Introduction
Amyotrophic lateral sclerosis (ALS) is a fatal
neurodegenerative disorder characterized by the progressive loss of
upper and lower motor neurons, leading to muscle weakness, atrophy,
and paralysis. As the disease progresses, patients experience
impairments in speech, swallowing, respiration, and limb function,
with most succumbing to respiratory failure within 3-5 years of
symptom onset (1,2). Clinical heterogeneity is a hallmark of
ALS, with bulbar- and limb-onset ALS being the two predominant
subtypes. Bulbar-onset ALS manifests with dysarthria and dysphagia
due to degeneration in the bulbar regions, while limb-onset ALS is
characterized by weakness and fasciculations in the extremities
(3-7).
These phenotypes differ not only in clinical manifestations but
also in prognosis; although less common, bulbar-onset ALS typically
exhibits rapid disease progression and is associated with shorter
survival (3-5).
Emerging evidence further suggests that subtype-specific
pathophysiological features involve distinct neural pathways and
genetic risk factors (8-11).
However, therapeutic strategies still primarily target ALS as a
single entity (12,13), overlooking potential
subtype-specific targets.
Dyslipidemia in ALS, characterized by altered
cholesterol and triglyceride levels, has been associated with both
neuroprotective and deleterious effects, suggesting a multifaceted
and context-dependent role in disease pathogenesis (14-18).
Some studies report that hyperlipidemia and a higher body mass
index may slow disease progression and improve survival (16,19),
whereas others associate lipid abnormalities with worse prognosis
(20-22).
These inconsistencies likely stem from variations in study design,
evaluated lipid markers, and patient cohorts. Notably, most studies
have treated ALS as a uniform disease, with limited consideration
of heterogeneity across clinical subtypes. This gap is significant
as bulbar- and limb-onset ALS exhibit distinct clinical
trajectories, and emerging evidence indicates potential differences
in metabolic mechanisms.
Most existing studies have been conducted in Western
populations (14-21),
raising uncertainties about the generalizability of the findings to
other regions. The regional and ethnic differences in both ALS
epidemiology and metabolic risk factors highlight the importance of
investigating non-Western cohorts (23-25).
While some studies have explored the lipid profiles in Chinese
patients with ALS, systematic analyses focusing on subtype-specific
associations remain scarce.
In this study, we investigated serum lipid profiles
in a Chinese ALS cohort, specifically exploring whether the
association with disease severity varied between bulbar- and
limb-onset subtypes. Through correlation analyses and multivariable
interaction models, our objective was to determine if lipid
parameters have subtype-specific effects on functional impairment.
These analyses provide new insights into the metabolic
heterogeneity of ALS and may contribute to the development of more
tailored prognostic assessments and management strategies across
clinical subtypes.
Materials and methods
Patients and ethical
considerations
This retrospective, cross-sectional study analyzed
clinical data from patients with ALS and age- and sex-matched
healthy controls treated between March 2022 and July 2024 at the
Hubei Provincial Hospital of Traditional Chinese Medicine, China.
The study 158 patients with ALS showing clinical and
electrophysiological signs of both upper and lower motor neuron
involvement, following the revised Gold Coast criteria (26). Exclusion criteria included patients
diagnosed with a history of neurological disorders that could
impact the assessment, such as brain injury, stroke,
alcohol/substance-related disorders, depression, and major
psychiatric conditions. Comprehensive demographic and clinical data
were collected during the initial visit. The severity of ALS was
evaluated using the revised ALS Functional Rating Scale (ALSFRS-R)
(27). Disease onset was defined as
the patient-reported time of the initial symptoms, i.e., the first
paresis in spinal-onset ALS or speech/swallowing disturbances in
bulbar-onset ALS. Additionally, 62 age- and sex-matched Healthy
controls (HCs) were retrospectively enrolled from the Hubei
Provincial Hospital of Traditional Chinese Medicine. All
participants provided written informed consent. The study received
approval from the Ethics Committee of the Hubei University of
Chinese Medicine (approval no. HBZY1022-C42-02). Fig. 1 illustrates the study design
flowchart.
Blood test
Blood samples from patients with ALS and healthy
individuals were collected between 7:00 a.m. and 10:00 a.m.
following an overnight fast of 8 h. Serum levels of total
cholesterol (TC), triglycerides (TG), high-density lipoprotein
(HDL), low-density lipoprotein (LDL), small dense LDL cholesterol
(sdLDL-c), apolipoprotein A-1 (ApoA1), and apolipoprotein B (ApoB)
were measured using an automatic biochemical analyzer (ADVIA 2400;
Siemens Healthcare Diagnostics Inc., Tarrytown, NY, USA), following
the manufacturer's protocols.
Statistical analysis
Descriptive statistics were employed to summarize
the patient characteristics. Continuous variables were assessed for
normality using the Shapiro-Wilk test. Normally distributed
variables were compared between subgroups utilizing an
independent-samples t-test, while non-normally distributed
variables were analyzed using the Mann-Whitney U test. Categorical
variables were compared employing the χ2 test. One-way
ANOVA with Tukey's post-hoc test was utilized for multiple group
comparisons, and Spearman's correlation analyses were conducted to
evaluate the relationships between serum lipid biomarkers and
disease severity.
To assess whether the relationships between lipid
parameters and ALS severity varied by clinical subtype, we
conducted distinct multivariable linear regression models with
ALSFRS-R score as the dependent variable. Each lipid parameter was
included in the model with an interaction term (phenotype x lipid
value). Phenotypes were categorized as 0=limb-onset and
1=bulbar-onset, while sex was categorized as 0=male and 1=female.
The models were adjusted for age, sex, body mass index (BMI), and
disease duration. Regression coefficients with 95% confidence
intervals and P-values were reported. Statistical analyses were
performed using JASP software (JASP Team, Amsterdam, Netherlands).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Demographic and clinical
characteristics
The characteristics of the patients with ALS and HCs
are summarized in Table I. The ALS
cohort comprised 158 patients (105 males, 53 females) with an
average age of 53.01±10.84 years at examination. Among these, 30
presented with bulbar onset, while 128 had limb onset. The most
prevalent age group was 49-59 years (38.6%). Only 22.8% of the
patients were from Hubei province, whereas the remaining 77.2% were
individuals from other regions of China who sought diagnosis and
treatment at the Hubei Provincial Hospital of Traditional Chinese
Medicine. The average age at symptom onset was 50.7±10.7 years. The
median diagnostic delay was 11 months, and the median disease
duration at assessment was 21.4 months.
 | Table IComparison of demographic and
clinical characteristics between patients with ALS and HCs. |
Table I
Comparison of demographic and
clinical characteristics between patients with ALS and HCs.
| Characteristic | Patients with ALS
(n=158) | HCs (n=62) |
|---|
| Sex,
male/female | 105/53 | 35/27 |
| BMI,
kg/m2 | 23.22±3.92 | 24.15±3.49 |
| Age at interview,
yearsa | 53.01±10.84 | 49.96±12.18 |
| Age of onset,
yearsa | 50.67±10.65 | NA |
| Site of onset,
spinal/bulbar | 128/30 | NA |
| Diagnostic delay,
monthsa | 11.0
(6.06-19.38) | NA |
| Disease duration,
monthsa | 21.4
(10.42-37.65) | NA |
| ALSFRS-R
scorea | 36.0
(30.0-41.0) | NA |
The control group comprised 62 individuals (35
males, 27 females) with a mean age of 49.96±12.18 years. No
significant variations in age or sex were observed between patients
and controls. The mean BMI was slightly lower in ALS patients
(23.22±3.92 kg·m-2) compared with controls (24.15±3.49
kg·m−2), although this variance was not statistically
significant.
Blood lipid levels in patients with
ALS
Compared with controls, patients with ALS exhibited
significantly elevated serum levels of TC, TG, LDL, sdLDL-c, ApoA1,
ApoB, and the TG/HDL ratio (Fig.
2). Stratification by disease duration revealed that patients
with a disease duration exceeding 12 months demonstrated higher
levels of TC, TG, LDL, sdLDL-c, and ApoB compared with both
controls and patients with a duration of less than 12 months.
