Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of
the most lethal and challenging malignancies, with rising incidence
and mortality rates. According to the Global Cancer Observatory
(GLOBOCAN) in 2022, pancreatic cancer was ranked 12th in terms of
global incidence and 6th in terms of cancer-related mortality.
Geographically, 45.5% cases were reported in Asia, followed by
Europe (28.7%), North America (13.1%), Latin America and the
Caribbean (8%), Africa (3.7%) and Oceania (0.95%) (1). GLOBOCAN projects that pancreatic
cancer will become the second leading cause of cancer-related
mortality in the United States by 2025(2). In Chile, pancreatic cancer accounted
for 5% cancer-related mortality in 2012, increasing to 5.6% in
2018, making it is the seventh leading cause of cancer-related
mortality (3). Despite significant
advances in cancer research, PDAC still has a 5-year survival rate
of only ~10% (4,5). PDAC is the most prevalent histological
type of pancreatic cancer, accounting for >90% of cases. Other
types include cystadenocarcinoma, acinar cell carcinoma and
neuroendocrine tumors (6).
According to age at diagnosis, pancreatic cancer is stratified into
early-onset pancreatic cancer (EOPC), defined as diagnosis before
50 years of age, and late-onset pancreatic cancer (LOPC), diagnosed
at 50 years of age or older (7).
Advances in understanding the molecular basis of PDAC and the
development of targeted therapies have provided new therapeutic
hope (7-9).
On molecular level, PDAC is caused by either inherited (germline)
or acquired (somatic) mutations. PDAC is primarily driven by four
genes, namely KRAS, CDKN2A (p16), TP53 and
SMAD4. The most frequently mutated gene is KRAS, a
member of the RAS family of small GTPases that plays a role in
early carcinogenesis (10).
Additionally, CDKN2A, a tumor suppressor gene responsible
for regulating the cell cycle, has been found to be inactivated in
~95% of cases, thereby accelerating cell cycle progression.
Similarly, TP53 has been shown to be mutated in 50-75%
tumors, causing it to lose the ability to repair DNA damage and
facilitate the proliferation of damaged cells. Furthermore, 50%
cases involve the inactivation of SMAD4, leading to alterations in
cell cycle regulation, constitutive activation of signaling
pathways and uncontrolled cell proliferation (9,10). The
present case of PDAC represents an early-onset presentation with a
distinctive molecular profile, notably characterized by the absence
of KRAS mutations.
Case report
A 45-year-old male with no significant medical
history presented to Indisa Clinic in Santiago, Chile, in October
2024 with persistent epigastric pain and mild jaundice. Physical
examination revealed a palpable mass in the epigastric region with
no other notable findings. Upper gastrointestinal endoscopy
revealed an infiltrating duodenal lesion and biopsy-confirmed
infiltrative adenocarcinoma. Laboratory results (Table I) indicated several genomic
abnormalities consistent with those observed in advanced pancreatic
cancer. Mild anemia is a common finding in patients with cancer and
was observed in this patient. Elevated total and direct bilirubin
levels, along with progressively increasing alkaline phosphatase
and high γ-glutamyl transferase levels, strongly suggest hepatic
dysfunction, likely secondary to bile duct obstruction. A low
albumin level was also noted, potentially reflecting malnutrition
or further liver involvement. Lactate dehydrogenase (LDH) levels
were within normal limits. Additionally, mild hyperglycemia was
present, possibly associated with altered cancer metabolism. Some
laboratory parameters were found to be within normal limits
(Table SI).
 | Table IVariants of uncertain significance in
a patient with early-onset of ductal pancreatic adenocarcinoma: A
genomic molecular study. |
Table I
Variants of uncertain significance in
a patient with early-onset of ductal pancreatic adenocarcinoma: A
genomic molecular study.
