Introduction
Type 2 diabetes mellitus (T2DM) is one of the
leading chronic diseases worldwide and continues to increase in
prevalence, particularly in low-income and minority populations. It
is estimated that over 37 million individuals in the United States
live with diabetes, of whom 90-95% have T2DM (1). Hispanic and Latino adults bear a
disproportionate share of this burden, often presenting with
earlier onset, higher rates of obesity and metabolic syndrome, and
poorer long-term outcomes compared with non-Hispanic White
populations (1,2). These disparities are influenced not
only by genetic predisposition but also by socioeconomic barriers,
healthcare access limitations, and cultural factors that affect
disease management and treatment adherence (3,4).
Achieving and maintaining adequate glycemic control
is fundamental to reducing the risk of both microvascular
complications (for example, nephropathy, retinopathy and
neuropathy) and macrovascular complications (for example,
cardiovascular disease and stroke) (5). Current guidelines from the American
Diabetes Association (ADA) and other international bodies recommend
a patient-centered, stepwise intensification of pharmacological
therapy, starting with metformin monotherapy and progressing to
dual or triple therapy, and eventually to insulin or more complex
combinations when needed (6). While
these guidelines provide a clear framework, real-world prescribing
patterns often diverge from this idealized stepwise approach, being
heavily influenced by patient-specific comorbidities, tolerability,
cultural factors that shape treatment decisions and adherence
(7), and systemic barriers to care.
These variations are particularly relevant in underserved Hispanic
communities, where evidence describing treatment pathways and their
real-world patterns and associated clinical profiles remains scarce
(8,9).
While glycated hemoglobin (HbA1c) is the most widely
accepted biomarker for assessing glycemic control (10), additional outcomes such as renal
function, measured by estimated glomerular filtration rate (eGFR)
and urine albumin-to-creatinine ratio (UACR), are essential to
understanding the broader impact of diabetes management (10,11).
This is particularly critical as diabetic kidney disease represents
one of the most severe and costly long-term complications of T2DM,
making renal status a key indicator of disease progression and
treatment context (12,13). Given that diabetic kidney disease is
one of the most severe and costly complications of T2DM, assessing
renal function parameters in relation to pharmacological strategies
provides descriptive insights into the clinical profiles observed
across treatment strategies (12,13).
Previous studies, such as pharmacist-led
interventions designed to improve adherence and
guideline-concordant care, have reported modest benefits in
glycemic control among Hispanic patients (8,9,14).
Building upon this, the present study addresses a critical gap by
providing a comprehensive, real-world evaluation of existing
pharmacological treatment patterns and their association with both
glycemic and renal function parameters. By characterizing
medication utilization in routine care, it was aimed to identify
key clinical factors associated with optimal glycemic control
within this high-risk, community-based clinical population
(15,16). By characterizing medication
utilization and identifying clinical factors associated with
optimal glycemic control in routine care, the present study
complements existing interventional studies and addresses an
important gap in the understanding of diabetes management within
this high-risk population.
Accordingly, the present study focuses exclusively
on Hispanic adults with T2DM attending a community-based primary
care clinic. The current objectives were threefold: i) to describe
real-world pharmacological treatment patterns in this population;
ii) to compare glycemic and renal profiles across different
pharmacological regimens as observed in real-world practice; and
iii) to identify demographic, clinical and therapeutic factors
independently associated with optimal glycemic control.
Materials and methods
Study design and setting
This observational, cross-sectional study analyzed
clinical and demographic data retrospectively extracted from
electronic medical records (EMR) of adult patients attending a
community-based primary care clinic (Caridad Center, Palm Beach,
Florida, USA) between 2022 and 2024. In addition, to contextualize
the study period, key descriptive indicators were summarized by
calendar year (2022, 2023, and 2024).
Participants and inclusion
criteria
Eligible participants were adults (≥18 years) who
received at least two medical visits at the clinic during the study
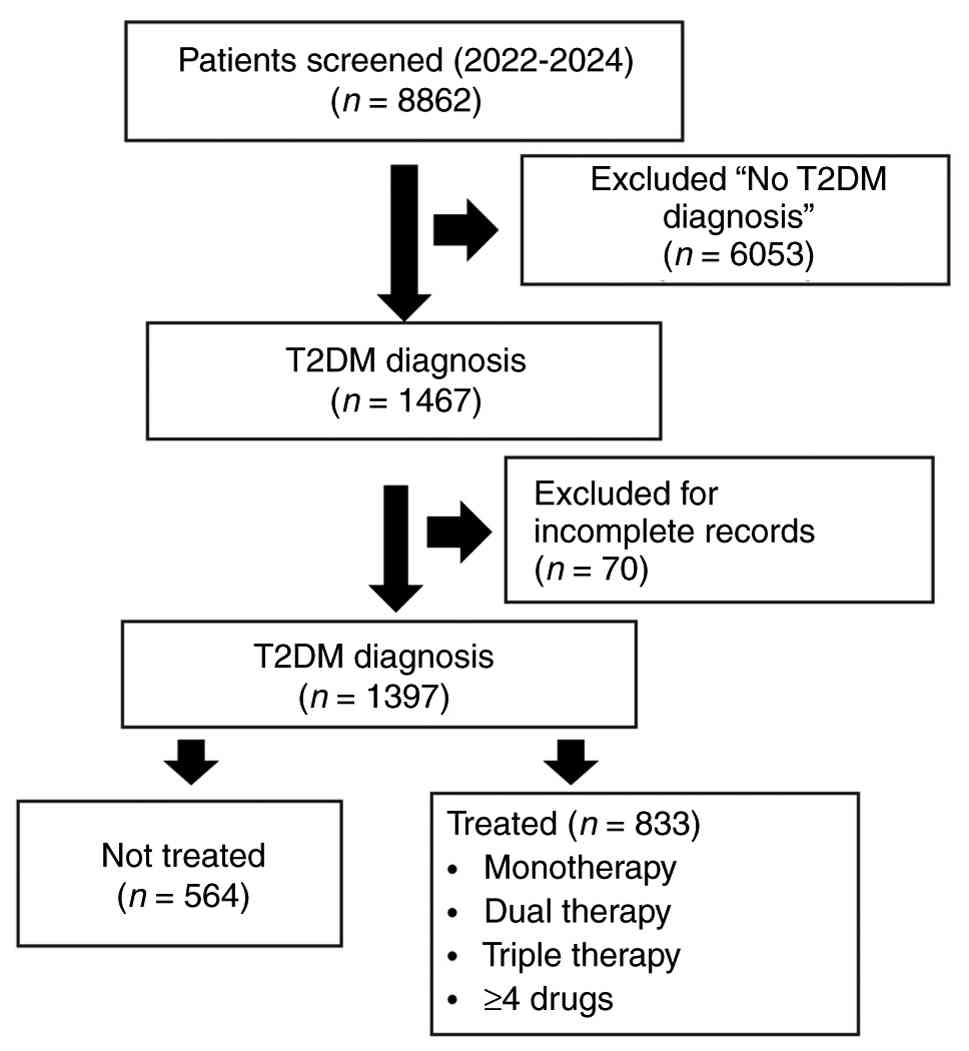
period. During the study period, 8,862 adult patients were
identified in the EMR. Of these, 1,397 met the criterion of having
a confirmed diagnosis of T2DM, resulting in a final analytical
sample of not-treated (n=564) and treated (n=833) (Fig. 1). For this analysis, only patients
with a confirmed diagnosis of T2DM were included (n=1,397).
Diagnosis was confirmed based on laboratory evidence (for example,
HbA1c ≥6.5% or fasting plasma glucose ≥126 mg/dl) consistent with
ADA criteria (3,17-19).
Patients with incomplete records, defined as missing essential
clinical or biochemical data, were excluded. Data quality was
independently validated by two clinically trained researchers, and
records with inconsistent or implausible values were systematically
removed to ensure accuracy and reliability. Importantly, clinical
staff at the clinic, who were not part of the research team,
conducted the initial de-identification of EMR prior to sharing
data for analysis, thereby ensuring blinding of the
investigators.
Data collection
Clinical, demographic and biochemical data were
systematically collected from the EMR. Demographic and
socioeconomic variables included age, sex, education level,
employment status, household composition and lifestyle behaviors
(alcohol and tobacco use). Clinical variables focused on
comorbidities, including hypertension, obesity, chronic kidney
disease (CKD), hyperlipidemia, cardiovascular disease,
depression/anxiety, and liver disease, which were used as
adjustment covariates in regression models (20,21).
