Introduction
Atrial fibrillation (AF) is the most common
tachyarrhythmia (1). The incidence
of AF is 3–5% in individuals aged >65 years and 9% in
individuals aged >80 years (2).
The incidence of cerebral apoplexy and heart failure is increased
in patients with AF since the heart atrium loses mechanical
function with slow blood flow (3,4). AF
is the strongest independent risk factor for cerebral apoplexy and
heart failure; 15% of cerebral apoplexy and 30% of heart failure
cases are associated with AF and the mortality rate of patients
with AF is three times the mortality rate of patients with sinus
rhythm (5). Compared with other
types of AF, chronic persistent AF is more complex and is difficult
to treat. According to the AF treatment guidelines produced by the
American College of Cardiology, American Heart Association and
European Society of Cardiology (ACC/AHA/ESC) in 2006, chronic AF is
defined as AF that persistently exists following drug therapy or
electroversion (6).
A great deal of attention has been paid to
radiofrequency catheter ablation for the treatment of chronic AF.
The relapse rate of radiofrequency catheter ablation is 20–60% in
the treatment of chronic AF (7–9). At
present, there is no standard radiofrequency catheter ablation
method for the treatment of chronic AF. The main methods of
radiofrequency catheter ablation for treating chronic AF include
pulmonary vein isolation (PVI), pulmonary vein antrum isolation
(PVAI), circumferential pulmonary vein isolation (CPVI), complex
fractionated atrial electrogram (CFAE) and PVI plus left atrial
ablation (LAA). There has been considerable debate about the
treatment of chronic AF with drugs or radiofrequency catheter
ablation.
In order to compare the therapeutic effects of drug
therapy and radiofrequency catheter ablation, as well as compare
different procedures of radiofrequency catheter ablation, papers
published in China and elsewhere between January 1, 2002 and May 1,
2012 concerning the treatment of chronic AF with drug or
radiofrequency catheter ablation were retrieved and then analyzed
with the RevMan 5.1 software package. This study discusses
different strategies for the treatment of chronic AF.
Materials and methods
Paper retrieval
Papers published in China and other countries
between January 1, 2002 and May 1, 2012, which reported the success
rates and relapse rates of drug therapy and catheter ablation for
the treatment of chronic AF were retrieved. Databases used were
Chinese HowNet, VIP, Wanfang, Medline, Wiley, SpringerLink, Google
Scholar and Science Direct.
The search items included the title, keyword and
abstract. The following English search terms and the corresponding
Chinese terms were used: atrial fibrillation, ablation/catheter
ablation, drugs/anti-arrhythmia and chronic atrial
fibrillation/permanent atrial fibrillation.
Inclusion and exclusion criteria
Inclusion criteria were as follows: i) papers
published in China and other countries between January 1, 2002 and
May 1, 2012; ii) randomized controlled trials (RCTs); iii) patients
with chronic AF; iv) clear diagnostic criteria: drugs did not
effectively maintain sinus rhythm or persistent AF for >7 days
repeated within 6 months; v) data collection with a scientific
method; vi) data analysis with a correct and scientific method;
vii) interventions including radiofrequency catheter ablation and
drug therapy; and viii) only one paper selected from several papers
about the same population. Exclusion criteria were as follows: i)
non-RCT; ii) data collection with a non-scientific method; iii)
data analysis with non-scientific method; iv) literature reviews
and v) repeated papers.
Quality evaluation
The quality of papers was evaluated according to the
quality evaluation criteria described in v.4.2.2 of the Cochrane
System Assessment handbook. The quality of papers was divided into
grades A, B and C based on the randomized method, hidden method,
double-blind method, loss of follow-up and exodus of patients from
the study. Grade A had low bias and the lowest possibility of bias,
and completely conformed to the four quality standards. The four
quality standards include randomized method, hidden method,
double-blind method, and loss of follow-up and exodus of patients.
Grade B had moderate bias and a moderate possibility of bias, and
partially conformed to ≥1 quality standards. Grade C had high bias
and a high possibility of bias, and did not conform to ≥1 quality
standards completely.
Statistical analysis
According to the requirements of the meta-analysis,
data processing was performed and a database was established. Data
analysis was performed with the RevMan 5.1 software package. The
therapeutic effects of catheter ablation and drug therapy for
chronic AF were analyzed, with the odds ratio (OR) as an effective
index, and the OR and 95% confidence interval (CI) were calculated.
Specific steps were as follows: i) OR served as the summary
statistic; ii) an homogeneity test was performed using the
χ2 test. If P>0.1, multiple independent studies had
homogeneity and OR was calculated with the fixed effect model. If
P≤0.1, multiple independent studies had heterogeneity and after
sensitivity or stratified analyses, the data had homogeneity; then
OR was calculated with the fixed effect model. Otherwise OR was
calculated with the random effect model; iii) the probability value
of the summary statistic was first obtained with the U test. If
P≤0.05 was considered to indicate a statistically significant
difference; iv) the publication bias was identified with funnel
plots. The funnel plots were generated by the RevMan 5.1 software
package, with OR values as the x-axis and with SE (log OR) as the
y-axis. The publication bias was evaluated by observing whether the
funnel plot was symmetrical.
Results
Paper retrieval
A total of 20 papers (10–29)
were used in this study. Of the 20 papers, eight compared drug
therapy with radiofrequency catheter ablation; five compared PVAI
and PVAI + CFAE, of which two papers also compared PVAI alone and
CFAE alone; four compared PVI + LAA and PVI; and three compared PVI
and CPVI (Table I).
 | Table I.Original papers included in this
meta-analysis. |
Table I.