| Gene | Result | Region | Transcript | Frequency (%) | Level of
mutation |
|---|
| MSH3 | p.A57_A62del
(c.162_179del) | EX1 | NM_002439.5 | 1.92 | Tier III |
| MUC1 | p.P143Q
(c.428C>A) | EX2 | NM_001371720.1 | 1.56 | Tier III |
| CHD1 | p.S1687F
(c.5060C>T) | EX36E | NM_001270.4 | 0.51 | Tier III |
| ZC3H7B | p.T833M (c.2498
C>T) | EX21 | NM_017590.6 | 0.24 | Tier III |
| MUC16 | p.S2342Ll
(c.7025C>T) | EX1 | NM_024690.2 | 0.16 | Tier III |
| ERBB2 | p.S413L
(c.1238C>T) | EX11 | NM_004448.4 | 0.14 | Tier III |
| AFDN | p.E838K
(c.2512G>A) | EX19 | NM_001386888.1 | 0.1% | Tier III |
An endoscopic ultrasound-guided biopsy was
performed. Histological examination using H&E (Sigma-Aldrich;
Merck KGaA) was performed. Tissue specimens were fixed in 10%
neutral buffered formalin at room temperature for 24-48 h, followed
by routine processing and paraffin embedding. Paraffin sections
were cut manually to a thickness of 4 µm and mounted on glass
slides. Staining was performed manually using commercially
available reagents (Sigma-Aldrich; Merck KGaA) according to the
manufacturer's instructions. Staining was performed using
hematoxylin for ~5 min and eosin for 1-2 min, both carried out at
room temperature. Histological assessment was performed using light
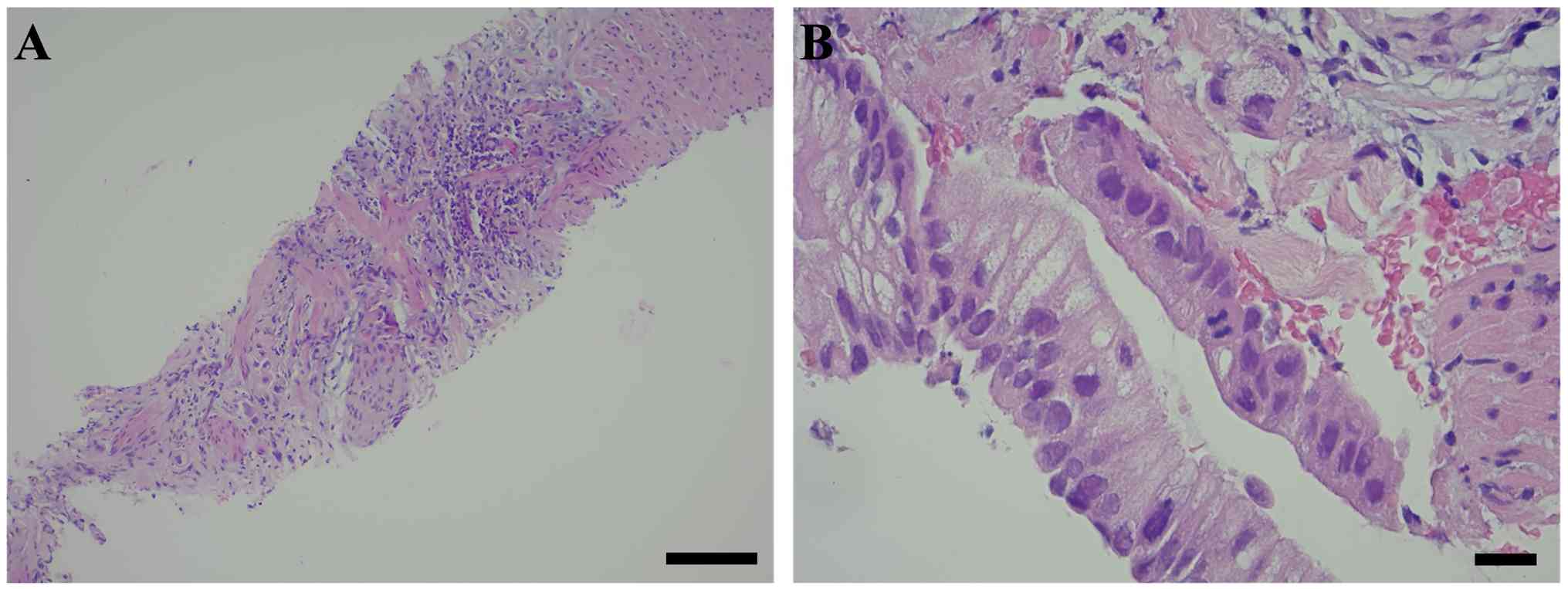
microscopy. Staining revealed fibroconnective tissue exhibiting a
desmoplastic reaction and a chronic non-specific inflammatory
response infiltrated by isolated clusters of epithelial neoplastic
malignant cells. No normal glandular pancreatic structures were
observed (Fig. 1A). At higher
magnification, another fragment of fibroconnective tissue was
noted, infiltrated by isolated neoplastic malignant epithelial
cells containing mucin-filled vacuoles (upper right), alongside
atypical glandular epithelium displaying architectural disarray and
high-grade nuclear and cytological atypia (upper left and central).