Biochemical markers included glycated hemoglobin (HbA1c) to
evaluate glycemic control, and renal function parameters, estimated
glomerular filtration rate (eGFR) and UACR (22). Data on duration of diabetes and
medication adherence were not consistently available and therefore
not analyzed. Healthcare utilization during the study period
(2022-2024) was recorded, including primary care visits, specialist
consultations, diabetes education sessions, mental health visits,
and case management encounters. These measures were primarily used
as covariates for adjustment in multivariable analyses.
Medication use and treatment
regimens
Medication data included prescribed glucose-lowering
agents: Insulin, biguanides (metformin), sulfonylureas,
sodium-glucose cotransporter-2 (SGLT2) inhibitors, glucagon-like
peptide-1 (GLP-1) receptor agonists (RAs), and dipeptidyl
peptidase-4 (DPP-4) inhibitors. Mineralocorticoid receptor
antagonists (MRAs) were also captured as a medication class
documented in the EMR and reported descriptively. Patients were
categorized by treatment regimen complexity into monotherapy, dual
therapy, triple therapy, or regimens with ≥4 medications, to assess
treatment intensity (23-26).
Fixed-dose combinations were counted according to the number of
active agents contained. Medication doses and adherence data were
not available.
Medication exposure was defined cross-sectionally
based on active prescriptions recorded during the full 2022-2024
period; therefore, year-stratified medication patterns are not
emphasized because prescriptions may span multiple calendar years
and do not capture initiation, discontinuation, or sequencing.
Information on treatment duration, sequencing of
therapies, prior medication changes, dosage adjustments, and
medication adherence was not consistently available in the EMR and
therefore could not be analyzed. As a result, medication classes
were treated as static exposure variables, and analyses stratified
by medication type reflect cross-sectional associations rather than
longitudinal treatment effects. Therefore, medication-related
findings should not be interpreted as evidence of therapeutic
effectiveness.
Study outcomes
The primary outcome was optimal glycemic control,
defined as an HbA1c ≤7% (27). For
patients with more than one HbA1c measurement during the study
period, the lowest recorded HbA1c value was selected to represent
their best-achieved glycemic status. HbA1c measurements per patient
during the study period were also counted. This approach was chosen
to descriptively capture the best glycemic value recorded during
the study period under routine clinical care. Secondary outcomes
included: i) Renal function parameters: Best-achieved eGFR and
UACR; ii) treatment regimen comparisons: Glycemic control rates
stratified by regimen complexity (monotherapy, dual, triple, ≥4
agents).
Covariates
Covariates included age, sex, tobacco and alcohol
use, employment status, comorbidities (obesity, hyperlipidemia,
hypertension and CKD), healthcare utilization variables, and use of
major medication classes (insulin, sulfonylureas, biguanides, GLP-1
RAs and SGLT2 inhibitors).
Statistical analysis
Descriptive statistics summarized demographic,
clinical, biochemical and treatment characteristics. Comparative
analyses were conducted using independent t-tests for continuous
variables and chi-square tests for categorical variables. Pairwise
comparisons of glycemic control rates across treatment regimens
were performed using Fisher's exact test (28). A multivariable logistic regression
model was used to identify independent factors associated with
optimal glycemic control (HbA1c ≤7%). Covariates included age, sex,
tobacco and alcohol use, employment status, comorbidities (obesity,
hyperlipidemia, hypertension and CKD), healthcare utilization
variables, and use of major medication classes (insulin,
sulfonylureas, biguanides, GLP-1 RAs and SGLT2 inhibitors). Results
were reported as adjusted odds ratios (aOR) with 95% confidence
intervals (CI). Variable selection followed a backward stepwise
approach (entry criterion P≤0.05; removal criterion P≥0.10).
Statistically significant difference was set at P<0.05
(two-tailed). Multivariable logistic regression models used a
complete-case approach (listwise deletion), and no imputation was
performed. Analyses were conducted using SPSS software (version 26;
IBM Corp.) (29).
Ethical approval
The present study was approved by the Florida
International University Institutional Review Board (approval no.
IRB-24-0488; Miami, USA). The requirement for individual informed
consent was waived due to the retrospective nature of the analysis.
All procedures complied with ethical standards for research
involving human subjects and ensured strict confidentiality of
patient data. The present study was not preregistered; analyses
should be interpreted as descriptive and exploratory.
Results
General characteristics of patients
with type 2 diabetes and comparison between medicated and
non-medicated
The study population included a total of 1,397
participants with type 2 diabetes. Demographic, clinical and
healthcare utilization characteristics among patients with type 2
diabetes (T2DM) according to their use of glucose-lowering
medications are presented in Table
I. Patients receiving pharmacological treatment were
significantly older and had a higher prevalence of comorbidities,
including CKD, hypertension, hyperlipidemia and obesity, compared
with non-medicated patients (all P<0.05). Furthermore, medicated
patients exhibited worse glycemic control, as reflected by higher
mean HbA1c levels, and lower renal function, indicated by reduced
eGFR values (both P<0.001). Healthcare utilization was
significantly greater among treated patients, who had more primary
care appointments, specialist visits, case management encounters,
mental health visits, and health education sessions. Although the
proportion of females was slightly lower in the medicated group
(59.5%) than in the non-medicated group (64.2%), this difference
was not statistically significant (P=0.083). During 2022-2024, the
number of HbA1c measurements per patient had a median of 2.0 (IQR
2), with minimum and maximum values of 1 and 13, respectively.
 | Table ICharacteristics of diabetic patients
according to use of glucose-lowering medication. |
Table I
Characteristics of diabetic patients
according to use of glucose-lowering medication.
| Clinicopathological
characteristics | No medication
(n=564) | Medication
(n=833) | P-value |
|---|
| Demographic
characteristics | | | |
|
Age,
yearsa | 50.17±14.41 | 53.08±11.30 | <0.001 |
|
Femaleb | 64.2% | 59.5% | 0.083 |
|
Hispanicb | 100% | 100% | |
|
High School
or Lessb | 86.5% | 83.6% | 0.644 |
| Employment
statusb | | | |
|
Unemployedb | 38.0% | 44.6% | 0.021 |
| Lifestyle
characteristics (%) | | | |
|
Alcohol
useb | 21.3% | 24.9% | 0.238 |
|
Tobaccob | 9.1% | 16.3% | 0.073 |
| Chronic
comorbiditiesb | | | |
|
Chronic
kidney disease b | 6.0% | 15.2% | <0.001 |
|
Hypertensionb | 38.1% | 56.5% | <0.001 |
|
Hyperlipidemiab | 51.4% | 66.7% | <0.001 |
|
Liver
diseaseb | 13.3% | 16.9% | 0.070 |
|
Cardiovascular
diseaseb | 2.7% | 4.1% | 0.183 |
|
Obesityb | 28.7% | 34.5% | 0.027 |
|
Depression/anxietyb | 11.7% | 14.8% | 0.111 |
| Healthcare
utilizationa | | | |
|
Primary care
appointmentsa | 0.60±2.15 | 1.49±3.48 | <0.001 |
|
Specialist
doctora | 9.40±9.22 | 17.17±16.34 | <0.001 |
|
Case
managementa | 1.79±2.72 | 3.73±4.21 | <0.001 |
|
Mental
healtha | 1.54±6.11 | 3.10±10.99 | 0.002 |
|
Health
educationa | 0.37±0.95 | 0.82±1.59 | <0.001 |
| Biochemical
markers | | | |
|
HbA1ca | 6.07±1.11 | 7.06±1.62 | <0.001 |
|
Estimated
glomerular filtration rate (ml/min/1.73 m²)a | 102.45±20.77 | 96.92±24.42 | <0.001 |
| Urine
albumin-to-creatinine ratioa | 63.46±155.82 | 69.19±202.28 | 0.864 |
| Total
cholesterol-to-high-density lipoprotein cholesterol
ratioa | 3.71±1.14 | 3.35±1.04 | <0.001 |
| HbA1c
≤7%b | 91.4% | 65.4% | <0.001 |
Pharmacological treatment patterns in
treated type 2 diabetes patients
Medication use among the treated subgroup (n=833) of
patients with T2DM is shown in Table
II. Within these patients, metformin was the most prescribed
medication, used by 86.4% of patients, followed by insulin (25.5%),
GLP-1 RAs (24.1%) and SGLT2 inhibitors (19.3%). Sulfonylureas were
prescribed to 17.4% of patients treated, while MRAs and DPP-4
inhibitors were less frequently used, at 2.2 and 0.1%,
respectively. Regarding therapy regimens, just over 50% of the
patients were on monotherapy (51.4%), while 28.9 and 14.1% were on
dual and triple therapy, respectively. A smaller proportion (4.6%)
received four or more medications.
 | Table IIMedication use patterns among
patients with diabetes. |
Table II
Medication use patterns among
patients with diabetes.
| Medication
class | Percentage (%) |
|---|
| Biguanides
(Metformin) | 86.43 |
| Insulin | 25.45 |
| GLP-1 receptor
agonists | 24.13 |
| SGLT2
inhibitors | 19.33 |
| Sulfonylureas | 17.41 |
| Mineralocorticoid
receptor antagonists | 2.16 |
| DPP-4
inhibitors | 0.12 |
| Number of
medications | Percentage (%) |
| 1
(Monotherapy) | 51.38 |
| 2 (Dual
therapy) | 28.93 |
| 3 (Triple
therapy) | 14.05 |
| ≥4 | 4.56 |
Glycemic control according to
therapeutic regimen and medication class
In Table III, the
proportion of patients achieving optimal glycemic control (HbA1c
≤7%) according to medication class and regimen complexity is shown.
