Introduction
Invasive fungal infections (IFIs) are serious
complications among patients admitted to surgical intensive care
units (ICUs), driven by profound alterations in host microbiota and
secondary immunosuppression. Surgical ICU patients face a unique
constellation of predisposing factors, including broad-spectrum
antibiotic use leading to dysbiosis, major surgical procedures that
disrupt natural barriers, indwelling intravascular catheters that
facilitate bloodstream invasion, mechanical ventilation that
impairs mucociliary clearance, and immunocompromising conditions
due to comorbidities or immunosuppressive therapy (1). Among the causative pathogens,
Candida species predominate, often resulting in substantial
morbidity, prolonged hospitalization, higher healthcare costs and
mortality rates that remain unacceptably high despite advances in
critical care (2).
IFI caused high rates of mortality in the surgical
ICU. The Extended Prevalence of Infection in Intensive Care/EPIC II
study, which collected data from 75 countries, reported that fungal
infections accounted for 19.4% of all ICU infections, with
Candida albicans as the most frequent species with
prevalence of 6.9 per 1,000 patients (3,4). A
previous study reported a candidiasis prevalence of 3.5% among ICU
patients with a mortality rate of 81.8% (5). Similarly, a study in a developing
country (Indonesia) showed that Candida species accounted
for 92.2% of ICU fungal infections, with a 50% mortality rate
(6). While incidence rates differ
substantially between developed and developing countries, mortality
remains high, ranging from 50 to 81.8% (3-6).
These findings may reflect the gap in diagnostic capabilities to
detect IFI between countries, but despite that, the treatment of
IFI is still a substantial issue given the high mortality rates.
These figures underscore the substantial burden of IFIs in surgical
ICUs and the pressing need for effective preventive strategies.
Antifungal prophylaxis has been proposed as an
intervention to reduce IFI incidence in high-risk surgical ICU
populations by suppressing fungal colonization, preventing invasive
disease, improving clinical outcomes, and reducing the economic
burden of prolonged and complex care (7). Treatment of fungal IFI often requires
weeks of antifungal administration in the ICU and increases the
risk of drug interactions since some commonly used agents, such as
azoles, could interfere with the metabolism of other drugs
(7). However, its use remains
controversial due to concerns about antifungal resistance, biofilm
formation, drug-related toxicity and cost implications (8-11).
Randomized controlled trials (RCTs) evaluating antifungal
prophylaxis in surgical ICU patients have yielded conflicting
results, with certain trials demonstrating notable reductions in
IFI incidence and others showing no meaningful benefit (8,9).
To date, to the best of our knowledge, no
meta-analysis has specifically examined the effectiveness of
antifungal prophylaxis in preventing IFIs in postoperative surgical
ICU patients, regardless of the underlying diagnosis. Existing
meta-analyses have largely focused on very specific population such
as patients with critical illness, hematologic malignancies or
hematopoietic stem cell transplantation, limiting their
applicability to surgical ICU settings (2,12). A
previous notable meta-analysis on fluconazole prophylaxis in
critically ill surgical patients reported reduced IFI incidence but
no survival advantage (12).
Given the heterogeneity and inconclusive nature of
the available evidence, there is a critical need for a
comprehensive systematic review and meta-analysis focused on
surgical ICU patients. Such an analysis could provide a robust
evidence base for clinical decision-making and policy development
regarding antifungal prophylaxis in this high-risk population. The
present study aimed to address this gap by analyzing data from RCTs
to evaluate the efficacy and safety of antifungal prophylaxis in
reducing IFI incidence and improving clinical outcomes in
postoperative surgical ICU patients, thereby guiding evidence-based
practice to enhance patient safety and optimize healthcare resource
utilization.
Materials and methods
Study design
The current systematic review and meta-analysis was
conducted in accordance with the Preferred Reporting Items for
Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines
(13). The study protocol was
prospectively registered in the International Prospective Register
of Systematic Reviews with registration no. CRD420251013674
(https://www.crd.york.ac.uk/PROSPERO/view/CRD420251013674).
Literature search strategy
A comprehensive literature search was performed
based on the Population, Intervention, Comparator, Outcome and
Study design criteria (13). The
search was conducted using PubMed (pubmed.ncbi.nlm.nih.gov), Scopus (scopus.com), Google Scholar (scholar.google.com) and ScienceDirect (www.sciencedirect.com) from database inception to
February 2025. The search strategy combined Medical Subject
Headings and free-text terms related to the population,
intervention, comparator and outcomes, with appropriate adjustments
for each database (Table SI). For
the population, search terms included ‘intensive care units,
surgical’, ‘surgical intensive care unit’, ‘SICU’, ‘critically ill
patients’, ‘postoperative patients’, ‘septic shock’, ‘multiple
organ dysfunction syndrome’, ‘mechanical ventilation’ and ‘total
parenteral nutrition’. For the intervention, terms such as
‘antifungal agents’, ‘antifungal prophylaxis’, ‘prophylactic
antifungal therapy’, ‘pre-emptive antifungal therapy’ and ‘fungal
infection prevention’ were applied. For comparator, terms such as
‘empirical antifungal therapy’ and ‘placebo’ were applied. For
outcomes, terms such as ‘mortality’, ‘incidence’, ‘adverse events’
and ‘length of stay’ were used. The search was not restricted by
language or the earliest publication date. The reference lists of
all included studies and relevant reviews were screened to identify
additional eligible articles.
Eligibility criteria
Studies were eligible if they enrolled patients aged
≥12 years admitted to a surgical ICU, evaluated antifungal
prophylaxis or pre-emptive therapy with agents such as azoles,
echinocandins or polyenes, and compared these interventions with a
placebo, no prophylaxis or an alternative antifungal agent. The
primary outcome of interest was the incidence of IFIs, including
candidemia and invasive candidiasis. Secondary outcomes included
all-cause mortality, length of ICU stay and adverse events. Only
RCTs were included. Studies were excluded if they involved
exclusively medical ICU patients, neonatal ICU patients or solely
immunosuppressed populations, unless these formed parts of a mixed
surgical ICU cohort. Observational studies, case reports, case
series, narrative reviews, editorials and in vitro or animal
studies were not considered. Studies that did not report the
incidence of IFIs or relevant clinical outcomes were also
excluded.
Study selection
Two authors independently screened the titles and
abstracts of all retrieved references to identify potentially
relevant studies. The full-text articles of these studies were then
assessed for eligibility according to the inclusion and exclusion
criteria (Table I). Any
disagreements between the authors were resolved through discussion
or consultation with a third author. The study selection process
was documented in a PRISMA flow diagram, which detailed the number
of records identified, screened and included, as well as the
reasons for exclusion at each stage.
 | Table IInclusion and exclusion criteria. |
Table I
Inclusion and exclusion criteria.
| Component | Inclusion
criteria | Exclusion
criteria |
|---|
| Population | Studies with
patients ≥12 years old in surgical ICUs. | Studies with
non-surgical ICU patients (such as medical ICU and neonatal ICU).
Studies with only immunosuppressed patients, unless part of a mixed
surgical ICU population. |
| Intervention | Studies evaluating
antifungal prophylaxis or pre-emptive therapy (fluconazole,
echinocandins and amphotericin B, among others). | Studies evaluating
treatment/therapy of fungal infections, not prophylaxis. Studies
focusing solely on antifungal de-escalation strategies. |
| Comparator | Placebo or no
antifungal prophylaxis. Comparisons between different antifungal
agents. | NA |
| Outcomes | Primary: Incidence
of fungal infection (such as candidemia and invasive candidiasis).
Secondary: Mortality, length of stay at the ICU and adverse
events. | Studies not
reporting fungal infection incidence or other clinically relevant
outcomes |
| Study design | RCTs. | Observational
studies, case reports, case series, commentaries, editorials,
narrative reviews, expert opinions, animal studies or in
vitro studies. |
| Publication
language | Any language
(English and non-English). | NA |
Data extraction and management
Data from eligible studies were extracted
independently by two authors using a standardized data extraction
form to ensure consistency. The extracted data included the
following information: i) Study characteristics, such as author,
year, country and setting; ii) patient demographics and clinical
characteristics; ii) details of the antifungal intervention,
including class, agent, dosage, route and duration; iv) comparator
details; and v) outcomes, including the incidence of IFIs,
mortality, length of stay at the ICU and adverse events such as
liver function abnormalities, when the data were available.
Discrepancies in the extracted data were resolved through
consensus.
Risk of bias assessment
The risk of bias for the included RCTs was assessed
using the Cochrane Risk of Bias 2.0 tool, which evaluates five
domains: Bias arising from the randomization process, bias due to
deviations from intended interventions, bias due to missing outcome
data, bias in measurement of the outcome and bias in selection of
the reported result (14). Each
domain was rated as ‘low risk’, ‘some concerns’ or ‘high risk’ of
bias. Visual summaries of the assessments were generated using the
Risk-of-Bias VISualization tool (version 0.3.0.900) (15).
