Introduction
Lung cancer is a prevalent malignant tumor, with an
incidence rate of 50.4/100,000 people in China in 2022(1). Non-small cell lung cancer (NSCLC)
accounts for ~85% of all lung cancer cases and has a 5-year
survival rate of 18% (1). The
treatment options for advanced NSCLC are limited, and targeted
drugs are typically the first-line choice (2). In recent years, immune checkpoint
inhibitors (ICIs) have facilitated a new era in tumor immunotherapy
via blockade therapy (3). Clinical
trials have demonstrated that the anti-programmed death 1 (PD-1)
monoclonal antibody exhibits significant therapeutic efficacy in
the treatment of numerous types of solid tumor, including NSCLC
(4-6).
However, certain patients receiving ICI treatment may experience
significant side effects, known as immune-related adverse events
(irAEs), which can damage different tissues or organs (7,8). In
2022, a multi-center study from China reported that among the
various side effects, ICI associated-myocarditis is a rare yet
lethal irAE, with an incidence of 1.14% and a mortality rate of
40-50% (9).
The symptoms of ICI associated-myocarditis are
diverse, with the most common clinical manifestations including
dyspnea, palpitations, chest pain and lower limb edema (10,11).
Mild cases typically present with non-specific symptoms, such as
general fatigue, nausea and discomfort, along with elevated cardiac
biomarkers but without any specific symptoms (12). By contrast, severe cases may exhibit
electrophysiological instability, fulminant myocarditis,
hemodynamic instability, cardiogenic shock and death (13). Compared with other irAEs,
ICI-associated myocarditis has a notably shorter onset time, with
symptoms typically emerging ~34 days after the first dose of
therapy (14,15). The typical pathological
characteristics of ICI-associated myocarditis are focal or diffuse
T cell infiltration, with CD8+ T cell infiltration
predominating in the majority of cases and CD4+ T cells
and macrophages being also prevalent (16). CD8+ T cells induce
myocardial injury via direct cytotoxic mechanisms, whereas
CD4+ T cells exacerbate the condition by modulating the
inflammatory microenvironment and activating CD8+ T
cells (17). The dynamic balance
and interaction between these cell types are crucial to the onset
and progression of ICI-associated myocarditis. Anti-PD-1 therapy
can interact with PD-L1, promoting the proliferation of
CD8+ T cells and enhancing their ability to kill cancer
cells (18). However, anti-PD-1 not
only blocks the negative regulatory signals of T cells to alleviate
immune suppression and enhance their anti-tumor effects, but also
intensifies normal immune responses, causing an imbalance in immune
tolerance and giving rise to autoimmune-like inflammatory reactions
in normal tissue (19). Therefore,
investigating the role of CD8+ T cells in the
development of anti-PD-1-induced myocarditis during NSCLC treatment
is key for improving the clinical management of ICI-associated
myocarditis.
The present study aimed to elucidate the role of
CD8+ T cells in anti-PD-1 antibody-induced myocarditis
in C57BL/6 mice with Lewis lung carcinoma (LLC).
Materials and methods
Cells, chemicals and antibodies
Mouse LLC cells (cat. no. CL-0140) were obtained
from Procell Life Science and Technology. Matrigel Matrix was
purchased from Corning, Inc. Glutamine, FBS, DMEM, streptomycin and
penicillin were obtained from Gibco (Thermo Fisher Scientific,
Inc.). DNase I, hyaluronidase and collagenase type V were obtained
from Sigma-Aldrich (Merck KGaA). Masson's trichrome (cat. no.
C0189S) and hematoxylin & eosin (HE) staining kit (cat. no.
C0105S) were purchased from Beyotime Institute of Biotechnology.
The total RNA Extraction kit (cat. no. LS1040) was acquired from
Promega Corporation. First Strand (cat. no. 330421) and QuantiNova
SYBR® Green PCR kits (cat. no. 208057) was obtained from
Qiagen GmbH. The FITC anti-mouse CD4 antibody (cat. no. 100405),
APC anti-mouse CD8a antibody (cat. no. 100711), Pacific Blue™
anti-mouse CD3 antibody (cat. no. 100213) and FITC anti-mouse CD68
antibody (cat. no. 137005; all 1:100) were obtained from BioLegend,
Inc. Anti-PD-1 antibody was purchased from BD Biosciences.
Orthotopic transplantation model with
LLC cells
LLC cells were cultured in DMEM supplemented with
glutamine (2 mM), streptomycin/penicillin (1%) and FBS (10%) with
5% CO2 at 37˚C. A total of 24 wild-type C57BL/6 male
mice (age, 6-8 weeks; weight, 18-22 g) were purchased from Vital
River Laboratories. A total of 18 CD8 knockout (CD8-/-)
C57BL/6 male mice (age, 6-8 weeks; weight, 18-22 g) were obtained
from Jackson Laboratories. The genotype of the wild-type and
CD8-/- mice was confirmed through PCR and agarose gel
electrophoresis (Fig. S1; Data S1).
All mice were housed in specific pathogen-free
environments with a temperature of 22±2˚C and a humidity of 50-60%
and a 12 h light-dark cycle. The mice had unrestricted access to
food and water. Following acclimatization, wild-type C57BL/6 mice
were randomly divided into four groups (n=6/group) as follows:
Control (sham-operated group), wild-type + anti-PD-1, wild-type LLC
and wild-type LLC + anti-PD-1. CD8-/- mice were randomly
separated into CD8-/- control, CD8-/- LLC and
CD8-/- LLC + anti-PD-1 groups. The orthotopic
transplantation model with LLC cells was established as previously
described (20). Following
anesthesia (50 mg/kg pentobarbital sodium), the mouse was
positioned on the operating table and the thoracic cavity was
exposed until the lung lobes were visible. Subsequently, LLC cell
suspension (1x105 cells) containing 50 µl Matrigel
Matrix was injected orthotopically into the left lobe of the lung.
After removing the needle, the incision was sutured. A total of 10
days after surgery, mice in anti-PD-1 groups were administered
anti-PD-1 antibody (200 µg) via intraperitoneal injection. The
control mice underwent the same surgery and were administered 50 µl
Matrigel Matrix or PBS vehicle on the same schedule. Animal health
and behavior were monitored each day. A decrease in normal body
weight >20% was defined as a humane endpoint; none of the mice
reached the humane endpoint. After 3 weeks, all mice were humanely
euthanized via intraperitoneal injection of 200 mg/kg sodium
pentobarbital. Tumor, left lung and heart tissue were collected.
The investigators were blinded to group identity. The present study
was performed in accordance with the National Institutes of Health
Guide for the Care and Use of Laboratory Animals (8th edition)
(21). The experimental protocols
were approved by the Experimental Animal Ethics Committee of Jinhua
Food and Drug Inspection and Testing Research Institute (Jinhua,
China; approval no. AL-JSYJ202023).
Histology
The tumor, left lung and left ventricle tissue were
fixed (4% paraformaldehyde at 25˚C for 12 h), dehydrated and
embedded in paraffin. Tissue samples were precisely sliced into
sections measuring 4 µm in thickness. Following dewaxing and
rehydration, sections were subjected to HE (hematoxylin for 5 min
and eosin for 2 min at room temperature) and Masson's trichrome
staining (Wiegert's iron hematoxylin for 8 min, Biebrich scarlet
for 5 min and aniline blue for 5 min at room temperature).