These findings are consistent with high-grade dysplasia,
potentially indicative of pancreatic intraepithelial neoplasia,
PanIN 3 (high-grade; Fig. 1B).
Radiological assessments indicated that the lesion
in the initial portion of the duodenum lacked a discernible
division plane, resulting in dilation of the central pancreatic
duct and mild tail atrophy. Vascular structures, including the
celiac trunk and superior mesenteric artery, appeared to maintain a
division plane with the neoplasm (Fig.
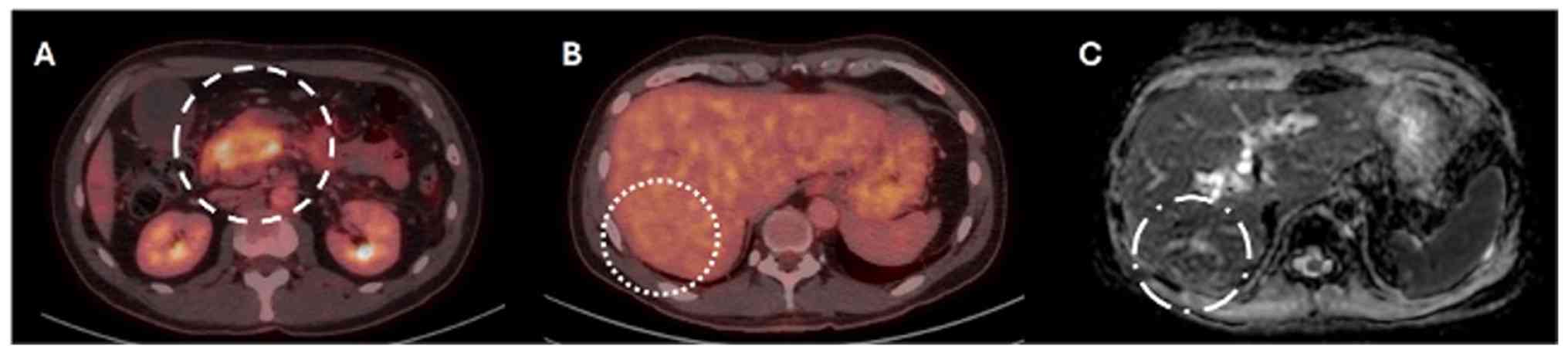
2A). PET-CT revealed three focal hypodense liver lesions in
segments VI, IVa, and II, all without metabolic activity (Fig. 2A). One lesion was suspicious for
metastasis on PET-CT (Fig. 2B,
dotted circle) and was subsequently confirmed on MRI, which
demonstrated ring-like enhancement following gadolinium
administration on ADC-weighted imaging (Fig. 2C, dotted circle). Several focal
hepatic lesions with a non-suspicious appearance, such as
hemangiomas, were also noted (Fig.
