Introduction
Schizophrenia is a severe, debilitating, psychiatric
disorder. Although the exact etiology of schizophrenia is unknown,
twin, family and adoption studies have provided consistent evidence
that genetic factors play a major role in the pathogenesis of
schizophrenia.
In a recent study, the data showed that
polymorphisms of several genes affect gene expression or the
function of the encoded protein in the human brain (1). Clinical, epidemiological,
neuroimaging and postmortem data suggest that schizophrenia is a
neurodevelopmental disorder, and that synaptic disturbances may
play a critical role in the development of the disease (2). In addition, it is hypothesized that a
loss of function of glial gap junctions may cause severe cognitive
impairment in schizophrenia. Thus, several gap junction proteins
may be capable of differentiating between the operation qualities
of the cognate synapses defined by the neurotransmitter types
(3). The major component for
barrier-forming tight junctions may play a crucial role in response
to the changing natural, physiological and pathological conditions
of schizophrenia (4).
In this study, we investigated whether par-3
partitioning defective 3 homolog (C. elegans) (PARD3)
is associated with schizophrenia in a Korean population.
Subjects and methods
Subjects
All subjects were ethnically Korean and unrelated to
each other. A total of 204 Korean schizophrenic patients [117 male,
41.1±9.6 years (mean age ± SD); 87 female, 42.6±11.5] and 351
control subjects (170 male, 43.8±6.6 years; 181 female, 44.2±5.8)
were enrolled in the study. All patients were diagnosed with
schizophrenia by three psychiatrists according to the Diagnostic
and Statistical Manual of Mental Disorders (4th edition) using all
available information from interviews and clinical records. All
patients were assessed using the Operational Criteria Checklist for
Psychotic Illness (OPCRIT). The OPCRIT was designed as a best
estimate diagnostic procedure, in which symptom and course features
are coded by an experienced clinician, allowing integration of the
relative prominence of clinical features over the entire course of
the illness. The control subjects were recruited after they had
been assessed as mentally healthy in a general health examination
program. Written informed consent was obtained from all subjects
involved in the study, and the study was approved by the Ethics
Review Committee of the Medical Research Institute, Kyung Hee
University Medical Center, Seoul, Republic of Korea. All studies
were carried out according to the guidelines of the Declaration of
Helsinki.
SNP selection and genotyping
We searched the SNPs of the PARD3 gene in the
SNP database of the National Center for Biotechnology Information
(http://www.ncbi.nlm.nih.gov/ SNP, BUILD
132). We selected nine SNPs [rs7075263 (intron), rs10827392
(intron), rs773970 (intron), rs2252655 (intron), rs10763984
(intron), rs3781128 (Ser889Ser), rs1936429 (intron), rs671228
(intron) and rs16935163 (intron)] in the PARD3 gene. SNP
genotyping was conducted using direct sequencing. For direct
sequencing, genomic DNA was amplified using polymerase chain
reaction (PCR). The PCR products were sequenced by an ABI PRISM
3730XL analyzer (PE Applied Biosystems, Foster City, CA, USA), and
sequence data were analyzed using SeqManII software (DNASTAR Inc.,
Madison, WI, USA).
Statistical analysis
The Chi-square test was used to assess
Hardy-Weinberg equilibrium (HWE). Genetic data were analyzed using
SNPStats (http://bioinfo.iconcologia.net/index.php?module=Snpstats),
Helixtree (Golden Helix Inc., Bozeman, MT, USA) and SPSS 18.0 (SPSS
Inc., Chicago, IL, USA). The analysis of SNPs was performed using
multiple logistic models (codominant 1, codominant 2, dominant,
recessive and log-additive models). The Haploview version 4.2
(5) was used for linkage
disequilibrium (LD) and haplo-type analysis between the nine
SNPs.
