Introduction
Lung cancer is a common type of malignant tumor,
which seriously impacts human health and quality of life due to its
high incidence and mortality rates (1). Approximately 80–85% of lung cancers
are non-small cell lung cancers (NSCLCs) and the majority of those
patients are diagnosed at the middle or late stage, which results
in a poor prognosis (2,3). Surgery is the predominant treatment
method for NSCLC, however, 60–70% of patients who receive surgery
exhibit postoperative recurrence and metastasis, and only 20%
survive (4,5). Therefore, the aim of current clinical
research is to reduce the recurrence rate and postoperative
metastasis, as well as elongate the survival time of patients.
Chemotherapy and radiotherapy, which have been adopted as basic
postoperative treatment strategies in NSCLC, reduce the local
recurrence rate in patients and extend their survival time to a
certain extent, however, the effect is limited (6,7). For
NSCLC patients, an effective immune system is significant for the
suppression of tumor recurrence and metastasis. As a result of
this, dendritic cell-cytokine induced killer (DC-CIK) cell-based
immunotherapy has been widely used in numerous tumor treatments and
has significantly prolonged patient survival time and improved
immune function (8–10). However, the effect of DC-CIK cell
immunotherapy on NSCLC, particularly stage III NSCLC, has not been
reported. Post-surgery NSCLC patients received GP chemotherapy and
DC-CIK cell immunotherapy in the present study.
Patients and methods
General information
Patients with stage III NSCLC (n=157) received
complete resection surgery between June 2010 and June 2013. The
inclusion criteria for the present study were as follows: i)
Patients were pathologically diagnosed with adenocarcinoma,
squamous cell carcinoma or adenosquamous-mixed NSCLC; ii) patients
were at stage IIIA according to the International Union Against
Cancer NSCLC criteria (7); iii)
patients were aged 30–78 years; iv) patients exhibited normal
function of the hematopoietic system, liver, kidney and heart; v)
Karnofsky performance status score of the patients was >60
points; and vi) patients provided a signed informed consent sheet.
The exclusion and rejection criteria were as follows: i) Those who
were not able to complete the entire course of treatment and ii)
patients who exhibited an adverse reaction to treatment. The 157
cases were randomly divided into a control group and an observation
group. The control group consisted of 78 cases, including 46 males
and 32 females (aged, 30–76 years; average age, 58.2±11.2 years).
The cytology types were as follows: Adenocarcinoma (n=41), squamous
cell carcinoma (n=28) and adenosquamous carcinoma (n=9). The
observation group consisted of 79 cases, including 49 male and 30
females (aged, 32–78 years; average age, 59.6±10.7 years). The
cytology types were as follows: Adenocarcinoma (n=44), squamous
cell carcinoma (n=27) and adenosquamous carcinoma (n=8). There was
no significant difference identified regarding age, gender or
cytology type between the control and observation groups. The
present study was conducted in accordance with the declaration of
Helsinki and approval was obtained from the Ethics Committee of
Henan University Huaihe Hospital (Kaifeng, China). Written informed
consent was obtained from all of the participants.
DC and CIK cell expansion
The mononuclear cells of the patients in the
observation group were isolated using a blood cell separator
(StatSpin, Norwood, MA, USA). Following purification using a
lymphocyte separation medium, the cells were cultured for 2 h to
allow pre-adhesion. The non-adherent cells were collected and the
adherent cells were treated with RPMI-1640 medium containing 1,000
U/ml granulocyte-macrophage colony-stimulating factor, 500 U/ml
interleukin (IL)-4 and 500 U/ml tumor necrosis factor (TNF)-α
(R&D Systems, Minneapolis, MN, USA), and the medium was
replaced every four days. The cells were cultured for 8–12 days and
were considered to be DCs. The non-adherent cells were cultured at
a concentration of 2.5×106 cells/ml and treated with
1,000 U/ml interferon (INF)-γ (R&D Systems). One day later the
cells were stimulated with 100 U/ml IL-1α, 500 U/ml IL-2 and 50
ng/ml cluster of differentiation (CD) 3 monoclonal antibodies
(R&D Systems), and the medium was changed every three days. The
cells were cultured for 8–12 days and were considered to be CIK
cells. The cultured DCs and CIK cells were collected on day nine,
mixed at a ratio of 1:5, co-cultured for three days and used for
reinfusion. Prior to cell reinfusion, the expression level of CD80,
83, 86, 1A and human leukocyte antigen (HLA)-DR (eBioscience,
SanDiego, CA, USA) in the DCs was detected using flow cytometry (BD
Biosciences, Franklin Lakes, NJ, USA). In addition, the expression
of CD3, 56 and 8 (eBioscience, San Diego, CA, USA) in the CIK cells
was analyzed.
