Introduction
Myocardial infarction following ischemia is a major
cause of mortality worldwide. Although early reperfusion is
necessary for myocardial salvage, reperfusion itself exacerbates
myocardial injury (1,2). Myocardial ischemia reperfusion (I/R)
injury causes negative events such as local inflammation and free
radical-mediated lipid peroxidation (3,4). The
latter is associated with deleterious effects on reversible
injuries of myocardial cells (5)
and contributes to cardiac dysfunction (6).
It is known that an increased formation of reactive
oxygen species (ROS) at the time of reperfusion plays a crucial
role in driving the inflammatory cascade (7). ROS activate nuclear factor κB
(NF-κB), a key transcription factor, and modulate the expression of
inflammation-related proteins, including tumor necrosis factor
(TNF)-α and interleukin (IL)-6, which are significant in the
development of myocardial I/R injury (8). With this background, pharmacological
intervention to scavenge ROS may have a double benefit in
myocardial I/R injury: it may diminish the progression of
myocardial damage by either attenuating ROS-induced lipid
peroxidation or by preventing the free radical-mediated
inflammation response.
Honokiol is an active component of Magnolia
officinalis (Houpo), a traditional Chinese herb used for the
treatment of various vascular diseases, including ischemia, heart
disease and stroke (9). Honokiol
has been shown to mitigate cerebral I/R injury through its
antioxidant and anti-inflammatory effects (10,11).
Early studies have also shown that honokiol is able to limit
infarct size and display anti-arrhythymic effects in rats with
acute myocardial infarction (12,13).
However, the mechanism remains unclear and there have been few
studies concerning honokiol in myocardial I/R injury. The present
study was designed to examine the effects of honokiol in a rat
model of myocardial I/R injury in vivo, and to investigate
its potential cardioprotective mechanisms.
Materials and methods
Animals
Sprague-Dawley rats (male, 250±20 g) were purchased
from Wuhan University Animal Center (Wuhan, China). This study
conformed to the Guide for the Care and Use of Laboratory Animals
of the National Institutes of Health (NIH Publication No. 80-23),
and the experimental procedures were approved by the Institutional
Animal Ethics Committee of Wuhan University.
In vivo myocardial I/R model
The in vivo myocardial I/R model was modified
from a previous study (14).
Briefly, rats were anesthetized with 1% pentobarbital sodium (40
mg/kg, intraperitoneally). The rats were intubated and mechanically
ventilated with room air using a rodent respirator (DV-2000,
Shanghai Jia Peng Technology Co., Ltd., Shanghai, China). A left
thoracotomy was carried out to expose the heart. A 5-0 silk
ligature was then passed under the left anterior descending (LAD)
coronary artery, and a small vinyl tube was placed on top of the
vessel to form a snare for the reversible coronary occlusion. After
a 30-min ischemia, the heart was reperfused for 2 h by releasing
the snare.
Experimental groups and protocols
A total of 50 rats were randomly divided into four
groups: sham (n=10), sham plus honokiol (sham-HK; n=10), I/R (n=15)
and I/R plus honokiol (I/R-HK; n=15). Honokiol (5 mg/kg;
Sigma-Aldrich, Inc., St. Louis, MO, USA) dissolved in dimethyl
sulfoxide (DMSO) was intraperitoneally administered to the rats 30
min prior to ischemia. The dose of honokiol was administered as
described previously (15,16).
Determination of infarct size
At the end of the experiment, the ligature around
the LAD was retied. A 2-ml dose of 2% Evans blue dye
(Sigma-Aldrich, Inc.) was administered intravenously to distinguish
between the perfused and non-perfused regions. The heart was
separated rapidly and cut into 3-mm thick slices parallel to the
atrioventricular groove. The slices were then placed in 1%
tetrazolium chloride solution (TTC; Sigma-Aldrich, Inc.) for 20 min
to identify the infarct zone. The weight of the infarct area
(white) and the area at risk (AAR; red) were measured. The infarct
size was expressed as a percentage of AAR mass.
Measurement of serum levels of creatine
kinase (CK) and lactate dehydrogenase (LDH)
Following a 2-h reperfusion, blood samples were
obtained and centrifuged. The serum levels of CK and LDH were
measured by a colorimetric method, using commercial kits (Nanjing
Jiancheng Bioengineering Institute, Nanjing, China) according to
the manufacturer’s instructions. The recorded values are presented
in U/l.