Among patients receiving monotherapy, the highest control rates
were observed for SGLT2 inhibitors (90.9%) or GLP-1 Ras (85.7%),
followed closely by biguanides (85.5%). Sulfonylureas showed
intermediate control rates, (62.5%), while insulin monotherapy
revealed notably lower rates of glycemic control (33.3%). Pairwise
comparisons of glycemic control among monotherapy regimens using
Fisher's exact test are demonstrated in Table IV. Higher proportions of glycemic
control were observed among patients receiving SGLT2 inhibitors or
GLP-1 RAs compared with those receiving insulin monotherapy;
however, these differences likely reflect variation in underlying
disease stage and treatment selection (P<0.001 for both
comparisons). Biguanides also showed a higher proportion of
glycemic control than insulin (P<0.001). Although sulfonylureas
showed intermediate glycemic control rates, differences compared
with other medications were not statistically significant,
highlighting intermediate control rates in monotherapy.
 | Table IIIPercentage of diabetic patients
achieving glycemic control (Hba1c ≤7%) by medication class and
regimen complexity. |
Table III
Percentage of diabetic patients
achieving glycemic control (Hba1c ≤7%) by medication class and
regimen complexity.
| | Percentage of
patients achieving glycemic control |
|---|
| | | In combination
with |
|---|
| Medication
class | Monotherapy | Dual therapy
Biguanides + | Dual therapy
Insulin + | Triple therapy
SGLT2 + Biguanides + |
|---|
| SGLT2
inhibitors | 90.90 | 61.90 | 25.00 | |
| Insulin | 33.30 | 12.80 | 33.30 | 50.00 |
| GLP-1 receptor
agonists | 85.70 | 76.70 | 66.70 | 88.90 |
| Sulfonylureas | 62.50 | 60.00 | - | 43.80 |
| Biguanides | 85.50 | 85.50 | 12.80 | - |
 | Table IVP-values from Fisher's Exact Test
comparing the proportion of patients achieving glycemic control
(HbA1c ≤7%) with monotherapy. |
Table IV
P-values from Fisher's Exact Test
comparing the proportion of patients achieving glycemic control
(HbA1c ≤7%) with monotherapy.
| Medication
class | SGLT2
inhibitors | Insulin | GLP-1 receptor
agonists | Sulfonylureas | Biguanides |
|---|
| SGLT2
inhibitors | - | 0.0027 | 1 | 0.2621 | 1 |
| Insulin | <0.001 | - | 0.0007 | 0.2191 | 0 |
| GLP-1 receptor
agonists | 1 | 0.0007 | - | 0.3045 | 1 |
| Sulfonylureas | 0.108 | 0.2191 | 0.3045 | - | 0.1042 |
| Biguanides | 1 | 0 | 1 | 0.1042 | - |
Differences in monotherapy by
comorbidity
Table V shows, for
each comorbidity, the proportion of patients with (Yes) or without
(No) the condition who receive monotherapy with each medication
class. Among patients with CKD, a significantly higher proportion
received SGLT2 inhibitors (20.5%) compared with those without CKD
(2.8%) (P<0.001). GLP-1 RAs are more frequently prescribed to
patients with obesity, with 13.1% of obese patients receiving this
therapy compared with 1.0% of non-obese patients (P<0.001).
Biguanide use is dominant among patients without CKD (85.7%) and
less common in those with CKD (38.5%) (P<0.001); similarly,
biguanides are more frequent in patients without hypertension than
in those with it. Sulfonylureas show no significant differences in
prescription rates between patients with or without the evaluated
comorbidities. Insulin monotherapy is characteristically prescribed
to patients with more complex clinical states, such as CKD and
possibly lower metabolic reserve, as suggested by the lower
prevalence of obesity among insulin users (1.4%) compared with
non-users (8.0%) (P=0.004). This is evidenced by the markedly
higher proportion of patients with CKD receiving insulin (25.6%)
vs. those without CKD (3.8%) (P<0.001). Additionally, insulin
use is significantly higher among unemployed patients (8.6%)
compared with employed ones (4.0%) (P=0.047), which may reflect
differences in clinical complexity, health care access, or other
unmeasured factors. These data suggest that prescribing patterns
reflect selective use of SGLT2 inhibitors and GLP-1 RAs in specific
clinical phenotypes (such as CKD and obesity) rather than uniform
superiority across disease stages, while insulin monotherapy is
commonly observed among patients with more advanced clinical
profiles of advanced diabetes, associated with more severe
comorbidities and complex patient profiles. Notably, patients with
regimens that include the use of insulin, either as monotherapy or
in combination with other medications, are those with the lowest
proportion of glycemic control (Table
III).
 | Table VProportion of patients receiving
monotherapy by medication class according to presence or absence of
comorbidities. |
Table V
Proportion of patients receiving
monotherapy by medication class according to presence or absence of
comorbidities.
| | | SGLT2 I | GLP-1 RA | Biguanides | Sulfonylureas | Insulin |
|---|
| Age | With | 57.5+12.0 | 51.6+9.0 | 52.4+11.8 | 53.8+12.2 | 54.6+15.7 |
| | Rest | 52.5+12.0 | 52.8+12.1 | 54.4+12.6 | 52.7+12.0 | 52.6+11.7 |
| | P | 0.077 | 0.705 | 0.178 | 0.742 | 0.424 |
| | | | | | | |
| Chronic kidney
disease | Yes | 20.5% | 7.7% | 38.5% | 7.7% | 25.6% |
| | No | 2.8% | 4.8% | 85.7% | 2.8% | 3.8% |
| | P | <0.001 | 0.442 | <0.001 | 0.112 | <0.001 |
| | | | | | | |
| HT | Yes | 7.0% | 6.0% | 75.3% | 4.7% | 7.0% |
| | No | 1.9% | 4.2% | 87.5% | 1.9% | 4.6% |
| | P | 0.010 | 0.976 | 0.001 | 0.102 | 0.298 |
| | | | | | | |
| Hyperlipidemia | Yes | 5.9% | 5.1% | 78.8% | 3.5% | 6.7% |
| | No | 2.3% | 5.1% | 85.2% | 2.8% | 4.5% |
| | P | 0.094 | 0.994 | 0.102 | 0.787 | 0.407 |
| | | | | | | |
| Obesity | Yes | 4.1% | 13.1% | 82.9% | 2.8% | 1.4% |
| | No | 4.5% | 1.0% | 78.6% | 3.5% | 8.0% |
| | P | 0.999 | <0.001 | 0.296 | 0.781 | 0.004 |
| | | | | | | |
| Unemployed | Yes | 3.1% | 1.9% | 81.5% | 3.1% | 8.6% |
| | No | 4.9% | 8.0% | 81.3% | 3.6% | 4.0% |
| | P | 0.427 | 0.011 | 0.999 | 0.999 | 0.047 |
Differences in glycemic control and
renal function across monotherapy regimens
HbA1c, eGFR and UACR among patients treated with
different monotherapy regimens are compared in Table VI. Significant differences in HbA1c
were observed, with the lowest mean values found in patients
treated with SGLT2 inhibitors (5.95%), GLP-1 RAs (6.28%), and
biguanides (6.38%). Patients treated with insulin (7.89%) and
sulfonylureas (7.92%) had significantly higher mean HbA1c levels,
indicating lower glycemic control (P<0.001 for comparisons). By
contrast, renal function parameters (eGFR and UACR) appeared
indicative of baseline patient characteristics rather than
therapeutic effects. Patients on insulin monotherapy had
significantly lower eGFR (67.7 ml/min/1.73 m²) compared with those
on biguanides (101.1 ml/min/1.73 m²; P<0.001) and GLP-1 RAs
(102.5 ml/min/1.73 m²; P<0.001). Patients receiving SGLT2
inhibitors showed the lowest eGFR (59.9 ml/min/1.73 m²), indicating
targeted prescription among patients with advanced renal
impairment. The UACR, reflecting renal damage, was significantly
elevated in patients treated with sulfonylureas (349.0 mg/g) and
insulin (148.6 mg/g), compared with notably lower values in those
receiving biguanides (30.6 mg/g; P<0.001), GLP-1 RAs (47.3
mg/g), and SGLT2 inhibitors (66.6 mg/g; P=0.047). In summary, it
can be stated that patients receiving insulin or sulfonylureas had
higher HbA1c levels and less favorable renal parameters; however,
these differences likely reflect more advanced disease stage and
selective prescribing rather than medication-related effects,
consistent with more advanced disease.