High-resolution images were captured using an OLYMPUS BX53 light
microscope (n=9 fields of view/mouse) and analyzed using ImageJ
version 1.54 (National Institutes of Health).
Flow cytometry
Heart tissue was dissected into small fragments (2
mm3) and subjected to enzymatic digestion in DMEM
enriched with DNase I (0.015 mg/ml), hyaluronidase (0.2 mg/ml) and
collagenase type V (0.5 mg/ml) for a duration of 2 h at 37˚C. Cell
suspensions were prepared by passing the tissues through a 70-µm
cell strainer. The single-cell suspensions were stained with
Pacific Blue™ anti-mouse CD3, FITC anti-mouse CD4, APC
anti-mouse CD8a and FITC anti-mouse CD68 antibody. Following 30 min
incubation in the dark at 4˚C, the cells were washed three times
with PBS, resuspended in PBS containing 1% FBS and analyzed using a
FACScan flow cytometer (BD Biosciences). Flow cytometric data were
analyzed using FlowJo software (version 10.8.1; BD
Biosciences).
Reverse transcription-quantitative
PCR
A total RNA extraction kit was used to extract the
total RNA from left ventricle tissue. According to the
manufacturer's protocol, RNA was used to produce cDNA using the
First Strand Kit, followed by PCR analysis using a QuantiNova
SYBR® Green PCR kit. The thermocycling conditions were
as follows: 95˚C for 5 min, followed by 40 cycles of denaturation
at 95˚C for 15 sec and annealing and extension at 60˚C for 40 sec.
Gene expression was assessed using the 2-ΔΔCq method
(22), with GAPDH serving as a
normalization control. Primer sequences are listed in Table I.
 | Table IPrimer sequences. |
Table I
Primer sequences.
| Gene | Sequence,
5'→3' |
|---|
| MCP-1 | Forward:
TGCCCTAAGGTCTTCAGCAC |
| | Reverse:
AAGGCATCACAGTCCGAGTC |
| IL-6 | Forward:
GACAAAGCCAGAGTCCTTCAGA |
| | Reverse:
TGTGACTCCAGCTTATCTCTTGG |
| IFN-γ | Forward:
CGGCACAGTCATTGAAAGCC |
| | Reverse:
TGCATCCTTTTTCGCCTTGC |
| TNF-α | Forward:
TGCCACAAGCAGGAATGAGA |
| | Reverse:
GACGTGGAAGTGGCAGAAGAG |
| GAPDH | Forward:
CTGCACCACCAACTGCTTAG |
| | Reverse:
GTCTGGGATGGAAATTGTGA |
Statistical analysis
Each experiment was performed in triplicate. All
data were analyzed with SPSS 23.0 statistical software (IBM Corp.)
and presented as the mean ± standard deviation. One-way ANOVA
followed by Tukey's post hoc test was used for data comparison.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Anti-PD-1 therapy improves the
histopathology of lung and tumor tissue but induces myocarditis in
wild-type mice
HE staining was utilized to investigate the
histopathological characteristics of tumor, lung and heart tissue
(Fig. 1A and B). Tumor cells exhibited an increase in
size, accompanied by a minor degree of hemorrhage and were
organized in a sheet-like arrangement. Some cell nuclei showed an
enlargement in volume, with an increased presence of mitotic
figures. Inflammatory infiltration was evident within the
interstitial space. Anti-PD-1 therapy inhibited tumor growth, as
evidenced by a loose distribution of tumor cells and decreased
numbers of mitotic figures. Hemorrhage was observed. The cellular
distribution appeared uneven, with a small number of lymphocytes
visible in the interstitial space. Infiltration of inflammatory
cells was not prominent and vacuolar changes were evident in the
cytoplasm. Compared with the control, LLC mice exhibited pronounced
lung tissue damage characterized by disorganized and collapsed
alveolar structures, extensive infiltration of inflammatory cells
and interstitial congestion (the black arrow; Fig. 1C). These histopathological changes
were mitigated by anti-PD-1 therapy (Fig. 1C). Morphology and structure of the
myocardial tissue in the control group were normal (Fig. 1D). The myocardial fibers exhibited a
neat arrangement, with clearly defined nuclei and no infiltration
of inflammatory cells. Similar histopathology was observed in LLC
mice. However, the intercellular spaces in the LLC + anti-PD-1
group were enlarged (the black arrow). Additionally, there was
infiltration of inflammatory cells (the red arrow). These results
indicated notable impairment of myocardial tissue following
treatment with anti-PD-1. In Masson's trichrome staining, red
represents myocardial cells, while blue represents collagen fiber.
Collagen fibers accounted for a relatively small proportion of
healthy myocardial tissue. The proportion of collagen fibers in
control and LLC mice groups was relatively small, however, LLC +
anti-PD-1 group exhibited a significant increase in blue-stained
area, characterized by extensive fibrotic tissue replacing normal
myocardial tissue (Fig. 1E).