Regimen
The control group patients were treated with
gemcitabine plus platinum (GP) following surgery as follows:
Gemcitabine (1,000 mg/m2), dissolved in 500 ml 5%
glucose and intravenously infused at Day 1 and Day 8; 30
mg/m2 cisplatin (Zhunzi H20010743; Jiangsu Stockhausen
Pharmaceutical Co., Ltd., Jiangsu, China) dissolved in 500 ml of 5%
glucose and intravenously injected at Days 1–3. The treatment cycle
was 21 days and each patient underwent four cycles of treatment.
The patients in the observation group received two cycles of GP
chemotherapy following surgery, at two-week intervals and
subsequently received DC-CIK cell therapy twice a week. Following
this, the patients received two cycles of GP chemotherapy, at
two-week intervals and received a further DC-CIK cell treatment. In
total, the patients in the observation group underwent four cycles
of GP chemotherapy and two cycles of DC-CIK treatment.
Observation indicators and evaluation
criteria
Analysis of immune function was performed as
follows: Blood (5 ml) was collected from the patients in the
control and observation groups prior to and one month following
treatment. The percentage of CD3+CD4+ and
CD3+CD8+ T lymphocytes and CD56+
natural killer (NK) cells in the peripheral blood, was analyzed
using flow cytometry to assess the cellular immune capacity of the
patients. The two groups were followed up for 36 months to observe
recurrence and metastasis. Disease-free survival time, cumulative
recurrence rate and cumulative survival rate were calculated in the
two groups. The time between surgery and recurrence or metastasis
was considered to be the disease-free survival time (11).
Statistical analysis
The data were analyzed using SPSS 17.0 statistical
software (SPSS Inc., Chicago, IL, USA) and the measurement data
were compared using Student’s t-test and were shown as means ±
standard deviation. The count data were compared using a
χ2 test and the cumulative recurrence rates, and
cumulative survival rates were compared using Kaplan-Meier curves
and the log-rank test. P<0.05 was considered to indicate a
statistically significant difference.
Results