Particularly, TG levels and the TG/HDL ratio were significantly
higher in patients with longer disease duration than in those with
shorter disease duration (Fig.
3).
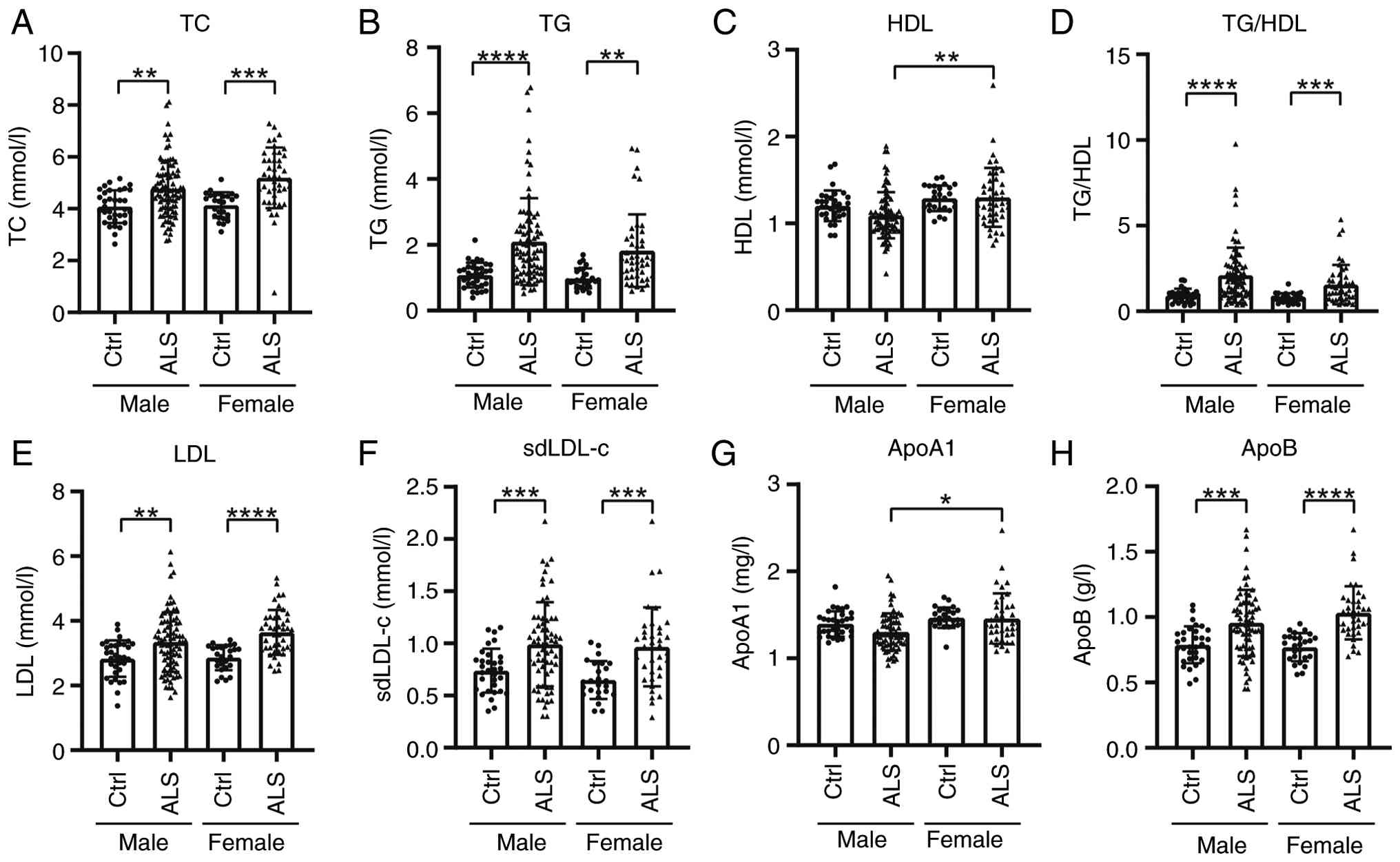 | Figure 2Serum lipid profiles in patients with
ALS by sex. Serum lipid levels of (A) TC, (B) TG, (C) HDL, (D)
TG/HDL, (E) LDL, (F) sdLDL-c, (G) ApoA1 and (H) ApoB in patients
with ALS and Ctrls, categorized by sex. ****P<0.0001;
***P<0.001; **P<0.01;
*P<0.05. ALS, amyotrophic lateral sclerosis; TC,
total cholesterol; TG, triglycerides; HDL, high-density
lipoprotein; LDL, low-density lipoprotein; sdLDL-c, small dense
LDL-cholesterol; Apo, apolipoprotein; Ctrl, control. |
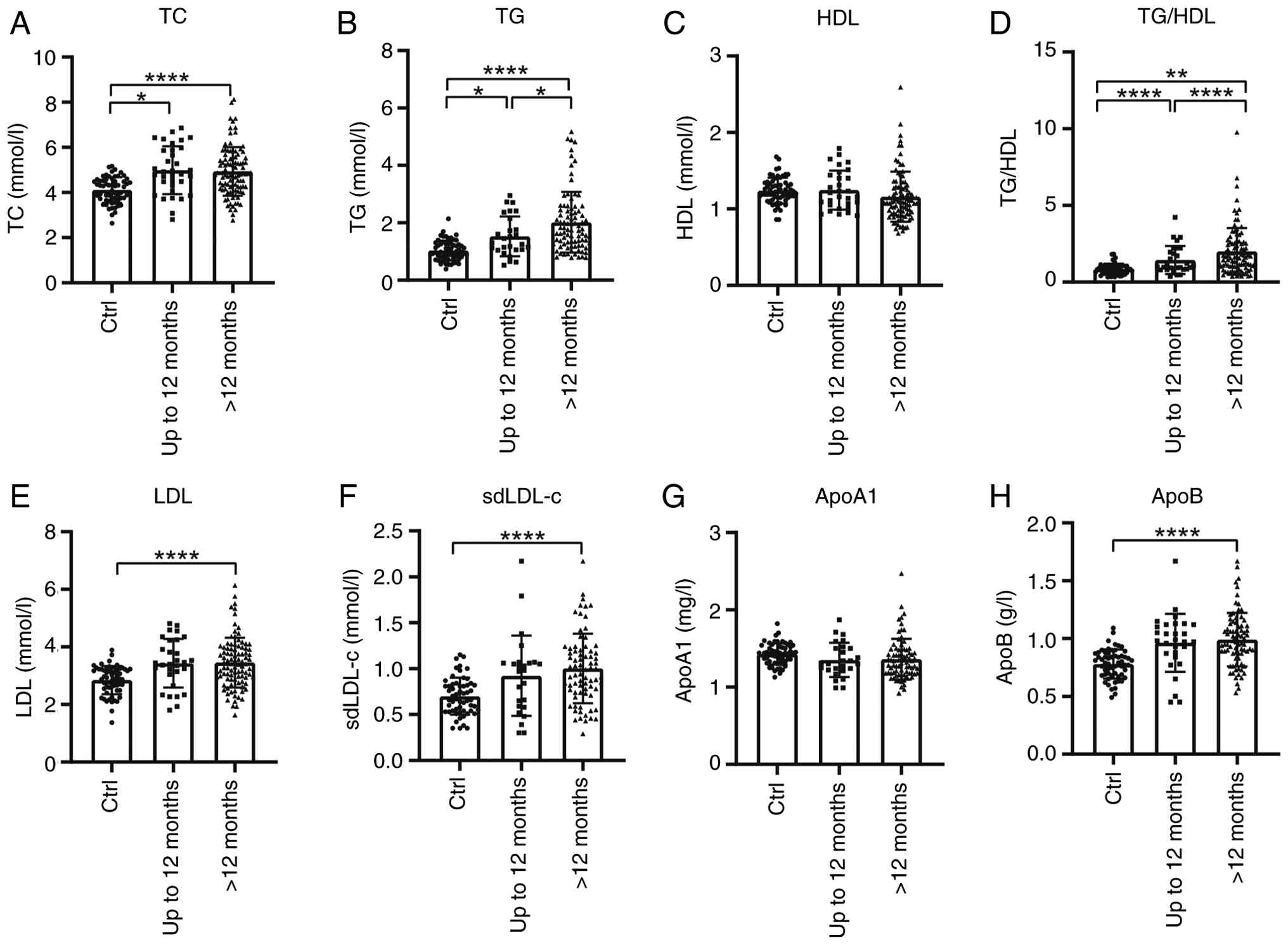 | Figure 3Serum lipid profiles in patients with
ALS based on disease duration. Serum lipid levels (A) TC, (B) TG
(B), (C) HDL, (D) TG/HDL, (E) LDL, (F) sdLDL-c, (G) ApoA1 and (H)
ApoB in patients with ALS and Ctrls, categorized by disease
duration (up to 12 months vs. >12 months).
