Introduction
Gastric cancer causes approximately 800,000 deaths
worldwide per year and is still one of the leading causes of
cancer-related death in the world (1). Most gastric cancers at an early stage
can be cured by surgical resection; however, patients with advanced
gastric cancers have worse prognosis than those with early stage
disease (2). Although metastasis
or relapse is the main cause of death for patients with gastric
cancer (3), the hematogenous
spread of malignant cells remains undetected at the time of initial
therapy. During the development of cancer, tumor cells may detach
from the primary tumor and disseminate into the lymph system and/or
blood circulation, and grow in the bone marrow, liver, kidney and
other organs, which is called micrometastasis (4). Micrometastasis is barely detected by
routine biochemical and histopathological assays or graphical
methods, such as X-ray, CT and MRI (3). Detection of circulating tumor cells
at the mRNA level [reverse transcription-polymerase chain reaction
(RT-PCR)] in blood samples of patients with cancer could serve as a
unique and easy diagnostic tool to predict cancer recurrence and to
monitor treatment effectiveness (5–7).
However, molecular marker(s) that detect circulating gastric cancer
cells for routine clinical use have not yet been identified. Hence,
the development of novel diagnostic molecular marker(s) to detect
circulating gastric cancer cells is an issue of great clinical
importance.
Carcinoembryonic antigen (CEA) is a well-known tumor
marker and has been used to detect small amounts of adenocarcinoma
cells in the blood, peritoneal wash or other body fluids (8–12).
However, the expression of CEA mRNA is not specific to cancer cells
and often produces false-positive results (13). Profiling of gene expression
patterns on genome-wide microarrays enables investigators to
perform comprehensive characterization of molecular activities in
cancer cells (14–17). Systematic analysis of expression
levels for thousands of genes is also a useful approach for
identifying molecular markers to detect small amounts of
circulating cancer cells (18). In
this study, we identified genes whose expression had been altered
during gastric carcinogenesis using genome-wide information
obtained from 8 cases on a microarray consisting of 30,000
transcribed elements. Based on the results of the microarray assay,
we identified five candidate genes for the specific detection of
circulating gastric cancer cells at the mRNA level. We suggest that
such information may lead ultimately to improve the prognosis of
patients with gastric cancers.
Materials and methods
Blood and tissue samples
Blood samples were obtained from 10 patients with
gastric cancer who underwent laparotomy and 4 healthy volunteers
after obtaining informed consent. Heparinized blood samples (5 ml)
from the 10 patients with gastric cancer were obtained from a
peripheral artery through a catheter used for monitoring arterial
blood pressure during surgical operation. Peripheral venous blood
was obtained from 4 healthy volunteers for control after discarding
the initial 10 ml of blood to protect the mixture from epithelial
cells. Clinicopathological characteristics of the 10 patients are
shown in Table I. Clinical stage
of each patient was judged according to the Union for International
Cancer Control (UICC) TNM classification. Among the 10 patients
with gastric cancer, 8 primary gastric cancer tissues and
corresponding non-cancerous gastric mucosae from surgically
resected tissues were obtained at Sapporo Medical University and
Douto Hospital after each patient had provided informed consent.
The samples that had been confirmed histologically as gastric
adenocarcinoma were used for microarray study. These samples were
immediately frozen and stored at −80°C. All cancer tissues were
obtained from the margin of the tumor mass, while non-cancerous
tissues were obtained from corresponding normal mucosae of the same
stomach. This study was approved by the Ethics Committee of Sapporo
Medical University, School of Medicine, Hokkaido, Japan.
 | Table I.Characteristics of patients included
in the nested RT-PCR analysis of PBMCs. |
Table I.
Characteristics of patients included
in the nested RT-PCR analysis of PBMCs.
| Parameters | No. of
patients |
|---|
| Gender
(male:female) | 5:5 |
| Age range
(average), in years | 41–82 (61.9) |
| Depth of tumor
invasion (T1:T2:T3:T4) | 1:6:2:1 |
| Lymph node
metastasis (N0:N1:N2:N3) | 4:3:2:1 |
| Distant metastasis
(M0:M1) | 10:0 |
| Liver metastasis
(H0:H1) | 10:0 |
| Peritoneal
metastasis (P0:P1) | 9:1 |
| Peritoneal lavage
cytology (CY0:CY1) | 9:1 |
| Stage
(I:II:III:IV) | 4:1:2:3 |
| Lymphatic invasion
(ly0:ly1–3) | 2:8 |
| Vessel invasion
(v0:v1–3) | 4:6 |
RNA extraction of blood samples
We prepared peripheral blood mononuclear cells
(PBMCs) using Ficoll (Amersham Biosciences, Buckinghamshire, UK)
and extracted total RNA using TRIzol (Invitrogen, Inc., Carlsbad,
CA, USA) according to the manufacturer’s instructions. Before the
synthesis of cDNA, deoxyribonuclease I (DNase I) (Nippon Gene,
Japan) was added to each sample of total RNA according to the
manufacturer’s instructions.
Analysis of microarray
Total RNA was extracted from each gastric tissue
using TRIzol according to the manufacturer’s instructions. To
guarantee the quality of RNAs, total RNA extracted from the
residual tissue of each case was electrophoresed on a denaturing
agarose gel, and the quality was confirmed by the presence of rRNA
bands. After treatment with DNase I, T7-based RNA amplification was
carried out as described previously with a few modifications
(19). Using 5 μg of total RNA
from each tissue sample as starting material, one round of
amplification was performed; the amount of each amplified RNA
(aRNA) was measured by a spectrophotometer. A mixture of normal
gastric mucosae from 8 patients was prepared as a universal control
and was amplified in the same manner; 2.5 μg of aRNAs from each
cancerous tissue and from the control was reversely transcribed in
the presence of Cy5-dCTP and Cy3-dCTP, respectively (15). AceGene 30K-1 Chip Version (Hitachi
Software Engineering Co., Japan) was used for microarray analysis.