Assay of malondialdehyde (MDA),
superoxide dismutase (SOD), catalase (CAT) and myeloperoxidase
(MPO)
The frozen samples of the ischemic zone were
homogenized and centrifuged. Subsequently, the supernatants were
collected for analysis of the MDA level, and SOD, CAT and MPO
activities. The measurements were obtained spectrophotometrically
with commercially available kits (Nanjing Jiancheng Bioengineering
Institute) according to the manufacturer’s instructions. The MDA
level was represented in nmol/mg protein. The activities of SOD and
CAT were expressed as U/mg protein. MPO activity was expressed as
U/mg wet tissue.
Determination of TNF-α and IL-6
The TNF-α and IL-6 levels in the cardiac tissues
were measured by enzyme-linked immunosorbent assay using specific
kits (Boster Biological Technology Company, Wuhan, China) according
to the manufacturer’s instructions. Values are expressed as pg/mg
protein.
NF-κB (p65) expression
The expresssion of NF-κB (p65) was evaluated by
western blotting. Ischemic cardiac tissues were homogenized and
nuclear proteins were extracted with a Nuclear-Cytosol Extraction
kit (Applygen Technology Inc., Beijing, China) according to the
manufacturer’s instructions. Once the protein concentration had
been determined by a bicinchoninic acid protein assay (Beyotime
Biotechnology, Inc., Nantong, China), proteins were separated on
15% SDS-polyacrylamide gels, transferred to a nitrocellulose
membrane, probed with the primary antibodies against NF-κB (p65;
1:500; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) and
β-actin (1:500; Santa Cruz Biotechnology, Inc.) and then incubated
with a goat anti-rabbit secondary antibody (1:1,000; Santa Cruz
Biotechnology, Inc.). Proteins bands were then visualized with an
enhanced chemiluminescence kit (Beyotime Biotechnology, Inc.).
Histopathological examination
Cardiac tissues were fixed in 10% formalin and
embedded in paraffin. The embedded tissues were sectioned and
stained with hematoxylin and eosin (H&E) for observation of any
myocardial neutrophil infiltration.
Statistical analysis
All results were expressed as the mean ± SD.
Differences between groups were analyzed by one-way ANOVA followed
by Bonferroni’s post hoc test. P<0.05 was considered to indicate
a statistically significant difference.
Results
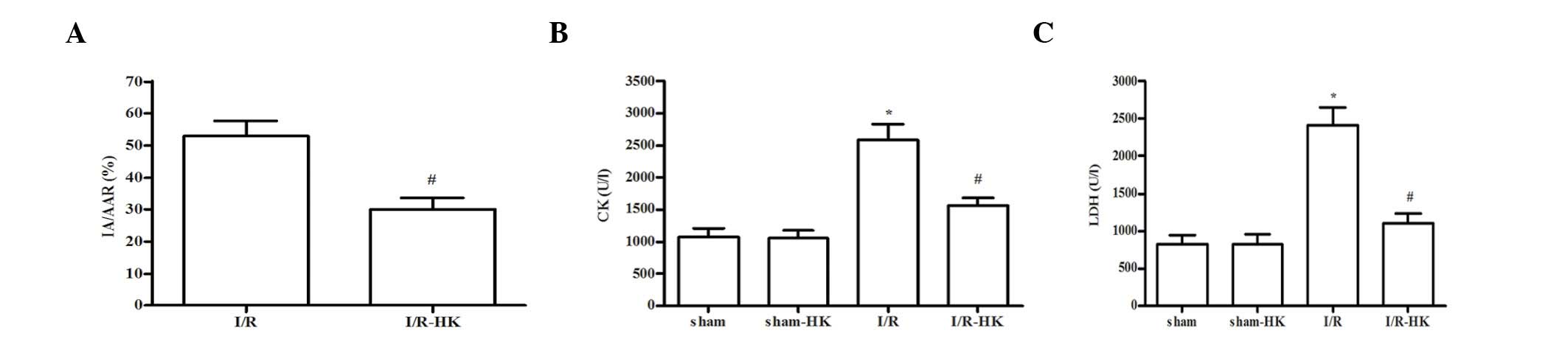
Myocardial infarct size
Following a 2-h reperfusion, honokiol treatment
significantly reduced myocardial infarct size compared with that in
the I/R group (P<0.05; Fig.1A).