Data synthesis and statistical
analysis
Meta-analysis was performed using RStudio and the
‘meta’ package version 8.2-1 (Posit Software, PBC). For dichotomous
outcomes, pooled risk ratios (RRs) with 95% CIs were calculated.
Heterogeneity among studies was assessed using the χ2
test and quantified with the I2 statistic. A
random-effects model was used for all meta-analyses. Subgroup
analyses were performed according to antifungal class (azole,
echinocandin and polyene) and prophylaxis type (prophylactic vs.
pre-emptive). Publication bias was assessed visually with funnel
plots and statistically analyzed using Egger's test. A
leave-one-out sensitivity analyses were conducted to evaluate the
robustness of the results by excluding studies at high risk of
bias. A leave-one-out sensitivity analysis was performed to
evaluate the influence of each individual study on the overall
pooled effect. The analysis sequentially excluded each RCT and
recalculated the pooled RR to determine whether the findings were
dependent on any single study. P<0.05 was considered to indicate
a statistically significant difference.
Results
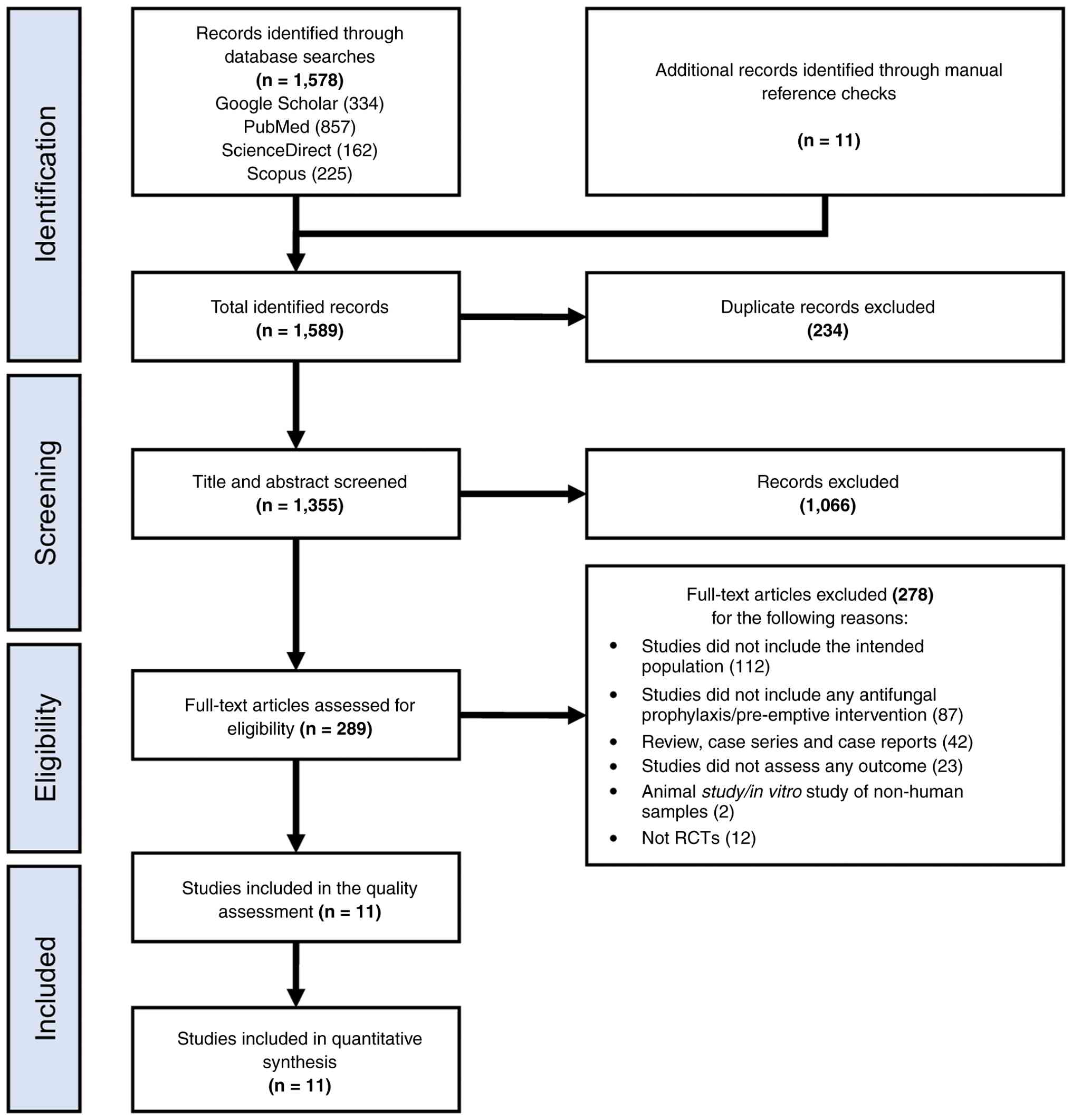
Systematic search and study
selection
The literature search identified a total of 1,578
records through database searches, with an additional 11 records
identified through manual reference checks. After removing
duplicates, 1,355 records remained for title and abstract
screening. Of these, 1,066 records were excluded due to not meeting
the eligibility criteria, leaving 289 articles for full-text
assessment. Following a detailed review, 278 articles were excluded
for reasons including non-RCT design, inappropriate patient
population, irrelevant intervention or absence of predefined
outcomes. Ultimately, 11 RCTs met the inclusion criteria and were
included in the meta-analysis (9,16-25).
The study selection process is presented as a PRISMA flow diagram
in Fig. 1.
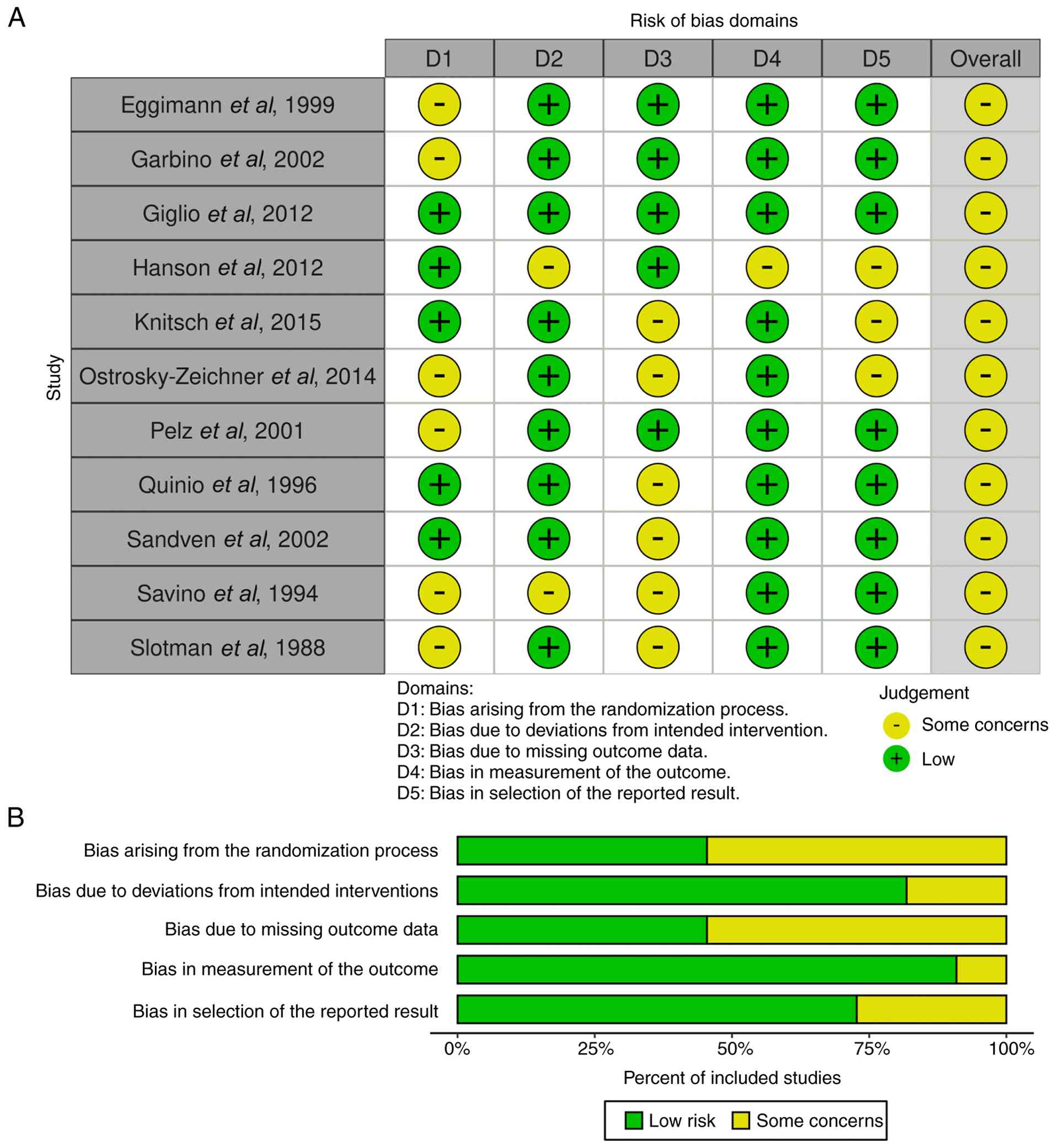
Quality assessment
All 11 included trials underwent risk of bias
assessment using the Cochrane Risk of Bias 2.0 tool. Most trials
exhibited low risk of bias across all domains, indicating good
methodological rigor. Several studies showed ‘some concerns’ in
several domains of evaluation. In total, 6 studies were evaluated
as exhibiting ‘some concerns’ in the randomization process due to
limitations in allocation concealment or absence of blinded outcome
assessment (domain 1) (16,17,20,21,24,25), 2
studies were evaluated as presenting ‘some concerns’ due to
deviations from the intended interventions (domain 2) (19,24), 6
studies exhibited ‘some concerns’ regarding missing outcomes
(domain 3), 1 study presented a possibility of bias in outcome
measurement due to limited information of the method used to detect
the fungal infection (domain 4) (19), and 3 studies showed ‘some concerns’
in the selection of the reported result due to limited availability
of outcomes in the registered clinical trial protocol (domain 5)
(9,19,20).