The procedures for hybridization, washing, photometric
quantification of signal intensities of each spot and normalization
of data were according to the manufacturer’s instructions. To
normalize the amount of mRNA between tumors and controls, the
fluorescence intensities of Cy5 (gastric cancer) and Cy3 (control)
for each target spot were adjusted so that the mean Cy5/Cy3 ratio
of 30,000 genes equaled 1. Genes were categorized into three groups
according to the cancer/normal ratio of their mean signal
intensity: up-regulated (expression ratio >5.0), down-regulated
(expression ratio <0.2) and unchanged expression (expression
ratio between 0.2 and 5.0).
Semi-quantitative RT-PCR
To validate the result of the microarray analysis,
we examined the expression levels of the genes up-regulated in
gastric cancer by means of semi-quantitative RT-PCR analysis. Total
RNAs (3 μg) extracted from each cancerous tissue and normal gastric
mucosa were reversely transcribed for single-stranded cDNAs using
oligo(dT)12–18 primer with Superscript II reverse transcriptase
(Life Technologies, Inc.). Each single-stranded cDNA was diluted
for subsequent PCR amplification. A housekeeping gene,
GAPDH, served as the internal control. The PCR reaction was
conducted at 95°C for 5 min, and then for 30 cycles at 95°C for 30
sec, 60°C for 30 sec and 72°C for 1 min followed by 72°C for 10
min, in the Gene Amp PCR System 9700 (Perkin-Elmer Applied
Biosystems, Foster City, CA, USA).
Nested RT-PCR using blood samples
We performed nested RT-PCR using total RNAs
extracted from PBMCs to accurately examine mRNA levels of the
candidate marker genes. Initially, RT-PCR was carried out as
described above. In nested RT-PCR, 1 ml of the initial PCR product,
4 ml of 10X PCR buffer, 200 mmol/l dNTP mixture, 0.2 mmol/l primers
and 1 unit Taq DNA polymerase (Takara) were added to a 40-ml
aliquot of the reaction mixture. The PCR reaction was conducted at
95°C for 5 min, and then 30 cycles at 95°C for 30 sec, 60°C for 30
sec and 72°C for 1 min followed by 72°C for 10 min, in the Gene Amp
PCR System 9700. The RT-PCR products were detected using 2% agarose
gel electrophoresis. The primer sequences are summarized in
Table II.
 | Table II.Primer sequences for
semi-quantitative nested RT-PCR. |
Table II.
Primer sequences for
semi-quantitative nested RT-PCR.
| Gene | Forward primer | Reverse primer |
|---|
| TSPAN8 |
5′-TCAACTTCTTGTTCTGGCTATGT-3′ |
5′-TATAGCTTTGGCATGGTCTCTGC-3′ |
| EPCAM |
5′-TGATCCTGACTGCGATGAGAGC-3′ |
5′-CAGCTTTCAATCACAAATCAGT-3′ |
| MMP12 |
5′-AACCAGCTCTCTGTGACCCCA-3′ |
5′-TCCAAGGATGTTAGGAAGCAAC-3′ |
| MMP7 |
5′-TCTCTGGACGGCAGCTATGCG-3′ |
5′-AATAAGACACAGTCACACCATAA-3′ |
| REG3A |
5′-GTATCTTGGATGCTGCTTTCCTG-3′ |
5′-GTATGACAAAATGAAGAGACTGA-3′ |
| HRH1 |
5′-TACAAGGCCGTACGACAACACT-3′ |
5′-TCTGCTGTTCTTCTATGGTGCCT-3′ |
| EGFR |
5′-ATGTCCCCACGGTACTTACTCCC-3′ |
5′-TCTTAACAATGCTGTAGGGGCTC-3′ |
| CK20 |
5′-TGGATTTCAGTCGCAGA-3′ |
5′-ATGTAGGGTTAGGTCATCAAAG-3′ |
| CEA |
5′-TTCTCCTGGTCTCTCAGCTGGG-3′ |
5′-AATGCTTTAAGGAAGAAGCAA-3′ |
Results
Identification of up- or down-regulated
genes in the gastric cancers
We extracted RNAs from eight primary gastric cancer
tissues and corresponding normal gastric mucosae as control, and
carried out gene expression analysis using a microarray consisting
of 30,000 genes or ESTs. We then selected genes from our data set
according to the criterion that the cancer/ normal ratio of the
mean signal intensity of a given gene was >5.0 or <0.2, and
53 genes were identified as up-regulated and 123 genes as
down-regulated in the gastric cancer tissues compared to the normal
gastric mucosa (Tables III and
IV). The up-regulated genes
represented a variety of functions, including genes associated with
signal-transduction pathways (SFRP4 and TSPAN8),
genes encoding transcription factors (TRIM33), genes
involved in various metabolic pathways (ADH4, USP33,
RNF128, MAN2A1, UBD and GCNT3),
apoptosis (SPP1 and RIPK2), chemokines
(CCL20), DNA replication and recombination (SNRPA1),
cell adhesion and cytoskeleton (LAMB3, EPCAM,
MMP7 and COL1A1), cell-cell signaling (CEACAM6
and CXCL9), cell cycle (CDC2, BUB1 and
CCNB2), cell proliferation (REG1B and REG3A),
or other functions (SPINK4, TMC5, LGALS2,
KYNU, DDX58, LY96, UMPS and
RNF157).
 | Table III.Genes up-regulated in advanced
gastric cancer. |
Table III.
Genes up-regulated in advanced
gastric cancer.
