they were asymptomatic intestinal carriers
since the incidence of fecal carriers is estimated to be 1-10% of
the population (11,18). In addition, head CT scans of all
patients described did not reveal signs of rhombencephalitis.
Perhaps, MRI would have been a better method to address this issue,
but it was not performed due to the patients' serious clinical
conditions.
Concerning Listeria meningitis, lumbar puncture is
essential for diagnosis since these patients are clinically
indistinguishable from those with more common pathogens (9). However, according to various studies,
the incidence of meningeal signs in patients with L.
monocytogenes is lower than in patients with other bacterial
CNS infections (19). Signs and
symptoms may include fever, headache, vomiting, diarrhea, and
altered mental status, such as those reported in these reports
(20). CSF analysis in most cases
shows pleocytosis with neutrophilia, reduced glucose
concentrations, and increased protein levels (21).
As stated for meningitis caused by other
microorganisms, even in neurolisteriosis, an earlier diagnosis
correlates with better outcomes and reduced neurological sequelae
rates (9).
Typical CSF findings of bacterial meningitis are
present in 77% of patients, and Gram staining of the CSF reveals
Gram-positive rods in only one-third of cases (21). Therefore, culture or PCR of CSF is
required to detect L. monocytogenes (sensitivity and
specificity of 80%) (9). Multiplex
PCR may reduce the appropriate treatment delay, improving results
and leading to the initiation of proper antibiotic therapy
(22). In our cases, RT-PCR
provided a positive result quickly for Listeria and specific
treatment was started immediately, whereas microbiological results
were confirmed later with positive CSF cultures, as the latter
represents the gold standard for assessing bacterial antibiotic
susceptibility.
First-line empiric treatment of bacterial meningitis
includes third-generation cephalosporins and vancomycin to target
the most common pathogens (9).
Unfortunately, despite their broad spectrum of action,
third-generation cephalosporins generally have no activity against
Listeria (23). The most effective
treatment for CNS listeriosis has not been determined as there are
no randomized controlled trials with an adequate number of
patients. Furthermore, an optimal treatment duration has not been
determined although a minimum of 21 days of therapy is recommended
for Listeria meningitis (9,24). Benzylpenicillin and ampicillin are
considered the core of any treatment regimen, either alone or in
combination with aminoglycosides (such as gentamicin) due to their
synergistic effect on bacterial killing (25).
Although there are no formal recommendations
regarding aminoglycoside administration in these patients due to
their adverse effects on renal function (9), to enhance ampicillin activity and to
improve clinical outcomes, all the subjects we described were
treated with gentamicin. The fifth patient received a short-term
reduced dose of gentamicin due to his impaired kidney function.
Corticosteroid therapy correlates with decreased CSF
inflammation, reversal of brain edema, and improved outcomes
(26). Dexamethasone administration
could reduce bacterial lysis, which enhances inflammation in the
subarachnoid space, resulting in higher morbidity and mortality
(26). Considering this,
dexamethasone administration is recommended for all meningitis of
unknown origin in adults due to its effect on edema, cerebral
vasculitis, altered cerebral blood flow, intracranial hypertension,
and neuronal damage (25).
Guidelines suggest the cessation of dexamethasone
administration if pathogens other than S. pneumoniae are
present in the culture (26-28)
since some cohorts, although small and heterogeneous, showed that
standard dexamethasone therapy had no benefits in terms of
mortality and morbidity in meningitis caused by other pathogens,
such as Neisseria meningitidis or Listeria
monocytogenes (29).
Due to the rarity of L. monocytogenes meningitis,
there is a significant lack of clinical trials and studies on
dexamethasone administration, and most of the literature consists
of case reports or retrospective studies (30). Therefore, it is difficult to make
evidence-based recommendations regarding this condition, and the
results of clinical studies are varied.