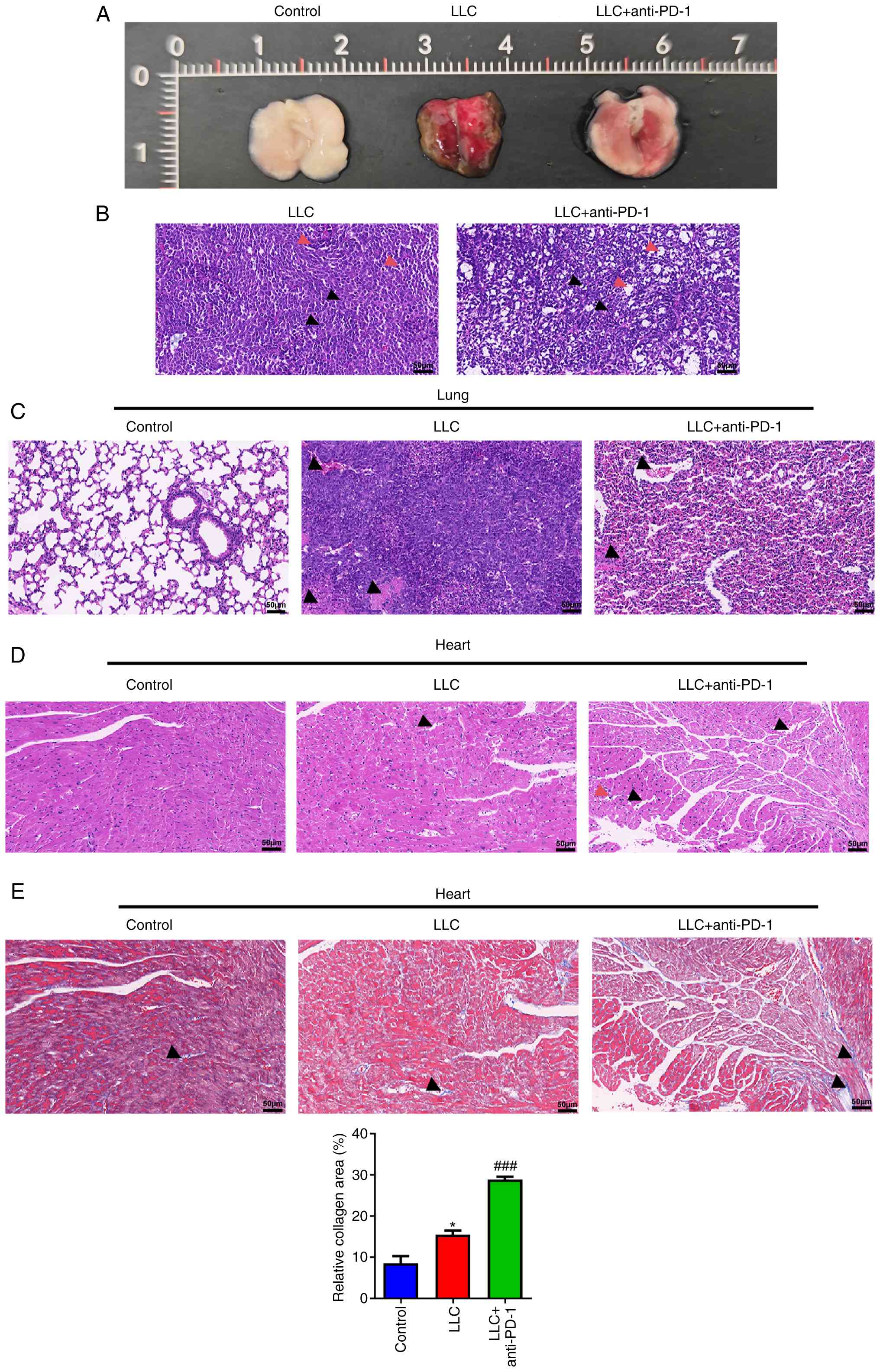 | Figure 1Anti-PD-1 therapy improves the
histopathology of lung and tumor tissue but induces myocarditis in
wild-type mice. (A) Representative photographs of the lungs
containing the tumors. HE staining was performed to examine the
histopathology of (B) tumor (black arrow, nucleus; red arrow,
inflammatory cell infiltration), (C) lung (arrow, interstitial
congestion) and (D) heart tissue (black arrow, intercellular space;
red arrow, inflammatory cell infiltration). (E) Masson's trichrome
staining was performed to examine the histopathology of heart
tissue. Arrow, collagen fiber. Scale bar, 50 µm.
*P<0.05 vs. control, ###P<0.001 vs.
LLC. HE, hematoxylin-eosin; LLC, Lewis lung carcinoma. |
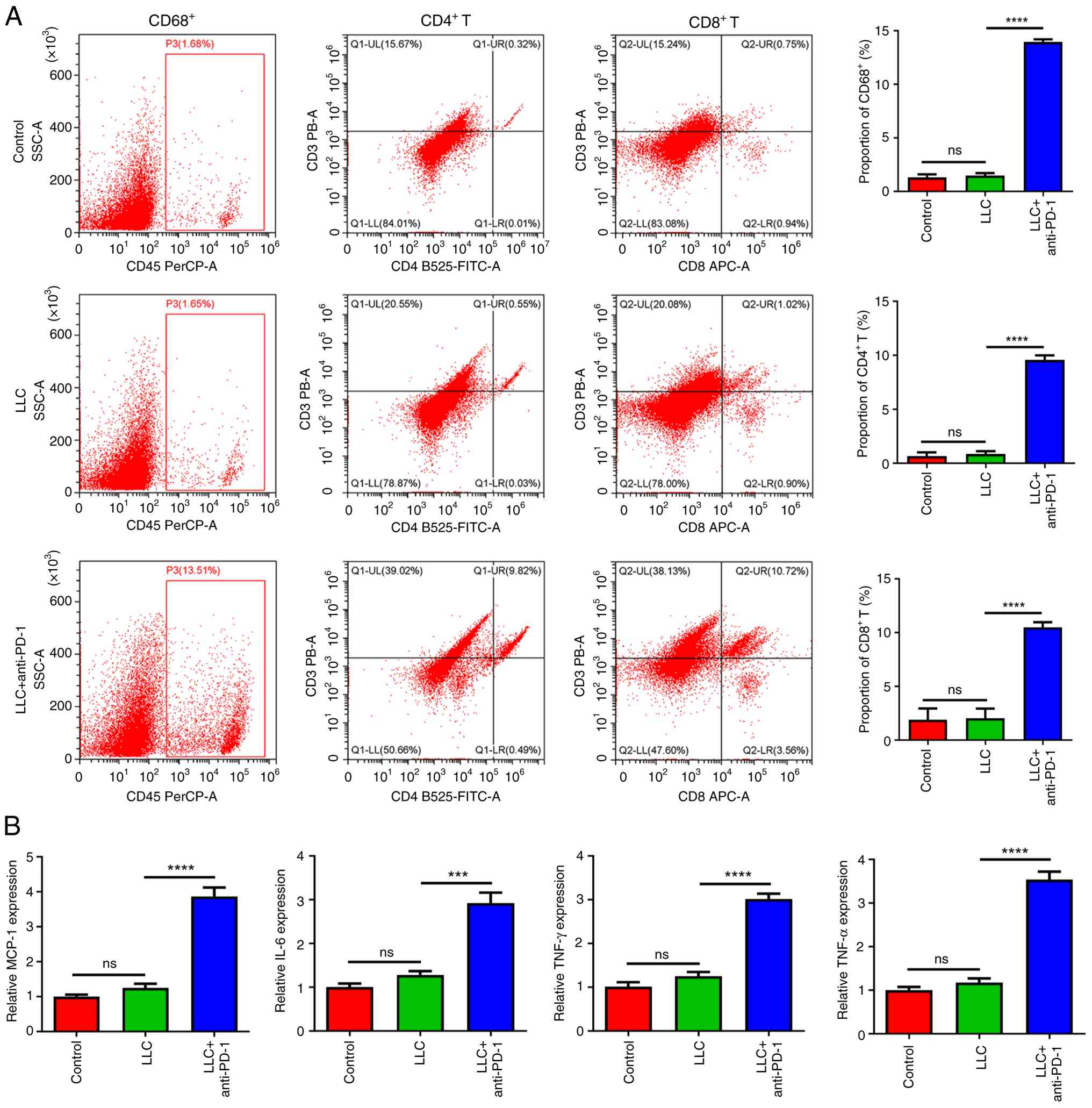
Anti-PD-1 therapy activates
CD8+ and CD4+ T cells and macrophages and
induces secretion of inflammatory cytokines in wild-type mice
Flow cytometry analysis indicated no significant
differences in the ratios of CD8+, CD4+ and
CD68+ (macrophage marker) cells between the control and
the LLC group (Fig. 2A). By
contrast, anti-PD-1 therapy notably increased the percentages of
CD8+ and CD4+ T cells and macrophages in
myocardial tissue of LLC mice (Fig.
2A). Furthermore, the expression of inflammation-associated
cytokines, including chemokine monocyte chemotactic protein-1
(MCP-1), IL-6, IFN-γ and tumor necrosis factor (TNF)-αwere
significantly increased by anti-PD-1 (Fig. 2B).