No trial was rated as having a high risk of bias. The graphical
traffic-light and summary plots in Fig.
2 provide an overview of domain-specific assessments.
Characteristics of the included
studies
The characteristics of the included studies are
presented in Table II. The
included RCTs had cumulative sample sizes ranging between 43 and
292 patients. Geographically, the studies spanned Europe and North
America with patient populations drawn from surgical ICUs following
major abdominal surgery, trauma surgery, gastrointestinal
perforations or complicated postoperative courses. The antifungal
agents evaluated included azoles (fluconazole), echinocandins
(caspofungin and micafungin) and polyenes (amphotericin B). The
duration of prophylaxis varied from 5 days to 4 weeks. All trials
reported IFI incidence as a primary outcome. Secondary outcomes
included mortality, adverse events and liver function
abnormalities.
 | Table IICharacteristics of included
studies. |
Table II
Characteristics of included
studies.
| | Sample size | | Age, years | |
|---|
| First author/s,
year | Country | Intervention
category | Drug class | Intervention and
dose | Control | Duration of
intervention | C | I | Total | Surgery type | C | I | Recorded risk
factors | (Refs.) |
|---|
| Eggimann et
al, 1999 | Switzerland | Prophylaxis | Azoles | Fluconazole 400 mg
i.v. once a day | Placebo | Given from the
admission to ICU until complete resolution of abdominal disease. If
fungal infection occurs, the treatment is switched to amphotericin
B. | 20 | 23 | 43 | GI | 57.0
(33.0-78.0) | 63.0
(21.0-82.0) | Diabetes,
corticosteroid treatment, antibiotic therapy, parenteral nutrition,
splenectomy | (16) |
| Garbino et
al, 2002 | Switzerland | Prophylaxis | Azoles | Fluconazole 100 mg
i.v., polymyxin B (37.5 mg), neomycin (250 mg) and vancomycin (250
mg) syrup; six times daily for the entire treatment. | Polymy xin B (37.5
mg), neomycin (250 mg) and vancomycin (250 mg) syrup; six times
daily without fluconazole | Given from the
admission to ICU until a fungal infection developed, withdrawal
from mechanical ventilation or suspicion of a serious adverse
event. | 101 | 103 | 204 | GI, CV, neuronal
and orthopedic | 55.9±18.0 | 52.9±19.0 | Diabetes, COPD CKD,
neurological disorder, cancer, chronic liver failure | (17) |
| Giglio et
al, 2012 | Italy | Prophylaxis | Polyenes | Nystatin
2x106 U/day administered three times daily via the
nasogastri tube | Placebo | At least 48 h
Criteria for stopping the prophylaxis are unknown. | 50 | 49 | 99 | GI, neuronal and
trauma | 58.0±19.0 | 54.0±22.0 | Diabetes,
corticosteroid treatment, neutropenia, antibiotic therapy,
parenteral nutrition, CKD,central venous catheter | (18) |
| Hanson et
al, 2012 | USA | Pre-emptive | Echinoc andins | Anidulafungin 100
mg i.v., single dose daily | Placebo | At least 14
days. | 17 | 47 | 64 | GI | 60.0
(22.0-82.0) | 58.0
(19.0-79.0) | Diabetes, CKD,
parenteral nutrition, antibiotics treatment | (19) |
| Knitsch et
al, 2015 | Germany | Pre-emptive | Echinoc andins | Micafungin 100 mg
i.v. daily | Placebo | 6 weeks unless the
subject experiences EOT event: Confirmed invasive candidiasis,
improvement in surgical condition (as indicated by recovery of
gastrointestinal function allowing enteral feeding of up to 50% of
daily calorie requirement), alternative antifungal treatment or
death. | 124 | 117 | 241 | GI | 63.0±15.8 | 61.0±14.8 | Not recorded | (9) |
| Ostrosky-Zeichner
et al, 2014 | USA | Pre-emptive and
prophylaxis | Echinoc andins | Caspofungin 70 mg
i.v. loading dose, followed by 50 mg i.v. once daily | Placebo | For the duration of