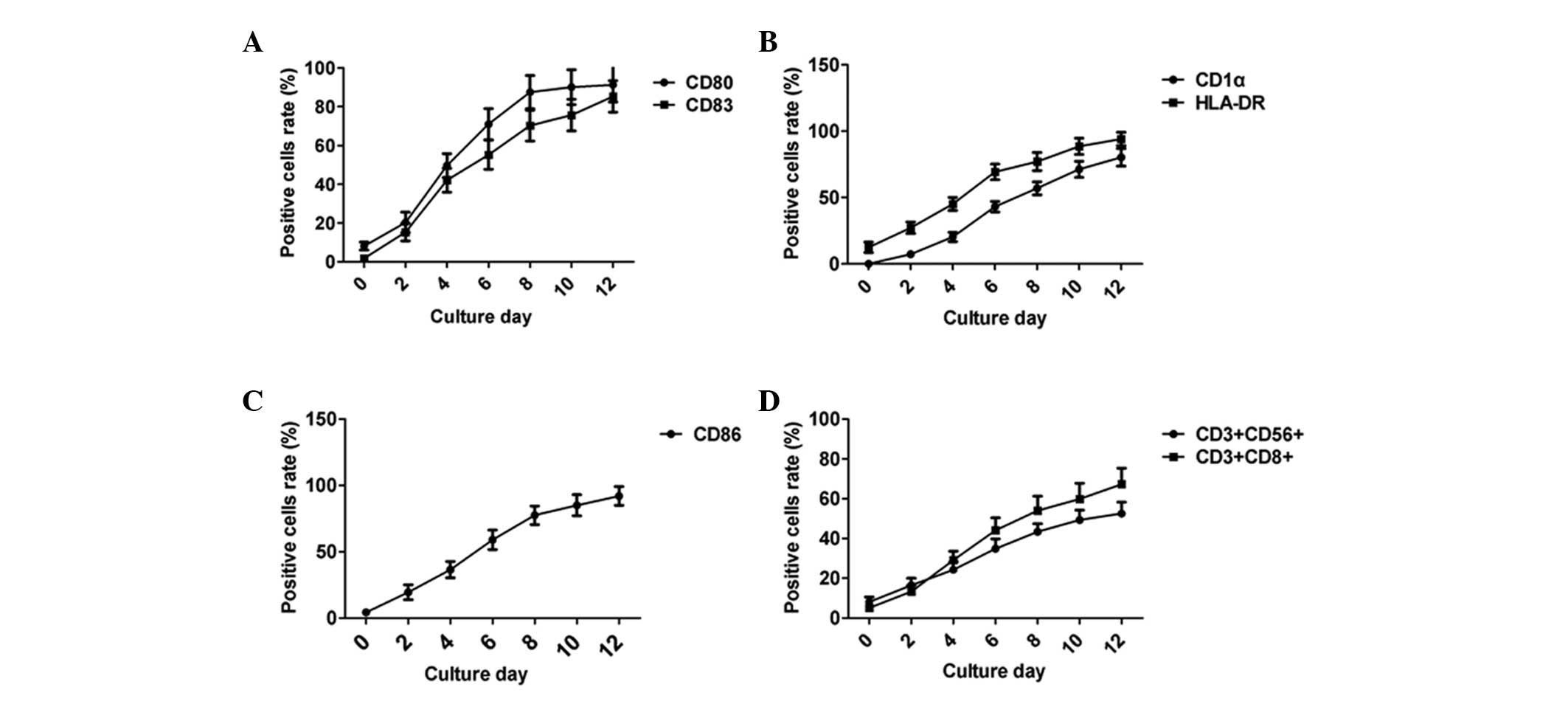
DC-CIK in vitro amplification
The DCs were amplified in vitro in accordance
with their culture time and the positive expression rates of the DC
surface markers, CD80, 83, 86, 1α and HLA-DR, gradually increased
(Fig. 1A). The number of
CD3+CD56+ and CD3+CD8+
double positive CIK cells increased in accordance with their
incubation time (Fig. 1B).
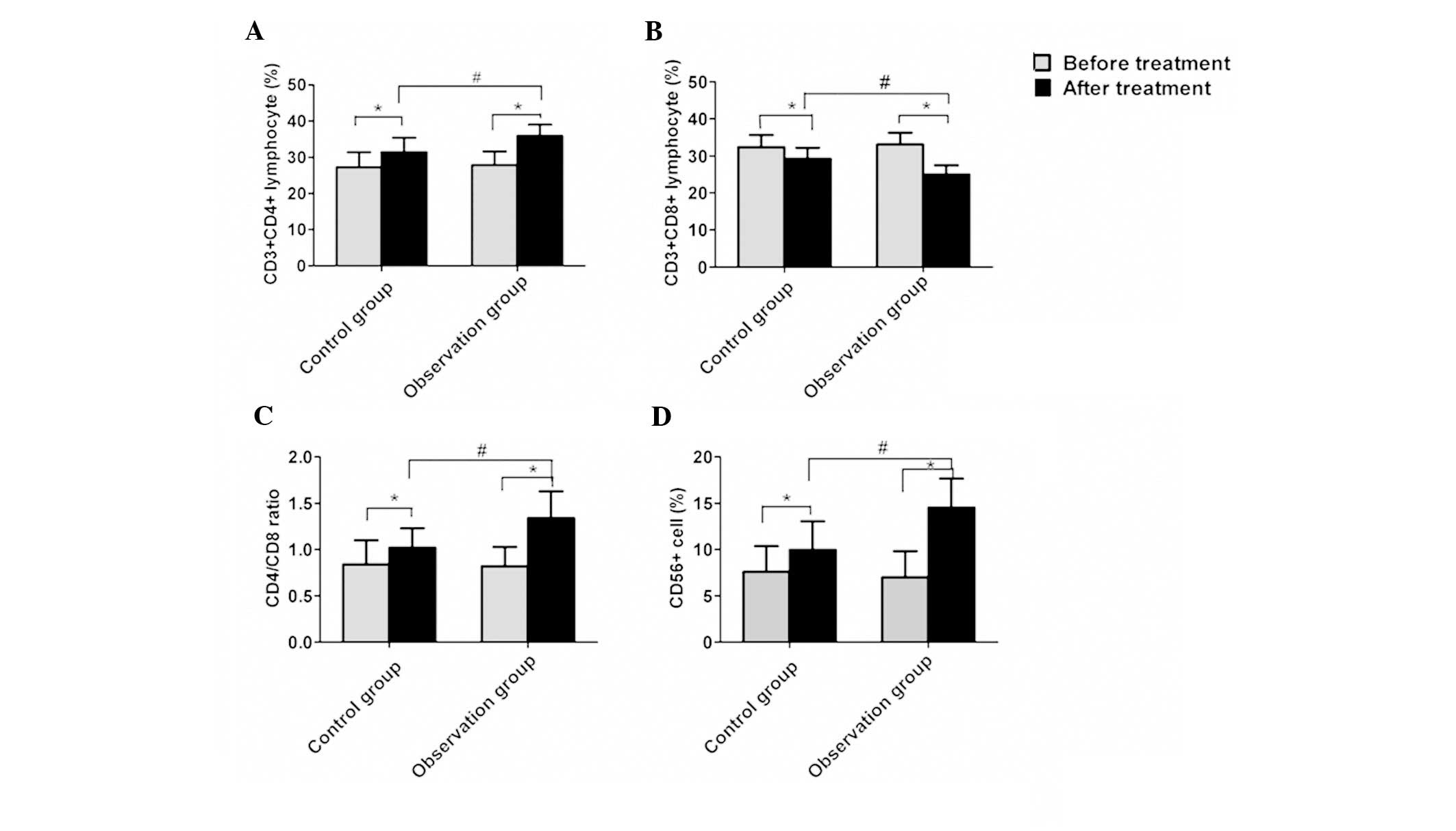
Comparison of postoperative cellular
immune function
The expression of CD3+CD4+ T
lymphocytes, the CD56+ NK cell percentage and the
CD4/CD8 ratio were observed to be significantly higher following
treatment compared with prior to treatment in the control and
observation groups, and the percentage of
CD3+CD8+ T lymphocytes significantly
decreased (Fig. 2, P<0.05). The
increase of the CD3+CD4+ T lymphocytes, the
CD56+ NK cell percentage and the CD4/CD8 ratio, and the
reduction of the CD3+CD8+ T lymphocyte
percentage in the peripheral blood were identified to be
significantly increased in the observation group compared with the
control group (P<0.05).