Anti-PD-1 therapy improves the
histopathology of tumor and lung tissue but does not induce
significant myocardial injury in CD8-/-
mice
After knocking out CD8 gene, tumor cells in LLC mice
were arranged in a dense, sheet-like pattern, exhibiting notable
proliferation and an increase in mitotic figures (Fig. 3A and B). The volume of the cell nucleus was
enlarged, with meganuclei and multinucleated tumor cells present.
Lymphocytes were found within the interstitial spaces. Following
anti-PD-1 treatment, tumor cells exhibited a decrease in size and
were arranged in loose pattern. There was also a notable decrease
in mitotic figures along with moderate infiltration of inflammatory
cells within the interstitium. No obvious pathological changes were
observed in lung tissue of mice in the control or anti-PD-1 groups
(Fig. 3C). Notably, the lung tissue
of CD8-/- LLC mice was severely damaged, with notable
inflammatory infiltration along with visible congestion and
thickening of the lung interstitium. However, anti-PD-1 therapy
alleviated pulmonary interstitial congestion and thickening. All
groups exhibited normal morphology and structure of the myocardial
tissue, with a well-organized arrangement of myocardial fibers,
distinct cell nuclei and an absence of inflammatory cell
infiltration (Fig. 3D). Notably,
the intercellular space in CD8-/- LLC + anti-PD-1 group
was observed to be slightly increased compared with the other
groups (Fig. 3D). Masson's
trichrome staining showed that a small amount of collagen fibers
was observed in control, anti-PD-1 and LLC groups (Fig. 3E). By contrast, the
CD8-/- LLC + anti-PD-1 group exhibited a significant
increase in collagen fibers (Fig.
3E).
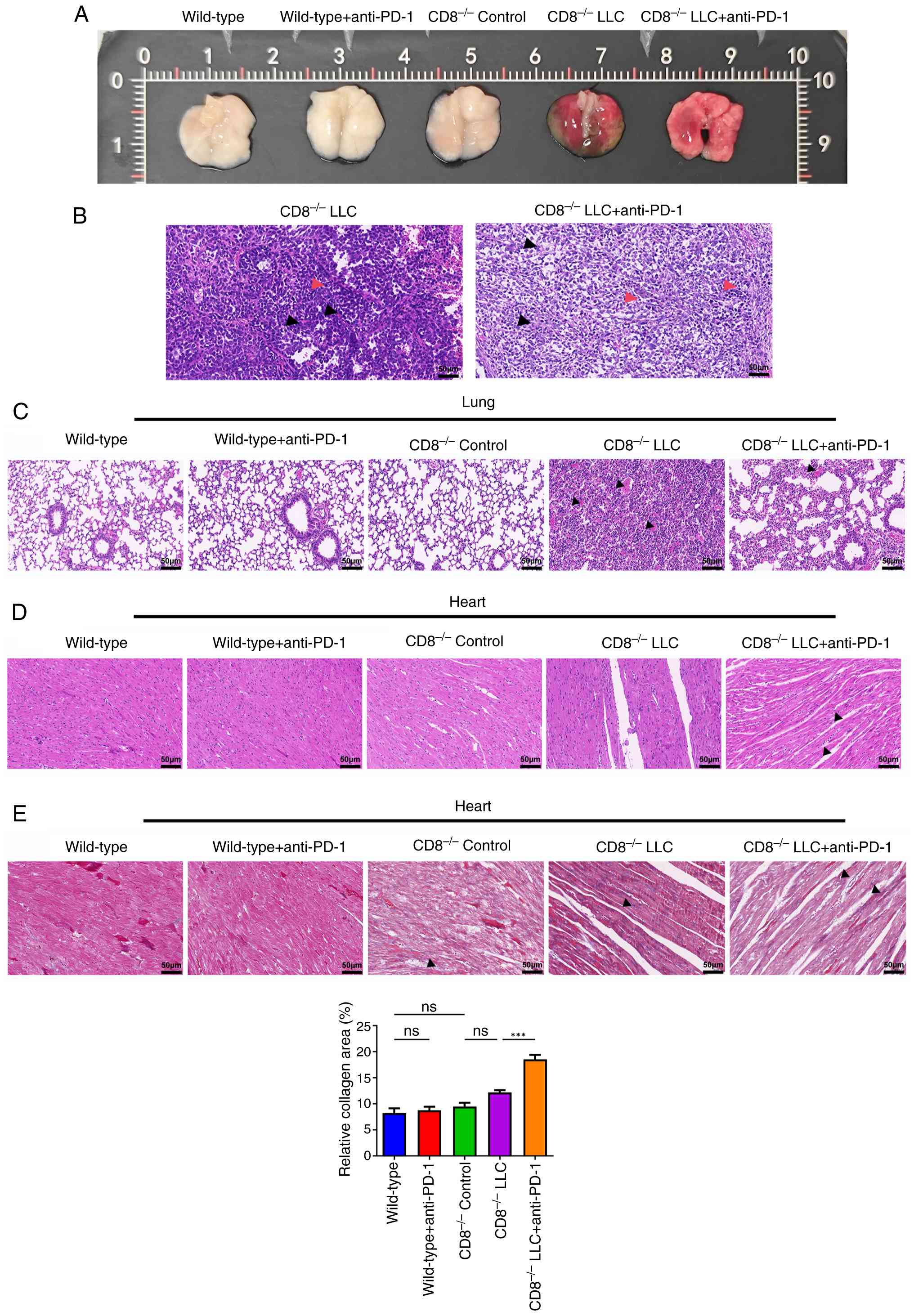 | Figure 3Effects of anti-PD-1 therapy on the
histopathology of tumor, lung and heart tissue in CD8-/-
mice. (A) Representative photographs of the lungs containing the
tumors. HE staining was performed to examine the histopathology of
(B) tumor (black arrow, nucleus; red arrow, inflammatory cell
infiltration), (C) lung (arrow, interstitial congestion) and (D)
heart tissue (black arrow, intercellular space; red arrow,
inflammatory cell infiltration). (E) Masson's trichrome staining
was performed to examine the histopathology of heart tissue. Arrow,
collagen fiber. Scale bar, 50 µm. ***P<0.001. HE,
hematoxylin-eosin; LLC, Lewis lung carcinoma; ns, not
significant. |
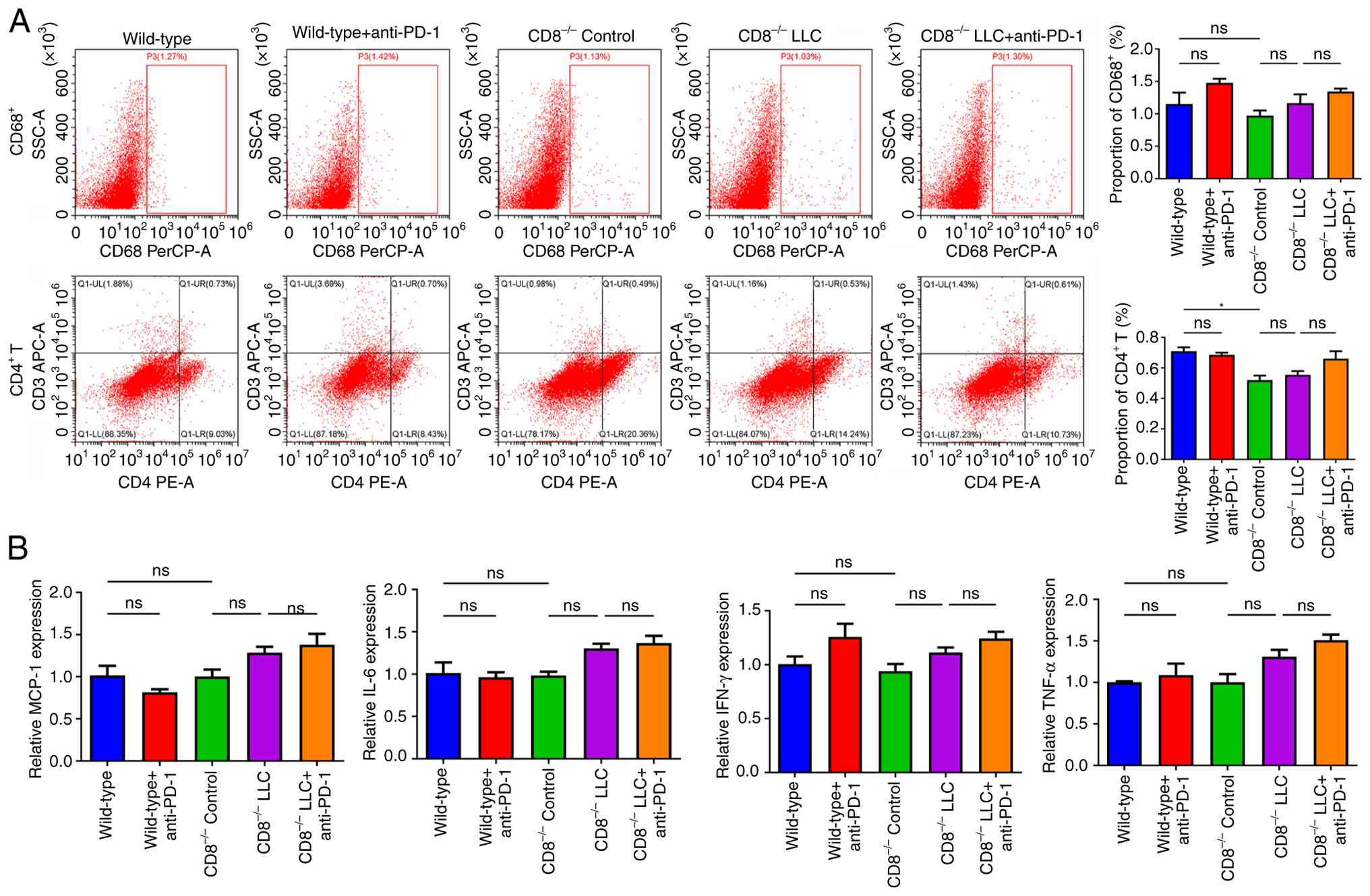
Anti-PD-1 therapy does not
significantly alter CD4+ T cell and macrophage levels or
inflammatory cytokine secretion in CD8-/-
mice
Ratios of CD4+ T cells and macrophages in
myocardial tissue were assessed. In CD8-/- mice, the
ratio of CD4+ T cells was significantly decreased
compared with that of the wild-type mice (Fig. 4A). There were no significant
differences between the other groups. Furthermore, expression of
MCP-1, IL-6, IFN-γ and TNF-α was not significantly different
between any groups (Fig. 4B).
Discussion
Initially considered to be a rare irAE with an
incidence rate ranging from 0.09 to 1.14%, recent studies have
indicated that ICI-associated myocarditis has a high fatality rate
(40-50%) (15,23). Al-Kindi and Oliveira (24) reported 250 cases of myocarditis
associated with ICI. The aforementioned findings indicated
increased reporting over time, with 18 cases documented between
2012 and 2015, followed by 70 cases in 2016 and 162 cases in
2017(24). Therefore, while