 | Table VIComparison of HbA1c, eGFR and UACR
values among patients treated with different monotherapy
regimens. |
Table VI
Comparison of HbA1c, eGFR and UACR
values among patients treated with different monotherapy
regimens.
| | | P-value
comparing |
|---|
| Parameter | | Mean ± SD | Sulfonylureas | Insulin | Biguanides | GLP-1 | SGLT2 |
|---|
| eGFR | Sulfonylureas | 90.22 | 39.66 | - | 0.131 | 0.999 | 0.999 | 0.039 |
| | Insulin | 67.66 | 43.36 | 0.131 | - | <0.001 | <0.001 | 0.999 |
| | Biguanides | 101.05 | 18.98 | 0.999 | <0.001 | - | 0.999 | <0.001 |
| | GLP-1RAs | 102.47 | 24.86 | 0.999 | <0.001 | 0.999 | - | - |
| | In SGLT2 | 59.90 | 34.98 | 0.039 | 0.999 | <0.001 | <0.001 | - |
| | All | 97.66 | 24.84 | 0.363 | <0.001 | <0.001 | 0.361 | <0.001 |
| UACR | Sulfonylureas | 349.00 | 412.07 | - | 0.116 | <0.001 | 0.095 | 0.095 |
| | Insulin | 148.60 | 324.62 | 0.116 | - | 0.107 | 0.999 | 0.999 |
| | Biguanides | 30.64 | 70.58 | <0.001 | 0.107 | - | 0.999 | 0.999 |
| | GLP-1RAs | 47.33 | 71.61 | 0.095 | 0.999 | 0.999 | - | - |
| | In SGLT2 | 66.60 | 84.68 | 0.047 | 0.999 | 0.999 | 0.999 | - |
| | All | 65.86 | 177.18 | <0.001 | 0.051 | <0.001 | 0.855 | 0.992 |
| HbGlu | Sulfonylureas | 7.92 | 2.25 | - | 0.999 | 0.001 | 0.004 | 0.002 |
| | Insulin | 7.89 | 1.67 | 0.999 | - | <0.001 | <0.001 | <0.001 |
| | Biguanides | 6.38 | 1.09 | <0.001 | <0.001 | - | 0.999 | <0.001 |
| | GLP-1RAs | 6.28 | 1.34 | 0.004 | <0.001 | 0.999 | - | - |
| | In SGLT2 | 5.95 | 1.14 | 0.002 | <0.001 | 0.999 | 0.999 | - |
| | All | 6.50 | 1.27 | <0.001 | <0.001 | <0.001 | 0.398 | 0.143 |
Glycemic control and renal function
parameters in dual and triple diabetes therapy regimens
Glycemic control, eGFR and UACR values among
patients on dual and triple diabetes medication regimens are
compared in Table VII. In dual
therapy, glycemic control was significantly more favorable in
regimens containing GLP-1 RAs, with a mean HbA1c of 6.68%, compared
with those without GLP-1 RAs (7.92%; P<0.001). Dual therapy
involving insulin was associated with significantly poorer glycemic
control (HbA1c, 8.51%) compared with those without insulin (7.07%;
P<0.001). Dual regimens including sulfonylureas did not show
significant glycemic advantages (7.41 vs. 7.58%; P=0.549). Triple
therapy regimens including sulfonylureas demonstrated significantly
poorer glycemic outcomes (HbA1c, 8.12%) compared with regimens
without sulfonylureas (7.50%; P=0.035). Although triple therapies
involving GLP-1 RAs had slightly improved glycemic control (7.55%)
compared with regimens without GLP-1 RAs (7.97%), this difference
was not statistically significant (P=0.138). Regarding renal
function parameters, patients receiving dual therapy with
biguanides had significantly improved eGFR (102.0 ml/min/1.73 m²
vs. 79.2 ml/min/1.73 m² without biguanides; P<0.001). Similarly,
dual therapy involving GLP-1 RAs demonstrated significantly
improved eGFR (104.5 ml/min/1.73 m² vs. 95.6 ml/min/1.73 m² without
GLP-1; P=0.018). Patients treated with dual therapies containing
SGLT2 inhibitors had significantly lower mean eGFR (81.4
ml/min/1.73 m² vs. 102.9 ml/min/1.73 m²; P<0.001), reflecting
their targeted use among patients with impaired renal function. The
UACR was significantly lower in dual therapies with GLP-1 RAs (24.1
mg/g vs. 95.2 mg/g; P=0.023) and biguanides (31.4 mg/g vs. 267.8
mg/g; P<0.001), showing favorable renal parameter profiles in
this cross-sectional comparison. In triple therapy, higher UACR
values among patients on SGLT2 inhibitors (88.7 mg/g vs. 32.0 mg/g
without SGLT2 inhibitors; P=0.044) likely reflect selective
prescribing for those with advanced renal disease.
 | Table VIIComparison of eGFR, UACR and HbA1c
levels among patients on dual and triple diabetes medication
regimens. |
Table VII
Comparison of eGFR, UACR and HbA1c
levels among patients on dual and triple diabetes medication
regimens.
| | | Dual Therapy
Including | Triple Therapy
Including |
|---|
| Parameter | With | No | Yes | P-value | No | Yes | P-value |
|---|
| eGFR | Sulfonylureas | 99.02±23.58 | 99.25±20.61 | 0.948 | 91.01±25.98 | 95.72±22.98 | 0.290 |
| | Insulin | 100.90±18.25 | 95.36±29.60 | 0.096 | 90.56±26.43 | 93.39±24.54 | 0.528 |
| | Biguanides | 79.18±37.24 | 102.01±18.05 | <0.001 | 61.50±24.15 | 94.28±24.03 | 0.005 |
| | GLP-1RAs | 95.59±25.34 | 104.47±14.19 | 0.018 | 87.61±27.35 | 95.16±23.50 | 0.080 |
| | In SGLT2 | 102.86±17.56 | 81.37±33.56 | <0.001 | 101.92±16.92 | 87.56±27.25 | 0.001 |
| UACR | Sulfonylureas | 49.88±110.02 | 41.96±103.01 | 0.712 | 73.80±156.50 | 55.64±163.10 | 0.566 |
| | Insulin | 35.82±81.95 | 66.67±137.48 | 0.089 | 43.92±79.67 | 79.82±180.64 | 0.236 |
| | Biguanides | 134.45±211.36 | 31.41±62.47 | <0.001 | 184.28±192.18 | 62.49±154.00 | 0.047 |
| | GLP-1RAs | 61.52±126.08 | 24.09±58.65 | 0.043 | 85.13±193.04 | 60.82±136.92 | 0.408 |
| | In SGLT2 | 27.84±58.51 | 139.00±199.76 | <0.001 | 31.97±117.37 | 86.36±169.99 | 0.047 |
| HbGlu | Sulfonylureas | 7.58±1.85 | 7.41±1.69 | 0.549 | 7.50±1.52 | 8.12±1.90 | 0.035 |
| | Insulin | 7.07±1.54 | 8.51±1.93 | <0.001 | 7.54±1.78 | 7.77±1.63 | 0.441 |
| | Biguanides | 7.52±1.53 | 7.53±1.84 | 0.984 | 6.50±0.78 | 7.77±1.69 | 0.037 |
| | GLP-1RAs | 7.92±1.89 | 6.68±1.22 | <0.001 | 7.97±2.00 | 7.55±1.46 | 0.138 |
| | In SGLT2 | 7.55±1.84 | 7.44±1.63 | 0.735 | 7.98±1.52 | 7.55±1.74 | 0.140 |
Factors associated with optimal
glycemic control (HbA1c ≤7%)
Factors significantly associated with optimal
glycemic control (defined as HbA1c ≤7%) among patients with
diabetes are presented in Table
VIII. In the multivariable logistic regression model, female
sex (aOR=3.246, 95% CI: 1.323-7.964; P=0.010) and frequent
specialist visits (more than nine visits, aOR=8.267, 95% CI:
2.344-29.164; P=0.001) were significantly associated with better
glycemic control. By contrast, insulin use (aOR=0.018, 95% CI:
0.005-0.060; P<0.001) and sulfonylurea use (aOR=0.072, 95% CI:
0.019-0.268; P<0.001) were strongly associated with poorer
glycemic control. These results suggest that insulin and
sulfonylureas are predominantly used among patients with more
advanced disease, greater therapeutic complexity, or non-optimal
therapeutic regimen, reflecting clinical challenges in achieving
optimal glucose control. Meanwhile, increased specialist care and
certain demographic factors (such as female sex) were important
factors associated with more favorable glycemic management.