No significant difference in AAR was observed between the two
groups (data not shown).
Serum levels of CK and LDH
The serum levels of CK and LDH were markedly
elevated in the I/R group compared with those in the sham group
(P<0.05). However, the elevation in CK and LDH levels was
significantly suppressed by honokiol adminstration (I/R-HK vs. I/R,
P<0.05). Honokiol did not affect the serum levels of LDH and CK
in the sham group (sham-HK vs. sham, P>0.05; Fig. 1B and C).
MDA, SOD, CAT and MPO in cardiac
tissues
The MDA level and MPO activity were notably
increased, while SOD and CAT activities were notably decreased in
cardiac tissues exposed to I/R injury (P<0.05). Honokiol
attenuated the I/R-induced increases in the MDA level and MPO
activity, while it inhibited the decreases in SOD and CAT
activities. Honokiol did not affect the MDA, SOD, CAT and MPO
levels in the sham group (sham-HK vs. sham, P>0.05; Table I).
 | Table IEffects of honokiol on MDA level and
SOD, CAT and MPO activities in cardiac tissues following I/R. |
Table I
Effects of honokiol on MDA level and
SOD, CAT and MPO activities in cardiac tissues following I/R.
| Group | MDA (nmol/mg
protein) | SOD (U/mg
protein) | CAT (U/mg
protein) | MPO (U/mg wet
tissue) |
|---|
| Sham | 2.3±0.4 | 154.2±9.6 | 55.4±4.1 | 1.3±0.2 |
| Sham-HK | 2.4±0.2 | 156.8±8.6 | 58.3±3.2 | 1.2±0.2 |
| I/R | 5.9±0.7a | 80.3±7.9a | 25.2±4.0a | 2.9±0.3a |
| I/R-HK | 3.3±0.4b | 137.1±12.0b | 44.6±4.8b | 1.8±0.2b |
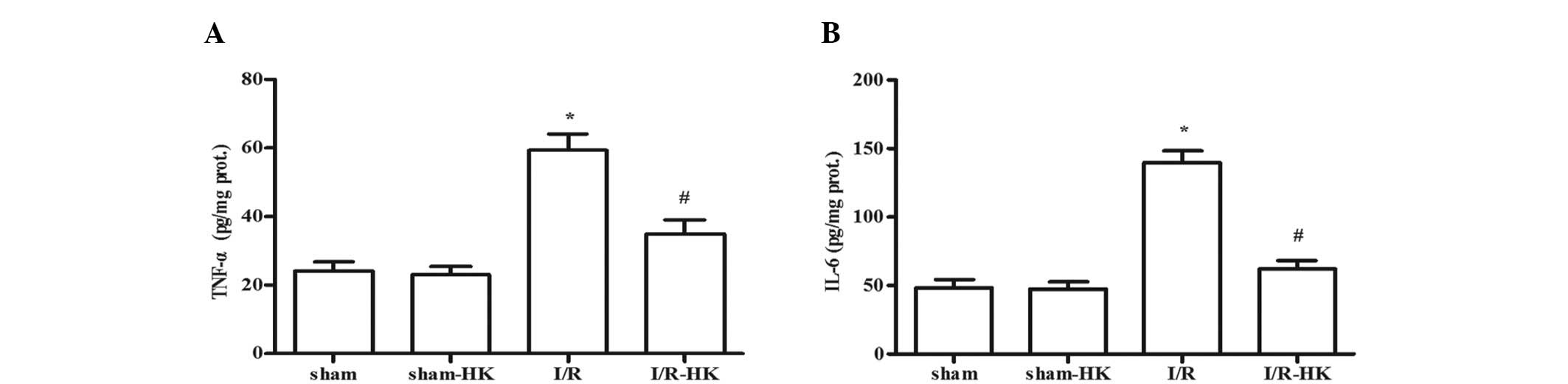
Levels of TNF-α and IL-6 in the
myocardium
TNF-α and IL-6 levels were greatly induced by I/R
compared with those in the sham group (P<0.05). Honokiol
significantly suppressed the increases in TNF-α and IL-6
(P<0.05) levels. The TNF-α and IL-6 levels in the sham group
were not altered by the honokiol treatment (sham-HK vs. sham,
P>0.05; Fig. 2).
