Introduction
The term Fournier's gangrene (FG) was used for the
first time in 1886 by the French venereologist Jean Fournier to
describe a necrotizing polymicrobial infection of the soft tissues
of the urogenital or anogenital area (1). This pathology is rare but potentially
fatal, accounting for 0.02% of hospital admissions, with an
incidence of 1.6 per 100,000 men and 0.25 per 100,000 women. The
literature describes a mortality rate ranging from 3 to 67% of
patients (2,3). Among the positive findings on physical
examination are edema, erythema, local heat and necrotic lesions in
the genital and anorectal region with rapidly progressive
worsening, expanding to the abdomen, flanks or thorax (3). FG is a life-threatening condition that
requires urgent medical attention and treatment. Certain relevant
aspects that must be considered in FG include rapid progression,
polymicrobial infection, predisposing factors (diabetes mellitus,
immunosuppression, obesity, peripheral vascular disease, alcohol
abuse, compromise of the immune system or blood supply to the
genital area), clinical presentation, systemic signs, diagnostic
imaging, early diagnosis and treatment, surgical intervention,
intensive care and support, and mortality risk. Given the critical
nature of FG, a multidisciplinary approach involving surgeons,
infectious disease specialists, critical care physicians and other
healthcare professionals is essential to achieve the best possible
outcomes (1-3).
Studies using bedside ultrasonography [point-of-care
ultrasound (PoCUS)] have described marked thickening of the scrotal