Currently, the prospective Multicentric
Observational NAtional Study on LISteriosis and ListeriA (MONALISA)
is the largest cohort of patients diagnosed with listeriosis, with
818 cases. However, only 252 cases were neurolisteriosis, and of
these, only 32 received dexamethasone. The study showed a
significant reduction in the survival of patients treated with
dexamethasone within 24 h of admission. However, the treated
population was small and this result is not from a clinical trial
(31).
An epidemiological study of two Dutch cohorts of
patients with neurolisteriosis described no association between
dexamethasone administration and poor outcomes. However, the rate
of dexamethasone administration was higher in the cohort with a
worse outcome (26).
Endorsing the idea of the beneficial effects of
corticosteroids, Pelegrín et al (32) analyzed 59 patients affected by
neurolisteriosis, 29 of those received adjuvant dexamethasone
administration. Although no significant differences in mortality or
major side effects were recorded and those patients who received
dexamethasone were more likely to present with fever at admission
than those who had did not, along with a higher number of cells in
the CSF, there was a trend toward fewer neurological sequelae in
the dexamethasone group. Similarly, Amaya-Villar et al
(33) described 43 patients with
neurolisteriosis, 21 of them received dexamethasone administration;
a higher survival rate was observed in patients treated with
adjunctive dexamethasone, although it was not statistically
significant (Table I).
 | Table ISummary of the clinical
characteristic and therapeutic management of patients with
neurolisteriosis in the previous literature and present study. |
Table I
Summary of the clinical
characteristic and therapeutic management of patients with
neurolisteriosis in the previous literature and present study.
| First author,
year | Number of
patients | Age, years | Sex (n) | Symptoms (n) | Risk factors
(n) | Targeted antibiotic
therapy (n) | DEX therapy
(n) | Outcome (n) | (Refs.) |
|---|
| Chau et al,
2010 | 1 | 30 | M | Fever, headache,
emesis, neck stiffness | None | None | Yes | Death | (34) |
| | 1 | 34 | F | Fever, headache,
emesis, neck stiffness | Raw milk and soft
cheese consumption | AMP 12 gr/die for
11 days | Yes | Complete
recovery | (34) |
| Barocci et
al, 2015 | 1 | 59 | F | Fever, headache,
altered mental status, altered consciousness, neck stiffness | HT, allo-SCT
complicated by cGvHD, soft cheese consumption | AMP 12 g daily and
RF 600 mg daily for 14 days, then AMC 1 g every 8 h + RF 600 mg
daily for 14 days | Yes | Recovery | (35) |
| Romero Gutiérrez
et al, 2012 | 1 | 55 | F | Fever, headache,
sleepiness, reduced mental aptitude, delayed response to stimuli,
neck stiffness, left hemiparesis | AA | CTX, AMP, VAN, ACV
(for concomitant HSV) encephalitis | Yes | Recovery | (36) |
| Charlier et
al, 2017 (MONNALISA cohort) | 252 | Median age 67 | M (152), F
(100) |
Encephalitis-associated symptoms (218),
meningeal involvement without encephalitis (34) | Alcohol abuse (32),
cirrhosis (20), DM (55), ESRD (4), SOC (49), HM (34), SOT (5),
asplenia (3), neutropenia (8), lympho penia (27), HIV (4), IBD
(12), rheumatic disorders (20), other auto-immune diseases (11),
age >70 years old (117); corticosteroids (48), anti-TNF
biotherapy (6), immunosuppressive therapy (71) | AMX (244), IPM
(10), GEN (200), TMP-SMX (42), RF (3), VAN (24), LZD (4), AMX+GEN
(192), AMX+TMP-SMX (37), No treatment (1). Mean antibi otic
duration 22 days. | Yes (32) | 17 of 32 patients
receiving dexamethasone survived, 157 of 216 of those who did not