Overall, these findings underscore the importance of targeted
healthcare resource utilization and careful medication selection in
optimizing glycemic outcomes among diabetic patients.
 | Table VIIIFactors associated with optimal
glycemic control (HbA1c≤7%) among patients with diabetes. |
Table VIII
Factors associated with optimal
glycemic control (HbA1c≤7%) among patients with diabetes.
| | Univariate
model | Multivariable
model |
|---|
| | | 95% C.I. | | | 95% C.I. | |
|---|
| | OR | Low | Upper. | Sig | aOR | Low | Upper | Sig |
|---|
| Age >52 | 1.042 | 0.797 | 1.364 | 0.762 | | | | |
| Female | 1.351 | 1.029 | 1.775 | 0.030 | 3.246 | 1.323 | 7.964 | 0.010 |
| Tobacco use | 0.423 | 0.210 | 0.852 | 0.016 | | | | |
| Alcohol use | 1.155 | 0.759 | 1.756 | 0.502 | | | | |
| Unemployed | 0.748 | 0.562 | 0.997 | 0.047 | | | | |
|
Obesity/overweight | 1.582 | 1.180 | 2.120 | 0.002 | | | | |
| Hyperlipidemia | 1.063 | 0.801 | 1.412 | 0.671 | | | | |
| Hypertension | 0.833 | 0.637 | 1.090 | 0.184 | | | | |
| Chronic kidney
disease | 0.652 | 0.447 | 0.950 | 0.026 | | | | |
| Primary care
appointments | 0.341 | 0.257 | 0.452 | 0.000 | | | | |
| Specialist
appointments (>9 visits) | 1.262 | 0.961 | 1.659 | 0.095 | 8.267 | 2.344 | 29.164 | 0.001 |
| Case management
visits | 0.643 | 0.485 | 0.854 | 0.002 | | | | |
| Mental health
appointments | 0.922 | 0.678 | 1.254 | 0.607 | | | | |
| Health education
sessions | 0.799 | 0.604 | 1.056 | 0.115 | | | | |
| Sulfonylurea
use | 0.274 | 0.186 | 0.403 | 0.000 | 0.072 | 0.019 | 0.268 | <0.001 |
| Biguanide use | 0.347 | 0.258 | 0.465 | 0.000 | | | | |
| SGLT2 inhibitor
use | 0.334 | 0.234 | 0.476 | 0.000 | | | | |
| Insulin use | 0.102 | 0.073 | 0.144 | 0.000 | 0.018 | 0.005 | 0.060 | <0.001 |
| GLP-1 receptor
agonist use | 0.481 | 0.345 | 0.670 | 0.000 | | | | |
Calendar-year descriptive patterns
(2022-2024)
Of the 1,397 patients included in the analysis, a
total of 4,099 HbA1c laboratory determinations were recorded during
the 2022-2024 study period. The annual distribution was 1,202
measurements in 2022 (29.3%), 1,347 in 2023 (32.9%), and 1,550 in
2024 (37.8%), demonstrating a progressive increase in the absolute
number of HbA1c assessments over time.
Regarding cumulative measurement frequency per
patient across the entire study period, 138 patients (9.9%) had one
determination, 564 (40.4%) had two, 327 (23.4%) had three, and 368
(26.3%) had four or more measurements.
Overall, these descriptive findings indicate an
increase in outpatient laboratory monitoring activity throughout
the study period. Although this pattern is consistent with the
gradual normalization of healthcare delivery following the acute
phase of the COVID-19 pandemic, no formal temporal trend analysis
was conducted; therefore, this interpretation should be considered
exploratory rather than causal evidence of post-pandemic recovery
or the absence of temporal sampling bias. During 2022-2024, the
number of HbA1c measurements per patient had a median of 2.0 (IQR
2), with minimum and maximum values of 1 and 13, respectively.
Association between glucose-lowering
medication type and healthcare utilization
Healthcare utilization patterns varied notably
according to the type of pharmacological treatment received by
patients with T2DM. Those treated with insulin, SGLT2 inhibitors,
or GLP-1 RAs exhibited significantly higher use of healthcare
services, including primary care, specialist consultations, case
management encounters and health education sessions (all
P<0.001) (Table IX). Insulin
users, in particular, showed the highest engagement in specialist
visits (28.21±21.51) and case management services (6.61±5.45),
reflecting insulin's typical use in patients with greater clinical
complexity who require closer follow-up. Similarly, patients
treated with SGLT2 inhibitors or GLP-1 RAs demonstrated substantial
healthcare resource use, which may reflect differences in clinical
complexity and prescribing patterns and the relatively recent
introduction of these therapies, which may necessitate more
intensive monitoring. Notably, mental health service utilization
was also higher among patients treated with insulin (P=0.005) and
GLP-1 RAs (P<0.001), possibly reflecting the psychosocial burden
associated with complex treatment regimens or disease severity. By
contrast, sulfonylureas and biguanides were associated with lower
use of specialist services and case management, suggesting their
preferential use in patients with less complicated disease profiles
or those with stable glycemic control. Particularly, metformin
users had significantly fewer specialist visits (P<0.001),
consistent with their common use as first-line therapy in routine
clinical practice (Table IX).
 | Table IXDifferences in healthcare utilization
by medication type among patients with diabetes receiving
pharmacological treatment. |
Table IX
Differences in healthcare utilization
by medication type among patients with diabetes receiving
pharmacological treatment.
| Medication
type | Service
utilization | Not receiving
medication | Receiving
medication | P-value |
|---|
| SGLT2
inhibitors | Primary care
visits | 1.25±3.12 | 2.48±4.58 | <0.001 |
| | Specialist
visits | 14.69±13.92 | 27.55±21.02 | <0.001 |
| | Case management
visits | 3.13±3.72 | 6.30±5.11 | <0.001 |
| | Mental health
visits | 2.83±10.64 | 4.26±12.31 | 0.138 |
| | Health education
sessions | 0.64±1.42 | 1.58±2.01 | <0.001 |
| Insulin | Primary care
visits | 1.14±2.97 | 2.51±4.53 | <0.001 |
| | Specialist
visits | 13.41±12.04 | 28.21±21.51 | <0.001 |
| | Case management
visits | 2.76±3.15 | 6.61±5.45 | <0.001 |
| | Mental health
visits | 2.48±10.03 | 4.93±13.29 | 0.005 |
| | Health education
sessions | 0.56±1.26 | 1.61±2.12 | <0.001 |
| GLP-1 receptor
agonists | Primary care
visits | 1.19±2.84 | 2.45±4.88 | <0.001 |
| | Specialist
visits | 15.19±15.27 | 23.44±17.97 | <0.001 |
| | Case management
visits | 3.15±3.96 | 5.60±4.45 | <0.001 |
| | Mental health
visits | 2.35±8.73 | 5.47±15.97 | <0.001 |
| | Health education
sessions | 0.63±1.38 | 1.44±2.02 | <0.001 |
| Sulfonylureas | Primary care
visits | 1.51±3.55 | 1.40±3.12 | 0.729 |
| | Specialist
visits | 17.72±16.91 | 14.62±13.07 | 0.038 |
| | Case management
visits | 3.94±4.38 | 2.78±3.14 | 0.002 |
| | Mental health
visits | 3.24±11.40 | 2.48±8.80 | 0.448 |
| | Health education
sessions | 0.90±1.69 | 0.47±0.94 | 0.003 |
| Biguanides
(Metformin) | Primary care
visits | 1.93±4.55 | 1.42±3.28 | 0.150 |
| | Specialist
visits | 26.59±21.45 | 15.70±14.87 | <0.001 |
| | Case management
visits | 5.59±5.45 | 3.45±3.91 | <0.001 |
| | Mental health
visits | 5.70±15.72 | 2.70±10.01 | 0.007 |
| | Health education
sessions | 0.99±1.64 | 0.80±1.58 | 0.232 |
Factors influencing diabetes
medication prescription
Factors independently associated with the
prescription of specific diabetes medications revealed distinct
clinical and healthcare engagement patterns (Table X). In the adjusted multivariable
logistic regression analysis, insulin use was significantly linked
to the presence of hypertension (aOR=2.94; 95% CI: 1.29-6.69;
P=0.010) and attendance at diabetes education sessions (aOR=4.89;
95% CI: 2.30-10.41; P<0.001). Conversely, insulin was less
likely to be prescribed in combination with sulfonylureas
(aOR=0.30; 95% CI: 0.10-0.87; P=0.027), likely reflecting
prescribing practices that aim to reduce therapeutic complexity and
the risk of hypoglycemia. The use of SGLT2 inhibitors was strongly
associated with hypertension (aOR=4.18; 95% CI: 1.53-11.47;
P=0.005), suggesting this is consistent with selective prescribing
patterns for patients with increased cardiovascular or renal risk.