fascia with edema, free unilateral or bilateral peri-testicular
fluid and areas of subcutaneous gas. PoCUS has the potential to
have a valuable role in the diagnosis of FG. Early diagnosis and
prompt treatment are crucial for improving patient outcomes and
reducing morbidity and mortality. Potential utilities of PoCUS in
the diagnosis of FG include rapid assessment, detection of gas in
soft tissues, visualization of fluid collections and abscesses,
monitoring disease progression, guiding surgical interventions and
screening in high-risk populations (4,5).
The present case report highlights the usefulness of
PoCUS in facilitating diagnostic and therapeutic decision-making in
a patient with FG in an emergency department.
Case report
A 63-year-old woman with a history of arterial
hypertension, type 2 diabetes mellitus and obesity consulted the
emergency department of the Hospital San Vicente Fundación
(Rionegro, Colombia) due to a 3-day history of pain in the left
buttock associated with changes in local inflammation and purulent
discharge, extending to the anorectal region. On admission, the
patient was hypotensive (mean arterial pressure, 50 mmHg; lower
limit of normal, 65 mmHg), with tachycardia, without fever and
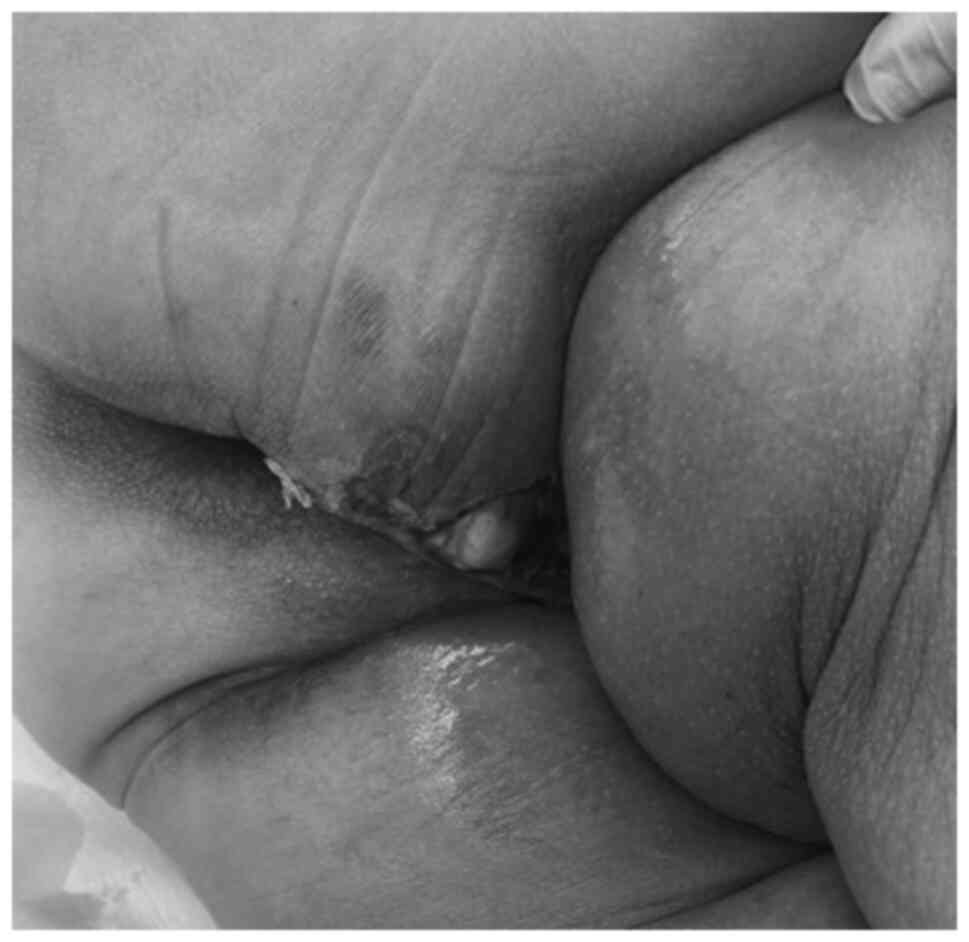
reporting intense pain in the affected area. On physical
examination, redness, flushes and local heat with an approximate
induration area of 15x10 cm were observed in the left gluteal
region, along with an ulcer with necrotic edges and purulent
discharge, without subcutaneous emphysema (Fig. 1).