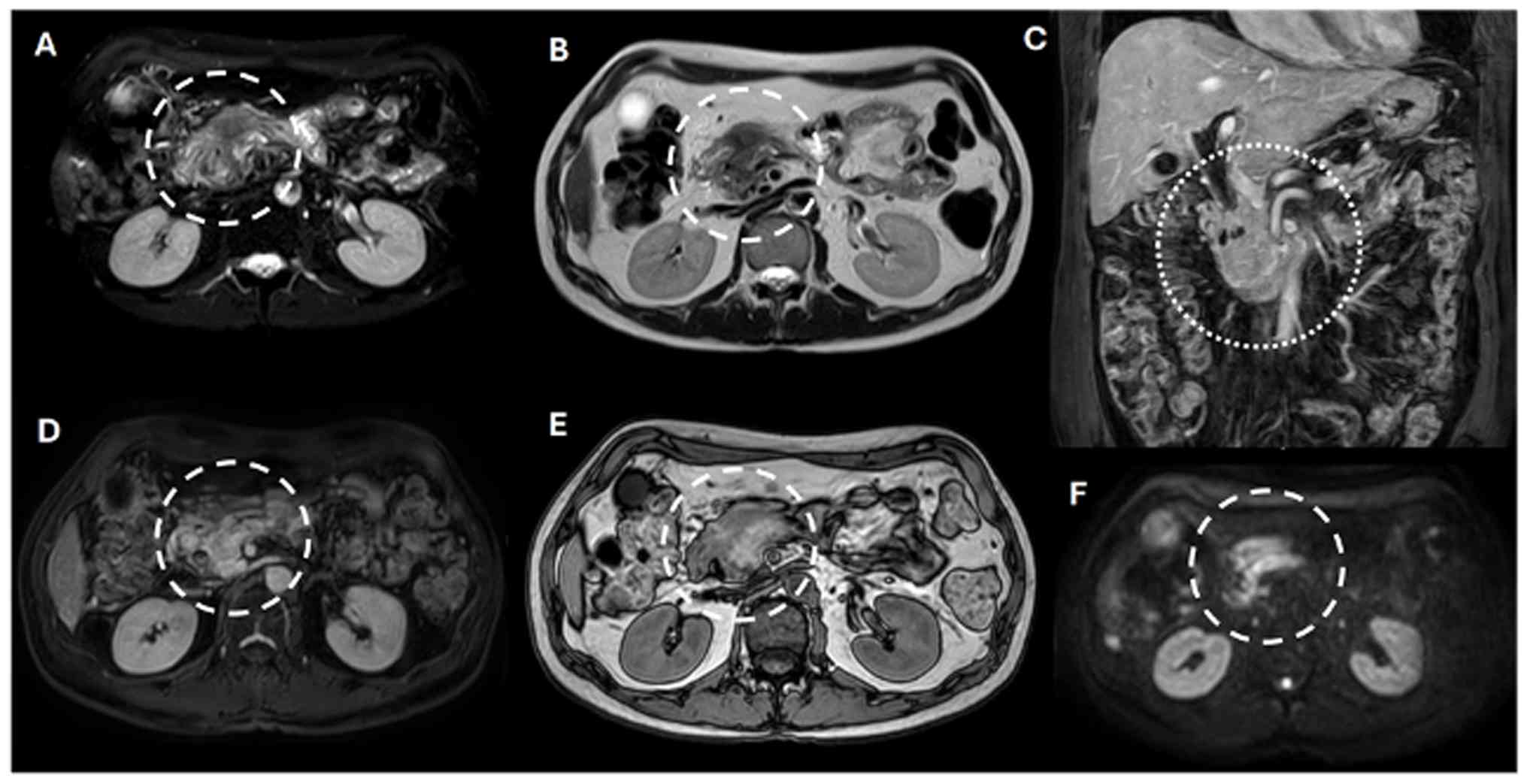
3). A mass was identified in the head of the pancreas (Fig. 3A, dashed circles), exhibiting
perivascular invasion that compromised the superior mesenteric
artery, inferior vena cava and abdominal aorta (Fig. 3C, dotted circle). The lesion
appeared hyperintense on T2-weighted images (Fig. 3A-C, circles). Dixon-weighted
sequence post-gadolinium administration demonstrated heterogeneous
enhancement, which was highly suggestive of an aggressive
neoplastic process (Fig. 3D, dashed
circle). The out-of-phase image from the dual fast-field echo
sequence displayed a heterogeneous signal intensity, indicating a
hemorrhagic component and/or associated vascular changes (Fig. 3E, dashed circle). Furthermore,
restricted diffusion of water molecules was observed on high-value
diffusion-weighted imaging, supporting this diagnosis (Fig. 3F, dashed circle). Collectively,
these imaging findings confirmed the diagnosis of an unresectable
PDAC (T4N0M1; Grade IV).