Prescription of GLP-1 RAs was significantly associated with
overweight or obesity (aOR=2.55; 95% CI: 1.30-5.00; P=0.006) and
participation in diabetes education programs (aOR=3.72; 95% CI:
1.84-7.52; P<0.001), consistent with their known benefits in
weight management and the role of patient engagement in complex
therapy adherence. By contrast, biguanides (metformin) were
significantly less likely to be prescribed in patients with
impaired renal function (aOR=0.34; 95% CI: 0.13-0.84; P=0.019), in
line with clinical guidelines recommending caution due to the risk
of lactic acidosis in this population.
 | Table XFactors associated with receiving
specific diabetes medications. |
Table X
Factors associated with receiving
specific diabetes medications.
| Medication
class | Associated
factor | Adjusted OR | 95% CI | P-value |
|---|
| Insulin | Hypertension | 2.941 | 1.292-6.693 | 0.010 |
| | ≥1 Diabetes
education visit | 4.893 | 2.301-10.407 | <0.001 |
| | Concomitant
sulfonylurea use | 0.297 | 0.102-0.871 | 0.027 |
| SGLT2
inhibitors | Hypertension | 4.184 | 1.527-11.465 | 0.005 |
| GLP-1 receptor
agonists | Obesity or
overweight | 2.554 | 1.304-5.003 | 0.006 |
| | ≥1 Diabetes
education visit | 3.722 | 1.843-7.515 | <0.001 |
| Biguanides
(Metformin) | Chronic kidney
disease or impaired eGFR | 0.335 | 0.134-0.836 | 0.019 |
These findings emphasize that prescription decisions
for diabetes medications are associated not only with comorbid
conditions such as hypertension, renal function and obesity but
also by healthcare engagement factors such as diabetes education
attendance. The observed patterns reflect a personalized approach
in pharmacological management, in which medication selection varies
with efficacy, safety and complexity according to each patient's
clinical profile (Table X).
Discussion
The present real-world analysis of 1,397 Hispanic
adults with T2DM provides critical insights into how
pharmacological therapies are utilized in a community setting,
revealing that prescription patterns are driven more by clinical
complexity and comorbidities, and other contextual factors
affecting treatment decisions and adherence than by a standardized,
stepwise approach. The findings of the present study contribute to
the growing body of evidence on diabetes management in minority
populations, while highlighting the importance of tailoring
therapeutic strategies to clinical complexity and healthcare
engagement.
Nearly 40% of diagnosed patients were not receiving
glucose-lowering medication at the time of data extraction. Whether
this reflects early-stage disease managed through lifestyle, loss
to follow-up, clinical inertia, or access barriers remains unclear.
As expected, metformin was the most commonly prescribed drug,
consistent with current international guidelines recommending it as
first-line therapy (19). Notably,
the relatively high use of insulin (25.5%), GLP-1 RAs (24.1%), and
SGLT2 inhibitors (19.3%) indicates increasing adoption of newer
agents in this population. However, the low use of DPP-4 inhibitors
and mineralocorticoid receptor antagonists reflects either
prescribing preferences, formulary restrictions, or access
barriers. The low utilization of DPP-4 inhibitors, for instance,
may reflect their modest glycemic efficacy compared with the robust
benefits of GLP-1 RAs and SGLT2 inhibitors, particularly regarding
weight and cardiorenal protection.
The data of the present study show that patients
receiving SGLT2 inhibitors, GLP-1 RAs, or metformin as monotherapy
were disproportionately represented among those with optimal
glycemic profiles, with >85% of patients achieving the HbA1c
goal of ≤7%. By contrast, insulin and sulfonylureas were associated
with poor glycemic outcomes, both as monotherapy and in combination
regimens. While this may reflect intrinsic limitations of these
therapies (30,31), it likely indicates confounding by
indication, namely, that these drugs are preferentially prescribed
to patients with advanced or more difficult-to-control diabetes
(32,33). This phenomenon, known as confounding
by indication, is critical for interpreting real-world
observational data. It means that the poor outcomes associated with
insulin and sulfonylureas do not necessarily reflect drug
ineffectiveness but rather serve as a marker for patients with a
longer disease duration, greater beta-cell failure, and higher
comorbidity burden, for whom these therapies are appropriately
reserved (32-34).
Although not directly modeled, descriptive
comparisons suggest that individuals requiring dual or triple
therapy tended to have poorer glycemic outcomes compared with those
on monotherapy, reinforcing that regimen complexity likely reflects
underlying disease severity rather than treatment failure. Within
dual therapy, combinations involving GLP-1 RAs and metformin were
more frequently observed among patients achieving glycemic targets;
however, this pattern likely reflects underlying differences in
disease stage and treatment selection rather than comparative
treatment effectiveness. In comparison, combinations including
insulin or sulfonylureas consistently yielded poorer glycemic
results (35,36).
Renal function parameters provided additional
insights into prescribing patterns. Patients receiving SGLT2
inhibitors had the lowest eGFR, consistent with selective
prescribing among individuals with established renal impairment.
Importantly, patients on biguanides or GLP-1 RAs exhibited more
favorable renal profiles (higher eGFR and lower UACR), in line with
evidence of nephroprotective effects of these agents. Conversely,
insulin and sulfonylurea users had worse renal parameters, again
reflecting their use in more complex or advanced disease. These
findings reinforce the clinical importance of considering renal
status in treatment selection and are consistent with existing
clinical trial evidence supporting the disease-modifying effects of
GLP-1 RAs and SGLT2 inhibitors (37,38).
Our multivariable analysis identified female sex and
frequent specialist visits as independent factors associated with
improved glycemic control. The association with sex may reflect sex
differences in health-seeking behavior, medication adherence, or
lifestyle practices, although further research is warranted
(39). The association between
specialist visits and improved glycemic control likely reflects
differences in healthcare access, monitoring intensity, disease
complexity, or patient engagement rather than a direct causal
effect of specialist care itself (40-42).
In addition, insulin and sulfonylureas users exhibited poorer
glycemic outcomes, likely reflecting more advanced or complex
disease status (43).
Healthcare utilization patterns varied by medication
class: Patients treated with insulin, SGLT2 inhibitors, or GLP-1
RAs had substantially higher healthcare engagement, including
specialist care and diabetes education (44). This may reflect that complex or
advanced cases are associated with greater resource allocation, but
also that proactive disease management, including structured
education, may be associated with the adoption of newer agents.
Importantly, prescription decisions were strongly associated with
comorbidities: Insulin and SGLT2 inhibitors were associated with
hypertension, GLP-1 RAs with obesity, and lower metformin use with
impaired renal function (45,46).
These findings are consistent with guideline-concordant,
individualized care, balancing efficacy, comorbidity profiles and
safety (47).
Taken together, the results of the present study
highlight critical opportunities for improving diabetes care among
Hispanic adults. First, the absence of pharmacotherapy in nearly
40% of patients may reflect lifestyle management, access barriers,
loss to follow-up, or early disease stage, although this cannot be
determined from available data (48-50).
Second, the observed glycemic and renal profiles among patients
receiving GLP-1 RAs and SGLT2 inhibitors, in conjunction with
established evidence from randomized trials, underscore the
importance of ensuring equitable access to these agents,
particularly given their proven cardiometabolic and renal benefits
(3,51). Finally, the strong association
between specialist engagement and glycemic control reinforces the
value of integrated, multidisciplinary care models tailored to
underserved populations.