receive dexamethasone survived | (31) |
| Koopmans et
al, 2013 | 30 | Median age 65 | M (15), F (15) | Headache (22), neck
stiffness (22), fever (27), altered mental status (21), coma (3),
focal neurologic deficits (13), aphasia (7), hemiparesis (2),
cranial nerve palsies (2) | Immunocompromised
(20) | AMX or penicillin
monotherapy (10), third generation cephalosporin monotherapy (5),
AMX or penicillin + third-generation cephalosporin (11), other
(4) | Yes (5) | Death (5), sequalae
(4) | (26) |
| | 62 | Median age 69 | M (39), F (23) | Headache (44), neck
stiffness (39), fever (52), altered mental status (38), coma (8),
focal neurologic deficits (21), aphasia (12), hemiparesis (2),
cranial nerve palsies (5) | Immunocompromised
(42) | AMX or penicillin
monotherapy (11), third-generation cephalosporin monotherapy (12),
AMX or penicillin + third-generation cephalosporin (36), other
(3). | Yes (44) | Death (22),
sequelae (12), | (26) |
| Pelegrìn et
al, 2014 | 59 | Median age 64 | M (41), F (18) | Fever (54),
meningeal signs (46), altered mental status (44), headache (42),
hemiparesis (5), cranial nerve palsy (12), focal signs (15),
seizures (7) | DM (14), Chronic
corticosteroid therapy (14), cirrhosis (6), SOC (4), HM (3),
Immunosuppression (5) | AMP (15), AMP +
Aminoglycosides (39). Median duration of therapy 21 days. | Yes (30) | Death (14),
sequelae (8) | (32) |
| Amaya-Villar et
al, 2010 | 43 | Median age 69 | M (24), F (19) | Fever (39),
headache (29), vomiting (20), neck stiffness (30), seizures (4),
focal neurological deficit (14), cerebellum dysfunction (5) | Immunocompromised
(29) | AMP + GEN (18) | Yes (21) | Death (12),
sequelae (5) | (33) |
| Present study | 5 | 69 | M | Cough, fever,
nausea, headache, decreased consciousness | None | AMP 3 gr x 4 i.v.
and GEN 400 mg i.v. for 21 days | Yes | Complete
recovery | |
| | | 39 | M | Fever, headache,
emesis, speech problems, dizziness, neck stiffness | None | AMP 3 gr x 4 i.v.,
GEN 4 mg/kg/day i.v. for 21 days | Yes | Complete
recovery | |
| | | 49 | M | Abdominal pain,
nausea, fever, photophobia, neck stiffness | UC | AMP 3 gr x 4 i.v.
and GEN 4 mg/kg/day i.v. for 21 days | Yes | Complete
recovery | |
| | | 67 | M | Headache, fever,
disorientation, neck stiffness | B-cell lymphoma,
hypoglobu linemia | AMP 3 gr x 4 i.v.
and GEN 4 mg/kg/day i.v. for 21 days | Yes | Complete
recovery | |
| | | 72 | M | Fever, photophobia,
confusion, neck stiffness | CKD, DM | AMP 3 gr x 4 i.v.
and GEN 4 mg/kg/day i.v. | Yes | Death | |
Overall, Chau et al (34), Barocci et al (35), and Romero Gutiérrez et al
(36) reported four cases of
Listeria meningitis. Of these, 3 of the 4 patients described were
female. The male patient had no risk factors for Listeria
infection. In all cases, ampicillin was the antibiotic of choice,
with or without other drugs. Out of 4 patients, 3 of them made a
complete recovery. One patient died on the 6th day after
admission, on the same day the CSF culture came back positive for
Listeria monocytogenes. In this case, ampicillin was not
included in the empiric antibiotic therapy, probably due to the
absence of risk factors for neurolisteriosis. Dexamethasone
administration was initiated from clinical presentation; however,
in 2 cases the dosage is unknown (35,36).
Despite current suggestions to stop corticosteroid
administration when Listeria monocytogenes is detected in
CSF (28), dexamethasone is still
commonly prescribed for Listeria CNS infections. In our
professional experience, as shown in this case series, 4 out of 5
patients treated with a concomitant low dose of dexamethasone made
a full recovery without sequelae.