Original papers included in this
meta-analysis.
| No. (ref.) | Publication date
(year) | First author | Title of paper |
|---|
| 1 (10) | 2010 | David J. Wilber | Comparison of
antiarrhythmic drug therapy and radiofrequency catheter ablation in
patients with paroxysmal atrial fibrillation: a randomized
controlled trial |
| 2 (11) | 2006 | Carlo Pappone | A randomized trial of
circumferential pulmonary vein ablation versus antiarrhythmic drug
therapy in paroxysmal atrial fibrillation: the APAF study |
| 3 (12) | 2011 | Carlo Pappone | Radiofrequency
catheter ablation and antiarrhythmic drug therapy: a prospective,
randomized, 4-year follow-up trial: the APAF study |
| 4 (13) | 2003 | R. Krittayaphong | A randomized clinical
trial of the efficacy of radiofrequency catheter ablation and
amiodarone in the treatment of symptomatic atrial fibrillation |
| 5 (14) | 2009 | Giovanni B.
Forleo | Catheter ablation of
atrial fibrillation in patients with diabetes mellitus type 2:
results from a randomized study comparing pulmonary vein isolation
versus antiarrhythmic drug therapy |
| 6 (15) | 2008 | Pierre Jaïs | Catheter ablation
versus anti-arrhythmic drugs for atrial fibrillation: the A4
study |
| 7 (16) | 2006 | Hakan Oral | Circumferential
pulmonary-vein ablation for chronic atrial fibrillation |
| 8 (17) | 2006 | Giuseppe Stabile | Catheter ablation
treatment in patients with drug-refractory atrial fibrillation: a
prospective, multi-centre, randomized, controlled study (Catheter
Ablation For The Cure Of Atrial Fibrillation Study) |
| 9 (18) | 2009 | Luigi Di Biase | Atrial fibrillation
ablation strategies for paroxysmal patients: randomized comparison
between different techniques |
| 10 (19) | 2011 | Minglong Chen | Randomized comparison
between pulmonary vein antral isolation versus complex fractionated
electrogram ablation for paroxysmal atrial fibrillation |
| 11 (20) | 2008 | Claude S. Elayi | Ablation for
longstanding permanent atrial fibrillation: results from a
randomized study comparing three different strategies |
| 12 (21) | 2009 | Hakan Oral | A randomized
assessment of the incremental role of ablation of complex
fractionated atrial electrograms after antral pulmonary vein
isolation for long-lasting persistent atrial fibrillation |
| 13 (22) | 2007 | Atul Verma | Efficacy of adjuvant
anterior left atrial ablation during intracardiac
echocardiography-guided pulmonary vein antrum isolation for atrial
fibrillation |
| 14 (23) | 2006 | Stephan Willems | Substrate
modification combined with pulmonary vein isolation improves
outcome of catheter ablation in patients with persistent atrial
fibrillation: a prospective randomized comparison |
| 15 (24) | 2005 | Mélèze Hocini | Techniques,
evaluation, and consequences of linear block at the left atrial
roof in paroxysmal atrial fibrillation: a prospective randomized
study |
| 16 (25) | 2004 | Michel
Haïssaguerre | Changes in atrial
fibrillation cycle length and inducibility during catheter ablation
and their relation to outcome |
| 17 (26) | 2006 | Imran Sheikh | Pulmonary vein
isolation and linear lesions in atrial fibrillation ablation |
| 18 (27) | 2008 | Martin Fiala | Pulmonary vein
isolation using segmental versus electro-anatomical circumferential
ablation for paroxysmal atrial fibrillation: over 3-year results of
a prospective randomized study |
| 19 (28) | 2005 | Martin R.
Karch | Freedom from atrial
tachyarrhythmias after catheter ablation of atrial fibrillation: a
randomized comparison between 2 current ablation strategies |
| 20 (29) | 2006 | Brian Nilsson | Recurrence of
pulmonary vein conduction and atrial fibrillation after pulmonary
vein isolation for atrial fibrillation: a randomized trial of the
ostial versus the extraostial ablation strategy |
Comparison of success rates between
catheter ablation and drug therapy
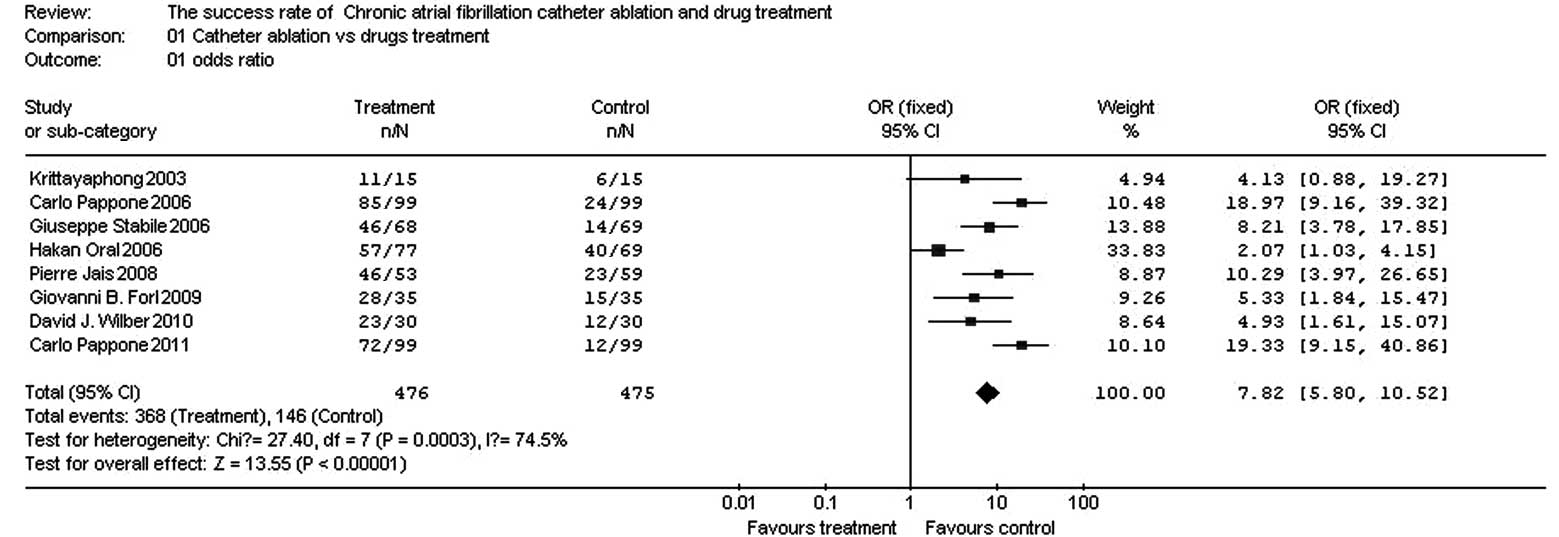
There were eight papers (10–17)
that compared the success rates of catheter ablation and drug
therapy. These papers included a total of 951 patients, with
catheter ablation as the test group and drug therapy as the control
group (Table II).