Results
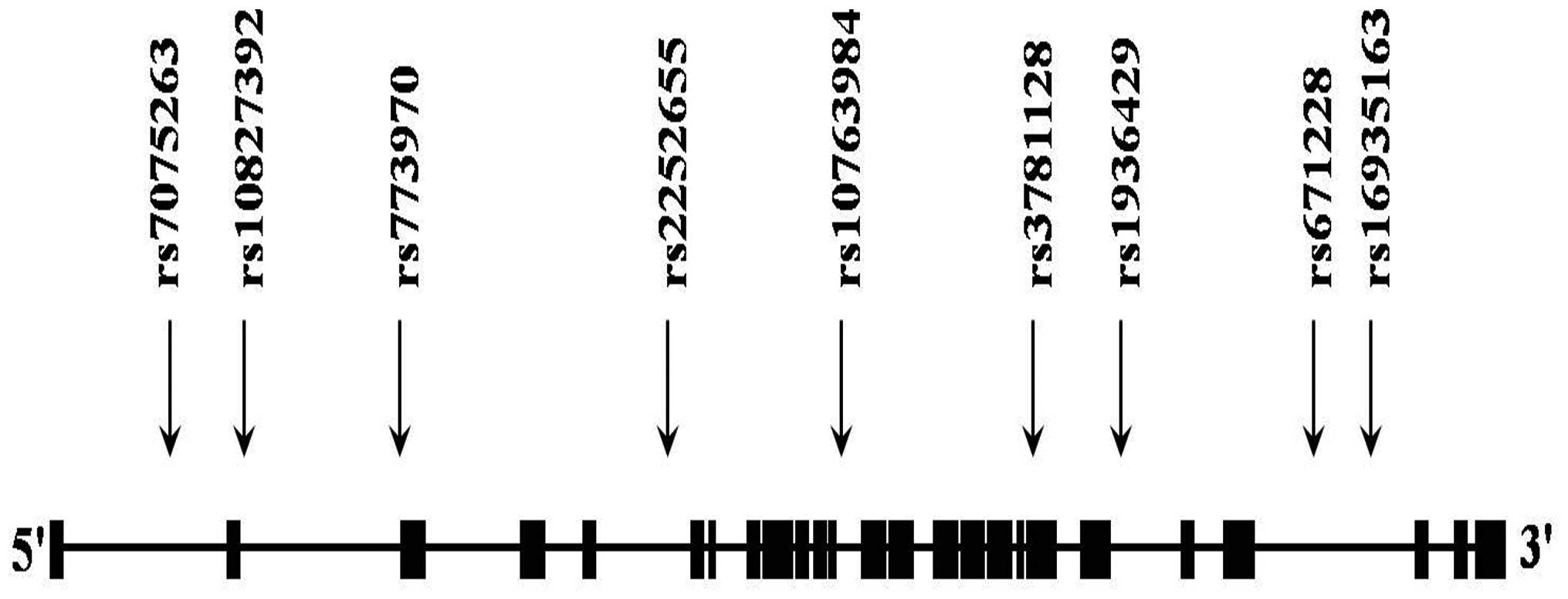
We selected nine SNPs of PARD3. The position
of the PARD3 SNPs is illustrated in Fig. 1A. Table I documents the allele frequencies
of the nine examined SNPs in schizophrenia patients and control
subjects. The HWEs of nine SNPs revealed no deviation in the
control groups, respectively (p>0.05, data not shown). Multiple
logistic regression analysis with adjustment for age and gender was
performed. Three SNPs were associated with schizophrenia
(rs3781128, OR=0.76, 95% CI=0.59–0.99, p=0.041; rs1936429, OR=0.75,
95% CI=0.58–0.97, p=0.030; rs16935163, OR=1.69, 95% CI=1.06–2.68,
p=0.028, respectively, Table I).
These results suggest that the C allele of rs3781128, the A allele
of rs1936429 and the C allele of rs16935163 may be risk factors for
schizophrenia.
 | Table I.Allele frequencies of polymorphisms of
the PARD3 gene in schizophrenia patients and control
subjects. |
Table I.
Allele frequencies of polymorphisms of
the PARD3 gene in schizophrenia patients and control
subjects.