clinically significant myocarditis is uncommon, it should not be
overlooked due to the rising number of patients with cancer
undergoing ICI therapy. The present study established an orthotopic
transplantation model with LLC cells. ICI associated-myocarditis
required the presence of CD8+ T cells to activate
CD4+ T cells and macrophages and induce inflammatory
responses.
LLC cell line was isolated from the epidermoid
carcinoma of the lung in mice. It is an essential tumor model in
studies of therapeutic effects (25), vessel formation (26) and metastasis (27). LLC-bearing mice have been employed
in experimental frameworks investigating NSCLC (28,29).
The histopathological analysis revealed an increase in the presence
of mitotic figures, along with pronounced infiltration of
inflammatory cells within the tumor tissue. Concurrently, the
structural integrity of lung tissue was compromised. These findings
suggested that the orthotopic transplantation model was
successfully established. Additionally, consistent with previous
studies (5,30), anti-PD-1 therapy decreased nuclear
division and infiltration of inflammatory cells and induced
necrosis of tumor cells. Clinical observational studies have
demonstrated that in patients with ICI associated-myocarditis,
there is significant infiltration of inflammatory cells within the
myocardial tissue and focal fibrosis is observed in some cases
(31,32). In the present study, the
histopathological examination of myocardial tissue revealed that
the use of anti-PD-1 resulted in fibrosis and infiltration of
inflammatory cells. In wild-type mice, levels of cytokines
associated with inflammation, such as MCP-1, IL-6, IFN-γ, and
TNF-α, were significantly elevated following anti-PD-1 treatment.
These cytokines are associated with immune-mediated heart damage.
Overactivated T cells and macrophages release notable amounts of
MCP-1, thereby establishing a positive feedback loop of
inflammation-chemotaxis that exacerbates myocardial injury
(33). IL-6 is a crucial
pro-inflammatory cytokine that can be secreted by T cells,
macrophages and cardiomyocytes. It plays an integral role in the
process of myocardial fibrosis (34). IFN-γ enhances the antigen
presentation capacity of macrophages, promotes a T helper 1-type
immune response and induces the expression of major
histocompatibility complex (MHC)-I molecules in cardiomyocytes,
rendering them susceptible to attack by CD8+ T cells
(12). TNF-α, secreted by T cells,
macrophages and cardiomyocytes, can directly induce myocardial cell
apoptosis and inhibit myocardial contractility (34). Collectively, the present findings
indicated that such treatment may damage the myocardium.
The heart is an organ with immune privilege, making
immune reactions within it particularly hazardous, as they can
result in life-threatening arrhythmia and severe heart failure
(15). Its lack of regenerative
ability, combined with a heightened risk of arrhythmia from minor
injuries and rich blood supply, renders the heart susceptible to
damage from the immune system (15). Histopathological examinations of
samples from patients with ICI associated-myocarditis show a
heightened presence of macrophages and T lymphocytes (including
CD8+ and CD4+ types) within the myocardial
tissue (12,35). Additionally, some reports have
demonstrated that lung biopsy samples from patients with NSCLC who
underwent anti-PD-1 therapy exhibit high infiltration of
CD8+ and CD4+ T cells (36,37).