****P<0.0001; **P<0.01;
*P<0.05. ALS, amyotrophic lateral sclerosis; TC,
total cholesterol; TG, triglycerides; HDL, high-density
lipoprotein; LDL, low-density lipoprotein; sdLDL-c, small dense
LDL-cholesterol; Apo, apolipoprotein; Ctrl, control. |
Correlation of total cholesterol with
ALS severity
A significant negative correlation was observed
between TC and ALSFRS-R scores (rs=-0.3113, P=0.0005),
indicating that higher cholesterol was associated with greater
functional impairment (Fig. 4). No
significant correlations were observed for TG, LDL, sdLDL-c, ApoA1,
ApoB, or the TG/HDL ratio at diagnosis (Fig. 4).
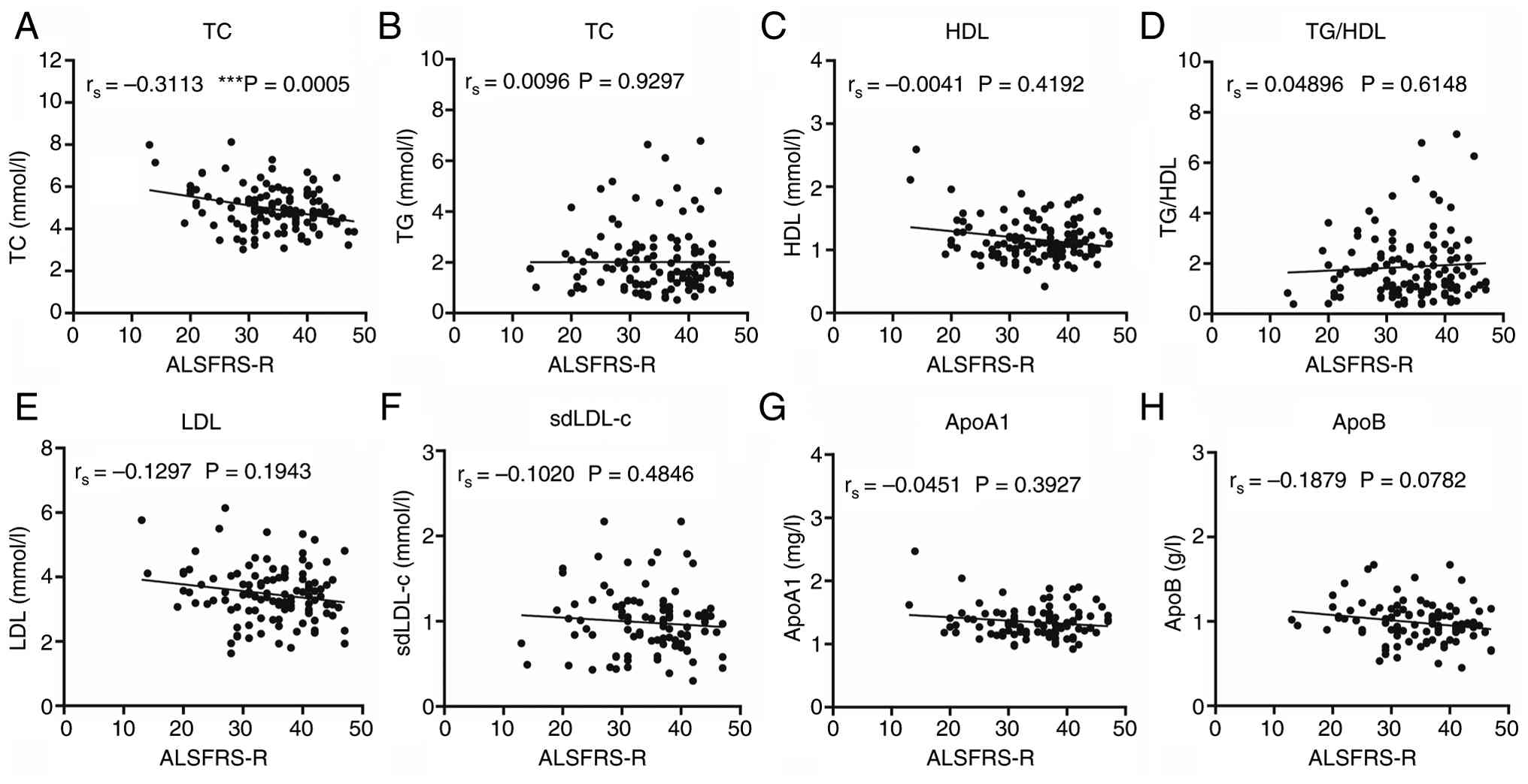 | Figure 4Correlations between serum lipid
levels and ALSFRS-R scores. Correlations between serum lipid levels
of (A) TC, (B) TG (B), (C) HDL, (D) TG/HDL, (E) LDL, (F) sdLDL-c,
(G) ApoA1 and (H) ApoB and ALSFRS-R scores at diagnosis. A
significant negative correlation is highlighted between (A) TC and
ALSFRS-R scores (rs=-0.3113; P=0.0005).
***P<0.001. ALSFRS-R, Amyotrophic Lateral Sclerosis
Functional Rating Scale; TC, total cholesterol; TG, triglycerides;
HDL, high-density lipoprotein; LDL, low-density lipoprotein;
sdLDL-c, small dense LDL-cholesterol; Apo, apolipoprotein. |
Blood lipid levels according to the
site of disease onset
To assess whether lipid profiles differed by
clinical phenotype, patients with ALS were categorized according to
the onset site. The limb-onset cohort comprised 87 males and 41
females, with a median age of 51.5 years, a diagnostic delay of 7.8
months, and a disease duration of 16.4 months at the time of
sampling. In contrast, the bulbar-onset group consisted of 18 males
and 12 females with a median age of 50.0 years, a diagnostic delay
of 8.4 months, and a disease duration of 26.6 months. Notably,
patients with bulbar-onset ALS exhibited a significantly prolonged
disease duration compared to those with limb-onset ALS (P=0.030),
while displaying slightly lower ALSFRS-R scores (P=0.111). There
were no significant differences were observed in sex distribution
(P=0.405), age (P=0.710), diagnostic delay (P=0.148) or BMI
(P=0.741) between the two subgroups (Table II).
 | Table IIDemographic and clinical
characteristics of the patients with ALS. |
Table II
Demographic and clinical
characteristics of the patients with ALS.
| | Phenotype | |
|---|
| Characteristic | Limb-onset ALS
(n=128) | Bulbar-onset ALS
(n=30) | P-value |
|---|
| Sex,
male/female | 87:41 | 18:12 | 0.405 |
| BMI,
kg/m2a | 23.37±4.29 | 22.68±3.34 | 0.741 |
| Age at interview,
yearsa | 51.5
(43.25-56.75) | 50.0
(44.0-57.0) | 0.710 |
| Diagnostic delay,
monthsa | 7.83
(4.03-14.88) | 8.4
(4.31-15.83) | 0.148 |
| Disease duration,
monthsa | 16.4
(4.94-29.54) | 26.6
(16.68-42.71) | 0.030 |
| ALSFRS-R
scorea | 40 (31-43) | 32 (2-41) | 0.111 |
| Increased total
cholesterol | 40.0% | 33.3% | - |
| Increased
triglycerides | 48.5% | 48.1% | - |
| Increased HDL | 17.2% | 18.5% | - |
| Increased LDL | 53.6% | 37.0% | - |
| Increased
sdLDL-c | 15.7% | 5.6% | - |
| Increased
ApoA1 | 13.1% | 14.3% | - |
| Increased ApoB | 7.1% | 4.8% | - |
Limb-onset patients had higher proportions of
elevated LDL and sdLDL-c levels compared with bulbar-onset patients
(Table II). Both subgroups
demonstrated significantly elevated TC, TG, and ApoB levels in
comparison to the controls, while HDL and ApoA1 levels were
similar. Notably, TG, sdLDL-c, and the TG/HDL ratio were elevated
in limb-onset patients compared with bulbar-onset patients
(Fig. 5).