stay in the ICU, maximum of 28 days. | 102 | 117 | 219 | GI, CV and
trauma | 56.7±16.6 | 58.2±17.6 | Metabolic disease,
CKD, pancreatitis, neurological disorder, infection | (20) |
| Pelz et al,
2001 | USA | Prophylaxis | Azoles | Fluconazole loading
dose, 800 mg; maintenance, 400 mg p.o. or i.v. once daily | Placebo | Administered until
initiation of empirical antifungals or ICU discharge. | 130 | 130 | 260 | Not specified | 66.0
(20.0-88.0) | 63.0
(18.0-92.0) | Diabetes,
antibiotic use, CKD, HIV infection | (21) |
| Quinio et
al, 1996 | France | Prophylaxis,
selective digestive decontamination | Polyenes | Colistin sulfate
(polymixin E; 10 mg/ml), gentamicin (8 mg/ml) and ampho-tericin B
(50 mg/ml) instilled through the nasogastric tube (10 ml) and
nostrils (2 ml in each) four times daily; a gel with antibiotics
was applied in the oroph-aryngeal cavity, four times daily | Placebo | Started within a
day in ICU until 24 h after extubation or beginning of enteral
feeding. | 72 | 76 | 148 | GI, thorax and head
trauma | 33.0±15.0 | 35.5±16.0 | Mechanical
ventilation, antibiotic use | (22) |
| Sandven et
al, 2002 | Norway | Prophylaxis | Azoles | Fluconazole 400 mg
i.v. intraoperatively | Placebo | Single dose,
intraoperatively | 56 | 53 | 109 | GI | 60.0
(15.0-87.0) | 68.0
(13.0-86.0) | Antibiotic use,
diabetes, cancer | (23) |
| Savino et
al, 1994 | USA | Prophylaxis | Azoles,
polyenes | Clotrimazole 10 mg
three times daily or ketoconazole 200 mg daily or nystatin
2,000,000 units four times daily | Placebo | Unknown. | 72 | 220 | 292 | Trauma and
non-trauma | 54.0±19.0 | Clotrimazole:
54.0±19.0; ketoconazole 57.0±18.0; nystatin: 53.0±20.0 | Ventilator,
antibiotic use | (24) |
| Slotman et
al, 1988 | USA | Prophylaxis | Azoles | Ketoconazole 200 mg
p.o. daily | Placebo | 21 days or until
discharged from surgical ICU. | 36 | 35 | 71 | GI, CV, thorax,
orthopedic, neuronal, genitourinary, trauma and other | 58.0
(18.0-91.0) | 57.0
(19.0-87.0) | Critically ill
surgical patients (MODS, sepsis, AKI, major burns or
malnutrition) | (25) |
Effect of antifungal prophylaxis or
pre-emptive therapy on fungal infection risk
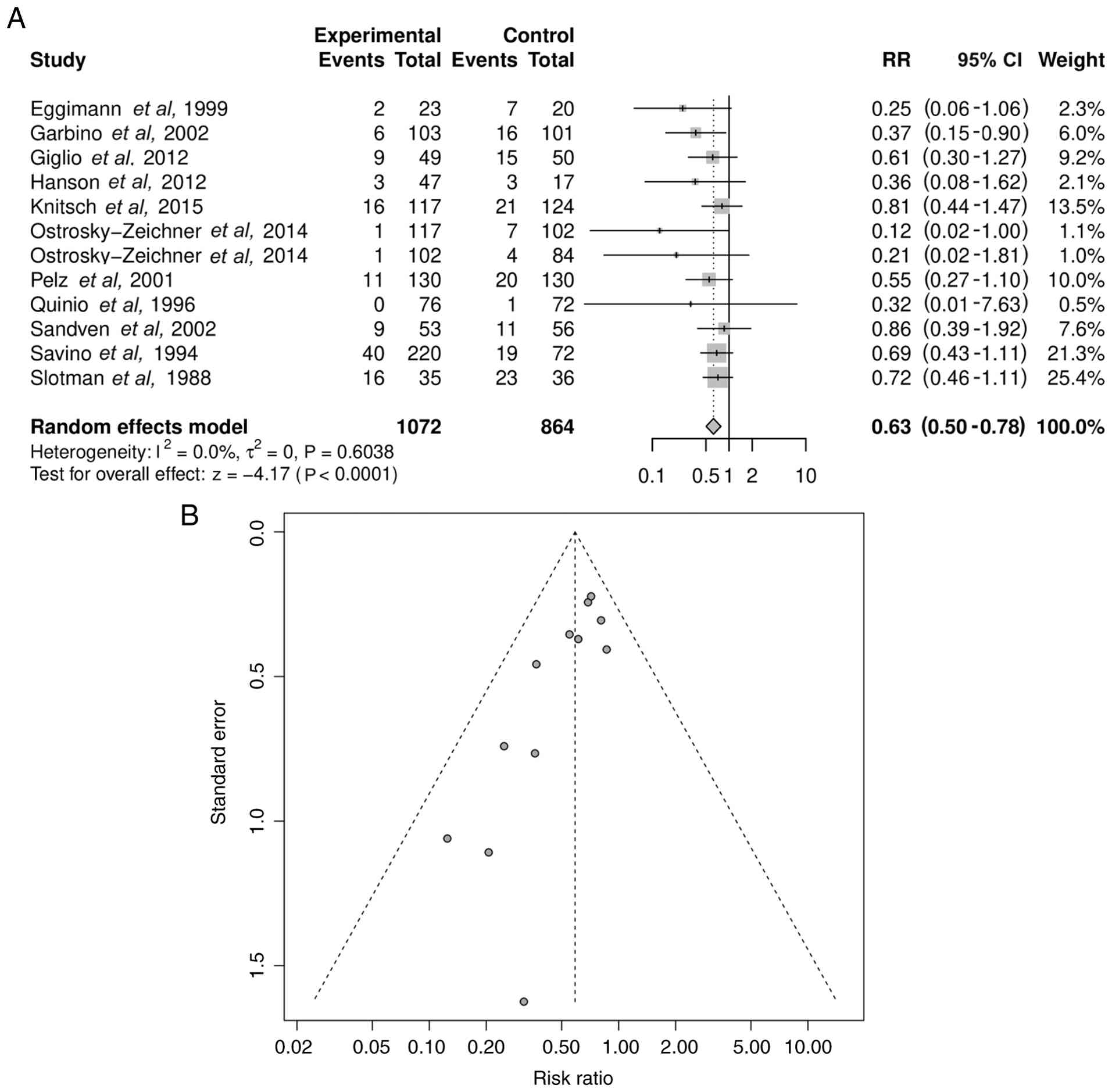
Meta-analysis of all 11 trials demonstrated a
statistically significant reduction in the incidence of IFIs in
patients receiving antifungal prophylaxis or pre-emptive therapy
compared with the controls (RR=0.63; 95% CI, 0.50-0.78;
P<0.0001; Fig. 3A).
Heterogeneity was low (I2=0.0%; τ2=0;
P=0.6038), supporting the robustness of the pooled estimate. The
forest plot (Fig. 3A) illustrates
the consistent direction of the effect across studies. The funnel
plot for this analysis (Fig. 3B)
appeared symmetrical, but Egger's test showed statistical evidence
of publication bias (P=0.0009).
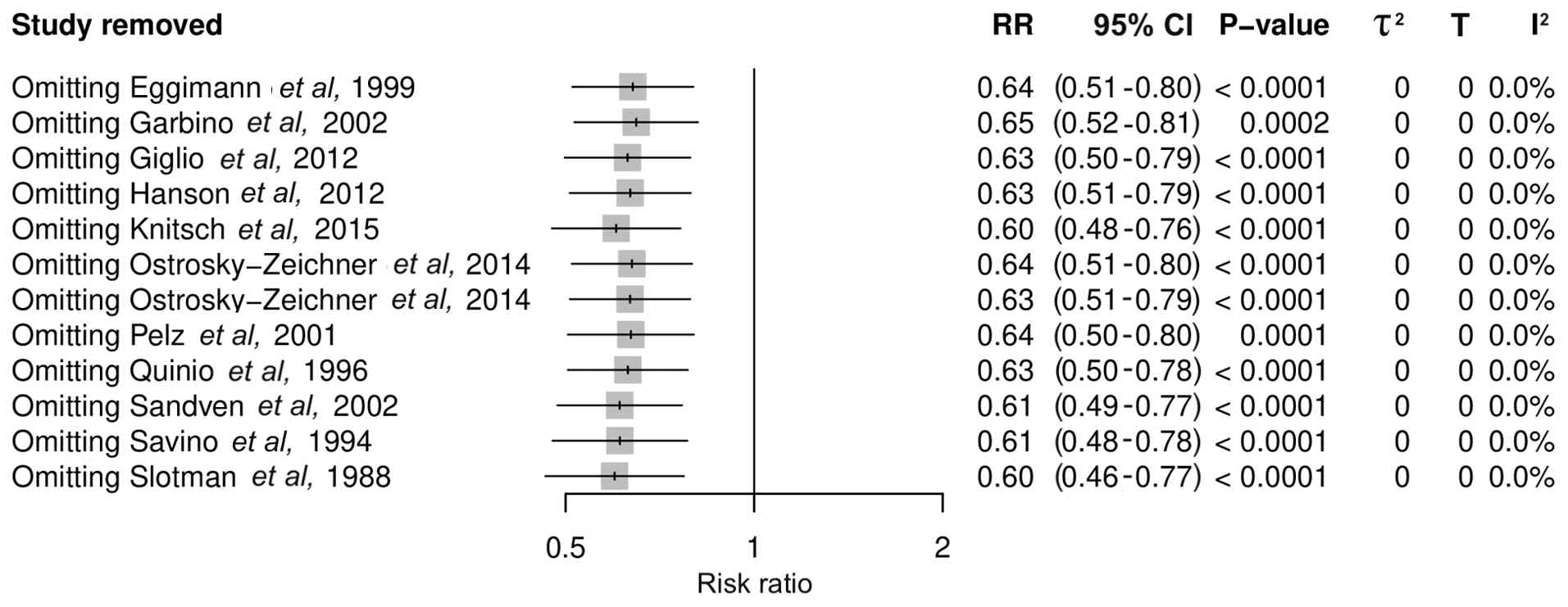
A leave-one-out sensitivity analysis was conducted
to assess the robustness of the primary pooled effect (Fig. 4). In this analysis, the study of
Ostrosky-Zeichner et al (20) was included twice since it presented
two analyses of different interventions, prophylaxis and
pre-emptive strategies. The omission of each individual study
sequentially did not change the effect estimate. The recalculated
RRs ranged from 0.60 to 0.65 and all 95% CIs remained below 1.0
(95% CI range, 0.48-0.81), with statistically significant results,
indicating a consistent protective effect. No single trial exerted
a disproportionate influence on the direction or magnitude of the
overall outcome. Heterogeneity remained negligible across all
iterations (I2=0%). These findings demonstrated that the
pooled effect was stable and not driven by any single study.