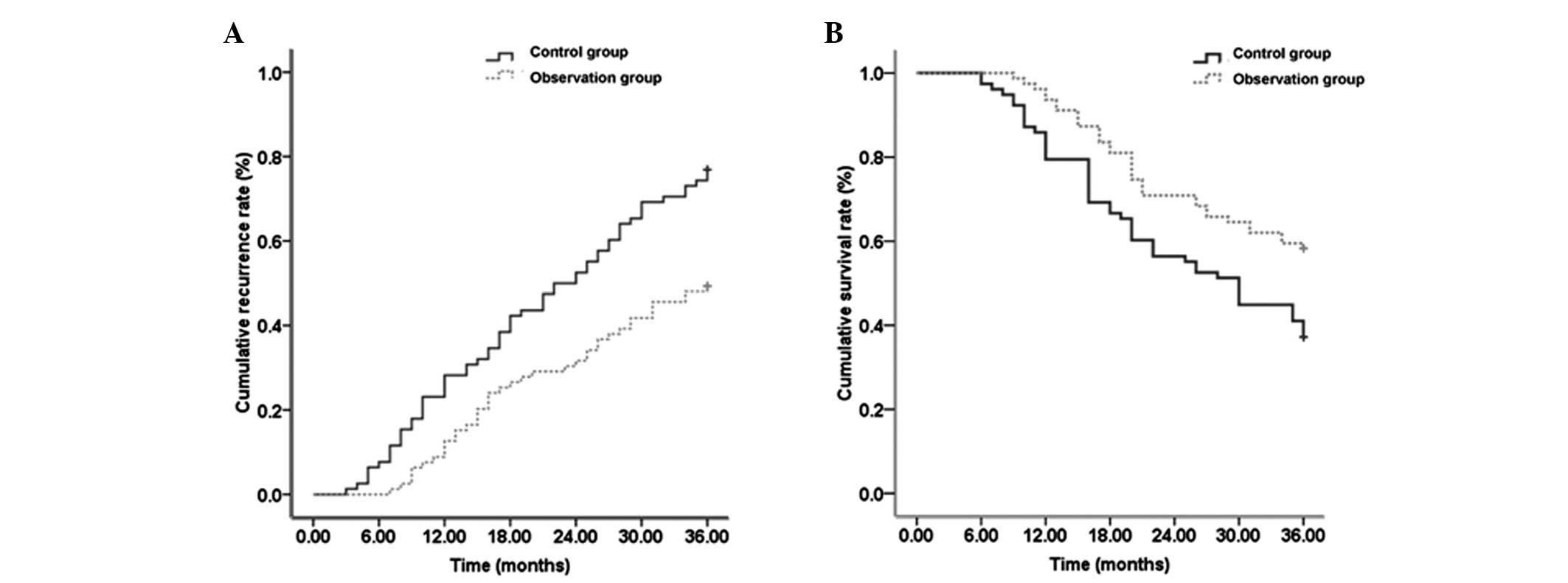
Comparison of the cumulative recurrence
rate and the cumulative survival rate
In the 36 month follow-up, the median survival time
of the patients without disease in the control and observation
group was 22 months (95% confidence interval (CI): 16.230–27.770
months) and 28 months (95% CI: 24.390–31.610 months), respectively,
which was identified to be a statistically significant difference
(P<0.05). The cumulative recurrence rates over the three years
that were observed in the control group were 28.21, 52.56 and
76.92% compared with 11.39, 30.38 and 47.37% in the observation
group. This identified that the cumulative recurrence rates in the
observation group were significantly lower than those of the
control group (Fig. 3A;
P<0.05). In addition, the cumulative survival rates over the
three years that were observed in the control group were 6.33,
29.11 and 58.23% compared with 37.14, 41.03 and 62.82% in the
observation group. This demonstrated that the cumulative survival
rates in the observation group were significantly higher than that
of the control group (Fig. 3B,
P<0.05).
Discussion
Previous studies have shown that in patients with
stage IIIA NSCLC, the median survival time and five-year survival
rates of patients that underwent surgery were similar to that of
the patients that were treated with GP chemotherapy, which
indicated that surgery alone does not result in a significantly
improved prognosis (12,13). Recurrence following surgery is the
leading cause of mortality in patients with NSCLC, 80% of which
develop distant metastases and 20% exhibit a local recurrence
(14). Therefore, optimizing the
postoperative treatment is critical to reduce the incidence of
postoperative recurrence and, thus, extend the survival time of
patients. Radiotherapy and chemotherapy are common methods of
treating patients with NSCLC; however, it has been found that
radiotherapy and chemotherapy do not alter the long-term outcome in
the majority of stage IIIA patients, which may be associated with a
plateau in the efficacy of these therapies (15). In recent years, as a result of
investigation into the immune function associated with cancer
development and metastasis, cellular-immunity based therapeutic
strategies have been gradually applied to certain malignant types
of tumors, including gastric, bladder and breast cancer, with
significant effects (16–18). DCs are primary antigen-presenting
cells within the body and are able to release large quantities of
cytokines, such as INF and IL-12, which mediate antitumor immune
responses via multiple pathways (19). CIK cells are a cell subset of
heterogeneous NK cell-like cells, which originate from peripheral
blood mononuclear cells and are induced by a variety of cytokines
in vivo. The CD16+CD56+ double
positive cell subset within CIK cells releases a large quantity of
antitumor-associated cytokines and exhibits a strong cytotoxic
effect on tumor cells independent of major histocompatibility
complex II (20). Therefore, in
the present study, NSCLC patients were treated with DC-CIK cell
immunotherapy on the basis of a GP regimen to investigate its
impact on recurrence and survival time.