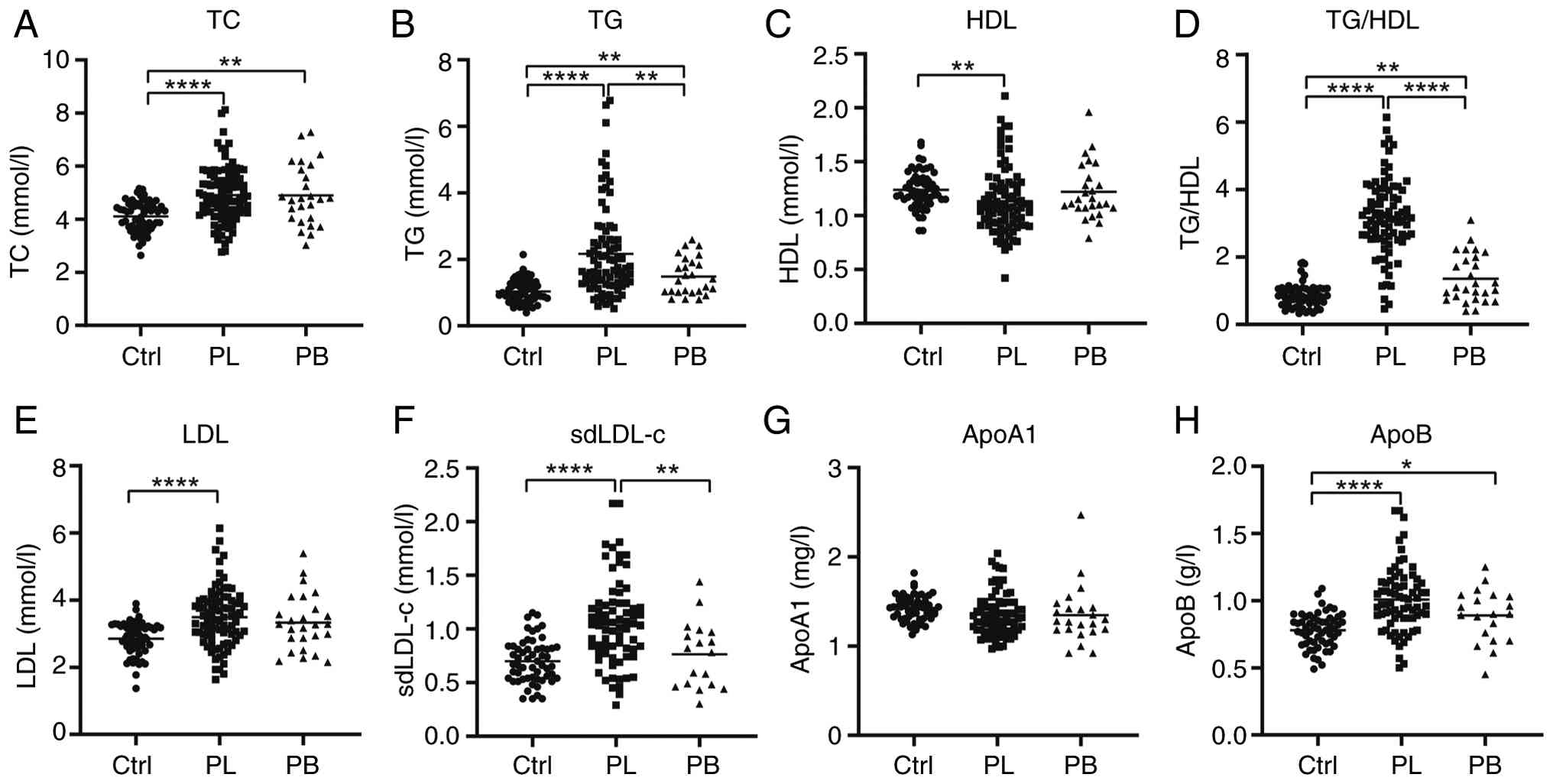 | Figure 5Subgroup analysis of serum lipids
levels in PL and PB. Serum lipid levels of (A) TC, (B) TG (B), (C)
HDL, (D) TG/HDL, (E) LDL, (F) sdLDL-c, (G) ApoA1 and (H) ApoB in PL
and PB compared to Ctrls. ****P<0.0001;
**P<0.01; *P<0.05. ALS, amyotrophic
lateral sclerosis; PL, patients with limb-onset ALS; PB, patients
with bulbar-onset ALS; TC, total cholesterol; TG, triglycerides;
HDL, high-density lipoprotein; LDL, low-density lipoprotein;
sdLDL-c, small dense LDL-cholesterol; Apo, apolipoprotein; Ctrl,
control. |
Associations between lipid profiles
and disease severity by subtypes
Correlation analyses indicated subtype-dependent
associations between lipids levels and ALS severity. In both
subgroups, higher TC was associated with lower ALSFRS-R scores
(limb-onset: rs=-0.2977, P=0.0028; bulbar-onset:
rs=-0.4036, P=0.0454). Among bulbar-onset patients, TG
(rs=0.4292, P=0.0255), sdLDL-c (rs=0.5158,
P=0.0285), and TG/HDL ratio (rs=0.4201, P=0.0291)
exhibited positive correlations with ALSFRS-R scores, whereas HDL
(rs=-0.3988, P=0.0394) and ApoA1 (rs=-0.4304,
P=0.0404) were inversely correlated. In limb-onset patients,
sdLDL-c (rs=-0.2451, P=0.0295) and ApoB
(rs=-0.2487, P=0.0255) showed negative correlations with
ALSFRS-R scores, while other lipid measures (TG, HDL, LDL) showed
no significant associations (Fig.
6).