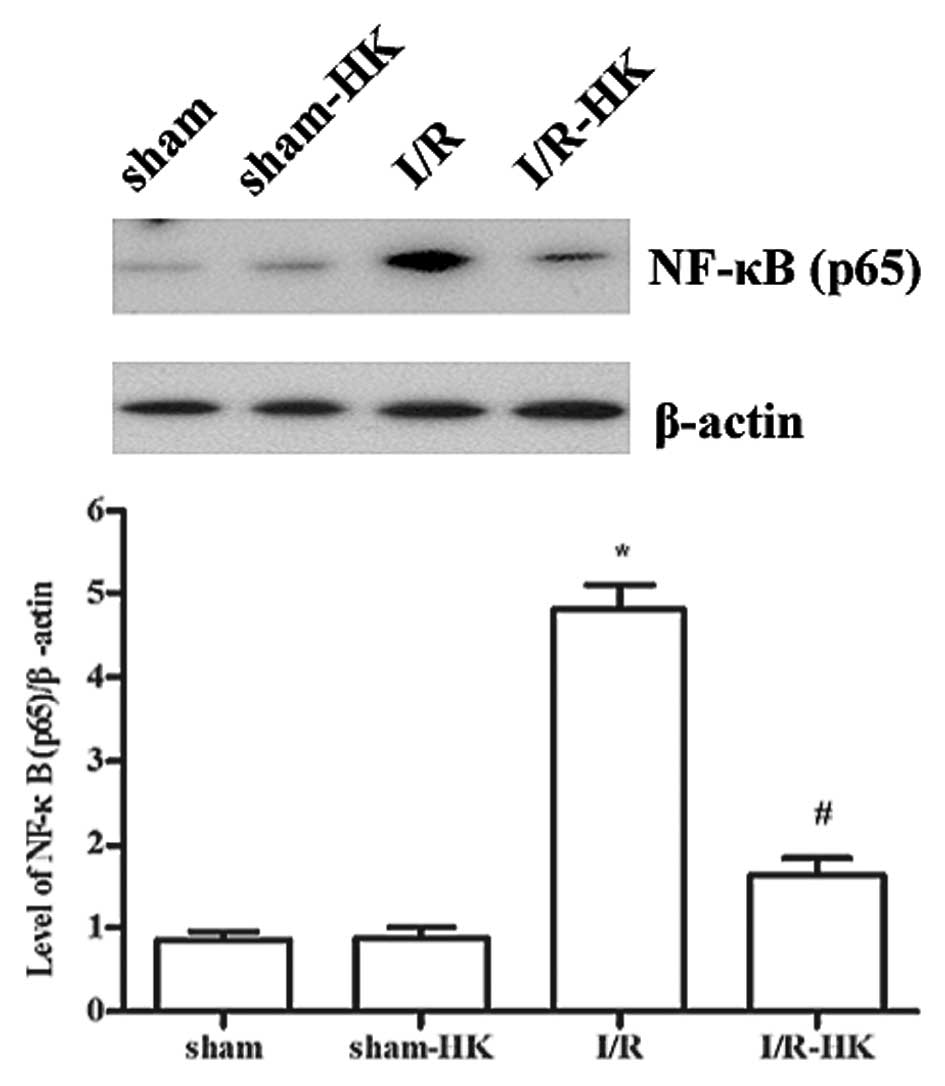
NF-κB (p65) expression
NF-κB expression was significantly upregulated
following I/R injury, however treatment with honokiol significantly
decreased the NF-κB expression (P<0.05). Honokiol did not affect
the expression of NF-κB in the sham group (sham-HK vs. sham,
P>0.05; Fig. 3).