| | Control
| Schizophrenia
| | |
|---|
| SNP | Allele | n | % | n | % | OR (95% CI) | P-value |
|---|
| rs7075263 | T | 623 | 88.7 | 354 | 86.8 | 1 | |
| Intron | C | 79 | 11.3 | 54 | 13.2 | 1.20 (0.83–1.74) | 0.33 |
| rs10827392 | T | 631 | 89.9 | 358 | 87.7 | 1 | |
| Intron | C | 71 | 10.1 | 50 | 12.3 | 1.24 (0.85–1.82) | 0.27 |
| rs773970 | G | 385 | 57.3 | 227 | 59.1 | 1 | |
| Intron | A | 287 | 42.7 | 157 | 40.9 | 0.93 (0.72–1.20) | 0.56 |
| rs2252655 | A | 343 | 50.7 | 216 | 53.5 | 1 | |
| Intron | G | 333 | 49.3 | 188 | 46.5 | 0.90 (0.70–1.15) | 0.38 |
| rs10763984 | C | 348 | 49.6 | 221 | 54.4 | 1 | |
| Intron | T | 354 | 50.4 | 185 | 45.6 | 0.82 (0.64–1.05) | 0.12 |
| rs3781128 | C | 435 | 62.5 | 272 | 68.7 | 1 | |
| Ser889Ser | T | 261 | 37.5 | 124 | 31.3 | 0.76 (0.59–0.99) | 0.041 |
| rs1936429 | A | 394 | 57.9 | 256 | 64.6 | 1 | |
| Intron | G | 286 | 42.1 | 140 | 35.4 | 0.75 (0.58–0.97) | 0.030 |
| rs671228 | C | 565 | 80.5 | 330 | 81.7 | 1 | |
| Intron | T | 137 | 19.5 | 74 | 18.3 | 0.93 (0.68–1.27) | 0.63 |
| rs16935163 | C | 658 | 94.3 | 361 | 90.7 | 1 | |
| Intron | A | 40 | 5.7 | 37 | 9.3 | 1.69 (1.06–2.68) | 0.028 |
Table II shows
genotype frequencies of the nine examined SNPs in schizophrenia
patients and control subjects. The seven SNPs among the
PARD3 polymorphisms showed significant associations with
schizophrenia [rs7075263, OR=6.00, 95% CI=1.21–26.98, p=0.028 in
codominant 2 (T/T versus T/T), OR=6.06, 95% CI=1.23–29.87, p=0.014
in recessive (T/T+C/T versus C/C); rs773970, OR=0.52, 95%
CI=0.30–0.89, p=0.014 in recessive (G/G+A/G versus A/A); rs2252655,
OR=0.58, 95% CI=0.37–0.91, p=0.016 in recessive (A/A+A/G versus
G/G); rs10763984, OR=0.53, 95% CI=0.34–0.84, p=0.005 in recessive
(C/C+C/T versus T/T); rs3781128, OR=0.65, 95% CI=0.45–0.95, p=0.026
in codominant 1 (C/C versus C/T), OR=0.66, 95% CI=0.46–0.94,
p=0.021 in dominant (C/C versus C/T+T/T); rs1936429, OR=0.57, 95%
CI=0.32–0.99, p=0.047 in codominant 2 (A/A versus G/G), OR=0.69,
95% CI=0.47–0.99, p=0.045 in dominant (A/A versus A/G+G/G),
OR=0.75, 95% CI=0.57–0.97, p=0.028 in log-additive (A/A versus A/G
versus G/G); and rs16935163, OR=1.68, 95% CI=1.02–2.76, p=0.044 in
dominant (C/C versus A/C+A/A), OR=1.74, 95% CI=1.08–2.81, p=0.024
in log-additive (C/C versus A/C versus A/A) models, respectively].
The rs10827392 and rs671228 SNPs had no differences between the
schizophrenia and control groups.
 | Table II.Genotype frequencies of polymorphisms
of the PARD3 gene in schizophrenia patients and control
subjects. |
Table II.
Genotype frequencies of polymorphisms
of the PARD3 gene in schizophrenia patients and control
subjects.
| | Control
| Schizophrenia
| | | |
|---|
| SNP | Genotype | n | % | n | % | Model | OR (95% CI) | P-value |
|---|
| rs7075263 | T/T | 274 | 78.1 | 156 | 76.8 | Codominant 1 | 0.96 (0.62–1.48) | 0.85 |
| intron | C/T | 75 | 21.4 | 40 | 19.7 | Codominant 2 | 6.00
(1.21–29.68) | 0.028 |
| C/C | 2 | 0.6 | 7 | 3.5 | Dominant | 1.09 (0.72–1.66) | 0.68 |
| | | | | | Recessive | 6.06
(1.23–29.87) | 0.014 |
| | | | | | Log-additive | 1.22 (0.84–1.77) | 0.30 |
| rs10827392 | T/T | 281 | 80.1 | 157 | 77.3 | Codominant 1 | 1.11