The diagnostic impression was septic shock due to
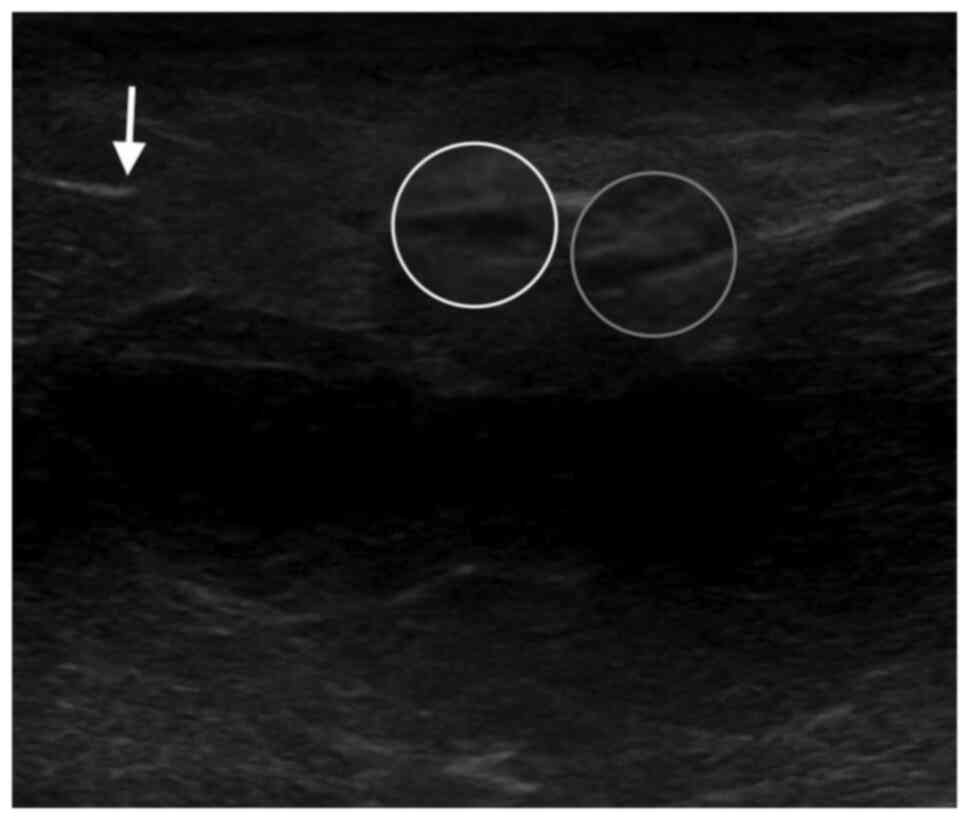
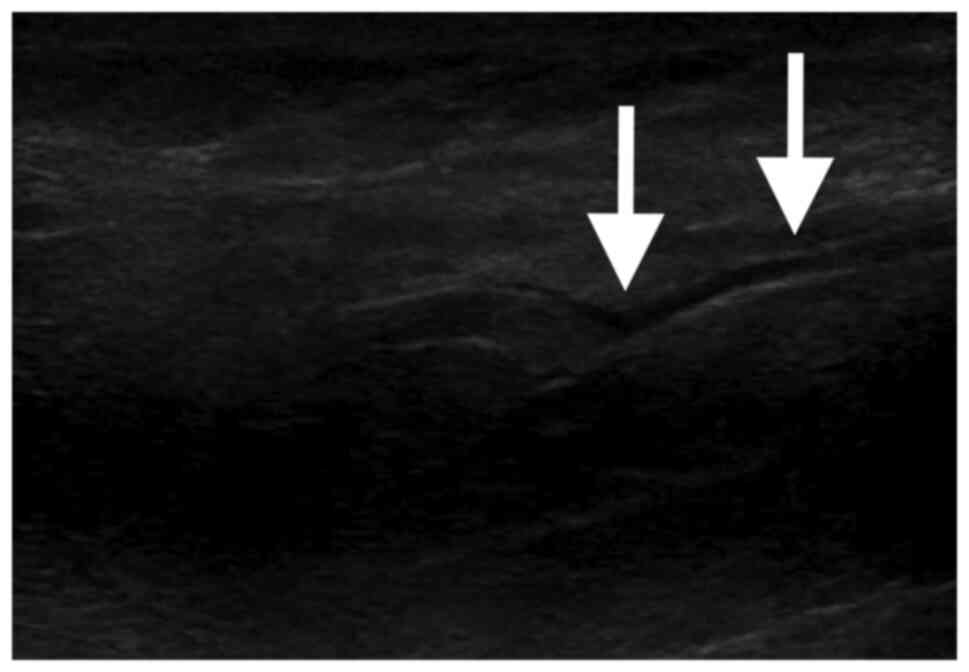
FG-type necrotizing soft tissue infection. PoCUS was performed,
revealing free fluid in the muscle planes, discontinuity of the
muscle fascia and the presence of gas in the subcutaneous cellular
tissue (Figs. 2 and 3). Pharmacological management was started
with intravenous crystalloids and antibiotic therapy with
piperacillin/tazobactam and vancomycin, together with prior blood
culture collection. The paraclinical tests on admission showed an
increase in acute phase reactants: Leukocytes, 17,400/µl (normal
range, 4,500-11,500/µl); neutrophils, 15,430/ml (normal range,
2,000-7,500/ml); and C-reactive protein, 33 mg/dl (normal range,
0.4-1 mg/dl). Hyperlactatemia (lactate, 2.2 mmol/l; normal range,
0.6-1.6 mmol/l), hyponatremia (sodium, 132 mmol/l; normal range,
135-145 mmol/l), creatinine abnormality (2.01 mg/dl; normal range,
0.57-1.11 mg/dl) and high urea nitrogen (60.5 mg/dl; normal range,
9-23 mg/dl) were found, findings that suggest a diagnosis of acute
kidney injury.
The patient was taken to surgery 2 h after admission
in accordance with the Surviving Sepsis guidelines (6). The patient exhibited necrosis in the
skin, subcutaneous cell tissue and fascia, with extension to the
right greater vaginal lip, without anorectal involvement. In the
hospital's surgical report and medical record of the patient,
abundant purulent material was described; washing and debridement
were performed and cultures were collected.