 | Table II.Papers comparing the success rates of
catheter ablation and drug therapy for treatment of chronic atrial
fibrillation. |
Table II.
Papers comparing the success rates of
catheter ablation and drug therapy for treatment of chronic atrial
fibrillation.
| Number | Reference | Publication date
(year) | First author | Test group (n) | Control group
(n) |
|---|
|
|
|---|
| Success | Total | Success | Total |
|---|
| 1 | 10 | 2010 | David J.
Wilber | 23 | 30 | 12 | 30 |
| 2 | 11 | 2006 | Carlo Pappone | 72 | 99 | 12 | 99 |
| 3 | 12 | 2011 | Carlo Pappone | 85 | 99 | 24 | 99 |
| 4 | 13 | 2003 | R.
Krittayaphong | 11 | 15 | 6 | 15 |
| 5 | 14 | 2009 | Giovanni B.
Forleo | 28 | 35 | 15 | 35 |
| 6 | 15 | 2008 | Pierre Jaïs | 46 | 53 | 23 | 59 |
| 7 | 16 | 2006 | Hakan Oral | 57 | 77 | 40 | 69 |
| 8 | 17 | 2006 | Giuseppe
Stabile | 46 | 68 | 14 | 69 |
In the eight papers, there were 476 patients in the
test group and 475 patients in the control group. The homogeneity
test (χ2=30.58, v=7, P<0.0001) demonstrated that the
eight papers had heterogeneity; therefore, the random effect model
was adopted. The OR value was 8.94 (95% CI, 4.70–17.02; z=6.68;
P<0.0001), suggesting that the success rate was significantly
higher for catheter ablation compared with that for drug therapy
(Fig. 1).
Comparison of relapse rates between
catheter ablation and drug therapy for treatment of chronic AF
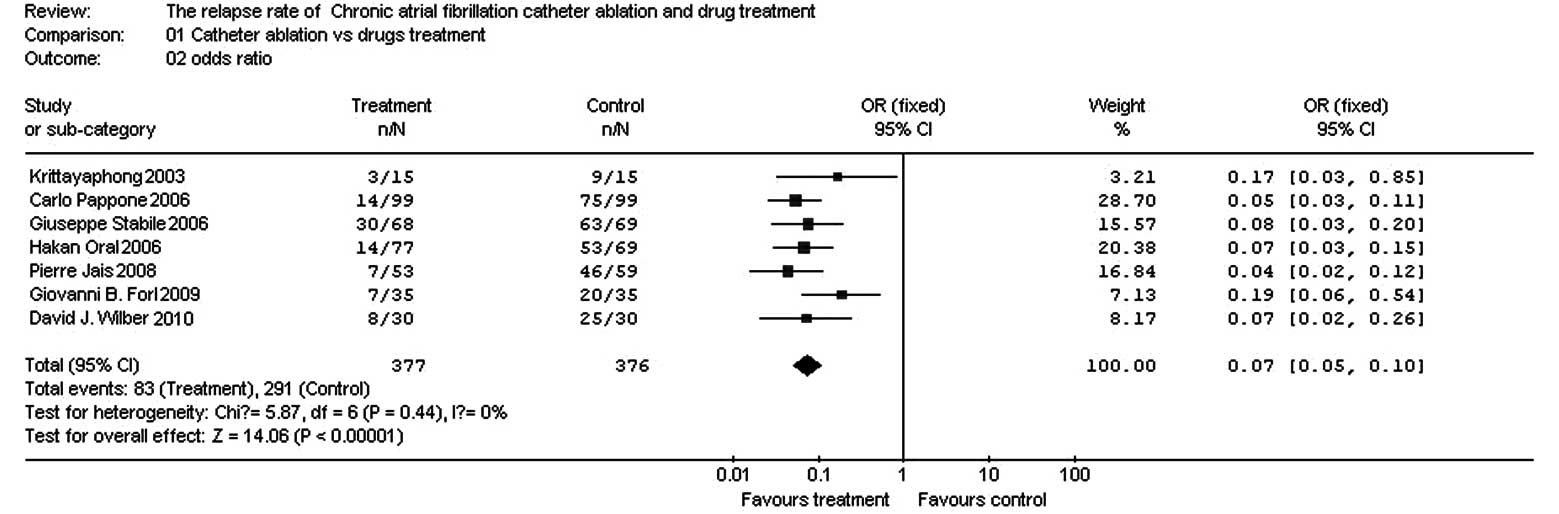
There were seven papers (10,11,13–17)
that compared the relapse rates of catheter ablation and drug
therapy for treatment of chronic AF. The seven papers included a
total of 753 patients, with catheter ablation as the test group and
drug therapy as the control group (Table III).
 | Table III.Papers comparing the relapse rates of
catheter therapy and drug therapy. |
Table III.
Papers comparing the relapse rates of
catheter therapy and drug therapy.
| Number | Reference | Publication date
(year) | First author | Test group (n) | Control group
(n) |
|---|
|
|
|---|
| Relapse | Total | Relapse | Total |
|---|
| 1 | 10 | 2010 | David J.