Although the subjects described had no signs of
encephalitis nor did they have any systemic complications, and the
number of reported cases is too small to make any statistical
considerations regarding the effectiveness and safety of
dexamethasone in these subjects, it does suggest that patients with
neurolisteriosis, in particular those with encephalitis, may
benefit from a low dose dexamethasone administration, especially
considering the effects on inflammations and edema. Presumably, the
successful outcomes described here are associated not only with
corticosteroid administration but also with the patients' mild
disease and the suitable antibiotic treatment.
In conclusion, despite the development of
antimicrobial therapy, the mortality and mobility rates of
neurolisteriosis remain high. Further studies on antibiotic
treatment as well as adjunctive therapies are needed to improve a
patient's morbidity and mortality. Dexamethasone has a central role
as both an adjunctive empiric therapy for patients affected by
meningitis of unknown origin and as a specific therapy for those
with pneumococcal meningitis. Concerning listeria meningitis,
dexamethasone administration should be carefully evaluated in
larger cohort studies and randomized clinical trials to better
assess its possible effectiveness and safety in these patients.
Acknowledgements
The authors would like to thank Dr Pietro Leanza,
University of Catania (Catania, Italy) for his assistance with the
editing of the manuscript.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
VM, AM, MC, FC, AZ, BMC, GN, and BC contributed to
the conception and design of the study. VM wrote the manuscript.
AM, MC, and FC searched the literature. AZ and BMC provided
clinical assistance to the patients and were responsible for the
pharmacological treatments. GN and BC revised the manuscript. GN
and BC confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
The patients signed written informed consent. Within
the consent, it was specified that data collected during the
diagnostic and therapeutic process can be anonymously used for
scientific purposes.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Pagliano P, Ascione T, Boccia G, De Caro F
and Esposito S: Listeria monocytogenes meningitis in the elderly:
Epidemiological, clinical and therapeutic findings. Le Infez Med.
24:105–111. 2016.PubMed/NCBI
|
|
2
|
Erdem H, Hargreaves S, Ankarali H,
Caskurlu H, Ceviker SA, Bahar-Kacmaz A, Meric-Koc M, Altindis M,
Yildiz-Kirazaldi Y, Kizilates F, et al: Managing adult patients
with infectious diseases in emergency departments: International
ID-IRI study. J Chemother. 33:302–318. 2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
El-Sokkary R, Uysal S, Erdem H, Kullar R,
Pekok AU, Amer F, Grgić S, Carevic B, El-Kholy A, Liskova A, et al:
Profiles of multidrug-resistant organisms among patients with
bacteremia in intensive care units: An international ID-IRI survey.
Eur J Clin Microbiol Infect Dis. 40:2323–2334. 2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
WHO. https://www.who.int/news-room/fact-sheets/detail/listeriosis.
|
|
5
|
Adriani KS, Brouwer MC, van der Ende A and
van de Beek D: Bacterial meningitis in pregnancy: Report of six
cases and review of the literature. Clin Microbiol Infect.
18:345–351. 2012.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Skogberg K, Syrjanen J, Jahkola M,
Renkonen OV, Paavonen J, Ahonen J, Kontiainen S, Ruutu P and
Valtonen V: Clinical presentation and outcome of listeriosis in
patients with and without immunosuppressive therapy. Clin Infect
Dis. 14:815–821. 1992.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Bula CJ, Bille J and Glauser MP: An
epidemic of food-borne listeriosis in Western Switzerland:
Description of 57 cases involving adults. Clin Infect Dis.
20:66–72. 1995.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Chen SY, Lee JJ, Chien CC, Tsai WC, Lu CH,
Chang WN and Lien CY: High incidence of severe neurological
manifestations and high mortality rate for adult Listeria
monocytogenes meningitis in Taiwan. J Clin Neurosci. 71:177–185.
2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
van de Beek D, Cabellos C, Dzupova O,
Esposito S, Klein M, Kloek AT, Leib SL, Mourvillier B, Ostergaard
C, Pagliano P, et al: ESCMID guideline: Diagnosis and treatment of
acute bacterial meningitis. Clin Microbiol Infect 22 Suppl.