The present study has several strengths, including
its large, community-based Hispanic cohort, comprehensive
characterization of pharmacological treatment patterns, and
simultaneous evaluation of glycemic and renal function parameters
within a real-world safety-net clinical setting. However, several
limitations warrant consideration. First, the cross-sectional
design precludes causal inference regarding the associations
observed between medication use and clinical outcomes. Second,
confounding by indication is likely, as medications such as insulin
and sulfonylureas are typically prescribed to patients with more
advanced or complex disease, which may partially explain observed
differences in glycemic control and comorbidity burden. Third,
medication exposure was defined cross-sectionally based on active
prescriptions recorded during the study period. Detailed
information on treatment duration, sequencing, dose adjustments,
switching patterns, and adherence was not available. Although the
clinic operates under a centralized multidisciplinary care model,
where patients receive comprehensive services within a single
healthcare system, reducing fragmentation, the absence of
longitudinal prescription data limits interpretation of
medication-stratified analyses and precludes assessment of
treatment dynamics over time. Fourth, the primary outcome was
defined using the lowest recorded HbA1c during the study period.
While this approach captures best-achieved glycemic control under
routine care, it may overestimate overall metabolic status and does
not necessarily reflect sustained glycemic control. Additionally,
outcome ascertainment may be influenced by measurement frequency,
although the distribution of HbA1c measurements per patient has
been reported to contextualize this definition. Fifth, the study
period (2022-2024) followed the acute phase of the COVID-19
pandemic. Although calendar-year summaries suggest progressive
normalization of laboratory monitoring, residual temporal effects
related to healthcare system adaptation cannot be entirely
excluded. Finally, although these findings provide important
insight into diabetes management within an underserved Hispanic
population receiving care in a community-based safety-net clinic,
caution is warranted when extrapolating results to other Hispanic
subgroups with different sociodemographic profiles or to healthcare
systems with distinct organizational structures and access
patterns.
In conclusion, the present real-world study of
Hispanic adults with T2DM demonstrates that pharmacological
treatment patterns are strongly associated with clinical
complexity, comorbidities, and healthcare engagement. The superior
glycemic and renal profiles associated with newer agents such as
GLP-1 RAs and SGLT2 inhibitors are heavily influenced by their
selective use in patients with less advanced disease, highlighting
significant confounding by indication. Importantly, specialist
engagement was independently associated with glycemic control;
however, this finding should be interpreted as a marker of
healthcare access, monitoring intensity, or disease complexity
rather than evidence of a direct therapeutic effect. These findings
underscore that addressing diabetes-related disparities in this
high-risk population may require a dual strategy: Ensuring
equitable access to effective modern therapies and emphasizing the
importance of structured healthcare engagement and coordinated care
delivery models to support patients with the most complex
needs.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Patient Centered
Outcomes Research Institute Engagement Award (grant no.
EACB-35014).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
IDE conceptualized and supervised the study and
conducted project administration. EW, ZB and IDE curated data. JDM,
CD, GAHF, FRL, LK, JM and YMR performed formal analysis. JDM and
IDE acquired funding. CD, AB, YMR and LPV conducted investigation.
JDM, CD, MH, GAHF, FRL, LK and AJP developed methodology. JDM, EW
and IDE provided resources. JDM, GAHF, JM, AB, YMR, LPV, EW and ZB
performed software analysis. Validation, JDM, CD, MH, GAHF, FRL,
LK, AJP and OAML validated data. OAML and IDE visualized data. GAHF
wrote the original draft, and wrote, reviewed and edited the
manuscript. All authors read and approved the final version of the
manuscript. IDE, JDM and GAHF confirm the authenticity of all the
raw data.
Ethics approval and consent to
participate
The present study was approved by the Florida
International University Institutional Review Board (approval no.
24-0488; Miami, USA). All procedures complied with ethical
standards for research involving human subjects and ensured strict
confidentiality of patient data. The requirement for individual
informed consent was waived due to the retrospective nature of the
analysis.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
GBD 2021 Diabetes Collaborators. Global,
regional, and national burden of diabetes from 1990 to. 2021, with
projections of prevalence to 2050: A systematic analysis for the
Global Burden of Disease Study 2021. Lancet. 402:203–234.
2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Zheng Y, Ley SH and Hu FB: Global
aetiology and epidemiology of type 2 diabetes mellitus and its
complications. Nat Rev Endocrinol. 14:88–98. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Elsayed NA, Aleppo G, Aroda VR, Bannuru
RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D,
Johnson EL, et al: Addendum. 2. Classification and diagnosis of
diabetes: Standards of care in diabetes-2023. Diabetes care
2023;46(Suppl. 1):S19-S40. Diabetes Care. 46(1715)2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gregg EW, Hora I and Benoit SR: Resurgence
in diabetes-related complications. JAMA. 321:1867–1868.
2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Edmonston D, Lydon E, Mulder H, Chiswell
K, Lampron Z, Marsolo K, Goss A, Ayoub I, Shah RC, Chang AR, et al:
Concordance with screening and treatment guidelines for chronic
kidney disease in type 2 diabetes. JAMA Netw Open.
7(e2418808)2024.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Alexander Fleming G, Petrie JR, Bergenstal
RM, Holl RW, Peters AL and Heinemann L: Diabetes digital app
technology: Benefits, challenges, and recommendations. A consensus
report by the european association for the study of diabetes (EASD)
and the American Diabetes Association (ADA) diabetes technology
working group. Diabetes Care. 43:250–260. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Caballero AE: The ‘A to Z’ of managing
type 2 diabetes in culturally diverse populations. Front Endocrinol
(Lausanne). 9(479)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
American Diabetes Association. Economic
costs of diabetes in the U.S. in 2017. Diabetes Care. 41:917–928.
2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Aguayo-Mazzucato C, Diaque P, Hernandez S,
Rosas S, Kostic A and Caballero AE: Understanding the growing
epidemic of type 2 diabetes in the Hispanic population living in
the United States. Diabetes Metab Res Rev. 35(e3097)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mahgoob MH and Moussa MM: Glycated albumin
versus HbA1c as indicators of glycemic control in type I diabetic
children with iron deficiency anemia. Clin Pediatr Endocrinol.
29:151–157. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
National Institute of Diabetes and
Digestive and Kidney Diseases (NIDDK): Quick Reference on UACR
& GFR. https://www.niddk.nih.gov/health-information/professionals/advanced-search/quick-reference-uacr-gfr
(03/07/2026]).
|
|
12
|
Thomas MC, Brownlee M, Susztak K, Sharma
K, Jandeleit-Dahm KA, Zoungas S, Rossing P, Groop PH and Cooper ME:
Diabetic kidney disease. Nat Rev Dis Primers.
1(15018)2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
de Boer IH, Khunti K, Sadusky T, Tuttle
KR, Neumiller JJ, Rhee CM, Rosas SE, Rossing P and Bakris G:
Diabetes management in chronic kidney disease: A consensus report
by the american diabetes association (ADA) and kidney disease:
Improving global outcomes (KDIGO). Diabetes Care. 45:3075–3090.
2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Narain KDC, Moreno G, Bell DS, Chen L,
Tseng CH, Follett RW, Skootsky S and Mangione CM: Pharmacist-Led
diabetes control intervention and health outcomes in hispanic
patients with diabetes. JAMA Netw Open. 6(e2335409)2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Baghikar S, Benitez A, Fernandez Piñeros
P, Gao Y and Baig AA: Factors impacting adherence to diabetes
medication among urban, low income Mexican-americans with diabetes.
J Immigr Minor Health. 21:1334–1341. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Narain KDC, Patel A, Skootsky S and
Mangione CM: Exploring the relationship between medication
adherence and diabetes disparities among hispanic patients in a
large health system. J Gen Intern Med. 39:1149–1155.
2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
American Diabetes Association.
Classification and diagnosis of diabetes: Standards of medical care
in diabetes-2019. Diabetes Care. 42 (Suppl 1):S13–S28.
2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Glycemic Targets: Standards of medical
care in diabetes-2018. Diabetes Care. 41 (Suppl 1):S55–S64.
2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pharmacologic approaches to glycemic
treatment: Standards of care in diabetes-2024. Diabetes Care. 47
(Suppl 1):S158–S178. 2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Koch-Weser S, Kennefick K, Tighiouart H,
Wong JB, Gordon EJ, Isakova T, Rifkin D, Rossi A, Weiner DE and
Ladin K: Development and validation of the rating of CKD knowledge
among older adults (Know-CKD) with kidney failure. Am J Kidney Dis.