Wilber | 8 | 30 | 25 | 30 |
| 2 | 11 | 2006 | Carlo Pappone | 14 | 99 | 75 | 99 |
| 3 | 13 | 2003 | R.
Krittayaphong | 3 | 15 | 9 | 15 |
| 4 | 14 | 2009 | Giovanni B.
Forleo | 7 | 35 | 20 | 35 |
| 5 | 15 | 2008 | Pierre Jaïs | 7 | 53 | 46 | 59 |
| 6 | 16 | 2006 | Hakan Oral | 14 | 77 | 53 | 69 |
| 7 | 17 | 2006 | Giuseppe
Stabile | 30 | 68 | 63 | 69 |
In the seven papers, there were 377 patients in the
test group and 376 patients in the control group. The homogeneity
test (χ2=5.87, v=6, P=0.44, P>0.10) demonstrated that
the seven papers had homogeneity; therefore, the fixed effect model
was adopted. The OR value was 0.07 (95% CI, 0.05–0.10; z=14.06;
P<0.0001), suggesting that the relapse rate was significantly
lower for catheter ablation compared with that for drug therapy
(Fig. 2).
Comparison of success rates between PVAI
and CFAE
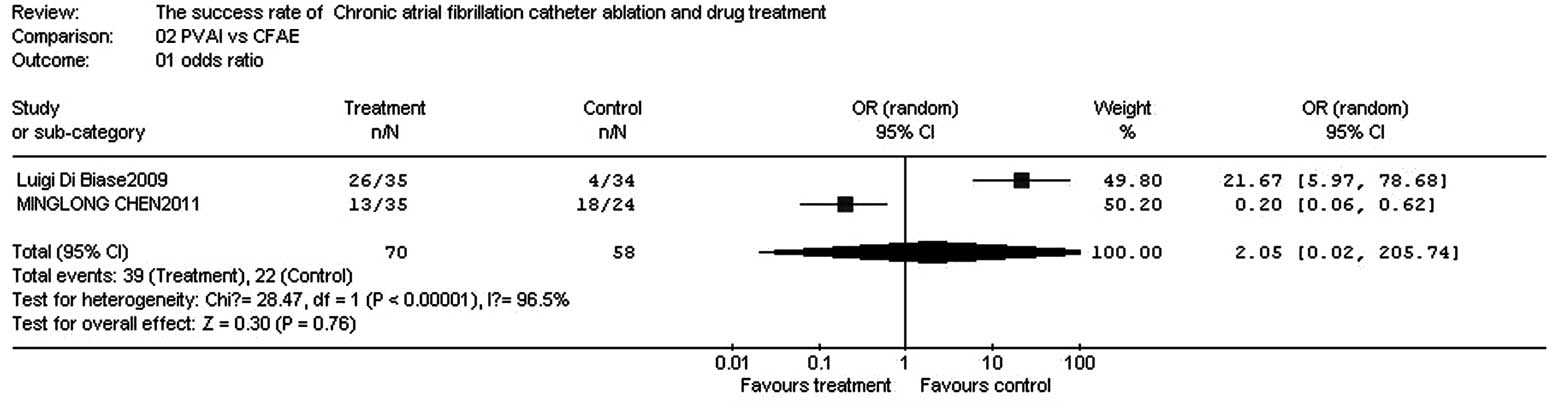
There were two papers (18,19)
comparing the success rates of PVAI and CFAE for treatment of
chronic AF. The two papers included a total of 128 patients, with
PVAI as the test group and CFAE as the control group (Table IV).
 | Table IV.Papers comparing the success rates of
PVAI and CFAE. |
Table IV.
Papers comparing the success rates of
PVAI and CFAE.
| Number | Reference | Publication date
(year) | First author | Test group (n) | Control group
(n) |
|---|
|
|
|---|
| Success | Total | Success | Total |
|---|
| 1 | 18 | 2009 | Luigi Di Biase | 26 | 35 | 4 | 34 |
| 2 | 19 | 2011 | Minglong Chen | 13 | 35 | 18 | 24 |
In the two papers, there were 70 patients in the
test group and 58 patients in the control group. The homogeneity
test (χ2=28.47, v=1, P=0.0000, P<0.10) demonstrated
that the two papers did not have homogeneity; therefore, the random
effect model was adopted. The OR value was 2.05 (95% CI,
0.06–205.74; z=0.30; P=0.76), suggesting that there was no
significant difference in success rates between PVAI and CFAE for
treatment of chronic AF (Fig.
3).