3:S37–S62. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lorber B: Listeria monocytogenes. In:
Mandell, Douglas, and Bennett's Principles and Practice of
Infectious Diseases, 9th edition. Bennett J, Dolin R and Blaser M
(eds). Elsevier/Saunders', Philadelphia, PA, 2020;.
|
|
11
|
Thønnings S, Knudsen JD, Schønheyder HC,
Søgaard M, Arpi M, Gradel KO and Østergaard C: Danish Collaborative
Bacteraemia Network (DACOBAN). Antibiotic treatment and mortality
in patients with Listeria monocytogenes meningitis or bacteraemia.
Clin Microbiol Infect. 22:725–730. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Garcia-Carretero R: Clinical features and
predictors for mortality in neurolisteriosis: An administrative
data-based study. Bacteria. 1:3–11. 2022.
|
|
13
|
Celesia BM, Marino A, Borracino S,
Arcadipane AF, Pantò G, Gussio M, Coniglio S, Pennisi A, Cacopardo
B and Panarello G: Successful extracorporeal membrane oxygenation
treatment in an acquired immune deficiency syndrome (AIDS) patient
with acute respiratory distress syndrome (ARDS) complicating
pneumocystis jirovecii Pneumonia: A challenging case. Am J Case
Rep. 21(e919570)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Celesia BM, Marino A, Del Vecchio RF,
Bruno R, Palermo F, Gussio M, Nunnari G and Cacopardo B: Is it safe
and cost saving to defer the CD4+ cell count monitoring
in stable patients on art with more than 350 or 500 cells/µl?
Mediterr J Hematol Infect Dis. 11(e2019063)2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Marino A, Cosentino F, Ceccarelli M,
Moscatt V, Pampaloni A, Scuderi D, D'Andrea F, Rullo EV, Nunnari G,
Benanti F, et al: Entecavir resistance in a patient with
treatment-naïve HBV: A case report. Mol Clin Oncol.
14(113)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Marino A, Scuderi D, Locatelli ME, Gentile
A, Pampaloni A, Cosentino F, Ceccarelli M, Celesia BM, Benanti F,
Nunnar G, et al: Modification of serum brain-derived neurotrophic
factor levels following Anti-HCV therapy with direct antiviral
agents: A new marker of neurocognitive disorders. Hepat Mon.
20(e95101)2020.
|
|
17
|
Marino A, Zafarana G, Ceccarelli M,
Cosentino F, Moscatt V, Bruno G, Bruno R, Benanti F, Cacopardo B
and Celesia BM: Immunological and clinical impact of DAA-Mediated
HCV Eradication in a Cohort of HIV/HCV coinfected patients:
Monocentric Italian experience. Diagnostics (Basel).
11(2336)2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Marino A, Munafò A, Zagami A, Ceccarelli
M, Di Mauro R, Cantarella G, Bernardini R, Nunnari G and Cacopardo
B: Ampicillin plus ceftriaxone regimen against enterococcus
faecalis endocarditis: A literature review. J Clin Med.
10(4594)2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pagliano P, Arslan F and Ascione T:
Epidemiology and treatment of the commonest form of listeriosis:
Meningitis and bacteraemia. Infez Med. 25:210–216. 2017.PubMed/NCBI
|
|
20
|
Hernandez-Milian A and Payeras-Cifre A:
What is new in listeriosis? Biomed Res Int.
2014(358051)2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Brouwer MC, van de Beek D, Heckenberg SG,
Spanjaard L and de Gans J: Community-Acquired Listeria
monocytogenes meningitis in adults. Clin Infect Dis. 43:1233–1238.
2006.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Mylonakis E, Hohmann EL and Calderwood SB:
Central nervous system infection with listeria monocytogenes: 33
Yearsʼ experience at a general hospital and review of 776 episodes
from the literature. Medicine (Baltimore). 77:313–336.