These findings align with the abnormally elevated levels of
CD4+ and CD8+ T cells and macrophages
observed in patients with ICI-induced myocarditis (16,17).
Modulation of immune checkpoints is a key mechanism for evading
immune surveillance by inhibiting activated T cells (38). Antibodies that suppress PD-1 improve
and restore the functionality of exhausted T cells and enhance
their anti-tumor immune response (39). Therefore, it was hypothesized that
the administration of anti-PD-1 therapy in lung cancer inhibits the
PD-1 signaling pathway, thereby alleviating T cell suppression and
enhancing the immune system capacity to target tumor cells. This
process leads to the activation and proliferation of immune cells
throughout the body, including CD8+ T cells.
Consequently, these activated immune cells may circulate
systemically and infiltrate myocardial tissue, resulting in
immune-mediated damage to cardiac structures. The present results
corroborated previous research findings, demonstrating notably
elevated ratios of CD4+ and CD8+ T cells and
macrophages in the myocardial tissues of mice treated with
anti-PD-1. By contrast, the number of these cells did not exhibit
significant increases in the absence of anti-PD-1 treatment. In
ICI-induced myocarditis, CD4+ T cells play promote
pathogenesis by regulating the inflammatory microenvironment and
co-activating CD8+ T cells (17). The present findings were consistent
with the aforementioned study highlighting that CD4+ T
cells assist CD8+ T cells in mediating the
immunopathological process of this condition. As key
antigen-presenting cells (APCs), macrophages (marked by CD68)
contribute to myocarditis by secreting pro-inflammatory cytokines
(40,41). The present study demonstrated that
anti-PD-1 therapy elevated macrophage and cytokine levels in LLC
mice. These results suggested that the cytokines established an
inflammation-chemotaxis feedback loop to amplify myocardial damage
and recruit CD8+ T cells. Jiménez-Alejandre et al
(16) noted dendritic cells (as
potent APCs) present myocardial or cross-reactive tumor antigens
via MHC molecules to prime CD4+ T cells and enhance
CD8+ T cell activation. In the present study, anti-PD-1
did not alter CD4+ T cell/macrophage infiltration or
cytokine levels in CD8-/- LLC mice compared with the
controls, indicating CD4+ T cell activation and APC
recruitment require CD8+ T cells to initiate the
pathogenic cascade of ICI-induced myocarditis. Anti-PD-1 therapy
was effective in inhibiting tumor growth and ameliorating lung
tissue damage. In the absence of CD8 gene, no significant
differences were observed in myocardial histopathology or
expression of MCP-1, IL-6, IFN-γ and TNF-α between groups.
CD8-/- LLC + anti-PD-1 mice showed an increase in
collagen fibers compared with CD8-/- LLC mice. These
results indicated the key role of CD8+ T cells in ICI
associated-myocarditis. There were no significant differences in
the ratios of CD4+ T and macrophages between groups,
which may indirectly suggest that the presence of CD8+ T
cells is a prerequisite for the activation of CD4+ T
cells and macrophages as well as subsequent inflammatory cell
infiltration.
The present study confirmed anti-PD-1 can trigger
myocardial inflammatory infiltration and fibrosis, consistent with
the study conducted by Johnson et al (35) that reported focal/diffuse T cell
infiltration and fibrosis in biopsy samples of patients with
myocarditis. CD8+ T cells serve as central pathogenic
mediators, aligning with the previous studies emphasizing
CD8+ T cell cytotoxicity and depletion abolishing
myocarditis (17,42). Furthermore, pro-inflammatory
cytokines (MCP-1, IL-6, IFN-γ, TNF-α) and CD68+
macrophages, key contributors to inflammation progression, were
identified, consistent with findings on cytokine elevation and
macrophage-mediated damage reported by Patel et al (43) and Wang et al (44). Additionally, the present study
demonstrated that CD8+ T cells serve as upstream
initiators of CD4+ T cell and macrophage activation and
identified CD8+ T cells as the common mediator linking
anti-tumor activity and cardiac toxicity, whereas prior clinical
studies have primarily focused on myocarditis incidence and
prognostic markers (45,46).
The present findings suggest potential for
advancements in clinical practice aimed at preventing or treating
myocarditis via regulation of CD8+ T cell activity. For
example, activation of CD8+ T cells depends on
costimulatory molecules, such as CD28/B7 and CD137/CD137L (47). Blocking their interaction may
inhibit the function of CD8+ T cells. Activated
CD8+ T cells exhibit high levels of pro-apoptotic
molecules, such as Fas. Inducing apoptosis of CD8+ T
cells may mitigate myocardial injury. Chimeric antigen
receptor-CD8+ T cells engineered to target tumor
antigens can be designed to minimize the risk of non-specific
myocardial damage. Regulation of the function and localization of
CD8+ T cells may prevent ICI-associated myocarditis and
treat lung cancer. The CCL5-CCR5 axis is activated in myocarditis
progression (48), whereas the
tumor microenvironment predominantly uses the CXCL9/CXCL10-CXCR3
axis to recruit anti-tumor CD8+ T cells (49). Consequently, CCR5 antagonists (such
as maraviroc) can be employed in combination therapy to selectively
inhibit the migration of self-reactive CD8+ T cells to
the heart without compromising the infiltration of CD8+
T cells within tumors. Directly injecting anti-PD-1 antibodies into
lung cancer lesions (pulmonary nodules) significantly enhances
local drug concentration within tumors, achieving levels 10-100
times higher than those attained through systemic administration,
while concurrently reducing peripheral blood drug concentrations
and minimizing the effects of CD8+ T cells on heart
(50).
The present study had limitations. First, the
specific mechanisms of CD8+ T cells on the activation of
CD4+ T cells and macrophages are unknown. Further
investigation into the role of CD4+ T cells and
macrophages in myocarditis is warranted. The sample size was
relatively small, potentially leading to insufficient statistical
power and an increased risk of false-negative or -positive results.
Additionally, the present study did not exclude the potential
effects of CD8-/- on other immune cells (such as natural
killer and regulatory T cells) or molecular pathways other than the
PD-1/PD-L1 pathway. For example, CD8-/- may alter the
immune balance in the tumor microenvironment, however the present
study did not measure associated markers (such as regulatory T
cells and IL-10) to exclude confounding factors. Certain non-T
cell-dependent mechanisms may be activated by anti-PD-1 treatment,
and further investigation of regulatory T cell and PD-L1 expression
levels is required.
In summary, the present study offers an initial
investigation into the molecular mechanisms underlying
CD8+ T cell functions in anti-PD-1 antibody-induced
myocarditis. Myocarditis induced by anti-PD-1 antibody required the
involvement of CD8+ T cells to activate both
CD4+ T cells and macrophages and stimulate inflammatory
responses. These findings suggest inhibition of CD8+ T
cells can attenuate ICI-associated myocarditis, which may
facilitate improved the treatment strategies for this
condition.
Supplementary Material
Genotyping strategy and results of
agarose gel electrophoresis. (A) Genotyping strategy. (B) Agarose
gel electrophoresis. WT, wild-type; KO, knockout; M, marker; F,
forward; R, reverse.