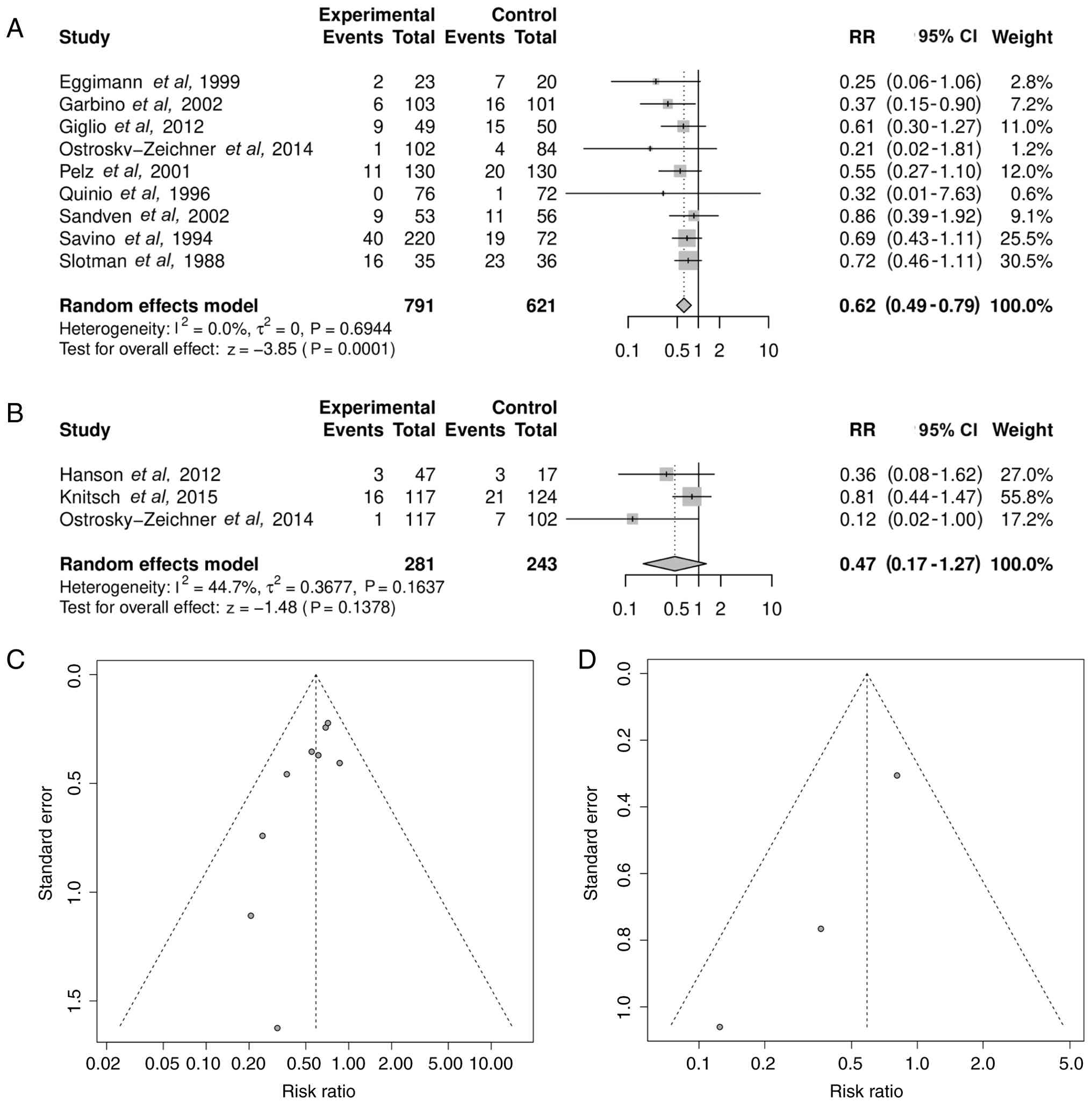
Subgroup analysis: Prophylaxis vs.
pre-emptive therapy
When stratified by strategy, prophylaxis
significantly reduced IFI risk but pre-emptive therapy did not.
Prophylaxis yielded a pooled RR of 0.62 (95% CI, 0.49-0.79;
P=0.0001) with non-significant heterogeneity (I2=0.0%;
τ²=0; P=0.6944), while pre-emptive therapy showed a pooled RR of
0.47 (95% CI, 0.17-1.27; P=0.1378) with moderate but
non-significant heterogeneity (I2=44.7%;
τ2=0.3677; P=0.1637) (Fig.
5A and B). The funnel plots for
each subgroup (Fig. 5C and D) generally demonstrated symmetrical
distribution, but there was a significant publication bias among
publications analyzed in Fig. 5D as
assessed by Egger's test (Fig. 5C,
P=0.1138; Fig. 5D, P=0.0236).
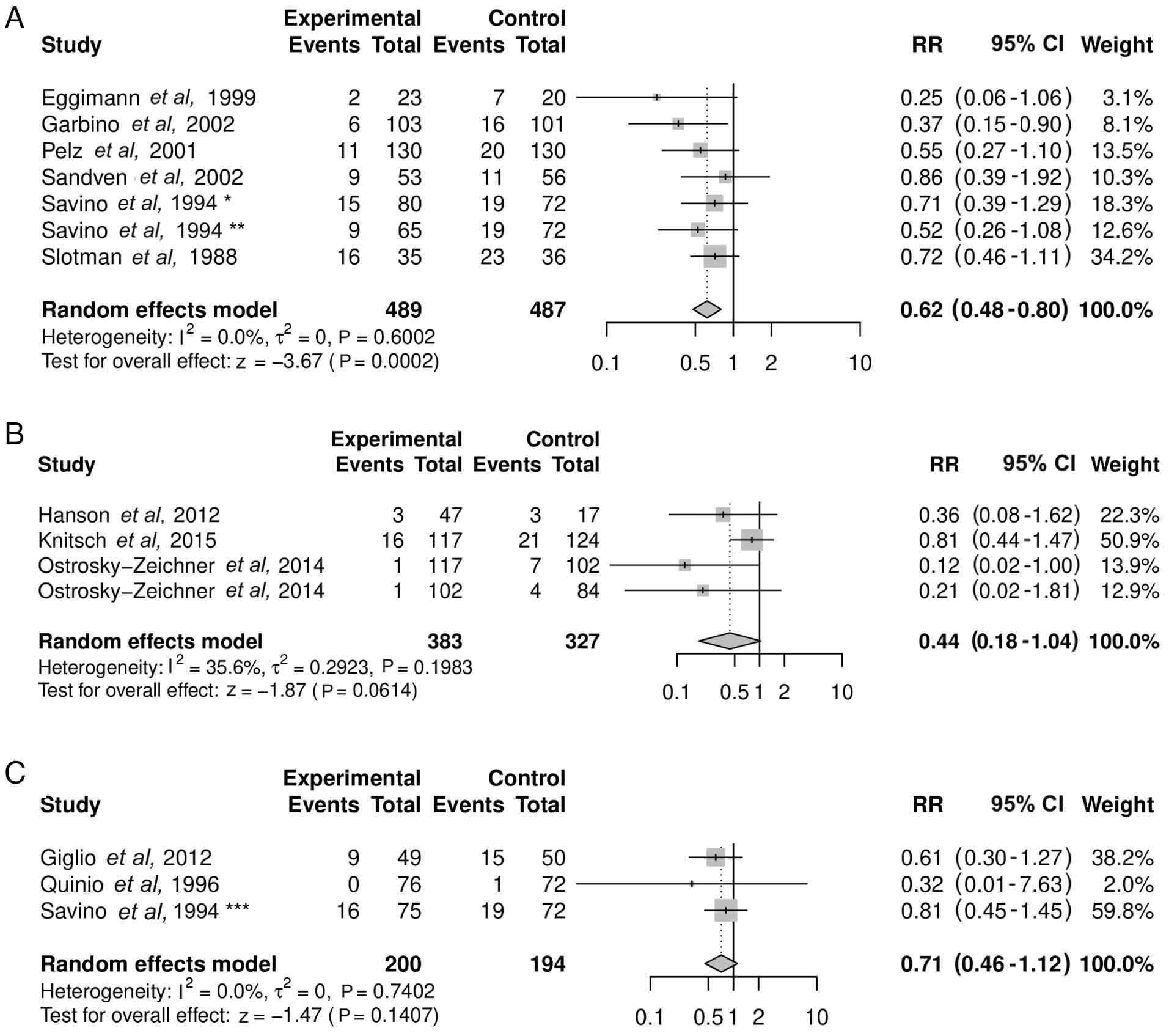
Subgroup analysis stratified by
antifungal class
Azole prophylaxis was associated with a significant
reduction in IFI risk (RR=0.62; 95% CI, 0.48-0.80; P=0.0002) and
low heterogeneity (I2=0.0%; τ2=0; P=0.6002)
(Fig. 6A). By contrast,
echinocandin prophylaxis (RR=0.44; 95% CI, 0.18-1.04; P=0.0614)
with heterogeneity of I2=35.6% (τ2=0.2923;
P=0.1983) (Fig. 6B) and polyene
prophylaxis (RR=0.71; 95% CI, 0.46-1.12; P=0.1407) with
heterogeneity of I2=0.0% (τ2=0; P=0.7402)
(Fig. 6C) were not significantly
associated with protection from IFI risk. In this analysis, the
study of Ostrosky-Zeichner et al (20) was included twice since it presented
analyses of two different interventions, prophylaxis and
pre-emptive strategies. This analysis also separated the study of
Savino et al (24) since by
three different types of intervention, clotrimazole, ketoconazole
and nystatin. The funnel plots for azoles (Egger's test P=0.0615),
echinocandins (Egger's test P=0.0207) and polyenes (Egger's test
P=0.4220) (Fig. 7A-C) were
symmetrical, indicating no strong evidence of publication bias
except for the studies that investigated echinocandins.