Genomic analysis was performed by BGI on circulating
tumor DNA (ctDNA) using the Sentis™ Cancer + Discovery
panel, a targeted 816-gene NGS assay on the DNBSEQ-G400/T7 platform
with high sequencing depth (≥2700X). Sequencing and library
preparation were performed according to the manufacturer's
protocols, and variants were interpreted using the BGI Shizhen
Database, which provides guidance on targeted therapy,
chemotherapy, immunotherapy, and hereditary tumor risk assessment.
KRAS mutations were not detected, but multiple variants of
uncertain significance (VUS) were detected in the following genes:
MSH3 (1.92%); MUC1 (1.56%), CHD1 (0.51%),
ZC3H7B (0.24%), MUC16 (0.16%), ERBB2 (0.14%)
and AFDN (0.12%). Furthermore, it revealed low
microsatellite stability (MSI-L/MSS) and low tumor mutational
burden (bTMB: 1.26 Muts/Mb). No pathogenic germline mutations were
identified in this patient (Table
I).
Given the advanced stage and initially unresectable
nature of the disease, first-line systemic therapy with eight
cycles of FOLFIRINOX was initiated, resulting in disease
stabilization without radiological disease progression.
Nevertheless, the rarity of this presentation and the limited
clinical benefit observed with chemotherapy alone led to a
reassessment of the therapeutic approach, resulting in a
reconsideration of surgical management. As the disease progresses,
these findings support the need to explore additional therapeutic
strategies, including human epidermal growth factor receptor 2
(HER2)-targeted approaches.
Discussion
PDAC is characterized by KRAS mutations,
which exhibit a high frequency of constitutive activation in 90% of
cases. The patient in this case report presented with advanced
clinical progression of pancreatic cancer, marked by obstructive
jaundice and abdominal pain radiating to the back. Among these
symptoms, abdominal pain tends to be the most prevalent, occurring
in 79% of cases (10). Physical
examination typically reveals a palpable epigastric mass in only 9%
of cases (11). Obstructive
jaundice results from common bile duct obstruction caused by a mass
in the pancreatic head (12). These
findings indicated an advanced stage of pancreatic cancer with
hepatic and systemic involvement, consistent with the PET-CT
results showing three hypodense focal lesions in liver segments VI,
IVa and II (≤9 mm) without increased metabolic activity.
Given the diagnostic and prognostic significance of
tumor markers in pancreatic cancer, CA19-9 is the most widely used
biomarker in clinical practice. In symptomatic patients, levels
>37 U/ml demonstrate a sensitivity of 79-80% and specificity of
82-90%, whilst values >100 U/ml reach a specificity of 98%
(13). However, its utility as a
screening tool in the general population is limited, with a
positive predictive value of only 0.9%. Additionally, ~10% of the
population lacks the Lewis blood group antigen, which is required
for CA19-9 production, leading to reduced sensitivity (14,15).
Although CA19-9 is not specific for the initial diagnosis, it can
also be elevated in other malignancies, including biliary, gastric,
colorectal, liver and lung cancers. Instead, its primary value lies
in disease monitoring, treatment response assessment and recurrence
detection. In the present case, the CA19-9 level was recorded at 1
U/ml, which is an unusually low value for symptomatic, unresectable
and metastatic pancreatic adenocarcinoma. MicroRNAs (miR) detected
in blood, such as miR-21, miR-155, miR-196a and miR-200c, have
shown significant diagnostic and prognostic potential in PDAC and
can be incorporated as complementary biomarkers (16).