The immune function of the two patient groups was
analyzed in the present study. Numerous studies have shown that the
immune system (particularly the cellular immune system in cancer
patients) exhibits severe defects, including a reduction of
CD3+CD4+ T cells, an increase in the number
of CD3+CD8+ T cells, a decrease in the
CD4+/CD8+ ratio and a depression of NK cell
killing activity (8). The present
study demonstrated that the percentage of T lymphocyte and NK cell
subsets in peripheral blood were severely abnormal in the
observation and control group patients. The number of
CD3+CD4+ T cells, the NK cell percentage and
the CD4+/CD8+ ratio were significantly higher
in the control and observation groups following treatment compared
with before treatment; conversely, the
CD3+CD8+ T cell percentage was significantly
decreased. Furthermore, the immune function of the observation
group patients following treatment was significantly improved
compared with the control group patients. The results indicated
that immune function was restored in the DC-CIK cell-therapy
treated patients. Enhancement of immune capacity considerably
improved the antitumor immune response, which efficiently killed
the residual tumor cells following surgery, in addition to
effectively preventing tumor metastasis and recurrence, as well as
prolonging the survival time of patients.
The present study further analyzed the effects of
DC-CIK cell therapy combined with GP chemotherapy on NSCLC
recurrence and survival time. The data demonstrated that the median
disease-free survival time of patients that underwent GP
chemotherapy alone was significantly shorter than that of patients
who received GP chemotherapy combined with DC-CIK cell therapy. The
three-year recurrence rate in the patients that received GP
chemotherapy combined with DC-CIK cell therapy (47.37%) was lower
than that observed in the GP chemotherapy alone group (76.92%),
while the three-year cumulative survival rate of the patients that
received GP chemotherapy combined with DC-CIK cell therapy (58.23%)
was higher compared with the GP chemotherapy-alone group (37.14%).
The results demonstrated that DC-CIK cell immunotherapy combined
with GP chemotherapy was significant in reducing recurrence and
extending the survival time of patients with NSCLC.
In conclusion, DC-CIK cell immunotherapy combined
with GP chemotherapy improved the postoperative immune function in
patients with NSCLC, reduced recurrence and extended the patient
survival time following surgery; therefore, it may be adopted in
the future for clinical use.
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2012. CA Cancer J Clin. 62:10–29. 2012. View Article : Google Scholar
|
|
2
|
Goldstraw P, Ball D, Jett JR, Le Chevalier
T, Lim E, Nicholson AG and Shepherd FA: Non-small-cell lung cancer.
Lancet. 378:1727–1740. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Naim Younes R, Gross JL, Abrao FG and
Rodrigues Pereira J: Impact of adjuvant chemotherapy in completely
resected stage IIIA non-small cell lung cancer. Minerva Chir.
68:169–174. 2013.
|
|
4
|
Filipits M and Pirker R: Predictive
markers in the adjuvant therapy of non-small cell lung cancer. Lung
Cancer. 74:355–363. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jemal A, Murray T, Ward E, et al: Cancer
statistics, 2005. CA Cancer J Clin. 55:10–30. 2005. View Article : Google Scholar
|
|
6
|
Pisters KM: Adjuvant chemotherapy for
non-small-cell lung cancer - the smoke clears. N Engl J Med.