(0.72–1.72) | 0.63 |
| intron | C/T | 69 | 19.7 | 42 | 20.7 | Codominant 2 | 7.50
(0.82–68.89) | 0.08 |
| C/C | 1 | 0.3 | 4 | 2.0 | Dominant | 1.20
(0.78–1.84) | 0.40 |
| | | | | | Recessive | 7.32
(0.80–67.16) | 0.044 |
| | | | | | Log-additive | 1.28
(0.86–1.91) | 0.22 |
| rs773970 | G/G | 108 | 32.1 | 55 | 28.8 | Codominant 1 | 1.36
(0.91–2.04) | 0.14 |
| intron | A/G | 169 | 50.3 | 116 | 60.7 | Codominant 2 | 0.63
(0.34–1.16) | 0.14 |
| A/A | 59 | 17.6 | 20 | 10.5 | Dominant | 1.16
(0.79–1.72) | 0.44 |
| | | | | | Recessive | 0.52
(0.30–0.89) | 0.014 |
| | | | | | Log-additive | 0.90
(0.68–1.18) | 0.44 |
| rs2252655 | A/A | 88 | 26.0 | 47 | 23.4 | Codominant 1 | 1.34
(0.87–2.07) | 0.18 |
| intron | A/G | 167 | 49.4 | 121 | 60.2 | Codominant 2 | 0.71
(0.41–1.22) | 0.22 |
| G/G | 83 | 24.6 | 33 | 16.4 | Dominant | 1.13
(0.75–1.71) | 0.56 |
| | | | | | Recessive | 0.58
(0.37–0.91) | 0.016 |
| | | | | | Log-additive | 0.87
(0.67–1.13) | 0.29 |
| rs10763984 | C/C | 87 | 24.8 | 49 | 24.3 | Codominant 1 | 1.21
(0.79–1.86) | 0.37 |
| intron | C/T | 174 | 49.6 | 122 | 60.4 | Codominant 2 | 0.61
(0.35–1.04) | 0.07 |
| T/T | 90 | 25.6 | 31 | 15.3 | Dominant | 1.01
(0.67–1.52) | 0.97 |
| | | | | | Recessive | 0.53
(0.34–0.84) | 0.005 |
| | | | | | Log-additive | 0.80
(0.62–1.04) | 0.10 |
| rs3781128 | C/C | 130 | 37.4 | 94 | 47.7 | Codominant 1 | 0.65
(0.45–0.95) | 0.026 |
| Ser889Ser | C/T | 175 | 50.3 | 82 | 41.6 | Codominant 2 | 0.68
(0.37–1.22) | 0.19 |
| T/T | 43 | 12.4 | 21 | 10.7 | Dominant | 0.66
(0.46–0.94) | 0.021 |
| | | | | | Recessive | 0.84
(0.48–1.48) | 0.55 |
| | | | | | Log-additive | 0.76
(0.58–1.00) | 0.045 |
| rs1936429 | A/A | 113 | 33.2 | 81 | 41.1 | Codominant 1 | 0.73
(0.49–1.07) | 0.11 |
| intron | A/G | 168 | 49.4 | 92 | 46.7 | Codominant 2 | 0.57
(0.32–0.99) | 0.047 |
| G/G | 59 | 17.4 | 24 | 12.2 | Dominant | 0.69
(0.47–0.99) | 0.045 |
| | | | | | Recessive | 0.68
(0.41–1.14) | 0.14 |
| | | | | | Log-additive | 0.75
(0.57–0.97) | 0.028 |
| rs671228 | C/C | 225 | 64.1 | 135 | 67.2 | Codominant 1 | 0.83
(0.56–1.22) | 0.35 |
| intron | C/T | 115 | 32.8 | 58 | 28.9 | Codominant 2 | 1.17
(0.45–3.00) | 0.75 |
| T/T | 11 | 3.1 | 8 | 4.0 | Dominant | 0.86
(0.59–1.25) | 0.43 |
| | | | | | Recessive | 1.24
(0.48–3.16) | 0.66 |
| | | | | | Log-additive | 0.92
(0.67–1.26) | 0.59 |
| rs16935163 | C/C | 309 | 88.5 | 163 | 82.3 | Codominant 1 | 1.58
(0.95–2.62) | 0.08 |
| intron | A/C | 40 | 11.5 | 33 | 16.7 | Codominant 2 | NA (0.00-NA) | NA |
| A/A | 0 | 0.0 | 2 | 1.0 | Dominant | 1.68
(1.02–2.76) | 0.044 |
| | | | | | Recessive | NA (0.00-NA) | 0.040 |
| | | | | | Log-additive | 1.74
(1.08–2.81) | 0.024 |
The LD block was estimated using Haploview version
4.2. The LD block did not be made among SNPs of PARD3
(Fig. 1B, data not shown).
Discussion
In this study, we found that PARD3 gene
polymorphisms were associated with schizophrenia. The allele
distributions of three SNPs (rs3781128, rs1936429 and rs16935163)
and genotype frequencies of seven SNPs (rs7075263, rs773970,
rs2252655, rs10763984, rs3781128, rs1936429 and rs16935163) were
significantly different between schizophrenia patients and control
subjects.