| No. | Accession no. | Gene symbol | Description |
|---|
| 1 | NM_006507 | REG1B | Regenerating
islet-derived 1 β (pancreatic stone protein, pancreatic thread
protein) |
| 2 | NM_004577 | PSPH | Phosphoserine
phosphatase |
| 3 | NM_138938 | REG3A | Regenerating
islet-derived 3 α |
| 4 | NM_014471 | SPINK4 | Serine peptidase
inhibitor, Kazal type 4 |
| 5 | NM_001105249 | TMC5 | Transmembrane
channel-like 5 |
| 6 | NM_002426 | MMP12 | Matrix
metallopeptidase 12 (macrophage elastase) |
| 7 | NM_002423 | MMP7 | Matrix
metallopeptidase 7 (matrilysin, uterine) |
| 8 | NM_002483 | CEACAM6 | Carcinoembryonic
antigen-related cell adhesion molecule 6 (non-specific cross
reacting antigen) |
| 9 | NM_001786 | CDC2 | Cell division cycle
2, G1 to S and G2 to M |
| 10 | NM_000582 | SPP1 | Secreted
phosphoprotein 1 (osteopontin, bone sialoprotein I, early
T-lymphocyte activation 1) |
| 11 | NM_004751 | GCNT3 | Glucosaminyl
(N-acetyl) transferase 3, mucin type |
| 12 | NM_000574 | CD55 | CD55 molecule,
decay accelerating factor for complement (Cromer blood group) |
| 13 | NM_002443 | MSMB | Microseminoprotein,
β |
| 14 | NM_004336 | BUB1 | UB1 budding
uninhibited by benzimidazoles 1 homolog (yeast) |
| 15 | NM_015017 | USP33 | Ubiquitin-specific
peptidase 33 |
| 16 | NM_004591 | CCL20 | Chemokine (C-C
motif) ligand 20 |
| 17 | NM_017633 | FAM46A | Family with
sequence similarity 46, member A |
| 18 | NM_000088 | COL1A1 | Collagen, type I, α
1 |
| 19 | XR_017717 |
ADAMTSL3 | ADAMTS-like 3 |
| 20 | NM_138938 | REG3A | Regenerating
islet-derived 3 α |
| 21 | NM_017934 | PHIP | Pleckstrin homology
domain interacting protein |
| 22 | XR_016124 | | Similar to
p21-activated kinase 2 |
| 23 | NM_006398 | UBD | Ubiquitin D |
| 24 | NM_002358 | MAD2L1 | MAD2 mitotic arrest
deficient-like 1 (yeast) |
| 25 | NM_002483 | CEACAM6 | Carcinoembryonic
antigen-related cell adhesion molecule 6 (non-specific
cross-reacting antigen) |
| 26 | NM_173164 | IPO9 | Importin 9 |
| 27 | NM_003014 | SFRP4 | Secreted
frizzled-related protein 4 |
| 28 | NM_004616 | TSPAN8 | Tetraspanin 8 |
| 29 | NM_002354 | EPCAM | Epithelial cell
adhesion molecule |
| 30 | NM_006498 | LGALS2 | Lectin,
galactoside-binding, soluble, 2 |
| 31 | NM_002372 | MAN2A1 | Mannosidase, α,
class 2A, member 1 |
| 32 | NM_003937 | KYNU | Kynureninase
(L-kynurenine hydrolase) |
| 33 | NM_003821 | RIPK2 |
Receptor-interacting serine-threonine
kinase 2 |
| 34 | NM_00108039 | ITGA7 | Integrin, α 7 |
| 35 | NM_000670 | ADH4 | Alcohol
dehydrogenase 4 (class II), π polypeptide |
| 36 | NM_014314 | DDX58 | DEAD
(Asp-Glu-Ala-Asp) box polypeptide 58 |
| 37 | NM_006418 | OLFM4 | Olfactomedin 4 |
| 38 | NM_198187.3 | ASTN2 | Astrotactin 2 |
| 39 | NM_015364 | LY96 | Lymphocyte antigen
96 |
| 40 | NM_000574 | CD55 | CD55 molecule,
decay accelerating factor for complement (Cromer blood group) |
| 41 | NM_018964 | SLC37A1 | Solute carrier
family 37 (glycerol-3-phosphate transporter), member 1 |
| 42 | NM_018455 | CENPN | Centromere protein
N |
| 43 | NM_001710 | CFB | Complement factor
B |
| 44 | NM_033020 | TRIM33 | Tripartite
motif-containing 33 |
| 45 | NM_003090 | SNRPA1 | Small nuclear
ribonucleoprotein polypeptide A' |
| 46 | NM_000373 | UMPS | Uridine
monophosphate synthetase (orotate phosphoribosyl transferase and
orotidine-5′-decarboxylase) |
| 47 | NM_144584 | C1orf59 | Chromosome 1 open
reading frame 59 |
| 48 | NM_052916.2 | RNF157 | Ring finger protein
157 |
| 49 | NM_006332 | IFI30 | Interferon,
γ-inducible protein 30 |
| 50 | NM_002416 | CXCL9 | Chemokine (C-X-C
motif) ligand 9 |
| 51 | NM_001017402 | LAMB3 | Laminin, β 3 |
| 52 | NM_004701 | CCNB2 | Cyclin B2 |
| 53 | NM_194463 | RNF128 | Ring finger protein
128 |
 | Table IV.Genes down-regulated in advanced
gastric cancer. |
Table IV.
Genes down-regulated in advanced
gastric cancer.