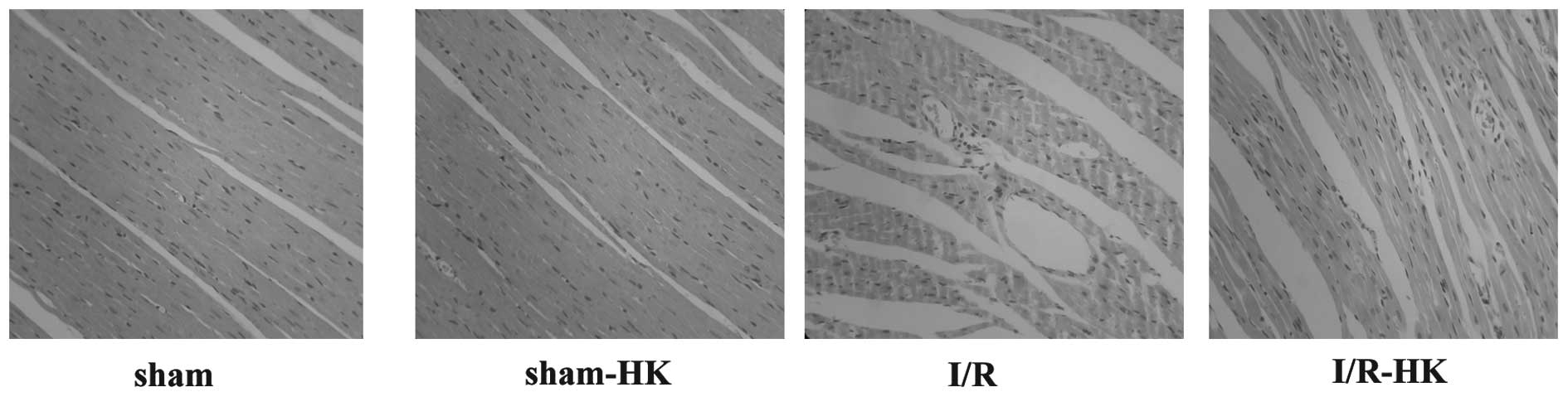
Histopathological examination of
myocardial tissues
No histological lesions were observed in the sham or
sham-HK groups. In the I/R group, there was evident structural
disorder, perivascular edema and neutrophil infiltration. However,
honokiol treatment markedly suppressed the I/R-induced myocardial
injury and neutrophil infiltration (Fig. 4).
Discussion
The major findings of the present study are as
follows. Firstly, honokiol pretreatment played a protective role
against myocardial I/R injury. Secondly, the protective effects of
honokiol during myocardial I/R injury were correlated with the
attenuation of oxidative stress and inflammation. Thirdly, the
antioxidant and anti-inflammatory effects of honokiol in myocardial
I/R may be due to the inhibition of the NF-κB pathway.
As a component of a traditional Chinese herb used in
various vascular diseases, honokiol has been demonstrated to
protect heart mitochondria against lipid peroxidation and to
diminish the myocardial infarct size of rats exposed to coronary
occlusion (12,17). However, whether honokiol has
beneficial effects on myocardial I/R remains unclear. The present
study revealed that honokiol pretreatment was able to protect rats
against myocardial I/R injury, as evidenced by a smaller infarct
size and reduced amounts of CK and LDH being released.
It is well documented that ROS play a pivotal role
in the development of myocardial I/R injury. ROS cause direct
damage to proteins and membranes as well as indirect damage through
activation of pro-apoptotic pathways (3). SOD and CAT, being key free radical
scavenging enzymes, are the primary line of defense against tissue
damage induced by ROS (18). MDA,
an significant product of lipid oxidation, is often used to
evaluate free radical-mediated myocardial cell injury (19). Reducing the elevation of MDA level
and suppressing the decrease of SOD and CAT activities has
cardioprotective effects in the ischemic-reperfused myocardium
(20). The present study revealed
that honokiol adminstration significantly reduced the elevation of
MDA level and attenuated the decrease of SOD and CAT activities in
ischemic-reperfused hearts. These data suggest that honokiol may
attenuate myocardial I/R-induced oxidative stress, which thus
contributes to the attenuation of myocardial damage.
Studies have demonstrated that inflammation is
associated with myocardial reperfusion injury, which is triggered
by ROS accumulation (21).
Pro-inflammatory cytokines and neutrophils play important roles in
the cardiac inflammatory response to I/R (4). TNF-α and IL-6 are pro-inflammatory
cytokines which contribute significantly to myocardial dysfunction
(22). Neutrophils are able to
release oxidants and proteases that damage tissues (23). Neutrophils also induce inflammatory
mediators that amplify neutrophil recruitment in the
ischemic-reperfused myocardium, thus expanding myocardial damage
(23). MPO is a neutrophil protein
whose activity may be considered to be an indicator for neutrophil
infiltration in injured tissues (24). In the present study, the generation
of TNF-α and IL-6, neutrophil infiltration and MPO activity were
all increased significantly in the rat hearts following I/R injury.