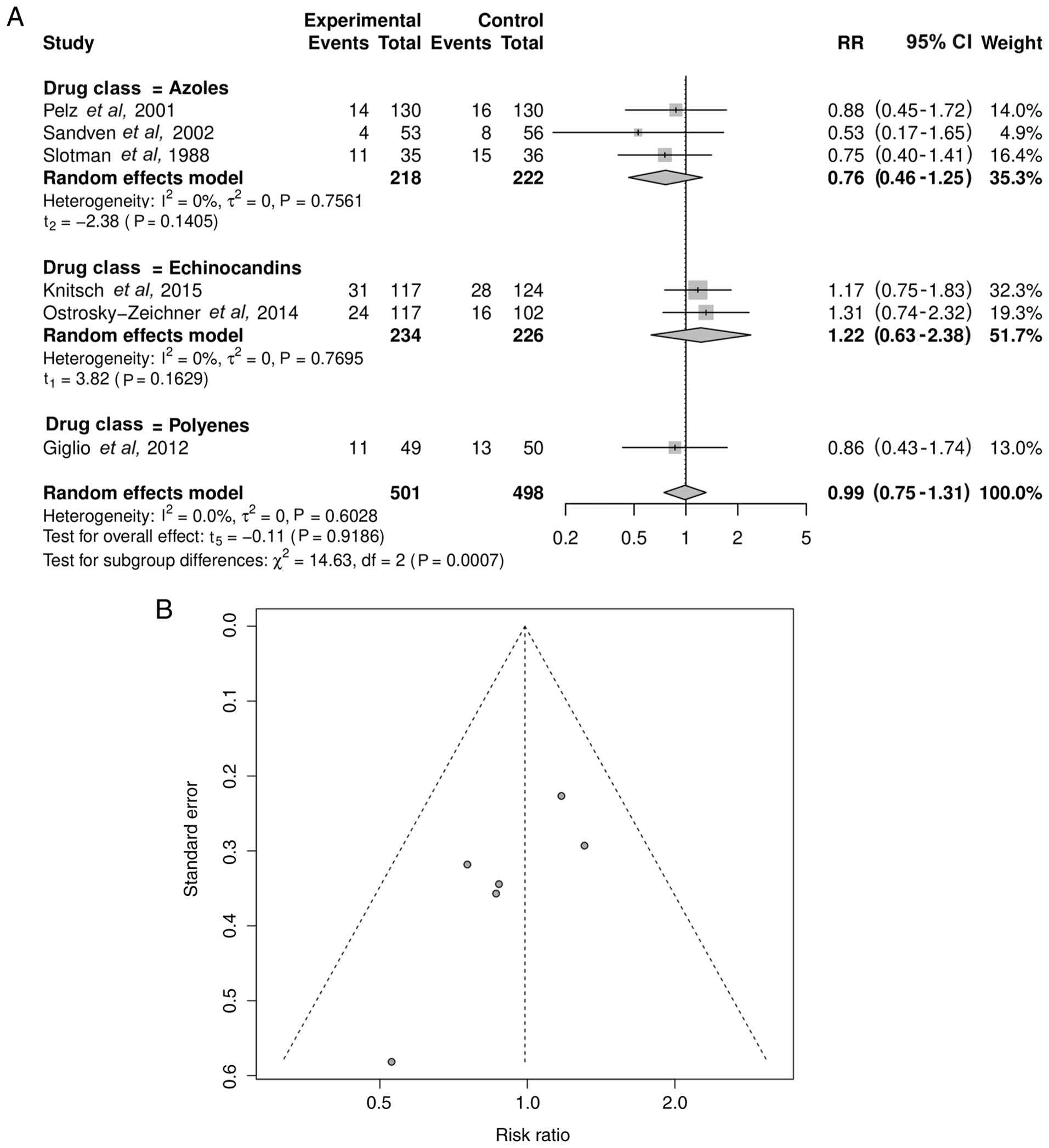
Effect on mortality
In total, 6 trials reported all-cause mortality.
Pooled analysis revealed no significant difference in the mortality
risk between the antifungal prophylaxis and control groups
(RR=0.99; 95% CI, 0.75-1.31; P=0.9186) with low heterogeneity
(I2=0.0%; τ2=0; P=0.6028) (Fig. 8A). The funnel plot for mortality
(Fig. 8B) was symmetrical (Egger's
test P=0.0563).
Severe adverse events and liver
function abnormalities
In total, 3 trials reported severe adverse events.
Pooled analysis revealed no significant association between
antifungal prophylaxis and the risk of severe adverse events
(RR=0.98; 95% CI, 0.70-1.36; P=0.8839) with heterogeneity of
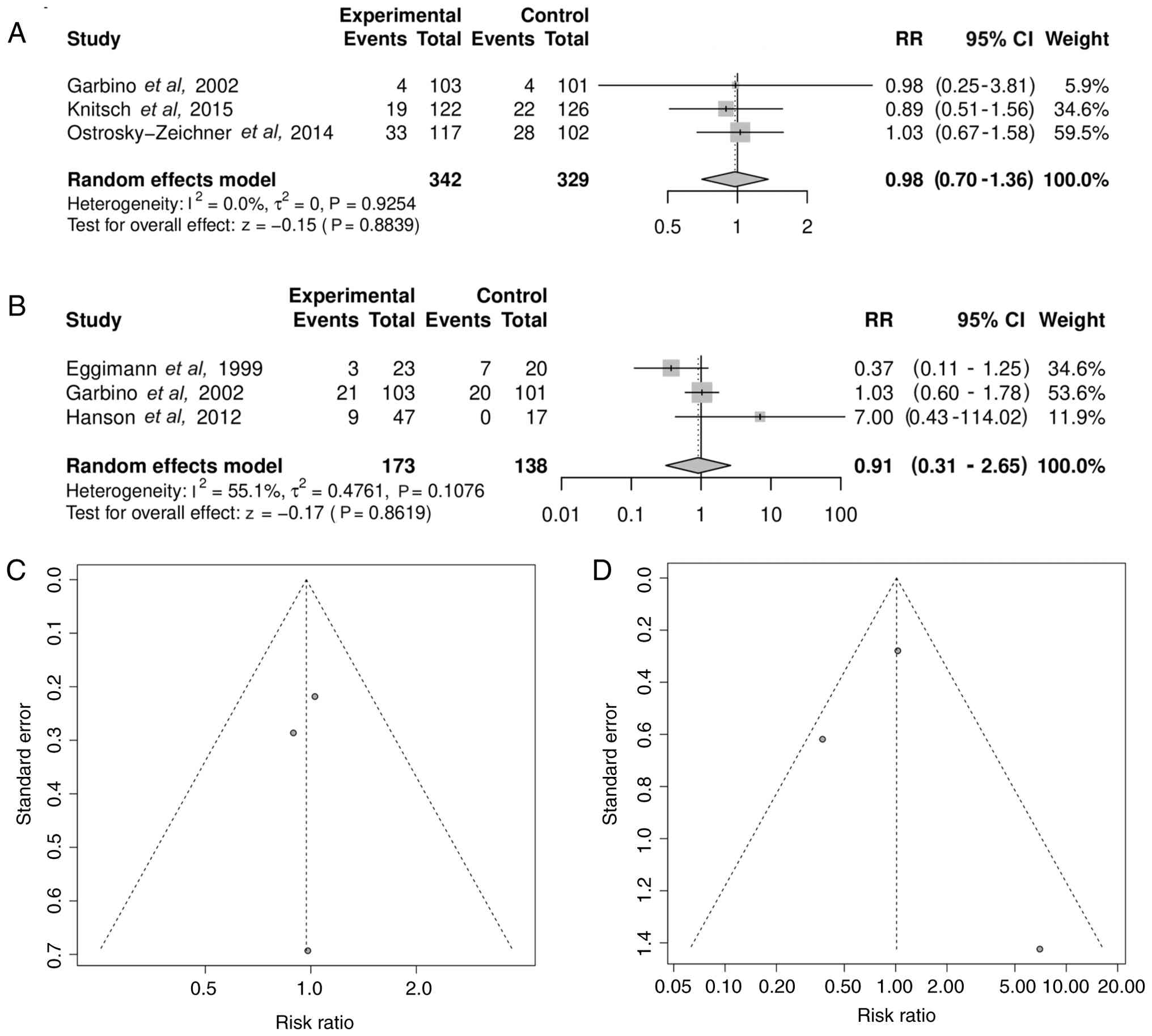
I2=0.0% (τ2=0; P=0.9254) (Fig. 9A). In total, 3 trials reported liver
function outcomes. The pooled RR for liver dysfunction was 0.91
(95% CI, 0.31-2.65; P=0.8619) with heterogeneity of
I2=55.1% (τ2=0.4761; P=0.1076) (Fig. 9B). The corresponding funnel plots
(Fig. 9C and D) showed a balanced distribution (Egger's
test P=0.8266 and 0.8852, respectively), suggesting no major
publication bias.
Discussion
The present systematic review and meta-analysis
demonstrated that antifungal prophylaxis (especially when
administered as prophylaxis but not as a pre-emptive strategy)
significantly reduced the risk of fungal infections in surgical ICU
patients without increasing the risk of severe adverse events or
hepatotoxicity. Across 11 RCTs, azoles were effective, while
polyenes and echinocandins did not confer a significant benefit.
Despite the reduction in IFI risk, no mortality benefit was
observed.
The relative risk reduction in fungal infection
incidence is clinically meaningful and mirrors outcomes reported in
populations with hematologic malignancies and transplantations
(1,26,27).