| No. | Accession no. | Gene symbol | Description |
|---|
| 1 | NM_004190 | LIPF | Lipase,
gastric |
| 2 | NM_020143 | PNO1 | Partner of NOB1
homolog (S. cerevisiae) |
| 3 | NM_000257 | MYH7 | Myosin, heavy chain
7, cardiac muscle, β |
| 4 | NM_015173 | TBC1D1 | TBC1 (tre-2/USP6,
BUB2, cdc16) domain family, member 1 |
| 5 | NM_005408 | CCL13 | Chemokine (C-C
motif) ligand 13 |
| 6 | NM_174929 | ZMIZ2 | Zinc finger,
MIZ-type containing 2 |
| 7 | NM_004747 | DLG5 | Discs, large
homolog 5 (Drosophila) |
| 8 | NM_024872.2 | DOK3 | Docking protein
3 |
| 9 | NM_201653 | CHIA | Chitinase,
acidic |
| 10 | NM_003893 | LDB1 | LIM domain binding
1 |
| 11 | NM_012455.2 | PSD4 | Pleckstrin and Sec7
domain containing 4 |
| 12 | NM_005213 | CSTA | Cystatin A (stefin
A) |
| 13 | NM_005416 | SPRR3 | Small proline-rich
protein 3 |
| 14 | NM_014989 | RIMS1 | Regulating synaptic
membrane exocytosis 1 |
| 15 | NM_001018005 | TPM1 | Tropomyosin 1
(α) |
| 16 | NM_213589 | RAPH1 | Ras association
(RalGDS/AF-6) and pleckstrin homology domains 1 |
| 17 | NM_004898 | CLOCK | Clock homolog
(mouse) |
| 18 | NM_013292 | | Fast skeletal
myosin light chain 2 |
| 19 | NM_020321 | ACCN3 | Amiloride-sensitive
cation channel 3 |
| 20 | NM_002754 | MAPK13 | Mitogen-activated
protein kinase 13 |
| 21 | NM_013443 |
ST6GALNAC6 | ST6
(α-N-acetyl-neuraminyl-2,3-β-galactosyl-1,3)-N-acetylgalactosaminide
α-2,6-sialyltransferase 6 |
| 22 | NM_001042453 | | Serine/threonine
protein kinase MST4 |
| 23 | NM_032646 | TTYH2 | Tweety homolog 2
(Drosophila) |
| 24 | NM_015089 | | p53-associated
parkin-like cytoplasmic protein |
| 25 | NM_003609 | HIRIP3 | HIRA interacting
protein 3 |
| 26 | NR_002219 | BIRC5 | Baculoviral IAP
repeat-containing 5 (survivin) |
| 27 | NM_000068 | CACNA1A | Calcium channel,
voltage-dependent, P/Q type, α 1A subunit |
| 28 | NM_203377 | MB | Myoglobin |
| 29 | NM_003768 | PEA15 | Phosphoprotein
enriched in astrocytes 15 |
| 30 | NM_053013 | ENO3 | Enolase 3 (β,
muscle) |
| 31 | XR_018802 | PI4K2A |
Phosphatidylinositol 4-kinase type 2
α |
| 32 | NM_003725 | HSD17B6 | Hydroxysteroid
(17-β) dehydrogenase 6 homolog (mouse) |
| 33 | NM_006063 | KBTBD10 | Kelch repeat and
BTB (POZ) domain containing 10 |
| 34 | NM_012288 | TRAM2 | Translocation
associated membrane protein 2 |
| 35 | NM_000730 | CCKAR | Cholecystokinin A
receptor |
| 36 | NM_000290 | PGAM2 | Phosphoglycerate
mutase 2 (muscle) |
| 37 | NM_199354 | PRB1 | Proline-rich
protein BstNI subfamily 1 |
| 38 | XR_019039 | ACTB | Actin, β |
| 39 | NM_006478 | GAS2L1 | Growth
arrest-specific 2 like 1 |
| 40 | NM_024674 | LIN28 | Lin-28 homolog
(C. elegans) |
| 41 | NM_001070 | TUBG1 | Tubulin, γ 1 |
| 42 | NM_015654 | NAT9 | N-acetyltransferase
9 |
| 43 | NM_003643 | GCM1 | Glial cells missing
homolog 1 (Drosophila) |
| 44 | NM_006901.2 | MYO9A | Myosin IXA |
| 45 | NM_017785 | CCDC99 | Coiled-coil domain
containing 99 |
| 46 | NM_025135 | FHOD3 | Formin homology 2
domain containing 3 |
| 47 | NM_022566 | MESDC1 | Mesoderm
development candidate 1 |
| 48 | NM_198255 | TERT | Telomerase reverse
transcriptase |
| 49 | NM_018231 | | Amino acid
transporter |
| 50 | NM_002458 | MUC5B | Mucin 5B,
oligomeric mucus/gel-forming |
| 51 | NM_001001522 | TAGLN | Transgelin |
| 52 | NM_002631 | PGD | Phosphogluconate
dehydrogenase |
| 53 | NM_006984 | CLDN10 | Claudin 10 |
| 54 | NM_004359 | CDC34 | Cell division cycle
34 homolog (S. cerevisiae) |
| 55 | NM_001824 | CKM | Creatine kinase,
muscle |
| 56 | NM_002274 | KRT13 | Keratin 13 |
| 57 | XR_019039 | ACTB | Actin, β |
| 58 | NM_000477 | ALB | Albumin |
| 59 | NM_001519.2 | BRF1 | BRF1 homolog,
subunit of RNA polymerase III transcription initiation factor IIIB
(S. cerevisiae) |
| 60 | NM_006790 | MYOT | Myotilin |
| 61 | NM_021948 | BCAN | Brevican |
| 62 | NM_001142404.1 | CD164 | CD164 molecule,
sialomucin |
| 63 | BC050364.1 | C7orf13 | Chromosome 7 open
reading frame 13 |
| 64 | NM_005177 |
ATP6V0A1 | ATPase,
H+ transporting, lysosomal V0 subunit a1 |
| 65 | NM_020393 | PGLYRP4 | Peptidoglycan
recognition protein 4 |
| 66 | XM_937007 | FRMPD3 | FERM and PDZ domain
containing 3 |
| 67 | NM_024754 | PTCD2 | Pentatricopeptide
repeat domain 2 |
| 68 | NM_001098511 | KIF2A | Kinesin heavy chain
member 2A |
| 69 | NM_025058 | TRIM46 | Tripartite
motif-containing 46 |
| 70 | AK126458.1 | MYO15B | Myosin XVB
pseudogene |
| 71 | NM_018659 | CYTL1 | Cytokine-like
1 |
| 72 | NM_002965 | S100A9 | S100 calcium
binding protein A9 |
| 73 | NM_032566 | SPINK7 | Serine peptidase
inhibitor, Kazal type 7 (putative) |
| 74 | NM_001669 | ARSD | Arylsulfatase
D |
| 75 | NM_206820 | MYBPC1 | Myosin binding
protein C, slow type |
| 76 | NM_003200 | TCF3 | Transcription
factor 3 (E2A immunoglobulin enhancer binding factors E12/E47) |
| 77 | NM_031413 | CECR2 | Cat eye syndrome
chromosome region, candidate 2 |
| 78 | NM_017539 | DNAH3 | Dynein, axonemal,
heavy chain 3 |
| 79 | NM_017426 | NUP54 | Nucleoporin 54
kDa |
| 80 | NM_002020 | FLT4 | Fms-related
tyrosine kinase 4 |
| 81 | NM_007320 | RANBP3 | RAN binding protein
3 |
| 82 | NM_005286 | NPBWR2 | Neuropeptides B/W
receptor 2 |
| 83 | NM_006428 | MRPL28 | Mitochondrial