In the immediate postoperative period, the patient
was taken to the intensive care unit, where septic shock management
was continued, including vasopressor support with norepinephrine
0.2 µg/kg/min and vasopressin 0.06 IU/min. Linezolid was adjusted
to vancomycin due to persistent renal damage. At 48 h after
admission, a new lavage and surgical debridement were performed,
revealing a large coverage defect that included the mons pubis,
running through the right labia majora to the inner face of the
buttock, and compromising the perineum and midline, with a necrotic
pocket and septate abscesses towards the posterior area.
At six days after admission, the patient was
transferred to the intermediate care unit. No positive
microbiological isolation was achieved in the blood cultures or
surgical cultures according to standard procedures (6) and a 14-day course of linezolid and
piperacillin/tazobactam was completed. Subsequently, the coverage
defect was corrected with local flap without complications,
achieving adequate metabolic control. The patient was discharged
after being hospitalized for 33 days, without any limitations in
mobility and under hemodynamic stability after completing
intrahospital therapy. After hospital discharge, the patient was
followed up for 6 weeks and exhibited a favorable healing
process.
Discussion
The diagnosis of FG in the emergency department is
challenging, as the clinical, laboratory and imaging findings are
not specific. Furthermore, patients may newly develop the disease
without having any of the conventionally described risk factors,
such as age, history of diabetes mellitus, alcoholism, obesity or
immunosuppression (3,7).
Approximate mortality rates of 20-50% have been
described, with the main sources of infection being the
gastrointestinal tract (30-50%), genitourinary system (20-40%),
skin lesions (20%) and idiopathic infections (<20%) (2,8). The
pathogens involved in these infections are microorganisms stemming
from the perineal and genital region, including aerobes
(Escherichia coli, Klebsiella pneumoniae,
Streptococcus aureus), anaerobes (Bacteroides
fragilis, Clostridium), other bacterial species such as
vibrio, streptococci, enterococci and corynebacteria, and fungal
agents such as Candida albicans and zygomycetes (2,4).
The infection compromises the superficial and deep
muscle fascia, facilitated by etiological factors and depending on
the patient's immune status and the route of entry of the
microorganism (8). These
pro-inflammatory mechanisms favor hypoxia, producing micro-infarcts
in the nervous tissue that result in high-intensity pain in the
affected area and necrosis (8).
In patients with suspected FG, it is necessary to
carry out a detailed anamnesis and physical examination of the
genital and perineal areas in search of wounds or infectious
processes that may go unnoticed. The findings in the initial stages
of the disease are edema (80%), disproportionate pain (79%) and
erythema (70%). Bullae and dermal necrosis, together with crepitus
in the abdominal region that may extend to the thorax, are findings
related to the late stages of the disease, the latter being highly
specific to Clostridium infection (3,7).
Finally, the presence of hypotension and septic shock are ominous
signs associated with high mortality and multi-organ failure, which
is the main cause of death in necrotizing soft tissue infections
(2,9).
The diagnosis of FG is mainly clinical and imaging
should not delay the surgical approach (7,9).
Images are useful when it is necessary to know the extension, or in
atypical presentations of the disease. According to the literature,
plain radiography has a sensitivity of 49% and a specificity of 94%
because gas formation is present in almost half of all patients
with FG. Computed tomography has a sensitivity of close to 90% and
a specificity of 93.3% for necrotizing soft tissue infections,
which allows for the determination of the possible extension of the
disease and other less obvious lesions. Magnetic resonance imaging
is another diagnostic modality; a sensitivity of 100% and a
specificity of 86% have been described for the detection of
necrotizing soft tissue infections. However, in the acute setting
of the emergency service, it is limited mainly by its high cost,
institutional availability and time spent performing the procedure
(10).
PoCUS has emerged as a useful tool for the
assessment of soft tissue inflammation. Certain studies have
recorded a sensitivity of 88% and a specificity of 93% for the
detection of necrotizing soft tissue infections (9-11).
Positive findings include the ‘cobblestone’ sign described at the
subcutaneous level, hyperechoic reverberation artifacts called
‘snow globe’ or ‘dirty shadow’, free fluid in the muscle fascia and
intrascrotal gas (4,5,9).
Furthermore, studies suggest that ultrasound may have better
diagnostic accuracy in patients with negative tomography or
magnetic resonance imaging findings, and may be used in
hemodynamically unstable patients and in cases where transport to a
radiology room is usually unsafe due to the patient's hemodynamic
condition (2,9,10).