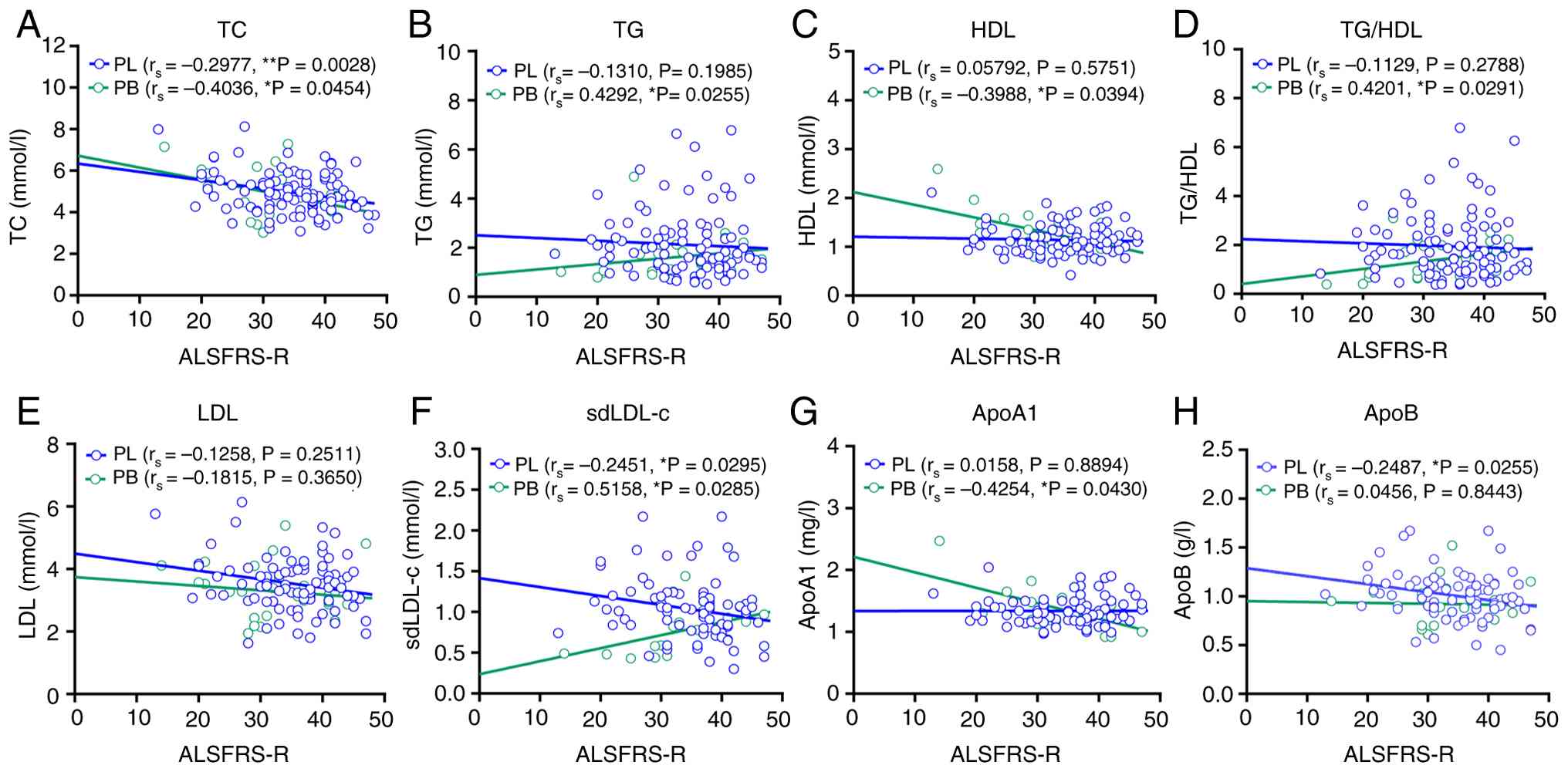 | Figure 6Correlations between serum lipid
levels and ALSFRS-R scores in PL and PB. Correlations between serum
lipid levels of (A) TC, (B) TG (B), (C) HDL, (D) TG/HDL, (E) LDL,
(F) sdLDL-c, (G) ApoA1 and (H) ApoB and ALSFRS-R scores in PL and
PB. **P<0.01; *P<0.05. ALS, amyotrophic
lateral sclerosis; PL, patients with limb-onset ALS; PB, patients
with bulbar-onset ALS; ALSFRS-R, ALS Functional Rating Scale; TC,
total cholesterol; TG, triglycerides; HDL, high-density
lipoprotein; LDL, low-density lipoprotein; sdLDL-c, small dense
LDL-cholesterol; Apo, apolipoprotein. |
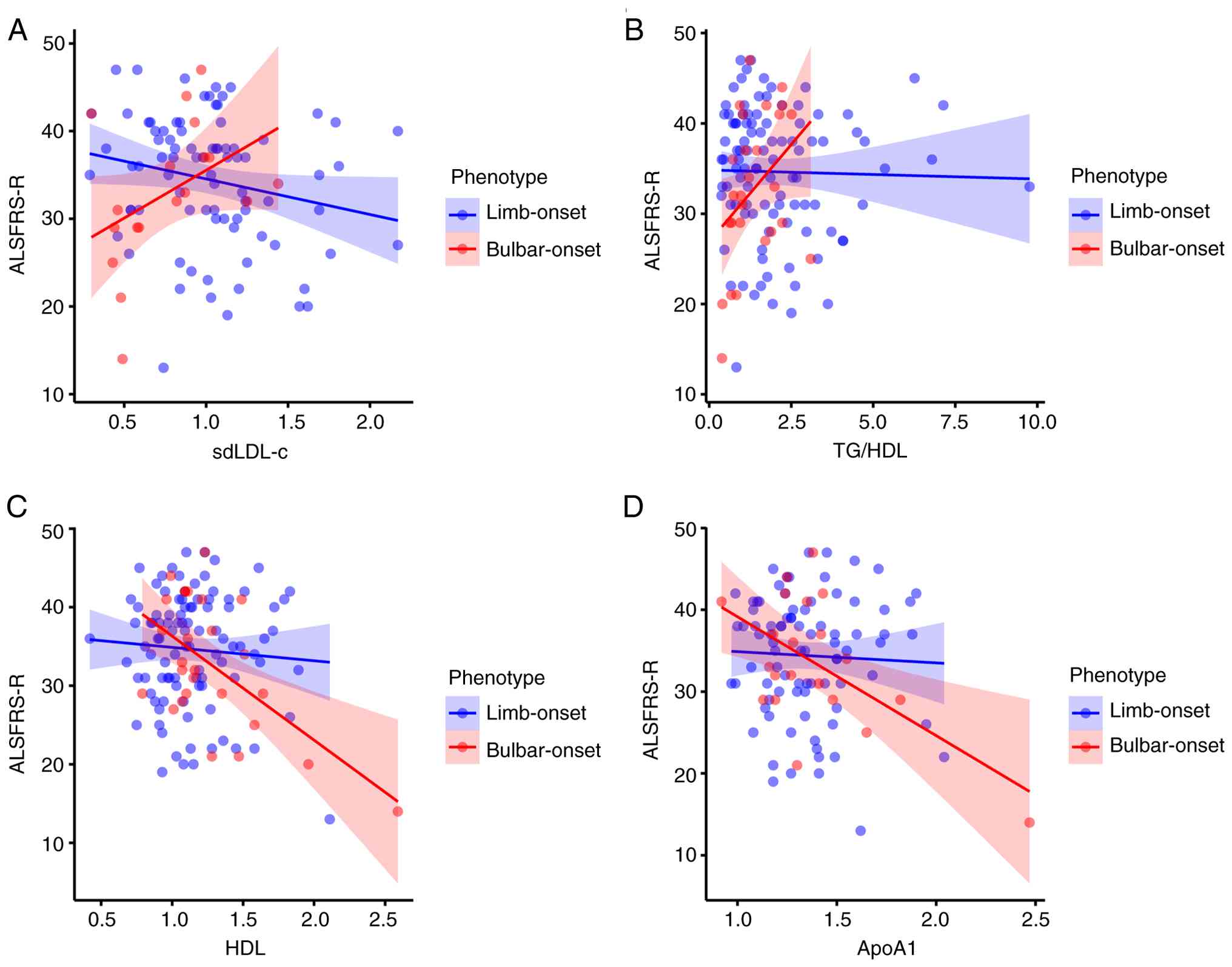
To further assess whether these associations
differed significantly between phenotypes, we conducted
multivariable linear regression analyses incorporating interaction
terms (phenotype x lipid level), and adjusting for age, sex, BMI,
and disease duration. Significant phenotype-lipid interactions were
identified for sdLDL-c (β=12.223, P=0.038), TG/HDL ratio (β=4.620,
P=0.019), ApoA1 (β=-13.618, P=0.020), and HDL (β=-11.963, P=0.006)
(Table III). Simple slope
analysis showed that higher sdLDL-c and TG/HDL ratios were
associated with better functional status in bulbar-onset patients
but not in limb-onset patients (Fig.
7A and B). In contrast, HDL and
ApoA1 levels exhibited stronger negative associations with ALSFRS-R
scores in bulbar-onset cases (Fig.