Genotype confirmation of wild-type
(WT) and CD8 knockout (CD8-/-) mice.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by The Science and
Technology Project of Jinhua City in China (grant no.
2022-3-146).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
MD and XW conceived and designed the study. HC, FL,
JS, SW, HX, RL, HZ and GC analyzed and interpreted data. HC wrote
the manuscript. MD and XW confirm the authenticity of all the raw
data. All authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Experiments for animals were conducted in compliance
with the National Institutes of Health Guide for the Care and Use
of Laboratory Animals, and approved by the Experimental Animal
Ethics Committee of Jinhua Food and Drug Inspection and Testing
Research Institute (Jinhua, China; approval no. AL-JSYJ202023).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Roller JF, Veeramachaneni NK and Zhang J:
Exploring the Evolving scope of neoadjuvant immunotherapy in NSCLC.
Cancers (Basel). 14(741)2022.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Li MSC, Mok KKS and Mok TSK: Developments
in targeted therapy & immunotherapy-how non-small cell lung
cancer management will change in the next decade: A narrative
review. Ann Transl Med. 30(358)2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Schoenfeld AJ, Lee SM, Doger de Spéville
B, Gettinger SN, Häfliger S, Sukari A, Papa S, Rodríguez-Moreno JF,
Graf Finckenstein F, Fiaz R, et al: Lifileucel, an autologous
tumor-infiltrating lymphocyte monotherapy, in patients with
advanced non-small cell lung cancer resistant to immune checkpoint
inhibitors. Cancer Discov. 14:1389–1402. 2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Qu J, Mei Q, Liu L, Cheng T, Wang P, Chen
L and Zhou J: The progress and challenge of anti-PD-1/PD-L1
immunotherapy in treating non-small cell lung cancer. Ther Adv Med
Oncol. 13(1758835921992968)2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Yin J, Wu Y, Yang X, Gan L and Xue J:
Checkpoint inhibitor pneumonitis induced by anti-PD-1/PD-L1 therapy
in non-small-cell lung cancer: Occurrence and mechanism. Front
Immunol. 13(830631)2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rittmeyer A, Barlesi F, Waterkamp D, Park
K, Ciardiello F, von Pawel J, Gadgeel SM, Hida T, Kowalski DM, Dols
MC, et al: Atezolizumab versus docetaxel in patients with
previously treated non-small-cell lung cancer (OAK): A phase 3,
open-label, multicentre randomised controlled trial. Lancet.
389:255–265. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhao Z, Wang X, Qu J, Zuo W, Tang Y, Zhu H
and Chen X: Immune-related adverse events associated with outcomes
in patients with NSCLC treated with anti-PD-1 inhibitors: A
systematic review and meta-analysis. Front Oncol.
11(708195)2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhang Q, Wang W, Yuan Q, Li L, Wang YC,
Chi CZ and Xu CH: Correlation between immune-related adverse events
and the efficacy of PD-1/PD-L1 inhibitors in the treatment of
non-small cell lung cancer: Systematic review and meta-analysis.
Cancer Chemother Pharmacol. 89:1–9. 2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Xie X, Wang L, Li Y, Xu Y, Wu J, Lin X,
Lin W, Mai Q, Chen Z, Zhang J, et al: Multi-organ immune-related
adverse event is a risk factor of immune checkpoint
inhibitor-associated myocarditis in cancer patients: A multi-center
study. Front Immunol. 13(879900)2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhou YW, Zhu YJ, Wang MN, Xie Y, Chen CY,
Zhang T, Xia F, Ding ZY and Liu JY: Immune checkpoint
inhibitor-associated cardiotoxicity: Current understanding on its
mechanism, diagnosis and management. Front Pharmacol.
10(1350)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chen X, Jiang A, Zhang R, Fu X, Liu N, Shi
C, Wang J, Zheng X, Tian T, Liang X, et al: Immune checkpoint
inhibitor-associated cardiotoxicity in solid tumors: Real-world
incidence, risk factors, and prognostic analysis. Front Cardiovasc
Med. 9(882167)2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Moslehi J, Lichtman AH, Sharpe AH,
Galluzzi L and Kitsis RN: Immune checkpoint inhibitor-associated
myocarditis: Manifestations and mechanisms. J Clin Invest.
131(e145186)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chen Y, Jia Y, Liu Q, Shen Y, Zhu H, Dong
X, Huang J, Lu J and Yin Q: Myocarditis related to immune
checkpoint inhibitors treatment: Two case reports and literature
review. Ann Palliat Med. 10:8512–8517. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Coustal C, Vanoverschelde J, Quantin X,
Lesage C, Michot JM, Lappara A, Ederhy S, Assenat E, Faure M, Issa
N, et al: Prognosis of immune checkpoint inhibitors-induced
myocarditis: A case series. J Immunother Cancer.
11(e004792)2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wang D, Bauersachs J and Berliner D:
Immune checkpoint inhibitor associated myocarditis and
cardiomyopathy: A translational review. Biology (Basel).
12(472)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Jiménez-Alejandre R, Ruiz-Fernández I and
Martín P: Pathophysiology of immune checkpoint inhibitor-induced
myocarditis. Cancers (Basel). 14(4494)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Gong J, Neilan TG and Zlotoff DA:
Mediators and mechanisms of immune checkpoint inhibitor-associated
myocarditis: Insights from mouse and human. Immunol Rev. 318:70–80.
2023.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Postow MA, Sidlow R and Hellmann MD:
Immune-related adverse events associated with immune checkpoint
blockade. N Engl J Med. 378:158–168. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Liu X, Wu W, Fang L, Liu Y and Chen W:
TNF-α inhibitors and other biologic agents for the treatment of
immune checkpoint inhibitor-induced myocarditis. Front Immunol.
13(922782)2022.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wang J, Zhou T, Sun Z, Ye T, Zhou S, Li J,
Liu Y, Kong L, Tang J, Liu D and Xing HR: Zeb1 regulates the
symmetric division of mouse lewis lung carcinoma stem cells through
numb mediated by miR-31. Int J Biol Sci. 14:1399–1410.
2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
National Research Council (USA): Guide for
the care and use of laboratory animals, 8th edition. National
Academies Press, Washington, DC, pp11-123, 2011.
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Xu L, Chen YK, Xiong L, Shen Y, Zhou Z,
Wang S and Xu X: A review of immune checkpoint inhibitor-associated
myocarditis: Epidemiology, pathogenesis, and biomarkers. Hum Vaccin
Immunother. 21(2512645)2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Al-Kindi SG and Oliveira GH: Reporting of
immune checkpoint inhibitor-associated myocarditis. Lancet.