Comparison of success rates between PVA I
and PVAI + CFAE
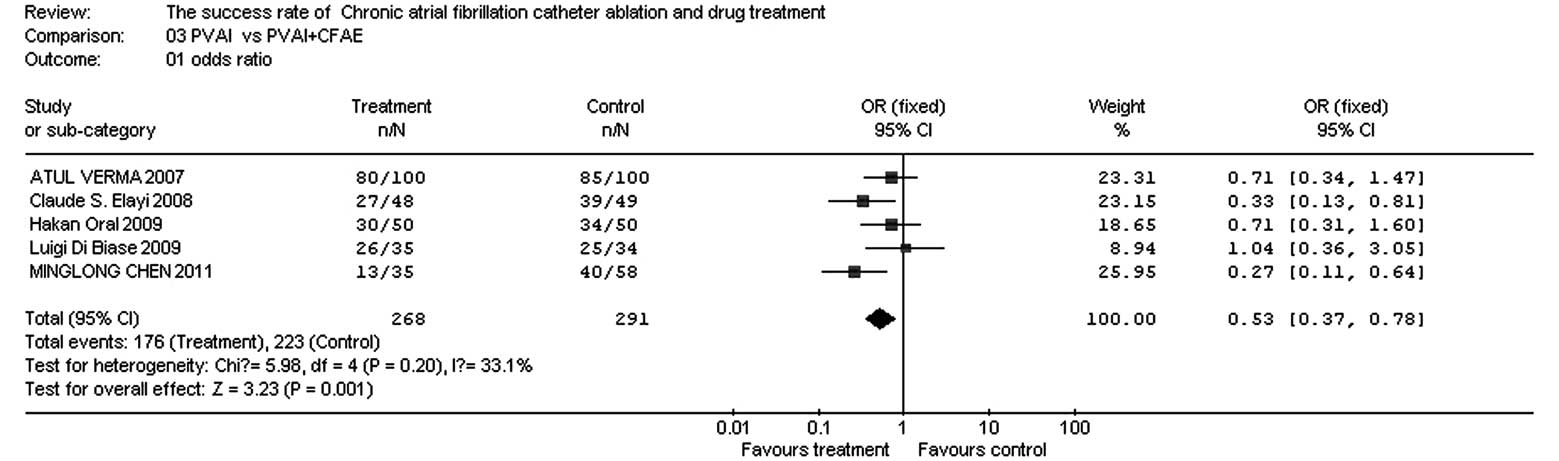
There were five papers (18–22)
comparing the success rates of PVAI and PVAI + CFAE for treatment
of chronic AF. The five papers included a total of 559 patients,
with PVAI as the test group and PVAI + CFAE as the control group
(Table V).
 | Table V.Papers comparing the success rates of
PVAI and PVAI + CFAE. |
Table V.
Papers comparing the success rates of
PVAI and PVAI + CFAE.
| Number | Reference | Publication date
(year) | First author | Test group (n) | Control group
(n) |
|---|
|
|
|---|
| Success | Total | Success | Total |
|---|
| 1 | 18 | 2009 | Luigi Di Biase | 26 | 35 | 25 | 34 |
| 2 | 19 | 2011 | Minglong Chen | 13 | 35 | 40 | 58 |
| 3 | 20 | 2008 | Claude S.
Elayi | 27 | 48 | 39 | 49 |
| 4 | 21 | 2009 | Hakan Oral | 30 | 50 | 34 | 50 |
| 5 | 22 | 2007 | Atul Verma | 80 | 100 | 85 | 100 |
In the five papers, there were 268 patients in the
test group and 291 patients in the control group. The homogeneity
test (χ2=5.98, v=4, P=0.20, P>0.10) demonstrated that
the papers had homogeneity; therefore, the fixed effect model was
adopted. The OR value was 0.53 (95% CI, 0.37–0.78; z=3.23;
P=0.001), suggesting that the success rate for treatment of chronic
AF was significantly higher for PVAI + CFAE than for PVAI (Fig. 4).
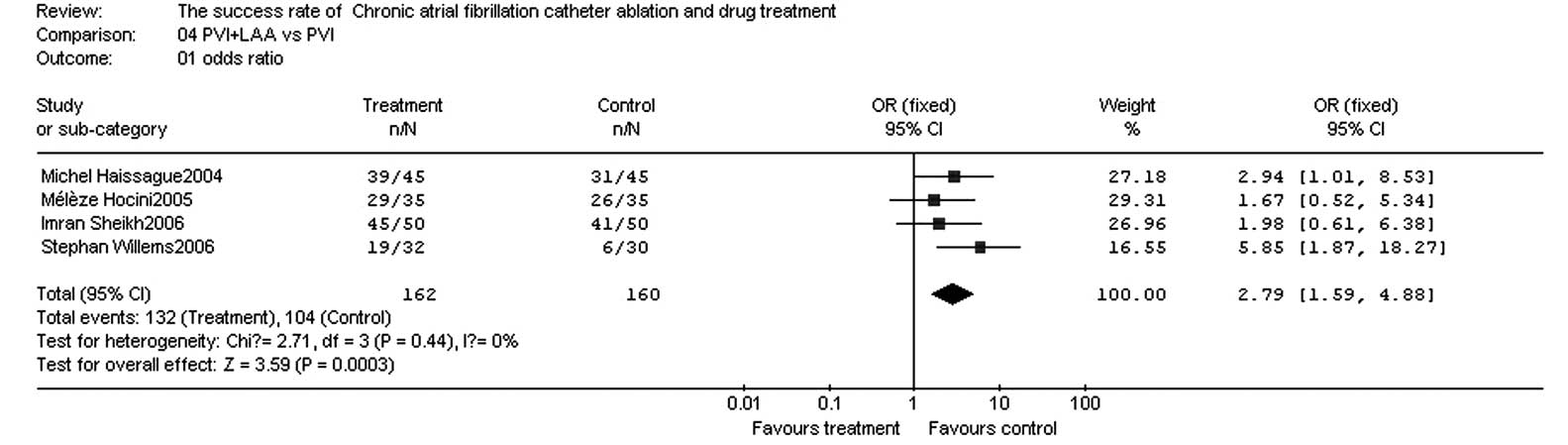
Comparison of success rates between PVI
and PVI + LAA
There were four papers (23–26)
in which the success rates of PVI and PVI + LAA in the treatment of
chronic AF were compared. These papers included a total of 322
patients, with PVI + LAA as the test group and PVI as the control
group (Table VI).
 | Table VI.Papers comparing the success rates of
PVI + LAA and PVI. |
Table VI.
Papers comparing the success rates of
PVI + LAA and PVI.
| Number | Reference | Publication date
(year) | First author | Test group (n) | Control group
(n) |
|---|
|
|
|---|
| Success | Total | Success | Total |
|---|
| 1 | 14 | 2006 | Stephan
Willems | 19 | 32 | 6 | 30 |
| 2 | 15 | 2005 | Mélèze Hocini | 39 | 45 | 31 | 45 |
| 3 | 16 | 2004 | Michel
Haïssaguerre | 29 | 35 | 26 | 35 |
| 4 | 17 | 2006 | Imran Sheikh | 45 | 50 | 41 | 50 |
In the four papers, there were 162 patients in the
test group and 160 patients in the control group. The homogeneity
test (χ2=2.71, v=3, P=0.44, P>0.10) demonstrated that
the papers had homogeneity; therefore, the fixed effect model was
adopted. The OR value was 2.79 (95% CI, 1.59–4.88; z=3.59;
P=0.0003), suggesting that the success rate was significantly
higher for PVI + LAA compared with that for PVI (Fig. 5).