7C and D). No significant
interactions were detected for TC, TG, ApoB, or LDL (Table III; Fig. S1).
 | Table IIIMultivariable linear regression
analyses of the interaction effects between ALS phenotype and lipid
parameters on functional severity (ALSFRS-R score). |
Table III
Multivariable linear regression
analyses of the interaction effects between ALS phenotype and lipid
parameters on functional severity (ALSFRS-R score).
| Model | Lipid
parameter | β for interaction
(phenotype x lipid) | 95% CI | P-value |
|---|
| 1 | TC | -0.288 | -2.654-2.079 | 0.810 |
| 2 | TG | 2.759 | -0.616-6.134 | 0.108 |
| 3 | HDL | -11.963 | -20.442 to
-3.484 | 0.006 |
| 4 | LDL | -0.534 | -4.144-3.075 | 0.770 |
| 5 | sdLDL-c | 12.223 | 0.713-23.732 | 0.038 |
| 6 | ApoA1 | -13.618 | -25.090 to
-2.146 | 0.020 |
| 7 | ApoB | 3.144 | -11.572-17.859 | 0.673 |
| 8 | TG/HDL ratio | 4.620 | 0.773-8.468 | 0.019 |
Together, these results suggest that certain lipid
parameters, particularly sdLDL-c, the TG/HDL ratio, HDL, and ApoA1,
may exhibit subtype-specific associations with ALS severity.
Discussion
This study demonstrates dyslipidemia in a Chinese
ALS cohort and reveals that the associations between lipid levels
and functional severity are modified by clinical subtype. In
bulbar-onset ALS, higher sdLDL-c levels and TG/HDL ratios were
associated with better functional status, whereas higher HDL and
ApoA1 levels tended to correlate with worse outcomes. In contrast,
in limb-onset ALS, higher sdLDL-c and ApoB levels were associated
with greater functional impairment, whereas other lipid measures
showed no significant association. These findings extend the
observations from Western cohorts and underscore the importance of
accounting for clinical heterogeneity when investigating metabolic
alterations in ALS.
Altered lipid metabolism has long been recognized as
a feature of ALS, reflecting the high energy demands of denervated
and atrophic muscle tissues (28).
Lipids serve as major energy substrates, and their imbalance may
influence both systemic metabolism and neuronal vulnerability
(29). Previous studies in Western
populations have reported inconsistent results regarding
dyslipidemia in patients with ALS. Some studies have linked
elevated lipid levels, particularly triglycerides, to improved
survival and slower progression, whereas others have found no
significant association or even adverse effects (21,22,30-32).
These inconsistencies may arise from methodological differences,
variation in disease stage, and unaccounted phenotypic diversity.
Evidence from Asian cohorts provides additional context for these
discrepancies. Early studies from southwestern China have reported
no significant lipid differences between patients with ALS and
controls (33). However, more
recent Chinese studies have observed negative correlations between
total cholesterol or LDL-c levels and ALSFRS-R scores, and between
ApoB or Lp(a) levels and respiratory function, suggesting that
lipid imbalance accompanies disease severity (34). Another large Chinese cohort found
lower HDL and total cholesterol levels but higher LDL/HDL ratios,
consistent with stage-related metabolic shifts (35). Our findings partially align with
these results but further reveal subtype-specific lipid signatures,
indicating that metabolic adaptations differ between bulbar- and
limb-onset ALS. Similar heterogeneity has been observed in
non-Western populations. In Japanese cohorts, higher total
cholesterol, LDL, and triglyceride levels were associated with
slower disease progression (36),
supporting our observation that elevated lipid levels may reflect
compensatory metabolic responses. Conversely, Nakamura et al
(25) reported that high HDL levels
predict poor prognosis, paralleling our finding of a negative
association between HDL and functional status. In South Korea,
lipid depletion, especially in men, suggests accelerated lipid
consumption (37), and CT-based
analyses have demonstrated that fat depletion (adipopenia)
independently predicts poor survival (38). Collectively, these findings,
together with our data, indicate that dyslipidemia is a shared
metabolic feature of ALS; however, its clinical implications vary
by ethnicity, sex, and phenotype, reflecting diverse adaptive
responses to neurodegeneration.
The inverse relationship between cholesterol and
ALSFRS-R scores observed in this study supports growing evidence
that cholesterol homeostasis may play a role in disease
progression. Cholesterol is a structural component of neuronal
membranes and a precursor of signaling molecules such as steroid
hormones and oxysterols (39,40).
Disturbances in cholesterol regulation have been implicated in ALS
through multiple molecular pathways. Dysregulation of the LXR/RXR
signaling pathways and polymorphisms in lipid-related genes such as
APOE and SREBP2 have been associated with altered
lipid metabolism and neuronal stress responses in ALS (41,42).
Furthermore, TDP-43, a central protein in ALS pathology, modulates
SREBP2-dependent cholesterol metabolism, suggesting a mechanistic
link between lipid dysregulation and neurodegeneration (43,44).
Large-scale genome-wide association studies have also identified
elevated total cholesterol as a potential risk factor for ALS
(45), further reinforcing the role
of lipid homeostasis in disease susceptibility.
Our subtype-specific analysis provides further
insight into metabolic diversity in ALS. In patients with
bulbar-onset ALS, we observed that higher levels of sdLDL-c and
TG/HDL ratio were associated with better functional status,
potentially indicating a compensatory lipid mobilization response
to the heightened metabolic stress in this subgroup (46). This observation aligns with the
concept that individuals with bulbar-onset ALS, who typically
experience faster disease progression, may rely on enhanced lipid
utilization to meet elevated energy demands (4,5).
Conversely, in limb-onset ALS, elevated levels of sdLDL-c and ApoB
were linked to poorer functional status, suggesting that lipid
accumulation in this subgroup could contribute to exacerbate the
disease burden. These opposing trends underscore the idea that
lipid dysregulation in ALS is not uniform, but is likely
context-dependent, with the clinical phenotype and metabolic
dynamics playing a critical role (46). Similarly, previous reports have
noted that elevated triglyceride level or BMI may predict longer
survival, whereas high HDL levels have been associated with poorer
outcomes (15,16,25).
This finding reinforces the notion that lipid dysregulation in ALS
is phenotype-specific with distinct metabolic responses to neuronal
degeneration in bulbar-onset and limb-onset ALS. Bulbar-onset ALS
may rely on lipid mobilization as an adaptive response to rapid
degeneration, whereas limb-onset ALS may exhibit lipid
accumulation, reflecting a passive metabolic adaptation to impaired
muscle function. These contrasting trends highlight the importance
of clinical heterogeneity when interpreting lipid alterations in
patients with ALS.
These findings emphasize the significance of
considering ALS as a metabolically heterogeneous disorder rather
than a singular disease entity. Lipid signatures specific to
subtypes could potentially function as biomarkers for monitoring
disease progression and categorizing patients in clinical studies.
Apart from their biomarker potential, lipid changes may hold
therapeutic relevance. Increasing evidence indicates that
maintaining metabolic balance can influence ALS outcomes.
Nutritional and metabolic strategies, such as high-calorie or
high-fat diets, have demonstrated positive effects in SOD1(G86R)
ALS mouse models and in small clinical trials, improving energy
balance, tolerance, and occasionally survival (47-49).
Recent reviews highlight that energy deficits and lipid depletion
accelerate neurodegeneration, while replenishing lipid availability
might offer neuroprotective effects (8,50).
Although our study did not establish causation, the identified
subtype-specific correlations suggest that personalized metabolic
or dietary interventions tailored to specific phenotypes could
complement future pharmacological approaches in ALS management.
Despite these insights, this study has several
limitations. First, the retrospective, cross-sectional design
limits causal inference. Since the data were extracted from
existing clinical records, the analysis has been subject to missing
information, unrecorded confounders, and potential selection bias.
Therefore, our findings should be viewed as hypothesis-generating
rather than confirmatory, pending validation in prospective
longitudinal cohorts. Second, the relatively small sample size of
the bulbar-onset subgroup (n=30) may have affected the statistical
stability of the subtype-specific associations. Given this modest
number, the observed trends should be interpreted with caution, as
they may reflect random variations rather than robust effects.