Surgical ICU patients face multiple predisposing factors, including
gastrointestinal surgery, central venous catheters, parenteral
nutrition, prolonged ventilation and broad-spectrum antibiotics
administration, which increase the susceptibility to IFIs (12,26,28).
The present findings suggest that targeted prophylaxis offers
substantial protection in this context.
Although prophylaxis was effective, pre-emptive
therapy did not reduce the IFI risk. This difference may be
attributed to earlier intervention, as pre-emptive approaches often
depend on the presence of biomarkers such as β-D-glucan or
galactomannan, which may allow limited fungal proliferation before
therapy begins (9,19,20).
These results align with the 2025 guideline by the European
Confederation of Medical Mycology, International Society of Human
and Animal Mycology (ISHAM) and American Society for Microbiology
(ASM) for the diagnosis and management of candidiasis (29). The guideline advised in favor of
prophylaxis for surgical ICU patients with high-risk factors, such
as anastomotic leaks, necrotizing pancreatitis or multifocal
Candida colonization, while noting that pre-emptive
strategies remain less validated outside controlled settings
(29).
While the findings of the present meta-analysis
demonstrated a clinically meaningful reduction in IFI risk, it is
important to interpret this benefit in the context of the broader
ecological implications of antifungal exposure. Beyond individual
patient outcomes, widespread or prolonged antifungal use may alter
the local microbial flora by selecting for resistant fungal
strains, disrupting commensal microbiota and increasing the
likelihood of future breakthrough infections with reduced treatment
responsiveness (30,31). Prolonged prophylaxis has been
associated with shifts in local epidemiology, including increased
colonization and infection by non-albicans Candida species
with reduced azole susceptibility (27,32).
Thus, although prophylaxis may be valuable in selected high-risk
surgical ICU patients, its implementation should remain
management-driven and supported by local surveillance data to
mitigate the long-term risk of resistance and preserve the efficacy
of antifungals.
Cost considerations are also relevant, especially in
low- and middle-income countries where access to treatments and
diagnostic capacity may be limited. When targeted to high-risk
surgical ICU patients, prophylaxis may remain cost-effective by
preventing IFIs and associated ICU resource utilization, but
broader indiscriminate use may not provide the same value. A
previous systematic review has also suggested that prophylaxis
treatment potentially brings cost benefits to ICU patients with
high-risk factors for IFIs (33).
Compared with a previous meta-analysis that focused primarily on
fluconazole and did not evaluate pre-emptive approaches or
echinocandin use, the present study offers a more updated and
comprehensive assessment and better reflects current therapeutic
options (12).
According to the present study, azoles, particularly
fluconazole, showed consistent efficacy in reducing IFI risk,
reinforcing their role as frontline prophylactic agents where local
resistance rates are low (16,17,21,23).
Although the limited number of included studies could not
demonstrate the effectiveness of echinocandins in the present
meta-analysis, echinocandins may also be beneficial and relevant in
the context of rising azole resistance; however, their high cost
limits routine prophylactic use (7,33). In
our study, polyenes did not exhibit a clear benefit. These findings
are consistent with those of previous studies supporting the
effectiveness of azoles in preventing candidemia in abdominal
surgical patients (26,34). ISHAM and ASM also recommend
fluconazole administration in selected high-risk patients but
caution should be taken against its widespread use due to concerns
of resistance to species such as Candida glabrata and
Candida krusei (29).
Echinocandins remain primarily recommended for the treatment of IFI
(29), although the present
analysis suggests that they may have prophylactic potential.
Therefore, further and more comprehensive studies are needed to
establish whether it is beneficial to start echinocandins earlier
in patients that have a high risk of IFI.
The results of the present meta-analysis revealed no
mortality benefit of the use of antifungal prophylaxis or
pre-emptive therapy. This is unsurprising, given the multifactorial
causes of death in critically ill patients, including bacterial
sepsis, multi-organ failure and postoperative complications
(35,36). Breakthrough infections in control
arms, when diagnosed and treated promptly, may also result in
minimized survival differences (35,36).
Furthermore, most RCTs were underpowered for mortality outcomes.
The 2025 guideline by ISHAM and ASM similarly emphasizes that
prophylaxis is unlikely to improve overall survival in ICU
populations, where mortality reflects a broad interplay of comorbid
conditions (29).
The lack of a demonstrable mortality benefit in the
present meta-analysis should be interpreted in the context of the
underlying complexity of critical illness. Most trials were not
statistically powered to detect differences in mortality, used IFI
incidence as the primary endpoint and enrolled relatively small
cohorts with limited follow-up (16,17,19,22,23).
Taken together, these factors make it biologically plausible that
antifungal prophylaxis substantially reduces IFI incidence without
translating into a measurable mortality advantage at the trial
level.
The safety outcomes were favorable, with no increase
in serious adverse events or hepatotoxicity. This supports
selective prophylaxis in well-defined risk groups, providing that
safety monitoring is maintained. Nevertheless, the potential for
antifungal resistance remains a critical concern.
Clinically, the present results support prophylaxis
for surgical ICU patients at high risk, for example, those with
complicated abdominal surgery, necrotizing pancreatitis or septic
shock requiring parenteral nutrition and central venous access.
Prophylaxis appears more dependable than pre-emptive strategy in
resource-limited settings where diagnostics are constrained.
The present study has a number of strengths:
Inclusion of only RCTs, rigorous risk of bias assessment, low
heterogeneity, and clinically meaningful subgroup analyses based on
antifungal class and strategy. To the best of our knowledge, this
is the first meta-analysis focused solely on surgical ICU patients,
rather than combined ICU populations. This distinction is
clinically meaningful, as surgical patients differ in baseline
risk, exposure to invasive procedures and epidemiology of fungal
colonization, making the findings of the present study more
directly applicable to real-world decision-making in perioperative
critical care. However, certain limitations should be acknowledged.
The number of echinocandin and polyene trials was small, limiting
statistical precision. All included studies were conducted in
high-income countries, restricting applicability to low- and
middle-income settings with differing epidemiology, diagnostic
capacity and management infrastructure. Important outcomes, such as
the length of stay at the ICU, cost-effectiveness and long-term
resistance were not reported in the included studies. Finally,
although publication bias was not apparent, undetected negative
studies cannot be excluded.
Future research should focus on large, multicenter
RCTs that directly compare antifungal classes, employ standardized
mortality endpoints, and evaluate cost-effectiveness. Trials in
low- and middle-income countries are particularly needed to
establish generalizability. Studies on biomarker-guided pre-emptive
strategies may clarify their role, especially in
resource-constrained environments. Integrating prophylaxis trials
into management frameworks and infection prevention programs will
enhance their real-world utility.
In conclusion, the present meta-analysis indicated
that antifungal prophylaxis reduced the incidence risk of IFIs in
high-risk surgical ICU patients without increasing the risk of
severe adverse events. Although no survival benefit was
demonstrated, the prevention of fungal infections justifies the use
of antifungal prophylaxis in carefully selected patients.
Implementation should remain targeted, guided by local resistance
patterns and embedded within antifungal management programs to
balance efficacy with the long-term risk of resistance.
Supplementary Material
Search strategy.
Acknowledgements
The authors would like to thank Ms. Rahmati Putri
Yaniafari (Nanyang Technological University, Singapore) for
providing access to several literature sources for the present
review.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
INK conceived the present study, and was involved in
the methodology (designed the protocol and research plan),
systematic search, data curation and formal analysis, writing the
original draft and study supervision. MAE was involved in data
extraction, methodology (designed the protocol and research plan),
formal analysis and reviewing and editing the manuscript. ED was
involved in quality assessment, formal analysis and reviewing and
editing the manuscript. IR performed data extraction, statistical
analysis and visualization of the forest plots. AMW was involved in
the literature search, data curation and reviewing and editing the
manuscript. EBK performed formal analysis, study supervision, wrote
the original manuscript and critically revised the manuscript.
DDCHR performed statistical analysis, interpreted the results, and
reviewed and edited the manuscript. EA was involved in methodology
(designed the protocol and research plan), data authentication,
formal analysis, providing resources, writing the original
manuscript and revising the manuscript. PDE conceived the present
study, and was involved in the methodology, systematic search,
reviewing and editing the manuscript, as well as study supervision.
INK, MAE and EBK confirm the authenticity of all the raw data. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools (Superhuman Platform, Inc.) were used to improve
the readability and language of the manuscript.
References
|
1
|
Muskett H, Shahin J, Eyres G, Harvey S,
Rowan K and Harrison D: Risk factors for invasive fungal disease in
critically ill adult patients: A systematic review. Crit Care.
15(R287)2011.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Thomas-Rüddel DO, Schlattmann P, Pletz M,
Kurzai O and Bloos F: Risk factors for invasive Candida
infection in critically ill patients: A systematic review and
meta-analysis. Chest. 161:345–355. 2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kett DH, Azoulay E, Echeverria PM and
Vincent JL: Extended Prevalence of Infection in ICU Study (EPIC II)
Group of Investigators. Candida bloodstream infections in
intensive care units: Analysis of the extended prevalence of
infection in intensive care unit study. Crit Care Med. 39:665–670.