PAR-3 is a scaffold-like PDZ-containing protein.
PAR-3 forms a complex with PAR-6 and atypical protein kinase C
(PAR-3-atypical protein kinase C-PAR-6 complex) and is associated
with the establishment of cell polarization (6,7).
Cell polarity has an important role in various cellular processes
such as cell migration, asymmetric cell division, and axon and
dendrite specification.
Chan et al reported that PAR-3 is directly
associated and recruits the p75 neurotrophin receptor to the axon
glial junction, forming a complex necessary for myelination
(8). It was found that depletion
of PAR-3 in mammalian epithelial cells profoundly disrupts tight
junction assembly (9). Synapses of
the junctions between nerve cells, through which the cells
communicate, are formed by the coordinated assembly and tight
attachment of presynaptic and postsynaptic specializations.
Therefore, tight junction protein-related studies suggest that
junctional protein polymorphisms are sufficient for a functional
postsynaptic response change (10–12).
Tight junctions play a major role in cell polarity
in vertebrate endothelial and epithelial cells. A protein complex
consisting of the cell polarity proteins PAR-3, PAR-6 and the
atypical protein kinase C localizes at tight junctions. It is
crucial for tight junction formation. It has been shown that PAR-3
associates with the junctional adhesion molecule (JAM). This
suggests that the ternary complex is targeted to tight junctions of
epithelial cells through PAR-3 binding to JAM (13).
In conclusion, this is the first report showing that
PARD3 is associated with the susceptibility to schizophrenia
in a Korean population. Our results suggest that PARD3 may
contribute to a genetic susceptibility to schizophrenia.
Acknowledgements
This research was supported by a grant
from Kyung Hee University (KHU-20090641).
References
|
1.
|
Kleinman JE, Law AJ, Lipska BK, et al:
Genetic neuropathology of schizophrenia: new approaches to an old
question and new uses for postmortem human brains. Biol Psychiatry.
69:140–145. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Faludi G and Mirnics K: Synaptic changes
in the brain of subjects with schizophrenia. Int J Dev Neurosci.
29:305–309. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Mitterauer B: Loss of function of glial
gap junctions may cause severe cognitive impairments in
schizophrenia. Med Hypotheses. 73:393–397. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Sun ZY, Wei J, Xie L, et al: The CLDN5
locus may be involved in the vulnerability to schizophrenia. Eur
Psychiatry. 19:354–357. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Barrett JC, Fry B, Maller J, et al:
Haploview: analysis and visualization of LD and haplotype maps.
Bioinformatics. 21:263–265. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Mizuno K, Suzuki A, Hirose T, et al:
Self-association of PAR-3-mediated by the conserved N-terminal
domain contributes to the development of epithelial tight
junctions. J Biol Chem. 278:31240–31250. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Zen K, Yasui K, Gen Y, et al: Defective
expression of polarity protein PAR-3 gene (PARD3) in esophageal
squamous cell carcinoma. Oncogene. 28:2910–2918. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Chan JR, Jolicoeur C, Yamauchi J, et al:
The polarity protein Par-3 directly interacts with p75NTR to
regulate myelination. Science. 314:832–836. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Chen X and Macara IG: Par-3 controls tight
junction assembly through the Rac exchange factor Tiam1. Nat Cell
Biol. 7:262–269. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Willott E, Balda MS, Fanning AS, et al:
The tight junction protein ZO-1 is homologous to the Drosophila
discs – large tumor suppressor protein of septate junctions. Proc
Natl Acad Sci USA. 90:7834–7838. 1993.
|
|
11.
|
Mino A, Ohtsuka T, Inoue E, et al:
Membrane-associated guanylate kinase with inverted orientation
(MAGI)-1/brain angiogenesis inhibitor 1-associated protein (BAP1)
as a scaffolding molecule for Rap small G protein GDP/GTP exchange
protein at tight junctions. Genes Cells. 5:1009–1016. 2000.
View Article : Google Scholar
|
|
12.
|
Biederer T, Sara Y, Mozhayeva M, et al:
SynCAM, a synaptic adhesion molecule that drives synapse assembly.
Science. 297:1525–1531. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Ebnet K, Aurrand-Lions M, Kuhn A, et al:
The junctional adhesion molecule (JAM) family members JAM-2 and
JAM-3 associate with the cell polarity protein PAR-3: a possible
role for JAMs in endothelial cell polarity. J Cell Sci.
116:3879–3891. 2003. View Article : Google Scholar : PubMed/NCBI
|