Considering the rapid hemodynamic deterioration of
the patient, the use of PoCUS may help guide a possible diagnosis
of FG. In addition, the use of intravenous contrast is
contraindicated in certain patients due to renal failure associated
with soft tissue sepsis, which may limit the use of contrast
imaging, and at certain hospitals nuclear magnetic resonance is not
available; for these reasons, PoCUS would be useful in
investigating some of the signs mentioned. Likewise, the usefulness
of PoCUS lies in the search for other identifiable causes, such as
epididymitis, orchitis, abscess and incarcerated or strangulated
hernia, among others. The sensitivity of PoCUS in necrotizing
fasciitis may range from 88 to 100%, with a specificity of 87.5 to
93% and a negative predictive value of 95.4% (12).
The availability of ultrasound at the patient's
bedside allows for the rapid diagnosis of infections, revealing
certain signs that may suggest them, such as the presence of gas or
fluid. Furthermore, significant time can be saved by making a
timely diagnosis of FG with PoCUS compared to taking the patient to
the radiological service and performing contrasting imaging
(13).
The therapeutic pillars for FG include adequate
hemodynamic resuscitation, emergent surgical debridement and the
early initiation of broad-spectrum parenteral antibiotic therapy
(14,15). Among the first-line antibiotics are
carbapenems (imipenem, meropenem) or beta-lactams with
beta-lactamase inhibitors (piperacillin/tazobactam), combined with
clindamycin plus drugs with a Gram-positive target spectrum, such
as vancomycin, daptomycin or linezolid. However, in scenarios where
the patient is at high risk of fungal infections, starting
amphotericin B or fluconazole should be considered (8,10). The
final disposition of these patients depends on their hemodynamic
status and comorbidities, although they generally require intensive
care unit stays in the hospital (16,17).
It is important to highlight that, although in the
present case the microbiological results were not positive either
at the emergency department or in the operating room, the
microbiological findings may help guide the patient's antibiotic
therapy. However, it has been reported that positivity for
microorganisms in cases of FG is relatively low (18). For this reason, in the current
setting, a broad-spectrum empirical therapy was chosen.
Advanced imaging, such as computer tomography, is
currently the gold standard for diagnosis; however, its application
may be difficult due to the instability of numerous affected
patients, as well as the additional time, costs and radiation and
contrast exposure involved (19).
PoCUS provides valuable real-time information to
surgeons, aiding in the planning of further surgical treatment for
patients. Its ability to provide rapid diagnosis, procedural
guidance, treatment monitoring, preoperative planning, bedside
assessment and enhancement of patient safety make it an
indispensable tool in surgical decision-making. By incorporating
PoCUS into their practice, surgeons may optimize patient care,
improve surgical outcomes and implement appropriate interventions
(20,21).
In conclusion, the use of ultrasonography in the
emergency setting is able to provide adequate diagnostic support
for necrotizing soft tissue infections, including FG. It is
necessary to carry out studies to evaluate its impact on patient
outcomes, such as the time from hospital admission to surgical
treatment, to establish it as a tool for systematic use in the
event of clinical suspicion in the emergency department.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
MZG, JCVR, SQV, DGO, DGA and CMA contributed to the
conception and design of the study. CMA wrote the manuscript. MZG,
JCVR, SQV, DGA and DGO searched the literature. MZG, JCVR, SQV, DGA
and DGO provided clinical assistance to the patient and were
responsible for the treatments. MZG and CMA revised the manuscript.
MZG and CMA confirm the authenticity of all the raw data. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
The Bioethics Committee of San Vicente Fundación
Hospital (Rionegro, Colombia) approved the publication of this
case.
Patient consent for publication
Written informed consent for the publication of
clinical details and images was obtained from the patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Horta R, Cerqueira M, Marques M, Ferreira
P, Reis J and Amarante J: Fournier's gangrene: From urological
emergency to plastic surgery. Actas Urol Esp. 33:925–929.