To further investigate the molecular profile of the
tumor and advance the diagnostic workup, liquid biopsy was
performed. The analysis identified seven mutations of uncertain
significance. Notably, the KRAS oncogene was not detected,
which was an atypical finding, since invasive ductal
adenocarcinomas exhibit the highest frequency of KRAS alterations,
with constitutive activation in ~90% of the cases. In the absence
of KRAS mutations, further liquid biopsy analysis identified
seven VUS. One such VUS was the MUC1 gene, in which the
amino acid proline, encoded by codon 143 exon 2, is substituted by
glutamine. MUC1 encodes mucin 1, a component of mucus that
lubricates and protects various body surfaces, including the
respiratory, digestive and reproductive tracts (17). This gene is expressed in nearly all
pancreaticobiliary and invasive PDACs. In tumor contexts, MUC1
protein expression is predominantly localized to the luminal
membrane of duct-forming regions but may also be detected in the
cytoplasm of poorly differentiated areas, a pattern associated with
increased tumor aggressiveness (18,19).
MUC1 homodimers interact with HER2 to facilitate constitutive
activation. Furthermore, they may stabilize HER2-HER3 and HER2-EGFR
heterodimers, thereby extending downstream signaling, even in the
absence of ligand stimulation, likely by enhancing the stability of
these receptor complexes (20). The
Cancer Genome Atlas reports that MUC1 mutations are present in
~0.56% pancreatic adenocarcinoma cases, underscoring the low
frequency of this gene mutation in this malignancy. It is also well
known that the frequency of HERBB2 overexpression in PDAC is low
(21). In a previous study of
37,864 cases, 2,072 (5.47%) were identified as pancreatic
adenocarcinomas, of which 2.7% tested positive for HER2. Mutations
in ERBB2 have been reported in 1.8% of various cancers according to
analyses of 7,300 solid tumor samples, with the highest prevalence
observed in breast, lung, ovarian, and colon cancers (22,23).
In the present case, the S413L mutation
(c.1238C>T) was identified in exon 11 of ERBB2, resulting in a
serine-to-leucine substitution due to a single nucleotide change.
This variant is classified as a VUS according to the American
College of Medical Genetics and Genomics criteria as implemented by
Franklin (https://franklin.genoox.com/) (24). Although it meets some pathogenic
indicators, such as PM2 (absence in healthy populations, extremely
low frequency) and PP2 (high prevalence of pathogenic missense
variants in ERBB2), there is no conclusive evidence that it is a
pathogenic mutation. Notably, to the best of our knowledge, this
mutation has not been previously reported in PDAC and refractory
metastatic breast cancer (25).
Consequently, therapeutic decisions should not be
based on this variant as there is no substantial evidence
supporting its pathogenicity or functional role in pancreatic
cancer. Future investigations into its relevance to tumor biology
and its association with targeted therapies could enhance its
clinical significance. Translational research and precision
medicine will be crucial to determine its potential as a future
biomarker. Notably, no mutations were identified in the four most
frequently described genes involved in PDAC tumorigenesis, which
may indicate the involvement of an alternative, non-canonical
molecular pathway in this context.
In conclusion, the present case report of an early
onset PDAC with atypical mutations and the absence of the four
central driver genes highlight an uncommon molecular profile. The
lack of mutations in KRAS, TP53, CDKN2A and
SMAD4, together with the presence of mutations in
ERBB2 S413L, MSH3 and MUC1/MUC16,
suggests a non-canonical oncogenic pathway driving tumor
progression independently of the currently known classical PDAC
mechanisms. In the present case, the exceptionally low levels of
CA19-9, despite metastatic disease, highlights known limitations of
this biomarker and suggests that alternative or complementary
biomarkers may be needed in selecting patient subsets. Furthermore,
the presence of multiple VUS, the lack of reported cases with the
same mutations in other pancreatic cancer reports, poor response to
FOLFIRINOX and the rapid clinical progression highlight the urgency
of exploring targeted therapies and reinforce the value of
precision medicine in advanced PDAC.
Supplementary Material
Laboratory test results across the
clinical course.
Acknowledgements
Not applicable.
Funding
Funding: The present study was funded by Fondo Nacional de
Desarrollo Científico y Tecnológico (FONDECYT; grant no.
1221499).
Availability of data and materials
The data generated in the present study may be found
in the NCBI Sequence Read Archive under SRA run accession no.
SRR36377907 or at the following URL: https://www.ncbi.nlm.nih.gov/sra/SRX31406456.