2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Vincent JL, Rello J, Marshall J, Silva E,
Anzueto A, Martin CD, Moreno R, Lipman J, Gomersall C, Sakr Y, et
al: International study of the prevalence and outcomes of infection
in intensive care units. JAMA. 302:2323–2329. 2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Maulana HR, Sumardi U and Koesoemadinata
RC: Invasive Candidiasis Prevalence Among Patients in Intensive
Care Unit, Hasan Sadikin Hospital. J Penyakit Dalam Indones.
6(3)2019.
|
|
6
|
Singh G, Pitoyo CW, Aditianingsih D and
Rumende CM: Profil pasien sakit kritis dengan penyakit jamur
invasif dini. Ina J CHEST Crit Emerg Med. 4:6–9. 2016.
|
|
7
|
Cataldo MA and Petrosillo N: Economic
considerations of antifungal prophylaxis in patients undergoing
surgical procedures. Ther Clin Risk Manag. 7:13–20. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Erb T, Mihai S, Strauß R, Herbst L,
Castellanos I, Diesch K, Cipa F, Bihlmaier K, Lang AK, Ganslmayer
M, et al: β-(1→3)-D-glucan- and mannan-guided early termination of
antifungal therapy in ICU patients: A randomized controlled study.
Antimicrob Agents Chemother. 67(e0072523)2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Knitsch W, Vincent JL, Utzolino S,
François B, Dinya T, Dimopoulos G, Özgüneş İ, Valía JC, Eggimann P,
León C, et al: A randomized, placebo-controlled trial of preemptive
antifungal therapy for the prevention of invasive candidiasis
following gastrointestinal surgery for intra-abdominal infections.
Clin Infect Dis. 61:1671–1678. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kadariswantiningsih IN, Empitu MA, Santosa
TI and Alimu Y: Antifungal resistance: Emerging mechanisms and
implications (review). Mol Med Rep. 32(247)2025.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Empitu MA, Kadariswantiningsih IN and
Shakri NM: Pharmacological strategies for targeting biofilms in
otorhinolaryngologic infections and overcoming antimicrobial
resistance (review). Biomed Rep. 22(95)2025.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Shorr AF, Chung K, Jackson WL, Waterman PE
and Kollef MH: Fluconazole prophylaxis in critically ill surgical
patients: A meta-analysis. Crit Care Med. 33:1928–1936.
2005.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372(n71)2021.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Jørgensen L, Paludan-Müller AS, Laursen
DRT, Savović J, Boutron I, Sterne JAC, Higgins JPT and Hróbjartsson
A: Evaluation of the Cochrane tool for assessing risk of bias in
randomized clinical trials: Overview of published comments and
analysis of user practice in Cochrane and non-Cochrane reviews.
Syst Rev. 5(80)2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
McGuinness LA and Higgins JPT:
Risk-of-bias VISualization (robvis): An R package and Shiny web app
for visualizing risk-of-bias assessments. Res Synth Methods.
12:55–61. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Eggimann P, Francioli P, Bille J,
Schneider R, Wu MM, Chapuis G, Chiolero R, Pannatier A, Schilling
J, Geroulanos S, et al: Fluconazole prophylaxis prevents
intra-abdominal candidiasis in high-risk surgical patients. Crit
Care Med. 27:1066–1072. 1999.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Garbino J, Lew DP, Romand JA, Hugonnet S,
Auckenthaler R and Pittet D: Prevention of severe Candida
infections in nonneutropenic, high-risk, critically ill patients: A
randomized, double-blind, placebo-controlled trial in patients
treated by selective digestive decontamination. Intensive Care Med.
28:1708–1717. 2002.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Giglio M, Caggiano G, Dalfino L, Brienza
N, Alicino I, Sgobio A, Favale A, Coretti C, Montagna MT, Bruno F
and Puntillo F: Oral nystatin prophylaxis in surgical/trauma ICU
patients: A randomised clinical trial. Critical Care.
16(R57)2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Hanson KE, Pfeiffer CD, Lease ED, Balch
AH, Zaas AK, Perfect JR and Alexander BD: β-D-glucan surveillance
with preemptive anidulafungin for invasive candidiasis in intensive
care unit patients: A randomized pilot study. PLoS One.
7(e42282)2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ostrosky-Zeichner L, Shoham S, Vazquez J,
Reboli A, Betts R, Barron MA, Schuster M, Judson MA, Revankar SG,
Caeiro JP, et al: MSG-01: A randomized, double-blind,
placebo-controlled trial of caspofungin prophylaxis followed by
preemptive therapy for invasive candidiasis in high-risk adults in
the critical care setting. Clin Infect Dis. 58:1219–1226.
2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Pelz RK, Hendrix CW, Swoboda SM,
Diener-West M, Merz WG, Hammond J and Lipsett PA: Double-blind
placebo-controlled trial of fluconazole to prevent Candidal
infections in critically ill surgical patients. Ann Surg.
233:542–548. 2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Quinio B, Albanèse J, Bues-Charbit M,
Viviand X and Martin C: Selective decontamination of the digestive
tract in multiple trauma patients. A prospective double-blind,
randomized, placebo-controlled study. Chest. 109:765–772.
1996.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sandven P, Qvist H, Skovlund E and
Giercksky KE: NORGAS Group and the Norwegian Yeast Study Group.
Significance of Candida recovered from intraoperative
specimens in patients with intra-abdominal perforations. Crit Care
Med. 30:541–547. 2002.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Savino JA, Agarwal N, Wry P, Policastro A,
Cerabona T and Austria L: Routine prophylactic antifungal agents
(clotrimazole, ketoconazole, and nystatin) in
nontransplant/nonburned critically ill surgical and trauma
patients. J Trauma. 36:20–26. 1994.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Slotman GJ, Burchard KW, D'arezzo A and
Gann DS: Ketoconazole prevents acute respiratory failure in
critically ill surgical patients. J Trauma. 28:648–654.
1988.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Playford EG, Webster AC, Sorrell TC and
Craig JC: Antifungal agents for preventing fungal infections in
non-neutropenic critically ill and surgical patients: Systematic
review and meta-analysis of randomized clinical trials. J
Antimicrob Chemother. 57:628–638. 2006.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang J, Zhou M, Xu JY, Zhou RF, Chen B and
Wan Y: Comparison of antifungal prophylaxis drugs in patients with
hematological disease or undergoing hematopoietic stem cell
transplantation: A systematic review and network meta-analysis.
JAMA Netw Open. 3(e2017652)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Vardakas KZ, Samonis G, Michalopoulos A,
Soteriades ES and Falagas ME: Antifungal prophylaxis with azoles in
high-risk, surgical intensive care unit patients: A meta-analysis
of randomized, placebo-controlled trials. Crit Care Med.
34:1216–1224. 2006.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Cornely OA, Sprute R, Bassetti M, Chen
SCA, Groll AH, Kurzai O, Lass-Flörl C, Ostrosky-Zeichner L,
Rautemaa-Richardson R, Revathi G, et al: Global guideline for the
diagnosis and management of candidiasis: an initiative of the ECMM
in cooperation with ISHAM and ASM. Lancet Infect Dis. 25:e280–e293.
2025.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Bohner F, Papp C and Gácser A: The effect
of antifungal resistance development on the virulence of
Candida species. FEMS Yeast Res. 22(foac019)2022.
|
|
31
|
Gow NAR, Johnson C, Berman J, Coste AT,
Cuomo CA, Perlin DS, Bicanic T, Harrison TS, Wiederhold N, Bromley
M, et al: The importance of antimicrobial resistance in medical
mycology. Nat Commun. 13(5352)2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Chatelon J, Cortegiani A, Hammad E, Cassir
N and Leone M: Choosing the right antifungal agent in ICU patients.
Adv Ther. 36:3308–3320. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Vianna CMDM, Mosegui GBG and Rodrigues
MPDS: Cost-effectiveness analysis and budgetary impact of
anidulafungin treatment for patients with candidemia and other
forms of invasive candidiasis in Brazil. Rev Inst Med Trop Sao
Paulo. 65(e9)2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Holzknecht BJ, Thorup J, Arendrup MC,
Andersen SE, Steensen M, Hesselfeldt P, Nielsen JM and Knudsen JD:
Decreasing candidaemia rate in abdominal surgery patients after
introduction of fluconazole prophylaxis*. Clin Microbiol
Infect. 17:1372–1380. 2011.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Girgin T, Sayur V, Güler E, Uç C, Göktepe
B, Ersin S, Uyar M and Sezer TÖ: Predictors of mortality in
surgical patients admitted to a tertiary intensive care unit. J
Clin Med. 14(6369)2025.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Ou L, Chen J, Hillman K, Flabouris A, Parr
M, Assareh H and Bellomo R: The impact of post-operative sepsis on
mortality after hospital discharge among elective surgical
patients: A population-based cohort study. Criti Care.
21(34)2017.PubMed/NCBI View Article : Google Scholar
|