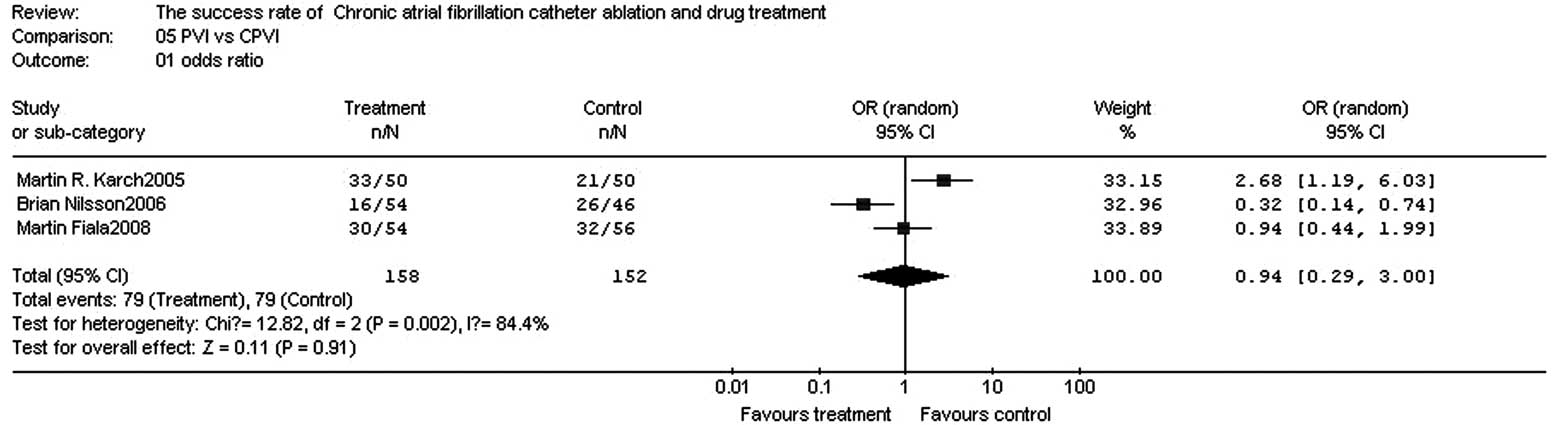
Comparison of success rates between PVI
and CPVI
There were three papers (27–29)
in which the success rates of PVI and CPVI were compared. The three
papers included a total of 310 patients, with PVI as the test group
and CPVI as the control group (Table
VII).
 | Table VII.Papers comparing the success rates of
PVI and CPVI. |
Table VII.
Papers comparing the success rates of
PVI and CPVI.
| Number | Reference | Publication date
(year) | First author | Test group (n) | Control group
(n) |
|---|
|
|
|---|
| Success | Total | Success | Total |
|---|
| 1 | 27 | 2008 | Martin Fiala | 30 | 54 | 32 | 56 |
| 2 | 28 | 2005 | Martin R.
Karch | 33 | 50 | 21 | 50 |
| 3 | 29 | 2006 | Brian Nilsson | 26 | 46 | 16 | 54 |
In the three papers, there were 150 patients in the
test group and 160 patients in the control group. The homogeneity
test (χ2=12.82, v=2, P=0.002, P<0.10) demonstrated
that the papers did not have homogeneity; therefore, the random
effect model was adopted. The OR value was 0.94 (95% CI, 0.29–3.00;
z=0.11; P=0.91), suggesting that there were no significant
differences in the success rates between PVI and CPVI for treatment
of chronic AF (Fig. 6).
Publication bias
All funnel plots comparing the success rates of
catheter ablation and drug therapy, PVAI and CFAE, PVAI and PVAI +
CFAE, PVI + LAA and PVI, and PVI and CPVI were essentially
symmetrical, and the majority of the points were located within the
95% CI. The funnel plot comparing the relapse rates of catheter
ablation and drug therapy was essentially symmetrical and the
majority of the points were located within the 95% CI.
Discussion
In this study, a meta-analysis of catheter ablation
and drug therapy for the treatment of chronic AF was performed
according to Preferred Reporting Items for Systematic Reviews and
Meta-Analyses (PRISMA). The results indicated that for the
treatment of chronic AF, the success rate is higher and the relapse
rate is lower for catheter ablation compared with that for drug
therapy. There were no significant differences between the success
rates of PVAI and CFAE, and PVI and CPVI; however, the success
rates were higher for PVAI + CFAE compared with that for PVAI, and
for PVI + LAA compared with that for PVI. The results of
publication bias indicated that the results of the meta-analysis
were stable and reliable, truly reflecting the status of catheter
ablation and drug treatment of chronic AF.
AF treatment includes rate control and rhythm
control. Although it has been reported that rate control is a
reasonable choice for the treatment of AF (30), rate control alone does not reduce
the risk of cerebral apoplexy and improve atrioventricular
synchrony. Antithrombotic therapy with warfarin decreases the
incidence of cerebral apoplexy and reduces the mortality rate;
however, there is the risk of bleeding, which requires long-term
monitoring of international normalized ratio (INR) of prothrombin
time with low patient compliance. Compared with rate control,
rhythm control decreases the mortality rate and the incidence of
transient ischemic attack (TIA), cerebral infarction (31), systemic embolism, hemorrhea and
heart failure (32). Therefore,
rhythm control is likely to be more effective than rate control.