ribosomal protein L28 |
| 84 | NM_014280.2 | DNAJC8 | DnaJ (Hsp40)
homolog, subfamily C, member 8 |
| 85 | NM_020679 | MIF4GD | MIF4G domain
containing |
| 86 | NM_001823 | CKB | Creatine kinase,
brain |
| 87 | NM_000477 | ALB | Albumin |
| 88 | NM_001927 | DES | Desmin |
| 89 | NM_005416 | SPRR3 | Small proline-rich
protein 3 |
| 90 | NM_022468 | MMP25 | Matrix
metallopeptidase 25 |
| 91 | NM_016599 | MYOZ2 | Myozenin 2 |
| 92 | NM_000243 | MEFV | Mediterranean
fever |
| 93 | NM_002272 | KRT4 | Keratin 4 |
| 94 | NM_003279 | TNNC2 | Troponin C type 2
(fast) |
| 95 | NM_006685 | SMR3B | Submaxillary gland
androgen regulated protein 3 homolog B (mouse) |
| 96 | NM_014760 | TATDN2 | TatD DNase domain
containing 2 |
| 97 | NM_006928 | SILV | Silver homolog
(mouse) |
| 98 | NM_016522 | | Neurotrimin |
| 99 | NM_000760 | CSF3R | Colony stimulating
factor 3 receptor (granulocyte) |
| 100 | NM_003167 | SULT2A1 | Sulfotransferase
family, cytosolic, 2A, dehydroepiandrosterone (DHEA)-preferring,
member 1 |
| 101 | NM_183360 | DTNB | Dystrobrevin,
β |
| 102 | NM_001711 | BGN | Biglycan |
| 103 | NM_023077 |
C1orf163 | Chromosome 1 open
reading frame 163 |
| 104 | NM_015926.4 | TEX264 | Testis expressed
264 |
| 105 | NM_006757 | TNNT3 | Troponin T type 3
(skeletal, fast) |
| 106 | NM_002675 | PML | Promyelocytic
leukemia |
| 107 | XR_018113 | GAPDH |
Glyceraldehyde-3-phosphate
dehydrogenase |
| 108 | NM_021245 | MYOZ1 | Myozenin 1 |
| 109 | NM_000383 | AIRE | Autoimmune
regulator |
| 110 | NM_006846 | SPINK5 | Serine peptidase
inhibitor, Kazal type 5 |
| 111 | XM_939725 | AP1S2 | Adaptor-related
protein complex 1, sigma 2 subunit |
| 112 | NM_024505 | NOX5 | NADPH oxidase,
EF-hand calcium binding domain 5 |
| 113 | NM_020145 | SH3GLB2 | SH3-domain
GRB2-like endophilin B2 |
| 114 | NM_016192 | TMEFF2 | Transmembrane
protein with EGF-like and two follistatin-like domains 2 |
| 115 | NM_006472 | TXNIP | Thioredoxin
interacting protein |
| 116 | NM_031866 | FZD8 | Frizzled homolog 8
(Drosophila) |
| 117 | NM_003808 | TNFSF13 | Tumor necrosis
factor (ligand) superfamily, member 13 |
| 118 | NM_015503 | SH2B1 | SH2B adaptor
protein 1 |
| 119 | NM_014047 |
C19orf53 | Chromosome 19 open
reading frame 53 |
| 120 | NM_022754 | SFXN1 | Sideroflexin 1 |
| 121 | NM_003061 | SLIT1 | Slit homolog 1
(Drosophila) |
| 122 | NM_003047 | SLC9A1 | Solute carrier
family 9 (sodium/hydrogen exchanger), member 1 (antiporter,
Na+/H+, amiloride sensitive) |
| 123 | NM_021991 | JUP | Junction
plakoglobin |
On the other hand, the down-regulated genes included
those associated with various metabolic pathways (CKM,
ARSD and BGN), small molecule transport
(ACCN3, ATP6V0A1 and SFXN1), signal
transduction (FLT4, NPBWR2, CSF3R and
FZD8), cell cycle regulation (TBC1D1, DLG5,
GAS2L1, CDC34 and SH2B1), cell adhesion
(RAPH1, CLDN10, BCAN, CD164 and
JUP), transcription factors (LDB1, CLOCK,
GCM1, BRF1, TCF3, PML and AIRE),
cell-cell signaling (PGD, S100A9, CCL13 and
RIMS1) or other functions.
Identification of candidate genes as
molecular markers for the detection of circulating gastric cancer
cells in human peripheral blood
Of the 53 genes that were up-regulated in the
gastric cancer compared to the normal gastric tissues, we
identified five candidate marker genes [tetraspanin 8
(TSPAN8), epithelial cell adhesion molecule (EPCAM),
matrix metallopeptidase 12 (MMP12), matrix metallopeptidase
7 (MMP7) and regenerating islet-derived 3 α (REG3A)]
for the detection of circulating gastric cancer cells in peripheral
blood in accordance with the following criteria: i) no or weak
expression in human normal tissues in the published database
(20), ii) no expression in PBMCs
from 4 healthy volunteers by nested RT-PCR. In addition to the
above five newly identified genes, we analyzed histamine receptor
H1 (HRH1) since a previous study reported that this gene was
overexpressed in gastric cancer cells, and the expression of this
gene satisfied the above criteria (21). Moreover, three candidate marker
genes [keratin 20 (CK20), epidermal growth factor receptor
(EGFR) and carcinoembryonic antigen (CEA)], which
have been reported to be promising markers for the detection of
cancer cells, were further analyzed (11,13,22,23).
Association of the expression of the nine
marker genes for the detection of circulating gastric cancer cells
with clinicopathological parameters by nested RT-PCR
We carried out semi-quantitative nested RT-PCR
analysis of the nine candidate marker genes for the detection of
circulating cancer cells using PBMCs of patients with gastric
cancer. Of the nine candidate genes, the expression of MMP12
and CEA mRNAs was positive in 40% of the patients with
gastric cancer. However, the expression of the other seven genes
was positive in ≤30% of the patients, respectively (Table V). We then investigated a combined
effect of the expression of the nine genes on the detection of
circulating cancer cells. Expression of one or more genes out of
the nine was detected in 80% of the patients with gastric cancer by
nested RT-PCR (Table VI).
 | Table V.Positive ratio of the nine marker
genes for detection of circulating gastric cancer cells. |
Table V.