83:569–577. 2024.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ikizler TA, Burrowes JD, Byham-Gray LD,
Campbell KL, Carrero JJ, Chan W, Fouque D, Friedman AN, Ghaddar S,
Goldstein-Fuchs DJ, et al: KDOQI clinical practice guideline for
nutrition in CKD: 2020 update. Am J Kidney Dis. 76 (3 Suppl
1):S1–S107. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Mahemuti N, Zou J, Liu C, Xiao Z, Liang F
and Yang X: Urinary Albumin-to-creatinine ratio in normal range,
cardiovascular health, and All-cause mortality. JAMA Netw Open.
6(e2348333)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sahoo J, Mohanty S, Kundu A and Epari V:
Medication adherence among patients of type II diabetes mellitus
and Its associated risk factors: A Cross-sectional study in a
tertiary care Hospital of Eastern India. Cureus.
14(e33074)2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Doya IF, Yahaya JJ, Ngaiza AI and
Bintabara D: Low medication adherence and its associated factors
among patients with type 2 diabetes mellitus attending Amana
Hospital in Dar es Salaam, Tanzania: A cross-sectional study. Int
Health. 16:200–207. 2024.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sugandh F, Chandio M, Raveena F, Kumar L,
Karishma F, Khuwaja S, Memon UA, Bai K, Kashif M, Varrassi G, et
al: Advances in the management of diabetes mellitus: A focus on
personalized medicine. Cureus. 15(e43697)2023.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kirkman MS, Briscoe VJ, Clark N, Florez H,
Haas LB, Halter JB, Huang ES, Korytkowski MT, Munshi MN, Odegard
PS, et al: Diabetes in older adults. Diabetes Care. 35:2650–2664.
2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Chen J, Yin D and Dou K: Intensified
glycemic control by HbA1c for patients with coronary heart disease
and Type 2 diabetes: A review of findings and conclusions.
Cardiovasc Diabetol. 22(146)2023.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Rosner B: Fundamentals of
biostatistics/Bernard Rosner. 7th edition. Boston, Brooks/Cole,
Cengage Learning, c2011., United States, 2011.
|
|
29
|
Dudley WN, Benuzillo JG and Carrico MS:
SPSS and SAS programming for the testing of mediation models. Nurs
Res. 53:59–62. 2004.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Scheen AJ and Lefèbvre PJ: Insulin versus
insulin plus sulfonylureas in type 2 diabetic patients with
secondary failure to sulfonylureas. Diabetes Res Clin Pract. 6
(Suppl):S33–S43. 1989.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Bailey TS and Mezitis NHE: Combination
therapy with insulin and sulfonylureas for type II diabetes.
Diabetes Care. 13:687–695. 1990.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kalra S, Bahendeka S, Sahay R, Ghosh S, Md
F, Orabi A, Ramaiya K, Al Shammari S, Shrestha D, Shaikh K, et al:
Consensus recommendations on sulfonylurea and sulfonylurea
combinations in the management of type 2 diabetes
mellitus-international task force. Indian J Endocrinol Metab.
22:132–157. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Naja K, Anwardeen N, Bashraheel SS and
Elrayess MA: Pharmacometabolomics of sulfonylureas in patients with
type 2 diabetes: A cross-sectional study. J Pharm Pharm Sci.
27(13305)2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Manta A and Peppa M: Sulfonylureas as
treatment choice in diabetes mellitus: Where are we now? Diabetes
Updates. 5:1–3. 2019.
|
|
35
|
Baccetti F, Crisafulli C, Andreozzi F,
Mannino GC, Nicolucci A, Michelli A, Miranda C, Candido R, Di
Bartolo P, Di Cianni G, et al: Profiles of sulfonylurea use in
diabetes mellitus type 2: An analysis of clinical practice over the
last 10 years. Diabetes Res Clin Pract. 214(111781)2024.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Feingold KR: Oral and Injectable
(Non-Insulin) Pharmacological Agents for the Treatment of type 2
diabetes. Endotext, Dec 29, 2025.
|
|
37
|
Pan HC, Chen JY, Chen HY, Yeh FY, Sun CY,
Huang TTM and Wu VC: GLP-1 receptor agonists' impact on
cardio-renal outcomes and mortality in T2D with acute kidney
disease. Nat Commun. 15(5912)2024.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Fadini GP, Longato E, Morieri ML, Bonora
E, Consoli A, Fattor B, Rigato M, Turchi F, Del Prato S, Avogaro A,
et al: Comparative renal outcomes of matched cohorts of patients
with type 2 diabetes receiving SGLT2 inhibitors or GLP-1 receptor
agonists under routine care. Diabetologia. 67:2585–2597.
2024.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Manteuffel M, Williams S, Chen W,
Verbrugge RR, Pittman DG and Steinkellner A: Influence of patient
sex and gender on medication use, adherence, and prescribing
alignment with guidelines. J Womens Health (Larchmt). 23:112–119.
2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Diaz-Martinez J, Kallus L, Levine HM,
Lavernia F, Pierre AJ, Mancilla J, Barthe A, Duran C, Kotzker W,
Wagner E and Hospital MM: Community-Engaged research (CEnR) to
address gaps in chronic kidney disease education among underserved
Latines-The CARE study. Int J Environ Res Public Health.
20(7026)2023.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Diaz-Martinez J, Delgado-Enciso I, Duran
C, Kallus L, Jean-Pierre A, Lopez B, Mancilla J, Madruga Y,
Hernandez-Fuentes GA, Kotzker W, et al: Patients' perspectives on
the acceptability and effectiveness of a community health
worker-led intervention to increase chronic kidney disease
knowledge and screening among underserved latine adults: The CARE
2.0 study. Behav Sci (Basel). 14(750)2024.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Taberna M, Gil Moncayo F, Jané-Salas E,
Antonio M, Arribas L, Vilajosana E, Peralvez Torres E and Mesía R:
The multidisciplinary team (MDT) approach and quality of care.
Front Oncol. 10(85)2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Sahin I, Bakiner O, Demir T, Sari R and
Atmaca A: Current position of gliclazide and sulfonylureas in the
contemporary treatment paradigm for type 2 diabetes: A scoping
review. Diabetes Therapy. 15:1687–1716. 2024.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Mahtta D, Ramsey DJ, Lee MT, Chen L, Al
Rifai M, Akeroyd JM, Vaughan EM, Matheny ME, Santo KRDE,
Navaneethan SD, et al: Utilization rates of SGLT2 inhibitors and
GLP-1 Receptor agonists and their Facility-level variation among
patients with atherosclerotic cardiovascular disease and type 2
diabetes: Insights from the department of veterans affairs.
Diabetes Care. 45:372–380. 2022.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Davies MJ, Aroda VR, Collins BS, Gabbay
RA, Green J, Maruthur NM, Rosas SE, Del Prato S, Mathieu C,
Mingrone G, et al: Management of hyperglycaemia in type 2 diabetes,
2022. A consensus report by the American Diabetes Association (ADA)
and the European Association for the Study of Diabetes (EASD).
Diabetologia. 65:1925–1966. 2022.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Rodriguez-Gutierrez R, Gionfriddo MR,
Ospina NS, Maraka S, Tamhane S, Montori VM and Brito JP: Shared
decision making in endocrinology: Present and future directions.
Lancet Diabetes Endocrinol. 4:706–716. 2016.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Freeman NLB, Browder SE and McGinigle KL:
Balancing evidence-based care with patient-centered individualized
care. J Vasc Surg Venous Lymphat Disord. 11:1089–1094.
2023.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Hill-Briggs F, Adler NE, Berkowitz SA,
Chin MH, Gary-Webb TL, Navas-Acien A, Thornton PL and Haire-Joshu
D: Social determinants of health and diabetes: A scientific review.
Diabetes Care. 44:258–279. 2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Kaushik P, Reed B, Kalirai S and
Perez-Nieves M: Challenges in insulin initiation among
Hispanics/Latinos with diabetes in the United States. Prim Care
Diabetes. 14:401–412. 2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Cubeddu LX and Hoffmann IS: One-hour
postload plasma glucose levels, a predictor of additional risk for
diabetes: Prevalence, mechanisms, and associated cardiovascular and
metabolic risk factors in hispanics. Metab Syndr Relat Disord.
8:395–402. 2010.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Neuen BL, Fletcher RA, Heath L, Perkovic
A, Vaduganathan M, Badve SV, Tuttle KR, Pratley R, Gerstein HC,
Perkovic V and Heerspink HJL: Cardiovascular, kidney, and safety
outcomes with GLP-1 receptor agonists alone and in combination with
SGLT2 inhibitors in type 2 diabetes: A systematic review and
Meta-analysis. Circulation. 150:1781–1790. 2024.PubMed/NCBI View Article : Google Scholar
|