392:382–383. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sun Z, Shen K, Xie Y, Hu B, He P, Lu Y and
Xue H: Shiquan Yuzhen Decoction inhibits angiogenesis and tumor
apoptosis caused by non-small cell lung cancer and promotes immune
response. Am J Transl Res. 13:7492–7507. 2021.PubMed/NCBI
|
|
26
|
Cui Y, Luo Y, Qian Q, Tian J, Fang Z, Wang
X, Zeng Y, Wu J and Li Y: Sanguinarine regulates tumor-associated
macrophages to prevent lung cancer angiogenesis through the
WNT/β-catenin pathway. Front Oncol. 12(732860)2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Huang F, Cao Y, Wang C, Lan R, Wu B, Xie
X, Hong J, Fu L and Wu G: PNMA5 promotes bone metastasis of
non-small-cell lung cancer as a target of BMP2 signaling. Front
Cell Dev Biol. 9(678931)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Chan YP, Chuang CH, Lee I and Yang NC:
Lycopene in combination with sorafenib additively inhibits tumor
metastasis in mice xenografted with lewis lung carcinoma cells.
Front Nutr. 9(886988)2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Han N, Yang ZY, Xie ZX, Xu HZ, Yu TT, Li
QR, Li LG, Peng XC, Yang XX, Hu J, et al: Dihydroartemisinin
elicits immunogenic death through ferroptosis-triggered ER stress
and DNA damage for lung cancer immunotherapy. Phytomedicine.
112(154682)2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wang P, Fang X, Yin T, Tian H, Yu J and
Teng F: Efficacy and safety of anti-PD-1 plus anlotinib in patients
with advanced non-small-cell lung cancer after previous systemic
treatment failure-A retrospective study. Front Oncol.
11(628124)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Patel RP, Parikh R, Gunturu KS, Tariq RZ,
Dani SS, Ganatra S and Nohria A: Cardiotoxicity of immune
checkpoint inhibitors. Curr Oncol Rep. 23(79)2021.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Nishikawa T, Inoue T, Otsuka T, Kuno I,
Kukita Y, Nakamura H, Ikeda Y, Yasui T, Shioyama W, Oka T, et al:
Prevalence and characteristics of immune checkpoint
inhibitor-related myocardial damage: A prospective observational
study. PLoS One. 17(e0275865)2022.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wu Y, Xu Y and Xu L: Drug therapy for
myocarditis induced by immune checkpoint inhibitors. Front
Pharmacol. 14(1161243)2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Ali A, Caldwell R, Pina G, Beinart N,
Jensen G, Yusuf SW, Koutroumpakis E, Hamzeh I, Khalaf S, Iliescu C,
et al: Elevated IL-6 and tumor necrosis factor-α in immune
checkpoint inhibitor myocarditis. Diseases. 12(88)2024.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Johnson DB, Balko JM, Compton ML, Chalkias
S, Gorham J, Xu Y, Hicks M, Puzanov I, Alexander MR, Bloomer TL, et
al: Fulminant myocarditis with combination immune checkpoint
blockade. N Engl J Med. 375:1749–1755. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Naidoo J, Cottrell TR, Lipson EJ, Forde
PM, Illei PB, Yarmus LB, Voong KR, Feller-Kopman D, Lee H, Riemer
J, et al: Chronic immune checkpoint inhibitor pneumonitis. J
Immunother Cancer. 8(e000840)2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Suresh K, Naidoo J, Zhong Q, Xiong Y,
Mammen J, de Flores MV, Cappelli L, Balaji A, Palmer T, Forde PM,
et al: The alveolar immune cell landscape is dysregulated in
checkpoint inhibitor pneumonitis. J Clin Invest. 129:4305–4315.
2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Yang K, Li J, Sun Z, Zhao L and Bai C:
Retreatment with immune checkpoint inhibitors in solid tumors: A
systematic review. Ther Adv Med Oncol.
12(1758835920975353)2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kim CG, Kim KH, Pyo KH, Xin CF, Hong MH,
Ahn BC, Kim Y, Choi SJ, Yoon HI, Lee JG, et al: Hyperprogressive
disease during PD-1/PD-L1 blockade in patients with non-small-cell
lung cancer. Ann Oncol. 30:1104–1113. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Heckmann MB, Finke D, Sauerbrey L, Frey N
and Lehmann LH: Increased expression of human endogenous retrovirus
K in endomyocardial biopsies from patients with cardiomyopathy-a
transcriptomics meta-analysis. BMC Genomics. 25(707)2024.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhang Y, Sun X, Jin Y, Chen K, Zhang L,
Gao X, Li M, Yuan Z, Jia J, Sun A and Ge J: Mitochondrial
transplantation augments the reparative capacity of macrophages
following myocardial injury. Adv Sci (Weinh).
12(e06337)2025.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kim H, Ahn HS, Hwang N, Huh Y, Bu S, Seo
KJ, Kwon SH, Lee HK, Kim JW, Yoon BK and Fang S: Epigenomic
landscape exhibits interferon signaling suppression in the patient
of myocarditis after BNT162b2 vaccination. Sci Rep.
13(8926)2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Patel T, Kelleman M, West Z, Peter A, Dove
M, Butto A and Oster ME: Comparison of multisystem inflammatory
syndrome in children-related myocarditis, classic viral
myocarditis, and COVID-19 vaccine-related myocarditis in children.
J Am Heart Assoc. 11(e024393)2022.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wang X, Chen J, Shen Y, Zhang H, Xu Y,
Zhang J and Cheng L: Baricitinib protects ICIs-related myocarditis
by targeting JAK1/STAT3 to regulate macrophage polarization.
Cytokine. 179(156620)2024.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Nishikawa T, Tamiya M, Ohta-Ogo K, Ikeda
Y, Hatakeyama K, Honma K, Yasui T, Shioyama W, Oka T, Inoue T, et
al: A case of lung cancer with very-late-onset immune checkpoint
inhibitor-related myocarditis. CJC Open. 4:651–655. 2022.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Nishikawa T, Kunimasa K, Ohta-Ogo K, Ikeda
Y, Yasui T, Shioyama W, Oka T, Honma K, Hatakeyama K, Kumagai T and
Fujita M: Sinus node dysfunction co-occurring with immune
checkpoint inhibitor-associated myocarditis. Intern Med.
61:2161–2165. 2022.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Flippe L, Bézie S, Anegon I and
Guillonneau C: Future prospects for CD8+ regulatory T
cells in immune tolerance. Immunol Rev. 292:209–224.
2019.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Valaperti A, Nishii M, Liu Y, Naito K,
Chan M, Zhang L, Skurk C, Schultheiss HP, Wells GA, Eriksson U and
Liu PP: Innate immune interleukin-1 receptor-associated kinase 4
exacerbates viral myocarditis by reducing CCR5(+) CD11b(+) monocyte
migration and impairing interferon production. Circulation.
128:1542–1554. 2013.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Lin W, Wang Y, Li M, Feng J, Yue Y, Yu J,
Hu Y and Suo Y: Tumor treating fields enhance anti-PD therapy by
improving CCL2/8 and CXCL9/CXCL10 expression through inducing
immunogenic cell death in NSCLC models. BMC Cancer.
25(489)2025.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Tan H, Shah NUH, He B, Liu T, Li T, Liu M,
Gu Y, Zhi C, Ou Y, Huang J, et al: Intratumoral delivery of
PD-1/PD-L1 and CTLA-4 inhibitors for recurrent/refractory solid
tumors: A proof-of-concept treatment strategy. Front Immunol.
16(1669924)2025.PubMed/NCBI View Article : Google Scholar
|