Positive ratio of the nine marker
genes for detection of circulating gastric cancer cells.
| Marker genes | TSPAN8 | EPCAM | HRH1 | CK20 | MMP12 | MMP7 | EGFR | REG3A | CEA |
|---|
| Positive cases
(%) | 20 | 30 | 30 | 20 | 40 | 10 | 10 | 20 | 40 |
 | Table VI.Number of positive genes in 10 cases
by nested RT-PCR. |
Table VI.
Number of positive genes in 10 cases
by nested RT-PCR.
| Cases | GC-1 | GC-2 | GC-3 | GC-4 | GC-5 | GC-6 | GC-7 | GC-8 | GC-9 | GC-10 |
|---|
| No. of positive
genes | 1 | 0 | 2 | 3 | 5 | 6 | 1 | 2 | 0 | 2 |
We further investigated the association of the
expression of the nine candidate marker genes with
clinicopathological parameters of the 10 cases. We focused on four
parameters: vascular invasion (v factor), lymphatic invasion (ly
factor), lymph node metastasis (N factor) and pathological stage
I–IV, and investigated the association of these parameters with the
total number of positive genes in the PBMCs of each patient
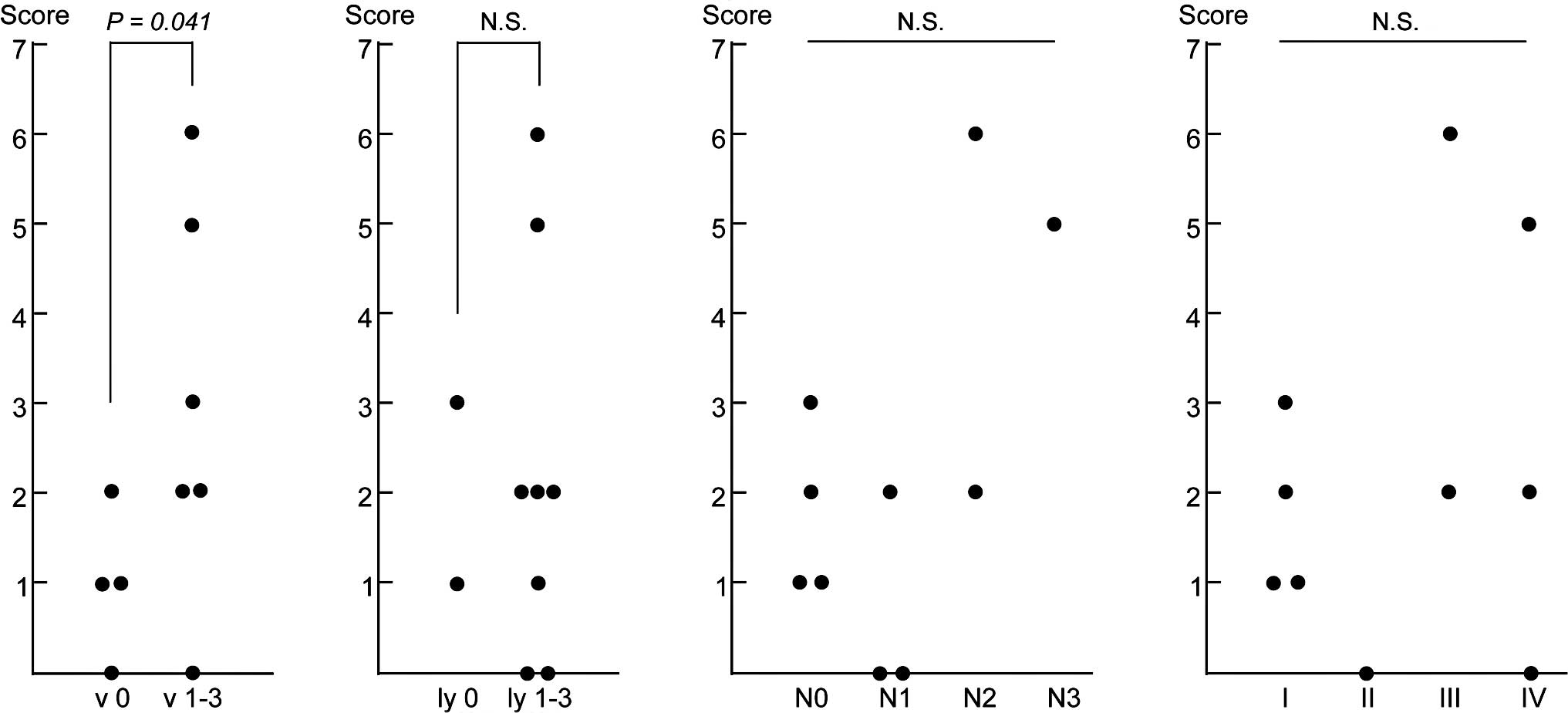
(Fig. 1). Of the four parameters,
the numbers of genes expressed in the PBMCs were ≤2 in all of the
vascular invasion-negative cases (v 0), while the numbers of genes
were ≥2 in 5 of 6 positive cases (v 1–3), exhibiting a significant
difference between the two groups (P=0.041; Fig. 1A). However, no significant
association was observed for the other three parameters (Fig. 1B–D), suggesting that the combined
expression analysis of the nine marker genes using PBMCs detected
micrometastasis through vascular invasion in the primary gastric
cancer tissues.
Discussion
Microarrays, at present, are widely used to analyze
expression of thousands of genes simultaneously in cancer tissues.
In the present study, we identified five genes (TSPAN8,
EPCAM, MMP12, MMP7 and REG3A) as
potential markers for the detection of circulating cancer cells in
the peripheral blood of patients with gastric cancer through
genome-wide gene expression profiling in combination with nested
RT-PCR. Some of these genes have previously been reported to be
up-regulated in gastric cancer cells; however, they have not
previously been designated for the detection of circulating gastric
cancer cells by nested RT-PCR. Furthermore, the combined expression
analysis of the five genes and four previously reported marker
genes, HRH1, EGFR, CK20 and CEA,
revealed that one or more mRNAs among the nine genes could be
detected in 80% of the patients with gastric cancer by nested
RT-PCR, suggesting that a set of nine marker genes is more
sensitive than a single marker gene for detection of circulating
gastric cancer cells. In this study, we did not investigate the
association of distant metastasis with expression of the nine
marker genes since no patients had distant metastasis among the 10
studied patients. Although we could not exclude false-positive
cases due to non-malignant epithelial cells which may have
contaminated the blood samples during collection and which may have
expressed the targeted transcripts (18), pathological v factor showed a
significant association with the total number of marker genes
expressed in the PBMCs of the patients. Hence, the set of nine
marker genes may be promising for the detection of minimal amounts
of circulating gastric cancer cells prior to the metastatic growth
of gastric cancer cells in organ(s).