Authors' contributions
MEA, CSMA, FGV, IS, INR and BGB were involved in the
conceptualization of the study. TDMG, MMM, AG, CS, FP and PA
contributed to the study methodology, including clinical data
collection, molecular analysis, and data interpretation. ACS, MGV,
FSC, IC, HB, MAS, JME, PAM and JAR, performed the investigations
and data acquisition. JAG and MG were involved in the study
conceptualization, wrote the original draft, and reviewed and
edited the manuscript to produce the final version. JAG and MG
confirm the authenticity of all the raw data. All authors have read
and approved the final manuscript.
Ethics approval and consent to
participate
This case report was performed following the ethical
standards of the ‘Declaration of Helsinki’ (1964) and its later
amendments.
Patient consent for publication
The patient provided written informed consent for
the publication of this case report and the accompanying diagnostic
images.
Competing interests
MG has been involved as a principal investigator in
clinical trials from Merck Sharp & Dohme, Bristol Myers Squibb,
Novartis, Roche, Astellas, Deciphera, Thermo Fisher Scientific, IMS
Health and Quintiles (IQVIA), Bayer, Principia, Covance,
Daiichi-Sankyo, Basilea, PRA-Exelisis, Syneos and Zimeworks. All
other authors declare that they have no competing interests.
References
|
1
|
International Agency for Research on
Cancer. Pancreas Fact Sheet. International Agency for Research on
Cancer. Lyon. Available from: https://gco.iarc.who.int/media/globocan/factsheets/cancers/13-pancreas-fact-sheet.pdf.
Accessed February 20, 2025.
|
|
2
|
International Agency for Research on
Cancer. Cancer Tomorrow. Global Cancer Observatory. Available from:
https://gco.iarc.who.int/tomorrow/en/dataviz/isotype?cancers=13&single_unit=50000&years=2050.
Accessed February 20, 2025.
|
|
3
|
Chilean Society of Gastroenterology. World
Pancreatic Cancer Day-Significant increase in pancreatic cancer in
the coming years: Is there a possibility of reversing this
situation? SCHGE, Santiago, 2024. Available from: https://sociedadgastro.cl/gastroweb/index.php/prensa/prensa-schge/572-dia-mundial-contra-el-cancer-de-pancreas-importante-incremento-en-el-cancer-de-pancreas-en-los-proximos-anos-existe-posibilidad-de-revertir-esta-situacion.
Accessed May 6, 2025.
|
|
4
|
Department of Health Statistics and
Information. Santiago: Ministry of Health of Chile (MINSAL).
Available from: https://informesdeis.minsal.cl/SASVisualAnalytics/?reportUri=%2Freports%2Freports%2Fbcf6e81f-d7f9-4f69-8703-9a83c3eb5da9§ionIndex=0&sso_guest=true&reportViewOnly=true&reportContextBar=false&sas-welcome=false.
Accessed October 10, 2023.
|
|
5
|
International Agency for Research on
Cancer. Chile fact sheet. Global Cancer Observatory. Available
from: https://gco.iarc.who.int/media/globocan/factsheets/populations/152-chile-fact-sheet.pdf.
Accessed February 20, 2025.
|
|
6
|
Puckett Y and Garfield K: Pancreatic
Cancer. In: StatPearls. StatPearls Publishing, Treasure Island, FL,
2025.
|
|
7
|
He TC, Li JA, Xu ZH, Chen QD, Yin HL, Pu
N, Wang WQ and Liu L: Biological and clinical implications of
early-onset cancers: A unique subtype. Crit Rev Oncol Hematol.
190(104120)2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hu ZI and O'Reilly EM: Therapeutic
developments in pancreatic cancer. Nat Rev Gastroenterol Hepatol.
21:7–24. 2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Goldman KC (ed): Tratado de Medicina
Interna. 27th edition. Elsevier, Amsterdam, pp1354-1357, 2024.
|
|
10
|
Luo J: KRAS mutation in pancreatic cancer.