Nonetheless, the internal consistency across analyses and
biological plausibility lend support to the reliability of the main
findings. Future studies with larger, multicenter cohorts are
required to confirm these subtype-specific metabolic patterns and
to assess their generalizability across populations. Finally, our
study focused on conventional lipid metrics; future research
incorporating advanced lipidomics and longitudinal designs is
crucial to unravel the precise temporal dynamics and molecular
mechanisms of lipid dysregulation across ALS subtypes.
In conclusion, our study identified dyslipidemia as
a prevalent feature of ALS and revealed subtype-specific
associations between lipid parameters and functional impairment.
These findings suggest that the metabolic pathways differ between
bulbar- and limb-onset ALS, reflecting distinct pathophysiological
mechanisms. Integrating metabolic profiling into ALS research may
improve the prognostic accuracy and support the development of
personalized management strategies tailored to each clinical
phenotype.
Supplementary Material
Interaction effects of additional
lipid parameters with ALS phenotype on ALSFRS-R scores. (A)
Interaction between phenotype and TC levels. (B) Interaction
between phenotype and TG levels. (C) Interaction between phenotype
and LDL levels. (D) Interaction between phenotype and ApoB levels.
Multivariable linear regression models included phenotype x lipid
interaction terms, adjusted for age, sex, body mass index and
disease duration. The red line represents the bulbar-onset
phenotype, and the blue line represents the limb-onset phenotype.
Shaded areas represent 95% confidence intervals. ALS, amyotrophic
lateral sclerosis; ALSFRS-R, ALS Functional Rating Scale; TC, total
cholesterol; TG, triglycerides; LDL, low-density lipoprotein; Apo,
apolipoprotein.
Acknowledgements
The authors would like to thank Dr YouYun Zhao
(Department of Clinical Laboratory, Hubei Provincial Hospital of
Traditional Chinese Medicine) for helping with the collection of
clinical data.
Funding
Funding: This work was supported by the National Natural Science
Foundation of China Grants (grant no. 82575246), the Hubei
Provincial Department of Education Science and Technology Research
Program Young Talent Project (grant no. Q20232010), the Hubei
University of Chinese Medicine Doctoral Startup Fund (grant no.
100501080740), the Key Research and Development Program of Hubei
Province (grant no. 2022BCA055) and the Hubei Provincial Hospital
of Traditional Chinese Medicine (grant no. 2021YJKT-24).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
LC conceived and designed the study, collected and
analyzed the data, interpreted the results and drafted the
manuscript. GC contributed to study design, data interpretation and
critically revised the manuscript. JZ and HH participated in data
acquisition and analysis, and contributed to manuscript revision.
AX and TZ assisted with data collection and organization, and
participated in data analysis. YZ and HL contributed to study
design and clinical data acquisition, and critically revised the
manuscript. LC, YZ and HL confirm the authenticity of all the raw
data. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The Ethical Committee of Hubei University of Chinese
Medicine approved this study (approval no. HBZY1022-C42-02). The
principal author has received consent forms from the participants
in this study and has them on file. All participants provided
written informed consent.
Patient consent for publication
Not applicable.
Competing interest
The authors declare that they have no competing
interests.
References
|
1
|
Feldman EL, Goutman SA, Petri S, Mazzini
L, Savelieff MG, Shaw PJ and Sobue G: Amyotrophic lateral
sclerosis. Lancet. 400:1363–1380. 2022.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Xu RS and Yuan M: Considerations on the
concept, definition, and diagnosis of amyotrophic lateral
sclerosis. Neural Regen Res. 16:1723–1729. 2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Todd TW and Petrucelli L: Amyotrophic
lateral sclerosis-insight into susceptibility. Nat Rev Neurol.
18:189–190. 2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Shellikeri S, Karthikeyan V, Martino R,
Black SE, Zinman L, Keith J and Yunusova Y: The neuropathological
signature of bulbar-onset ALS: A systematic review. Neurosci
Biobehav Rev. 75:378–392. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Yunusova Y, Plowman EK, Green JR, Barnett
C and Bede P: Clinical measures of bulbar dysfunction in ALS. Front
Neurol. 10(106)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Goutman SA, Savelieff MG, Jang DG, Hur J
and Feldman EL: The amyotrophic lateral sclerosis exposome: Recent
advances and future directions. Nat Rev Neurol. 19:617–634.
2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Turner MR, Wicks P, Brownstein CA,
Massagli MP, Toronjo M, Talbot K and Al-Chalabi A: Concordance
between site of onset and limb dominance in amyotrophic lateral
sclerosis. J Neurol Neurosurg Psychiatry. 82:853–854.
2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Ludolph A, Dupuis L, Kasarskis E, Steyn F,
Ngo S and McDermott C: Nutritional and metabolic factors in
amyotrophic lateral sclerosis. Nat Rev Neurol. 19:511–524.
2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Holdom CJ, Steyn FJ, Henderson RD, McCombe
PA, Rogers ML and Ngo ST: Biofluid biomarkers of amyotrophic
lateral sclerosis. In: Neurodegenerative Diseases Biomarkers.
Neuromethods. Peplow PV, Martinez B and Gennarelli TA (eds). Vol
173. Humana, New York, NY, pp263-306, 2022.
|
|
10
|
Hu Y, Chen W, Wei C, Jiang S, Li S, Wang X
and Xu R: Pathological mechanisms of amyotrophic lateral Sclerosis.
Neural Regen Res. 19:1036–1044. 2024.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Swinnen B and Robberecht W: The phenotypic
variability of amyotrophic lateral sclerosis. Nat Rev Neurol.
10:661–670. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zeng Q, Shen J, Chen K, Zhou J, Liao Q, Lu
K, Yuan J and Bi F: The alteration of gut microbiome and metabolism
in amyotrophic lateral sclerosis patients. Sci Rep.
10(12998)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Mead RJ, Shan N, Reiser HJ, Marshall F and
Shaw PJ: Amyotrophic lateral sclerosis: A neurodegenerative
disorder poised for successful therapeutic translation. Nat Rev
Drug Discov. 22:185–212. 2023.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ingre C, Chen L, Zhan Y, Termorshuizen J,
Yin L and Fang F: Lipids, apolipoproteins, and prognosis of
amyotrophic lateral sclerosis. Neurology. 94:e1835–e1844.
2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chełstowska B, Barańczyk-Kuźma A and
Kuźma-Kozakiewicz M: Dyslipidemia in patients with amyotrophic
lateral sclerosis-a case control retrospective study. Amyotroph
Lateral Scler Frontotemporal Degener. 22:195–205. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Michels S, Kurz D, Rosenbohm A, Peter RS,
Just S, Bäzner H, Börtlein A, Dettmers C, Gold HJ, Kohler A, et al:
Association of blood lipids with onset and prognosis of amyotrophic
lateral sclerosis: Results from the ALS Swabia registry. J Neurol.
270:3082–3090. 2023.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xia K, Klose V, Högel J, Huang T, Zhang L,
Dorst J, Fan D and Ludolph A: Lipids and amyotrophic lateral
sclerosis: A two-sample Mendelian randomization study. Eur J
Neurol. 30:1899–1906. 2023.PubMed/NCBI View Article : Google Scholar
|
|
18
|
González De Aguilar JL: Lipid biomarkers
for amyotrophic lateral sclerosis. Front Neurol.
10(284)2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dupuis L, Corcia P, Fergani A, Gonzalez De
Aguilar JL, Bonnefont-Rousselot D, Bittar R, Seilhean D, Hauw JJ,
Lacomblez L, Loeffler JP and Meininger V: Dyslipidemia is a
protective factor in amyotrophic lateral sclerosis. Neurology.
70:1004–1009. 2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Chiò A, Calvo A, Ilardi A, Cavallo E,
Moglia C, Mutani R, Palmo A, Galletti R, Marinou K, Papetti L and
Mora G: Lower serum lipid levels are related to respiratory
impairment in patients with ALS. Neurology. 73:1681–1685.
2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Rafiq MK, Lee E, Bradburn M, McDermott CJ
and Shaw PJ: Effect of lipid profile on prognosis in the patients
with amyotrophic lateral sclerosis: Insights from the olesoxime
clinical trial. Amyotroph Lateral Scler Frontotemporal Degener.