1998.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Temple ME and Nahata MC: Treatment of
listeriosis. Ann Pharmacother. 34:656–661. 2000.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Lan ZW, Xiao MJ, Guan YL, Zhan YJ and Tang
XQ: Detection of Listeria monocytogenes in a patient with
meningoencephalitis using next-generation sequencing: A case
report. BMC Infect Dis. 20(721)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Arslan F, Meynet E, Sunbul M, Sipahi OR,
Kurtaran B, Kaya S, Inkaya AC, Pagliano P, Sengoz G, Batirel A, et
al: The clinical features, diagnosis, treatment, and prognosis of
neuroinvasive listeriosis: A multinational study. Eur J Clin
Microbiol Infect Dis. 34:1213–1221. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Koopmans MM, Brouwer MC, Bijlsma MW,
Bovenkerk S, Keijzers W, van der Ende A and van de Beek D: Listeria
monocytogenes sequence type 6 and increased rate of unfavorable
outcome in meningitis: Epidemiologic cohort study. Clin Infect Dis.
57:247–253. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
de Gans J and van de Beek D: European
Dexamethasone in Adulthood Bacterial Meningitis Study
Investigators. Dexamethasone in adults with bacterial meningitis. N
Engl J Med. 347:1549–1556. 2002.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Brouwer MC, McIntyre P, Prasad K and van
de Beek D: Corticosteroids for acute bacterial meningitis. Cochrane
Database Syst Rev: Jun 13, 2013 (Epub ahead of print).
|
|
29
|
Nau R, Djukic M, Spreer A and Eiffert H:
Bacterial meningitis: New therapeutic approaches. Expert Rev Anti
Infect Ther. 11:1079–1095. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Polkowska A, Toropainen M, Ollgren J,
Lyytikäinen O and Nuorti JP: Bacterial meningitis in Finland,
1995-2014: A population-based observational study. BMJ Open.
7(e015080)2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Charlier C, Perrodeau É, Leclercq A,
Cazenave B, Pilmis B, Henry B, Lopes A, Maury MM, Moura A, Goffinet
F, et al: Clinical features and prognostic factors of listeriosis:
The MONALISA national prospective cohort study. Lancet Infect Dis.
17:510–519. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Pelegrín I, Moragas M, Suárez C, Ribera A,
Verdaguer R, Martínez-Yelamos S, Rubio-Borrego F, Ariza J,
Viladrich PF and Cabellos C: Listeria monocytogenes
meningoencephalitis in adults: Analysis of factors related to
unfavourable outcome. Infection. 42:817–827. 2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Amaya-Villar R, García-Cabrera E,
Sulleiro-Igual E, Fernández-Viladrich P, Fontanals-Aymerich D,
Catalán-Alonso P, Rodrigo-Gonzalo de Liria C, Coloma-Conde A,
Grill-Díaz F, Guerrero-Espejo A, et al: Three-year multicenter
surveillance of community-acquired listeria monocytogenes
meningitis in adults. BMC Infect Dis. 10(324)2010.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Chau TT, Campbell JI, Schultsz C, Chau NV,
Diep TS, Baker S, Chinh NT, Farrar JJ and van Doorn HR: Three adult
cases of Listeria monocytogenes meningitis in Vietnam. PLoS Med.
7(e1000306)2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Barocci S, Mancini A, Canovari B, Petrelli
E, Sbriscia-Fioretti E, Licci A, D'Addesa S, Petrini G, Giacomini
M, Renzi A, et al: Listeria monocytogenes meningitis in an
immunocompromised patient. New Microbiol. 38:113–118.
2015.PubMed/NCBI
|
|
36
|
Romero Gutiérrez J, Garcia Peña J, Rodrigo
Casanova P and Aguilera Celorrio L: Listeria monocytogenes and
Herpes simplex type 1, associated in a meningoencephalitis episode
in an immunodepressed patient. Rev Esp Anestesiol Reanim.
59:227–228. 2012.PubMed/NCBI View Article : Google Scholar : (In Spanish).
|