Semin Oncol. 48:10–18. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Park W, Chawla A and O'Reilly EM:
Pancreatic cancer: A review. JAMA. 326:851–862. 2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Porta M, Fabregat X, Malats N, Guarner L,
Carrato A, de Miguel A, Ruiz L, Jariod M, Costafreda S, Coll S, et
al: Exocrine pancreatic cancer: Symptoms at presentation and their
relation to tumour site and stage. Clin Transl Oncol. 7:189–197.
2005.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kleeff J, Korc M, Apte M, La Vecchia C,
Johnson CD, Biankin AV, Neale RE, Tempero M, Tuveson DA, Hruban RH
and Neoptolemos JP: Pancreatic cancer. Nat Rev Dis Primers.
2(16022)2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kamisawa T, Wood LD, Itoi T and Takaori K:
Pancreatic cancer. Lancet. 388:73–85. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ye C, Sadula A, Ren S, Guo X, Yuan M, Yuan
C and Xiu D: The prognostic value of CA19-9 response after
neoadjuvant therapy in patients with pancreatic cancer: A
systematic review and pooled analysis. Cancer Chemother Pharmacol.
86:731–740. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Parra-Robert M, Santos VM, Canis SM, Pla
XF, Fradera JMA and Porto RM: Relationship between CA 19.9 and the
lewis phenotype: Options to improve diagnostic efficiency.
Anticancer Res. 38:5883–5888. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ozawa H, Takahashi K, Motegi T, Jacobi J,
Bhattarchya A, Shigeta K, Takamori S, Onishi M, Luan Z, Fukuda K,
et al: Targeting KRAS inhibitor-resistant pancreatic cancer with an
MUC1-C antibody-drug conjugate. Clin Cancer Res. 31:5246–5260.
2025.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hull A, Li Y, Bartholomeusz D, Hsieh W,
Tieu W, Pukala TL, Staudacher AH and Bezak E: Preliminary
development and testing of C595 radioimmunoconjugates for targeting
MUC1 cancer epitopes in pancreatic ductal adenocarcinoma. Cells.
11(2983)2022.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Basturk O, Hong SM, Wood LD, Adsay NV,
Albores-Saavedra J, Biankin AV, Brosens LA, Fukushima N, Goggins M,
Hruban RH, et al: A revised classification system and
recommendations from the Baltimore consensus meeting for neoplastic
precursor lesions in the pancreas. Am J Surg Pathol. 39:1730–1741.
2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Raina D, Uchida Y, Kharbanda A, Rajabi H,
Panchamoorthy G, Jin C, Kharbanda S, Scaltriti M, Baselga J and
Kufe D: Targeting the MUC1-C oncoprotein downregulates HER2
activation and abrogates trastuzumab resistance in breast cancer
cells. Oncogene. 33:3422–3431. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yan M, Schwaederle M, Arguello D, Millis
SZ, Gatalica Z and Kurzrock R: HER2 expression status in diverse
cancers: Review of results from 37,992 patients. Cancer Metastasis
Rev. 34:157–164. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chou A, Waddell N, Cowley MJ, Gill AJ,
Chang DK, Patch AM, Nones K, Wu J, Pinese M, Johns AL, et al:
Clinical and molecular characterization of HER2
amplified-pancreatic cancer. Genome Med. 5(78)2013.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Chmielecki J, Ross JS, Wang K, Frampton
GM, Palmer GA, Ali SM, Palma N, Morosini D, Miller VA, Yelensky R,
et al: Oncogenic alterations in ERBB2/HER2 represent potential
therapeutic targets across tumors from diverse anatomic sites of
origin. Oncologist. 20:7–12. 2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Richards S, Aziz N, Bale S, Bick D, Das S,
Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, et al:
Standards and guidelines for the interpretation of sequence
variants: A joint consensus recommendation of the American college
of medical genetics and genomics and the association for molecular
pathology. Genet Med. 17:405–424. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Park YH, Shin HT, Jung HH, Choi YL, Ahn T,
Park K, Lee A, Do IG, Kim JY, Ahn JS, et al: Role of HER2 mutations
in refractory metastatic breast cancers: Targeted sequencing
results in patients with refractory breast cancer. Oncotarget.
6:32027–32038. 2015.PubMed/NCBI View Article : Google Scholar
|