Among the five marker genes which were newly
identified in the microarray analysis, we identified epithelial
cell adhesion molecule (EPCAM) which is a member of a family
of type I membrane proteins and pan-epithelial differentiation
antigen expressed in many types of carcinomas (24–28).
Magnetic beads or structures coated with EPCAM monoclonal
antibodies have been recently used for circulating cancer cell
separation (29–31). Although we did not compare the
accuracy of the detection of gastric cancer cells by these methods
to that of nested RT-PCR since we did not conduct the former
assays, 30% of patients with gastric cancer exhibited
EPCAM-positivity in PBMCs by nested RT-PCR. Further clinical
study investigating the relationship between the clinical outcome
of patients and EPCAM expression in PBMCs by nested RT-PCR
may clarify whether this method could be clinically applied for the
detection of circulating gastric cancer cells. Two matrix
metalloproteinases, MMP7 and MMP12, were among the
five marker genes which were newly identified in this study. MMPs
are a family of zinc-dependent proteolytic enzymes capable of
cleaving extracellular matrix proteins, and the expression of MMPs
in cancer tissue has been reported to be associated with the risk
of metastasis (32–38). These two MMPs may play important
roles in tumor invasion and the formation of metastasis in gastric
cancer.
In conclusion, five novel marker genes were
designated for the detection of circulating gastric cancer cells.
The nested RT-PCR analysis for the set of nine marker genes,
TSPAN8, EPCAM, MMP12, MMP7,
REG3A, HRH1, EGFR, CK20 and CEA,
using PBMCs of patients with gastric cancer may provide the
potential for the detection of circulating gastric cancer cells
prior to the formation of metastasis in other organs. Our data
suggest that early detection and personalized therapy for gastric
cancers, by prescribing the appropriate treatment to patients with
a high risk of metastasis, may be achievable by utilizing specific
sets of marker genes according to the approach shown here.
Acknowledgements
We thank Tomohisa Furuhata, Yasutoshi
Kimura, Chikashi Kihara, Kenji Okita and Noriko Nishikawa for the
helpful discussions.
References
|
1.
|
Thun MJ, DeLancey JO, Center MM, Jemal A
and Ward EM: The global burden of cancer: priorities for
prevention. Carcinogenesis. 31:100–110. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Ott K, Lordick F, Blank S and Buchler M:
Gastric cancer: surgery in 2011. Langenbecks Arch Surg. Jan.
14–2011.(E-pub ahead of print).
|
|
3.
|
Chen XM, Chen GY, Wang ZR, Zhu FS, Wang XL
and Zhang X: Detection of micrometastasis of gastric carcinoma in
peripheral blood circulation. World J Gastroenterol. 10:804–808.
2004.PubMed/NCBI
|
|
4.
|
Joyce JA and Pollard JW:
Microenvironmental regulation of metastasis. Nat Rev Cancer.
9:239–252. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Vardakis N, Messaritakis I, Papadaki C, et
al: Prognostic significance of the detection of peripheral blood
CEACAM5 mRNA-positive cells by real-time polymerase chain reaction
in operable colorectal cancer. Clin Cancer Res. 17:165–173. 2011.
View Article : Google Scholar
|
|
6.
|
Miyazono F, Natsugoe S, Takao S, et al:
Surgical maneuvers enhance molecular detection of circulating tumor
cells during gastric cancer surgery. Ann Surg. 233:189–194. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Pantel K, Brakenhoff RH and Brandt B:
Detection, clinical relevance and specific biological properties of
disseminating tumour cells. Nat Rev Cancer. 8:329–340. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Qiu MZ, Li ZH, Zhou ZW, et al: Detection
of carcinoembryonic antigen messenger RNA in blood using
quantitative real-time reverse transcriptase-polymerase chain
reaction to predict recurrence of gastric adenocarcinoma. J Transl
Med. 8:1072010. View Article : Google Scholar
|
|
9.
|
Guadagni F, Kantor J, Aloe S, et al:
Detection of blood-borne cells in colorectal cancer patients by
nested reverse transcription-polymerase chain reaction for
carcinoembryonic antigen messenger RNA: longitudinal analyses and
demonstration of its potential importance as an adjunct to multiple
serum markers. Cancer Res. 61:2523–2532. 2001.
|
|
10.
|
Ikeguchi M and Kaibara N: Detection of
circulating cancer cells after a gastrectomy for gastric cancer.
Surg Today. 35:436–441. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Dardaei L, Shahsavani R, Ghavamzadeh A, et
al: The detection of disseminated tumor cells in bone marrow and
peripheral blood of gastric cancer patients by multimarker (CEA,
CK20, TFF1 and MUC2) quantitative real-time PCR. Clin Biochem.
44:325–330. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Katsuragi K, Yashiro M, Sawada T, Osaka H,
Ohira M and Hirakawa K: Prognostic impact of PCR-based
identification of isolated tumour cells in the peritoneal lavage
fluid of gastric cancer patients who underwent a curative R0
resection. Br J Cancer. 97:550–556. 2007. View Article : Google Scholar
|
|
13.
|
Mori K, Aoyagi K, Ueda T, et al: Highly
specific marker genes for detecting minimal gastric cancer cells in
cytology negative peritoneal washings. Biochem Biophys Res Commun.