The conventional methods of rhythm control include anti-arrhythmic
drugs, direct current countershock and the surgical maze procedure.
However, these methods are limited in clinical practice due to
their therapeutic effects and safety. In the past 20 years, a great
deal of attention has been paid to catheter ablation for the
treatment of AF. In the AF treatment guidelines established by
ACC/AHA/ESC in 2008, catheter ablation is suitable for patients who
exhibit no therapeutic effects following treatment with class I or
III anti-arrhythmic drugs, are unable to tolerate the side-effects
of drugs or have symptomatic heart failure or low cardiac output
prior to the use of anti-arrhythmic drugs.
The pathogenesis of AF is not completely clear. At
present, its mechanism mainly includes triggering factors and an
electrical substrate (or atrial substrate). The majority of the
triggering factors are located in the pulmonary veins and superior
vena cava; however, a few triggering factors are located in the
crista terminalis, coronary sinus, Marshall ligaments and atrial
posterior wall. Triggering factors are also called triggering foci.
Electrical substrate refers to the changes in electrophysiological
characteristics to maintain AF and mainly includes electrical
reconstitution, anatomical reconstitution, reconstitution of
autonomic nerves and reconstitution of the renin-angiotensin
system. Atrial dilatation, myocarditis, myocardial fibrosis and
increased autonomic nervous tension all serve as triggering factors
and/or an electrical substrate to lead to AF. Triggering factors
and the electrical substrate may be located in the same place or at
different locations. Radiofrequency ablation is used to treat AF
through the isolation of triggering factors and interference with
the electrical substrate (33,34).
The limitations of the current study are that only a
small number of papers were included in the study, unpublished
papers from conferences were not included and the quality control
standards were not completely uniform. Future meta-analyses should
include a greater number of RCTs.
Our results suggest that catheter ablation is more
effective than drug therapy in the treatment of AF. There are no
significant differences in success rates between two single
procedures for catheter ablation; however, the success rate is
higher in PVAI + CFAE compared with that in PVAI, and in PVI + LAA
compared with that in PVI.
References
|
1.
|
Furberg CD, Psaty BM, Manolio TA, et al:
Prevalence of atrial fibrillation in elderly subjects (the
Cardiovascular Health Study). Am J Cardiol. 74:236–241. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Kannel WB, Wolf PA, Benjamin EJ and Levy
D: Prevalence, incidence, prognosis, and predisposing conditions
for atrial fibrillation: population-based estimates. Am J Cardiol.
82:2N–9N. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Singer DE: Overview of the randomized
trials to prevent stroke in atrial fibrillation. Ann Epidemiol.
3:563–567. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Kazemian P, Oudit G and Jugdutt BI: Atrial
fibrillation and heart failure in the elderly. Heart Fail Rev.
17:597–613. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Fuster V, Rydén LE, Cannom DS, et al:
ACCF/AHA/HRS focused updates incorporated into the ACC/AHA/ESC 2006
Guidelines for the management of patients with atrial fibrillation:
a report of the American College of Cardiology Foundation/American
Heart Association Task Force on Practice Guidelines developed in
partnership with the European Society of Cardiology and in
collaboration with the European Heart Rhythm Association and the
Heart Rhythm Society. J Am Coll Cardiol. 57:e101–e198. 2011.
|
|
6.
|
Fuster V, Rydén LE, Cannom DS, et al:
ACC/AHA/ESC 2006 Guidelines for the Management of Patients with
Atrial Fibrillation: a report of the American College of
Cardiology/American Heart Association Task Force on Practice
Guidelines and the European Society of Cardiology Committee for
Practice Guidelines (Writing Committee to Revise the 2001
Guidelines for the Management of Patients With Atrial
Fibrillation): developed in collaboration with the European Heart
Rhythm Association and the Heart Rhythm Society. Circulation.
114:e257–e354. 2006.
|
|
7.
|
Joshi S, Choi AD, Kamath GS, et al:
Prevalence, predictors, and prognosis of atrial fibrillation early
after pulmonary vein isolation: findings from 3 months of
continuous automatic ECG loop recordings. J Cardiovasc
Electrophysiol. 20:1089–1094. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Takahashi Y, Takahashi A, Kuwahara T, et
al: Clinical characteristics of patients with persistent atrial
fibrillation successfully treated by left atrial ablation. Circ
Arrhythm Electrophysiol. 3:465–471. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Li C, Ding X, Zhang J, et al: Does the
E/e’ index predict the maintenance of sinus rhythm after catheter
ablation of atrial fibrillation? Echocardiography. 27:630–636.
2010.
|
|
10.
|
Wilber DJ, Pappone C, Neuzil P, et al:
Comparison of antiar-rhythmic drug therapy and radiofrequency
catheter ablation in patients with paroxysmal atrial fibrillation:
a randomized controlled trial. JAMA. 303:333–340. 2010. View Article : Google Scholar
|
|
11.
|
Pappone C, Augello G, Sala S, et al: A
randomized trial of circumferential pulmonary vein ablation versus
antiarrhythmic drug therapy in paroxysmal atrial fibrillation: the
APAF study. J Am Coll Cardiol. 11:2340–2347. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Pappone C, Vicedomini G, Augello G, et al:
Radiofrequency catheter ablation and antiarrhythmic drug therapy: a
prospective, randomized, 4-year follow-up trial: the APAF study.
Circ Arrhythm Electrophysiol. 4:808–814. 2011.PubMed/NCBI
|
|
13.
|
Krittayaphong R, Raungrattanaamporn O,
Bhuripanyo K, et al: A randomized clinical trial of the efficacy of
radiofrequency catheter ablation and amiodarone in the treatment of
symptomatic atrial fibrillation. J Med Assoc Thai. 86(Suppl 1):
S8–S16. 2003.PubMed/NCBI
|
|
14.
|
Forleo GB, Mantica M, De Luca L, et al:
Catheter ablation of atrial fibrillation in patients with diabetes
mellitus type 2: results from a randomized study comparing
pulmonary vein isolation versus antiarrhythmic drug therapy. J
Cardiovasc Electrophysiol. 20:22–28. 2009. View Article : Google Scholar
|
|
15.
|
Jaïs P, Cauchemez B, Macle L, et al:
Catheter ablation versus antiarrhythmic drugs for atrial
fibrillation: the A4 study. Circulation. 118:2498–2505.