352:2640–2642. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ost D, Goldberg J, Rolnitzky L and Rom WN:
Survival after surgery in stage IA and IB non-small cell lung
cancer. Am J Respir Crit Care Med. 177:516–523. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhong GC, Yan B, Sun Y, et al: Clinical
efficacy of immunotherapy of dendritic cell and cytokine-induced
killer cell combined with chemotherapy for treatment of multiple
myeloma. Zhonghua Xue Ye Xue Za Zhi. 33:1000–1003. 2012.(In
Chinese).
|
|
9
|
Zhan HL, Gao X, Pu XY, Li W, Li ZJ, Zhou
XF and Qiu JG: A randomized controlled trial of postoperative tumor
lysate-pulsed dendritic cells and cytokine-induced killer cells
immunotherapy in patients with localized and locally advanced renal
cell carcinoma. Chin Med J (Engl). 125:3771–3777. 2012.
|
|
10
|
Shi SB, Ma TH, Li CH and Tang XY: Effect
of maintenance therapy with dendritic cells: cytokine-induced
killer cells in patients with advanced non-small cell lung cancer.
Tumori. 98:314–319. 2012.PubMed/NCBI
|
|
11
|
Sargent D, Shi Q, Yothers G, et al;
Adjuvant Colon Cancer End-points (ACCENT) Group. Two or three year
disease-free survival (DFS) as a primary end-point in stage III
adjuvant colon cancer trials with fluoropyrimidines with or without
oxaliplatin or irinotecan: data from 12,676 patients from MOSAIC,
X-ACT, PETACC-3, C-06, C-07 and C89803. Eur J Cancer. 47:990–996.
2011.
|
|
12
|
Fidler MJ, Kim AW, Zusag T and Bonomi P:
Treatment of locally advanced non-small cell lung cancer. Clin Adv
Hematol Oncol. 7:455–464. 479–480. 2009.
|
|
13
|
Juretic A, Sobat H and Samija M: Combined
modality therapy of non-small cell lung cancers. Ann Oncol.
10(Suppl 6): 93–98. 1999. View Article : Google Scholar
|
|
14
|
Seder CW, Allen MS, Cassivi SD, et al:
Stage IIIA non-small cell lung cancer: morbidity and mortality of
three distinct multimodality regimens. Ann Thorac Surg.
95:1708–1716. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Carney DN: Lung cancer - time to move on
from chemotherapy. N Engl J Med. 346:126–128. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yuan XK, Zhao XK, Xia YC, Zhu X and Xiao
P: Increased circulating immunosuppressive
CD14+HLA-DR−/low cells correlate with
clinical cancer stage and pathological grade in patients with
bladder carcinoma. J Int Med Res. 39:1381–1391. 2011.PubMed/NCBI
|
|
17
|
Ren J, Di L, Song G, et al: Selections of
appropriate regimen of high-dose chemotherapy combined with
adoptive cellular therapy with dendritic and cytokine-induced
killer cells improved progression-free and overall survival in
patients with metastatic breast cancer: reargument of such
contentious therapeutic preferences. Clin Transl Oncol. 15:780–788.
2013.
|
|
18
|
Shi L, Zhou Q, Wu J, Ji M, Li G, Jiang J
and Wu C: Efficacy of adjuvant immunotherapy with cytokine-induced
killer cells in patients with locally advanced gastric cancer.
Cancer Immunol Immunother. 61:2251–2259. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sangiolo D, Mesiano G, Carnevale-Schianca
F, Piacibello W, Aglietta M and Cignetti A: Cytokine induced killer
cells as adoptive immunotherapy strategy to augment graft versus
tumor after hematopoietic cell transplantation. Expert Opin Biol
Ther. 9:831–840. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zheng YW, Li RM, Zhang XW and Ren XB:
Current adoptive immunotherapy in non-small cell lung cancer and
potential influence of therapy outcome. Cancer Invest. 31:197–205.
2013. View Article : Google Scholar : PubMed/NCBI
|