2009.PubMed/NCBI View Article : Google Scholar : (In Spanish).
|
|
2
|
Montrief T, Long B, Koyfman A and Auerbach
J: Fournier gangrene: A review for emergency clinicians. J Emerg
Med. 57:488–500. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Molla YD, Assefa MA and Abraha AY:
Fournier's gangrene with retroperitoneal extension, a case report.
Int J Surg Case Rep. 105(107984)2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Coyne C, Mailhot T and Perera P: Diagnosis
of Fournier's gangrene on bedside ultrasound. West J Emerg Med.
15(122)2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kim DJ and Kendall JL: Fournier's gangrene
and its characteristic ultrasound findings. J Emerg Med.
44:e99–e101. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Pelletier J, Gottlieb M, Long B and
Perkins JC Jr: Necrotizing soft tissue infections (NSTI): Pearls
and pitfalls for the emergency clinician. J Emerg Med. 62:480–491.
2022.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Evans L, Rhodes A, Alhazzani W, Antonelli
M, Coopersmith CM, French C, Machado FR, Mcintyre L, Ostermann M,
Prescott HC, et al: Surviving sepsis campaign: International
guidelines for management of sepsis and septic shock 2021. Crit
Care Med. 49:e1063–e1143. 2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Auerbach J, Bornstein K, Ramzy M, Cabrera
J, Montrief T and Long B: Fournier gangrene in the emergency
department: Diagnostic dilemmas, treatments and current
perspectives. Open Access Emerg Med. 12:353–364. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Stevens DL and Bryant AE: Necrotizing
soft-tissue infections. N Engl J Med. 377:2253–2265.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Castleberg E, Jenson N and Dinh VA:
Diagnosis of necrotizing fasciitis with bedside ultrasound: The
STAFF Exam. West J Emerg Med. 15:111–113. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kehrl T: Point-of-care ultrasound
diagnosis of necrotizing fasciitis missed by computed tomography
and magnetic resonance imaging. J Emerg Med. 47:172–175.
2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yen ZS, Wang HP, Ma HM, Chen SC and Chen
WJ: Ultrasonographic screening of clinically-suspected necrotizing
fasciitis. Acad Emerg Med. 9:1448–1451. 2002.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Levenson RB, Singh AK and Novelline RA:
Fournier gangrene: Role of imaging. Radiographics. 28:519–528.
2008.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Nawijn F, Smeeing DPJ, Houwert RM, Leenen
LPH and Hietbrink F: Time is of the essence when treating
necrotizing soft tissue infections: A systematic review and
meta-analysis. World J Emerg Surg. 15(4)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Eckmann C and Montravers P: Current
management of necrotizing soft-tissue infections. Curr Opin Infect
Dis. 34:89–95. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Peetermans M, de Prost N, Eckmann C,
Norrby-Teglund A, Skrede S and De Waele JJ: Necrotizing skin and
soft-tissue infections in the intensive care unit. Clin Microbiol
Infect. 26:8–17. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Sartelli M, Guirao X, Hardcastle TC,
Kluger Y, Boermeester MA, Raşa K, Ansaloni L, Coccolini F,
Montravers P and Abu-Zidan FM: , et al: 2018 WSES/SIS-E
consensus conference: Recommendations for the management of skin
and soft-tissue infections. World J Emerg Surg.
13(58)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Lyons NB, Cohen BL, O'Neil CF Jr, Ramsey
WA, Proctor KG, Namias N and Meizoso JP: Short versus long
antibiotic duration for necrotizing soft tissue infection: A
systematic review and meta-analysis. Surg Infect (Larchmt).
24:425–432. 2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ramm L, Guidry K, Cirilli A, Kurkowski E
and Yu C: Critical point-of-care ultrasound diagnosis of Fournier's
gangrene: A case report. Clin Pract Cases Emerg Med. 6:57–60.
2022.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Pek JH and Teo LYL: Point-of-care
ultrasound in the evaluation of patients with left ventricular
assist devices at the emergency department. J Emerg Med.
62:348–355. 2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Shaahinfar A and Ghazi-Askar ZM:
Procedural applications of point-of-care ultrasound in pediatric
emergency medicine. Emerg Med Clin North Am. 39:529–554.
2021.PubMed/NCBI View Article : Google Scholar
|