Each of these changes were inhibited by honokiol pretreatment. This
suggests that honokiol protected the myocardium from inflammation
during I/R insult. Such protection may contribute to the survival
of the myocardial cells, as demonstrated by our results.
NF-κB (p65) is an early redox-sensitive
transcriptional factor which regulates the inflammation-related
gene expression involved in myocardial I/R (25). NF-κB activation induces
pro-inflammatory proteins and neutrophil infiltration, resulting in
injury to endothelial and myocardial cells (26). Blocking NF-κB activation reduces
myocardial infarcts following I/R injury (27,28).
Honokiol has been reported to exert anti-inflammatory effects and
protect endothelial cells through the inhibition of the NF-κB
pathway (29,30). In the present study, pretreatment
with honokiol notably reduced myocardial I/R-induced NF-κB
activation, which was associated with the attenuation of oxidative
stress and inflammation. These results indicate that the
suppression of NF-κB by honokiol may be a potential mechanism of
its cardioprotective action.
In summary, the present study showed that honokiol
had a protective effect in a rat model of myocardial I/R injury,
possibly by reducing oxidative stress and inflammation. However,
only the beneficial role during the early phase of myocardial
reperfusion was investigated. Future studies to investigate the
effects of honokiol on heart failure and long-term survival
postinfarction would be worthwhile.
Acknowledgements
This study was supported by the
National Natural Science Foundation of China (No. 30872426) and the
Natural Science Foundation of HuBei Province of China (No.
2010CBD00403).
References
|
1
|
Braunwald E: Myocardial reperfusion,
limitation of infarct size, reduction of left ventricular
dysfunction, and improved survival. Should the paradigm be
expanded? Circulation. 79:441–444. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Matsumura K, Jeremy RW, Schaper J and
Becker LC: Progression of myocardial necrosis during reperfusion of
ischemic myocardium. Circulation. 97:795–804. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Braunersreuther V and Jaquet V: Reactive
oxygen species in myocardial reperfusion injury: from
physiopathology to therapeutic approaches. Curr Pharm Biotechnol.
13:97–114. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Eltzschig HK and Eckle T: Ischemia and
reperfusion - from mechanism to translation. Nat Med. 17:1391–1401.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Takano H, Zou Y, Hasegawa H, Akazawa H,
Nagai T and Komuro I: Oxidative stress-induced signal transduction
pathways in cardiac myocytes: involvement of ROS in heart diseases.
Antioxid Redox Signal. 5:789–794. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lakshmi SV, Padmaja G, Kuppusamy P and
Kutala VK: Oxidative stress in cardiovascular disease. Indian J
Biochem Biophys. 46:421–440. 2009.
|
|
7
|
Toufektsian MC, Boucher FR, Tanguy S,
Morel S and de Leiris JG: Cardiac toxicity of singlet oxygen:
implication in reperfusion injury. Antioxid Redox Signal. 3:63–69.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lu X, Liu H, Wang L and Schaefer S:
Activation of NF-kappaB is a critical element in the antiapoptotic
effect of anesthetic preconditioning. Am J Physiol Heart Circ
Physiol. 296:H1296–H1304. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liou KT, Lin SM, Huang SS, Chih CL and
Tsai SK: Honokiol ameliorates cerebral infarction from
ischemia-reperfusion injury in rats. Planta Med. 69:130–134. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liou KT, Shen YC, Chen CF, Tsao CM and
Tsai SK: Honokiol protects rat brain from focal cerebral
ischemia-reperfusion injury by inhibiting neutrophil infiltration
and reactive oxygen species production. Brain Res. 992:159–166.
2003. View Article : Google Scholar
|
|
11
|
Chen CM, Liu SH and Lin-Shiau SY:
Honokiol, a neuroprotectant against mouse cerebral ischaemia,
mediated by preserving Na+, K+-ATPase activity and mitochondrial
functions. Basic Clin Pharmacol Toxicol. 101:108–116.