313:931–937. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Zembutsu H, Ohnishi Y, Daigo Y, et al:
Gene-expression profiles of human tumor xenografts in nude mice
treated orally with the EGFR tyrosine kinase inhibitor ZD1839. Int
J Oncol. 23:29–39. 2003.PubMed/NCBI
|
|
15.
|
Zembutsu H, Ohnishi Y, Tsunoda T, et al:
Genome-wide cDNA microarray screening to correlate gene expression
profiles with sensitivity of 85 human cancer xenografts to
anticancer drugs. Cancer Res. 62:518–527. 2002.PubMed/NCBI
|
|
16.
|
Zembutsu H, Suzuki Y, Sasaki A, et al:
Predicting response to docetaxel neoadjuvant chemotherapy for
advanced breast cancers through genome-wide gene expression
profiling. Int J Oncol. 34:361–370. 2009.PubMed/NCBI
|
|
17.
|
Zembutsu H, Yanada M, Hishida A, et al:
Prediction of risk of disease recurrence by genome-wide cDNA
microarray analysis in patients with Philadelphia
chromosome-positive acute lymphoblastic leukemia treated with
imatinib-combined chemotherapy. Int J Oncol. 31:313–322. 2007.
|
|
18.
|
Obermayr E, Sanchez-Cabo F, Tea MK, et al:
Assessment of a six gene panel for the molecular detection of
circulating tumor cells in the blood of female cancer patients.
BMC. 10:6662010.PubMed/NCBI
|
|
19.
|
Luo L, Salunga RC, Guo H, et al: Gene
expression profiles of laser-captured adjacent neuronal subtypes.
Nat Med. 5:117–122. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Saito-Hisaminato A, Katagiri T, Kakiuchi
S, Nakamura T, Tsunoda T and Nakamura Y: Genome-wide profiling of
gene expression in 29 normal human tissues with a cDNA microarray.
DNA Res. 9:35–45. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Hasegawa S, Furukawa Y, Li M, et al:
Genome-wide analysis of gene expression in intestinal-type gastric
cancers using a complementary DNA microarray representing 23,040
genes. Cancer Res. 62:7012–7017. 2002.PubMed/NCBI
|
|
22.
|
Amin AT, Shiraishi N, Ninomiya S, Tajima
M, Inomata M and Kitano S: Increased mRNA expression of epidermal
growth factor receptor, human epidermal receptor, and survivin in
human gastric cancer after the surgical stress of laparotomy versus
carbon dioxide pneumoperitoneum in a murine model. Surg Endosc.
24:1427–1433. 2010. View Article : Google Scholar
|
|
23.
|
Mori M, Mimori K, Inoue H, et al:
Detection of cancer micrometastases in lymph nodes by reverse
transcriptase-polymerase chain reaction. Cancer Res. 55:3417–3420.
1995.PubMed/NCBI
|
|
24.
|
Trebak M, Begg GE, Chong JM, Kanazireva
EV, Herlyn D and Speicher DW: Oligomeric state of the colon
carcinoma-associated glycoprotein GA733-2 (Ep-CAM/EGP40) and its
role in GA733-mediated homotypic cell-cell adhesion. J Biol Chem.
276:2299–2309. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Maghzal N, Vogt E, Reintsch W, Fraser JS
and Fagotto F: The tumor-associated EpCAM regulates morphogenetic
movements through intracellular signaling. J Cell Biol.
191:645–659. 2010. View Article : Google Scholar
|
|
26.
|
Du W, Ji H, Cao S, et al: EpCAM: a
potential antimetastatic target for gastric cancer. Dig Dis Sci.
55:2165–2171. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Kuhn S, Koch M, Nubel T, et al: A complex
of EpCAM, claudin-7, CD44 variant isoforms, and tetraspanins
promotes colorectal cancer progression. Mol Cancer Res. 5:553–567.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Mukherjee S, Richardson AM,
Rodriguez-Canales J, et al: Identification of EpCAM as a molecular
target of prostate cancer stroma. Am J Pathol. 175:2277–2287. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Sha MY, Xu H, Natan MJ and Cromer R:
Surface-enhanced Raman scattering tags for rapid and homogeneous
detection of circulating tumor cells in the presence of human whole
blood. J Am Chem Soc. 130:17214–17215. 2008. View Article : Google Scholar
|
|
30.
|
Myung JH, Launiere CA, Eddington DT and
Hong S: Enhanced tumor cell isolation by a biomimetic combination
of E-selectin and anti-EpCAM: implications for the effective
separation of circulating tumor cells (CTCs). Langmuir.
26:8589–8596. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Hosokawa M, Hayata T, Fukuda Y, et al:
Size-selective microcavity array for rapid and efficient detection
of circulating tumor cells. Anal Chem. 82:6629–6635. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Shi WD, Meng ZQ, Chen Z, Lin JH, Zhou ZH
and Liu LM: Identification of liver metastasis-related genes in a
novel human pancreatic carcinoma cell model by microarray analysis.
Cancer Lett. 283:84–91. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Szarvas T, Becker M, vom Dorp F, et al:
Matrix metallo-proteinase-7 as a marker of metastasis and predictor
of poor survival in bladder cancer. Cancer Sci. 101:1300–1308.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Oshima T, Akaike M, Yoshihara K, et al:
Clinicopathological significance of the gene expression of matrix
metalloproteinase-7, insulin-like growth factor-1, insulin-like
growth factor-2 and insulin-like growth factor-1 receptor in
patients with colorectal cancer: insulin-like growth factor-1
receptor gene expression is a useful predictor of liver metastasis
from colorectal cancer. Oncol Rep. 20:359–364. 2008.
|
|
35.
|
Fang YJ, Lu ZH, Wang GQ, et al: Elevated
expressions of MMP7, TROP2, and survivin are associated with
survival, disease recurrence, and liver metastasis of colon cancer.
Int J Colorectal Dis. 24:875–884. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Kerkela E, Ala-aho R, Klemi P, et al:
Metalloelastase (MMP-12) expression by tumour cells in squamous
cell carcinoma of the vulva correlates with invasiveness, while
that by macrophages predicts better outcome. J Pathol. 198:258–269.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Balaz P, Friess H, Kondo Y, Zhu Z,
Zimmermann A and Buchler MW: Human macrophage metalloelastase
worsens the prognosis of pancreatic cancer. Ann Surg. 235:519–527.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Hofmann HS, Hansen G, Richter G, et al:
Matrix metalloproteinase-12 expression correlates with local
recurrence and metastatic disease in non-small cell lung cancer
patients. Clin Cancer Res. 11:1086–1092. 2005.PubMed/NCBI
|