2008.PubMed/NCBI
|
|
16.
|
Oral H, Pappone C, Chugh A, et al:
Circumferential pulmonary-vein ablation for chronic atrial
fibrillation. N Engl J Med. 2:934–941. 2006. View Article : Google Scholar
|
|
17.
|
Stabile G, Bertaglia E, Senatore G, et al:
Catheter ablation treatment in patients with drug-refractory atrial
fibrillation: a prospective, multi-centre, randomized, controlled
study (Catheter Ablation For The Cure Of Atrial Fibrillation
Study). Eur Heart J. 27:216–221. 2006. View Article : Google Scholar
|
|
18.
|
Di Biase L, Elayi CS, Fahmy TS, et al:
Atrial fibrillation ablation strategies for paroxysmal patients:
randomized comparison between different techniques. Circ Arrhythm
Electrophysiol. 42:113–119. 2009.
|
|
19.
|
Chen M, Yang B, Chen H, et al: Randomized
comparison between pulmonary vein antral isolation versus complex
fractionated electrogram ablation for paroxysmal atrial
fibrillation. J Cardiovasc Electrophysiol. 22:973–981. 2011.
View Article : Google Scholar
|
|
20.
|
Elayi CS, Verma A, Di Biase L, et al:
Ablation for longstanding permanent atrial fibrillation: results
from a randomized study comparing three different strategies. Heart
Rhythm. 5:1658–1664. 2008. View Article : Google Scholar
|
|
21.
|
Oral H, Chugh A, Yoshida K, et al: A
randomized assessment of the incremental role of ablation of
complex fractionated atrial electrograms after antral pulmonary
vein isolation for long-lasting persistent atrial fibrillation. J
Am Col Cardiol. 53:782–789. 2009. View Article : Google Scholar
|
|
22.
|
Verma A, Patel D, Famy T, et al: Efficacy
of adjuvant anterior left atrial ablation during intracardiac
echocardiography-guided pulmonary vein antrum isolation for atrial
fibrillation. J Cardiovasc Electrophysiol. 18:151–156. 2007.
View Article : Google Scholar
|
|
23.
|
Willems S, Klemm H, Rostock T, et al:
Substrate modification combined with pulmonary vein isolation
improves outcome of catheter ablation in patients with persistent
atrial fibrillation: a prospective randomized comparison. Eur Heart
J. 27:2871–2878. 2006. View Article : Google Scholar
|
|
24.
|
Hocini M, Jaïs P, Sanders P, et al:
Techniques, evaluation, and consequences of linear block at the
left atrial roof in paroxysmal atrial fibrillation: a prospective
randomized study. Circulation. 112:3688–3696. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Haïssaguerre M, Sanders P, Hocini M, et
al: Changes in atrial fibrillation cycle length and inducibility
during catheter ablation and their relation to outcome.
Circulation. 109:3007–3013. 2004.PubMed/NCBI
|
|
26.
|
Sheikh I, Krum D, Cooley R, et al:
Pulmonary vein isolation and linear lesions in atrial fibrillation
ablation. J Interv Card Electrophysiol. 17:103–109. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Fiala M, Chovancík J, Nevralová R, et al:
Pulmonary vein isolation using segmental versus electroanatomical
circumferential ablation for paroxysmal atrial fibrillation: over
3-year results of a prospective randomized study. J Interv Card
Electrophysiol. 22:13–21. 2008.
|
|
28.
|
Karch MR, Zrenner B, Deisenhofer I, et al:
Freedom from atrial tachyarrhythmias after catheter ablation of
atrial fibrillation: a randomized comparison between 2 current
ablation strategies. Circulation. 111:2875–2880. 2005. View Article : Google Scholar
|
|
29.
|
Nilsson B, Chen X, Pehrson S, et al:
Recurrence of pulmonary vein conduction and atrial fibrillation
after pulmonary vein isolation for atrial fibrillation: a
randomized trial of the ostial versus the extraostial ablation
strategy. Am Heart J. 152:537–538. 2006. View Article : Google Scholar
|
|
30.
|
Chung MK, Shemanski L, Sherman DG, et al:
Functional status in rate-versus rhythm-control strategies for
atrial fibrillation: results of the Atrial Fibrillation Follow-up
Investigation of Rhythm Management (AFFIRM) Functional Status
Substudy. J Am Coll of Cardiol. 46:1891–1899. 2005. View Article : Google Scholar
|
|
31.
|
Reynolds MR, Gunnarsson CL, Hunter TD, et
al: Health outcomes with catheter ablation or antiarrhythmic drug
therapy in atrial fibrillation: results of a propensity-matched
analysis. Circ Cardiovasc Qual Outcomes. 5:171–181. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Ogawa S, Yamashita T, Yamazaki T, et al:
Optional treatment stategy for patients with paroxysmal atrial
fibrillation: J-RHYTHM study. Circ J. 73:242–248. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Tan HW, Wang XH, Shi HF, et al: Left
atrial wall thickness: anatomic aspects relevant to catheter
ablation of atrial fibrillation. Chin Med J (Engl). 125:12–15.
2012.PubMed/NCBI
|
|
34.
|
Hunter RJ, Liu Y, Lu Y, et al: Left atrial
wall stress distribution and its relationship to electrophysiologic
remodeling in persistent atrial fibrillation. Circ Arrhythm
Electrophysiol. 5:351–360. 2012. View Article : Google Scholar : PubMed/NCBI
|