16:478–484. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Abdel-Khalik J, Yutuc E, Crick PJ,
Gustafsson JÅ, Warner M, Roman G, Talbot K, Gray E, Griffiths WJ,
Turner MR and Wang Y: Defective cholesterol metabolism in
amyotrophic lateral sclerosis. J Lipid Res. 58:267–278.
2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chiò A, Logroscino G, Traynor BJ, Collins
J, Simeone JC, Goldstein LA and White LA: Global epidemiology of
amyotrophic lateral sclerosis: A systematic review of the published
literature. Neuroepidemiology. 41:118–130. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Xu L, Chen L, Wang S, Feng J, Liu L, Liu
G, Wang J, Zhan S, Gao P and Fan D: Incidence and prevalence of
amyotrophic lateral sclerosis in urban China: A national
population-based study. J Neurol Neurosurg Psychiatry. 91:520–525.
2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Nakamura R, Kurihara M, Ogawa N, Kitamura
A, Yamakawa I, Bamba S, Sanada M, Sasaki M and Urushitani M:
Investigation of the prognostic predictive value of serum lipid
profiles in amyotrophic lateral sclerosis: Roles of sex and
hypermetabolism. Sci Rep. 12(1826)2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Shefner JM, Al-Chalabi A, Baker MR, Cui
LY, de Carvalho M, Eisen A, Grosskreutz J, Hardiman O, Henderson R,
Matamala JM, et al: A proposal for new diagnostic criteria for ALS.
Clin Neurophysiol. 131:1975–1978. 2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Cedarbaum JM, Stambler N, Malta E, Hilt D,
Thurmond B and Nakanishi A: The ALSFRS-R: A revised ALS functional
rating scale that incorporates assessments of respiratory function.
BDNF ALS study group (phase III). J Neurol Sci. 169:13–21.
1999.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tracey TJ, Kirk SE, Steyn FJ and Ngo ST:
The role of lipids in the central nervous system and their
pathological implications in amyotrophic lateral sclerosis. Semin
Cell Dev Biol. 112:69–81. 2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Burg T and Van Den Bosch L: Abnormal
energy metabolism in ALS: A key player? Curr Opin Neurol.
36:338–345. 2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Delaye JB, Patin F, Piver E, Bruno C,
Vasse M, Vourc'h P, Andres CR, Corcia P and Blasco H: Low IDL-B and
high LDL-1 subfraction levels in serum of ALS patients. J Neurol
Sci. 380:124–127. 2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Dorst J, Kühnlein P, Hendrich C, Kassubek
J, Sperfeld AD and Ludolph AC: Patients with elevated triglyceride
and cholesterol serum levels have a prolonged survival in
amyotrophic lateral sclerosis. J Neurol. 258:613–617.
2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Paganoni S, Deng J, Jaffa M, Cudkowicz ME
and Wills AM: Body mass index, not dyslipidemia, is an independent
predictor of survival in amyotrophic lateral sclerosis. Muscle
Nerve. 44:20–24. 2011.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Huang R, Guo X, Chen X, Zheng Z, Wei Q,
Cao B, Zeng Y and Shang H: The serum lipid profiles of amyotrophic
lateral sclerosis patients: A study from south-west China and a
meta-analysis. Amyotroph Lateral Scler Frontotemporal Degener.
16:359–365. 2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Hu N, Zhang L, Shen D, Yang X, Cui L and
Liu M: Impacts of serum biomarkers regarding glucose, lipid and
protein on progression and survival of amyotrophic lateral
sclerosis: A Chinese cohort study. Neurol Asia. 29:385–396.
2024.
|
|
35
|
Chen X, Wei QQ, Chen Y, Cao B, Ou R, Hou
Y, Yuan X, Zhang L, Liu H and Shang H: Clinical disease stage
related changes of serological factors in amyotrophic lateral
sclerosis. Amyotroph Lateral Scler Frontotemporal Degener.
20:53–60. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Ikeda K, Hirayama T, Takazawa T, Kawabe K
and Iwasaki Y: Relationships between disease progression and serum
levels of lipid, urate, creatinine and ferritin in Japanese
patients with amyotrophic lateral sclerosis: A cross-sectional
study. Intern Med. 51:1501–1508. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yang JW, Kim SM, Kim HJ, Kim JE, Park KS,
Kim SH, Lee KW and Sung JJ: Hypolipidemia in patients with
amyotrophic lateral sclerosis: A possible gender difference? J Clin
Neurol. 9:125–129. 2013.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Choi SJ, Yoon SH, Sung JJ and Lee JH:
Association between fat depletion and prognosis of amyotrophic
lateral sclerosis: CT-based body composition analysis. Ann Neurol.
94:1116–1125. 2023.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Hartmann H, Ho WY, Chang JC and Ling SC:
Cholesterol dyshomeostasis in amyotrophic lateral sclerosis: Cause,
consequence, or epiphenomenon? FEBS J. 289:7688–7709.
2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Yang ST, Kreutzberger AJB, Lee J,
Kiessling V and Tamm LK: The role of cholesterol in membrane
fusion. Chem Phys Lipids. 199:136–143. 2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mouzat K, Molinari N, Kantar J, Polge A,
Corcia P, Couratier P, Clavelou P, Juntas-Morales R, Pageot N,
Lobaccaro JA, et al: Liver X receptor genes variants modulate ALS
phenotype. Mol Neurobiol. 55:1959–1965. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Gu D, Zhou F, You H, Gao J, Kang T, Dixit
D, Wu Q, Yang K, Ci S, Shan D, et al: Sterol regulatory
element-binding protein 2 maintains glioblastoma stem cells by
keeping the balance between cholesterol biosynthesis and uptake.
Neuro Oncol. 25:1578–1591. 2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Ho WY, Chang JC, Lim K, Cazenave-Gassiot
A, Nguyen AT, Foo JC, Muralidharan S, Viera-Ortiz A, Ong SJM, Hor
JH, et al: TDP-43 mediates SREBF2-regulated gene expression
required for oligodendrocyte myelination. J Cell Biol.
220(e201910213)2021.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Scotter EL, Chen HJ and Shaw CE: TDP-43
proteinopathy and ALS: Insights into disease mechanisms and
therapeutic targets. Neurotherapeutics. 12:352–363. 2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Zeng P and Zhou X: Causal effects of blood
lipids on amyotrophic lateral sclerosis: A Mendelian randomization
study. Hum Mol Genet. 28:688–697. 2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Rosina M, Scaricamazza S, Fenili G, Nesci
V, Valle C, Ferri A and Paronetto MP: Hidden players in the
metabolic vulnerabilities of amyotrophic lateral sclerosis. Trends
Endocrinol Metab. 36:1029–1042. 2025.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Dupuis L, Oudart H, René F, Gonzalez de
Aguilar JL and Loeffler JP: Evidence for defective energy
homeostasis in amyotrophic lateral sclerosis: Benefit of a
high-energy diet in a transgenic mouse model. Proc Natl Acad Sci
USA. 101:11159–11164. 2004.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Wills AM, Hubbard J, Macklin EA, Glass J,
Tandan R, Simpson EP, Brooks B, Gelinas D, Mitsumoto H, Mozaffar T,
et al: Hypercaloric enteral nutrition in patients with amyotrophic
lateral sclerosis: A randomised, double-blind, placebo-controlled
phase 2 trial. Lancet. 383:2065–2072. 2014.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Dorst J, Dupuis L, Petri S, Kollewe K,
Abdulla S, Wolf J, Weber M, Czell D, Burkhardt C, Hanisch F, et al:
Percutaneous endoscopic gastrostomy in amyotrophic lateral
sclerosis: A prospective observational study. J Neurol.
262:849–858. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Nelson AT and Trotti D: Altered
bioenergetics and metabolic homeostasis in amyotrophic lateral
sclerosis. Neurotherapeutics. 19:1102–1118. 2022.PubMed/NCBI View Article : Google Scholar
|