2007.PubMed/NCBI
|
|
12
|
Tsai SK, Huang CH, Huang SS, Hung LM and
Hong CY: Antiarrhythmic effect of magnolol and honokiol during
acute phase of coronary occlusion in anesthetized rats: influence
of L-NAME and aspirin. Pharmacology. 59:227–233. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tsai SK, Huang SS and Hong CY: Myocardial
protective effect of honokiol: an active component in Magnolia
officinalis. Planta Med. 62:503–506. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jin YC, Kim CW, Kim YM, et al:
Cryptotanshinone, a lipophilic compound of Salvia
miltiorrriza root, inhibits TNF-alpha-induced expression of
adhesion molecules in HUVEC and attenuates rat myocardial
ischemia/reperfusion injury in vivo. Eur J Pharmacol. 614:91–97.
2009.PubMed/NCBI
|
|
15
|
Chiang J, Shen YC, Wang YH, et al:
Honokiol protects rats against eccentric exercise-induced skeletal
muscle damage by inhibiting NF-kappaB induced oxidative stress and
inflammation. Eur J Pharmacol. 610:119–127. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huang KH, Weng TI, Huang HY, et al:
Honokiol attenuates torsion/detorsion-induced testicular injury in
rat testis by way of suppressing endoplasmic reticulum
stress-related apoptosis. Urology. 79:967.e965–e911. 2012.
View Article : Google Scholar
|
|
17
|
Lo YC, Teng CM, Chen CF, Chen CC and Hong
CY: Magnolol and honokiol isolated from Magnolia officinalis
protect rat heart mitochondria against lipid peroxidation. Biochem
Pharmacol. 47:549–553. 1994.
|
|
18
|
Dhalla NS, Elmoselhi AB, Hata T and Makino
N: Status of myocardial antioxidants in ischemia-reperfusion
injury. Cardiovasc Res. 47:446–456. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zheng W, Huang LZ, Zhao L, et al:
Superoxide dismutase activity and malondialdehyde level in plasma
and morphological evaluation of acute severe hemorrhagic shock in
rats. Am J Emerg Med. 26:54–58. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang X, Yu Y, Ji L, Liang X, Zhang T and
Hai CX: Alpha-lipoic acid protects against myocardial
ischemia/reperfusion injury via multiple target effects. Food Chem
Toxicol. 49:2750–2757. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Altavilla D, Deodato B, Campo GM, et al:
IRFI 042, a novel dual vitamin E-like antioxidant, inhibits
activation of nuclear factor-kappaB and reduces the inflammatory
response in myocardial ischemia-reperfusion injury. Cardiovasc Res.
47:515–528. 2000. View Article : Google Scholar
|
|
22
|
Feldman AM, Combes A, Wagner D, et al: The
role of tumor necrosis factor in the pathophysiology of heart
failure. J Am Coll Cardiol. 35:537–544. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wu X, Zhang B, Fan R, et al: U50,488H
inhibits neutrophil accumulation and TNF-alpha induction induced by
ischemia-reperfusion in rat heart. Cytokine. 56:503–507. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Abe M, Takiguchi Y, Ichimaru S, Kaji S,
Tsuchiya K and Wada K: Different effect of acute treatment with
rosiglitazone on rat myocardial ischemia/reperfusion injury by
administration method. Eur J Pharmacol. 589:215–219. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Van der Heiden K, Cuhlmann S, Luong le A,
Zakkar M and Evans PC: Role of nuclear factor kappaB in
cardiovascular health and disease. Clin Sci (Lond). 118:593–605.
2010.PubMed/NCBI
|
|
26
|
Valen G, Yan ZQ and Hansson GK: Nuclear
factor kappa-B and the heart. J Am Coll Cardiol. 38:307–314. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kawano S, Kubota T, Monden Y, et al:
Blockade of NF-kappaB improves cardiac function and survival after
myocardial infarction. Am J Physiol Heart Circ Physiol.
291:H1337–H1344. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Guo J, Jie W, Kuang D, et al:
Ischaemia/reperfusion induced cardiac stem cell homing to the
injured myocardium by stimulating stem cell factor expression via
NF-kappaB pathway. Int J Exp Pathol. 90:355–364. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lee J, Jung E, Park J, et al:
Anti-inflammatory effects of magnolol and honokiol are mediated
through inhibition of the downstream pathway of MEKK-1 in NF-kappaB
activation signaling. Planta Med. 71:338–343. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sheu ML, Chiang CK, Tsai KS, et al:
Inhibition of NADPH oxidase-related oxidative stress-triggered
signaling by honokiol suppresses high glucose-induced human
endothelial cell apoptosis. Free Radic Biol Med. 44:2043–2050.
2008. View Article : Google Scholar
|