Introduction
The farnesoid X receptor (FXR), a member of the
nuclear receptor superfamily, was initially isolated in the liver
and identified as a bile acid sensor (1–5). FXR
plays a critical role in the regulation of bile acid, cholesterol,
triglyceride and glucose homeostasis (6–9). For
example, ablation of the FXR in C57BL/6 mice was shown to result in
severe hepatic cholestasis, liver steatosis and insulin resistance
(6,9).
Previous studies have also hypothesized that FXR is
involved in the regulation of tumorigenesis (10–13).
One study demonstrated that male and female FXR knockout mice
spontaneously developed liver tumors, which was accompanied with
liver injury and inflammation (10). Loss of the FXR in
ApcMin/+ and chronic colitis mouse models of intestinal
tumorigenesis was shown to result in early mortality and increased
tumor progression via the promotion of Wnt signaling by
infiltrating neutrophils and macrophages and proinflammatory
cytokine production (11). In
addition, FXR agonists have been shown to reduce liver and
intestine tumor growth and metastasis in an orthotopic mouse
xenograft model (12).
Furthermore, downregulation of FXR has been associated with
multiple malignant clinicopathological characteristics in human
hepatocellular carcinoma (13),
indicating that FXR functions as an important tumor suppressor.
However, whether FXR affects prostate cancer cell proliferation
remains unknown. The aim of the present study was to investigate
the roles and molecular mechanisms of FXR in prostate cancer cell
proliferation.
Materials and methods
Cell culture and tissue samples
LNcaP cells were purchased from the American Type
Culture Collection (Rockville, MD, USA). Cells were culture in
Dulbecco’s modified Eagle’s medium (DMEM; Gibco-BRL, Beijing,
China) supplemented with 10% fetal bovine serum (Gibco-BRL). After
seeding in the 96- or 6-well plates for 24 h, cells were treated
with chenodeoxycholic acid (CDCA) (5 μM), GW4064 (2 μM) or vehicle
control (DMSO). Small interfering RNA oligos targeting FXR or
negative control (NC) were obtained from Genepharm Company
(Shanghai, China). For the cell transfection experiments, LNcaP
cells were grown to 70–80% confluence in six-well plates. The cells
were transiently transfected using Lipofectamine 2000 (Invitrogen
Life Technologies, Carlsbad, CA, USA). Prostate cancer tissues and
adjacent normal tissues were collected from patients undergoing
routine therapeutic surgery at the Department of Urology Surgery in
Huashan Hospital Affiliated to Fudan University (Shanghai, China).
All the samples were collected from patients that provided informed
consent, and the experimental procedures were approved by the
Institutional Review Board of Huashan Hospital Affiliated to Fudan
University.
mRNA isolation and quantitative
polymerase chain reaction (PCR)
Total RNA was obtained from the tissue samples, and
cells were harvested using TRIzol kits (Invitrogen Life
Technologies). Quantitative PCR was performed using an Applied
Biosystems 7900 Real-time PCR System (Shanghai, China) and a
TaqMan Universal PCR Master Mix (Takara, Dalian, China),
according to the manufacturer’s instructions.
Bromodeoxyuridine (BrdU) assays
A cell proliferation enzyme-linked immunosorbent
assay (ELISA; BrdU kit; Beyotime Institute of Biotechnology,
Shanghai, China) was used to analyze the incorporation of BrdU
during DNA synthesis, according to the manufacturer’s instructions.
All the experiments were performed in triplicate, and the
absorbance was measured at 450 nm using the Spectra Max 190 ELISA
reader (Molecular Devices, Sunnyvale, CA, USA).
Western blot analysis
Proteins were separated by 10% SDS-PAGE and
transferred to nitrocellulose membranes (Amersham Bioscience,
Little Chalfont, UK). Following blocking with 10% nonfat milk in
phosphate-buffered saline, the membranes were immunoblotted with
antibodies as indicated, followed by horseradish
peroxidase-conjugated secondary antibodies (Cell Signaling
Technology, Inc., Beverly, MA, USA). The signals were detected
using a SuperSignal West Pico Chemiluminescent Substrate kit
(Pierce Biotechnology, Inc., Rockford, IL, USA), according to
manufacturer’s instructions. Anti-FXR, -PTEN and -Akt antibodies
were purchased from Abcam (Cambridge, MA, USA). Protein expression
levels of GAPDH were used as an internal control.
Statistical analysis
Data are expressed as the mean ± standard error of
the mean from at least three separate experiments. Differences
between the groups were analyzed using the Student’s t-test, where
P<0.05 was considered to indicate a statistically significant
difference. Differences between the groups were analyzed by
two-tailed Student’s t tests using SPSS version 13.0 (SPSS, Inc.,
Chicago, IL,USA).
Results
FXR activation inhibits cell
proliferation
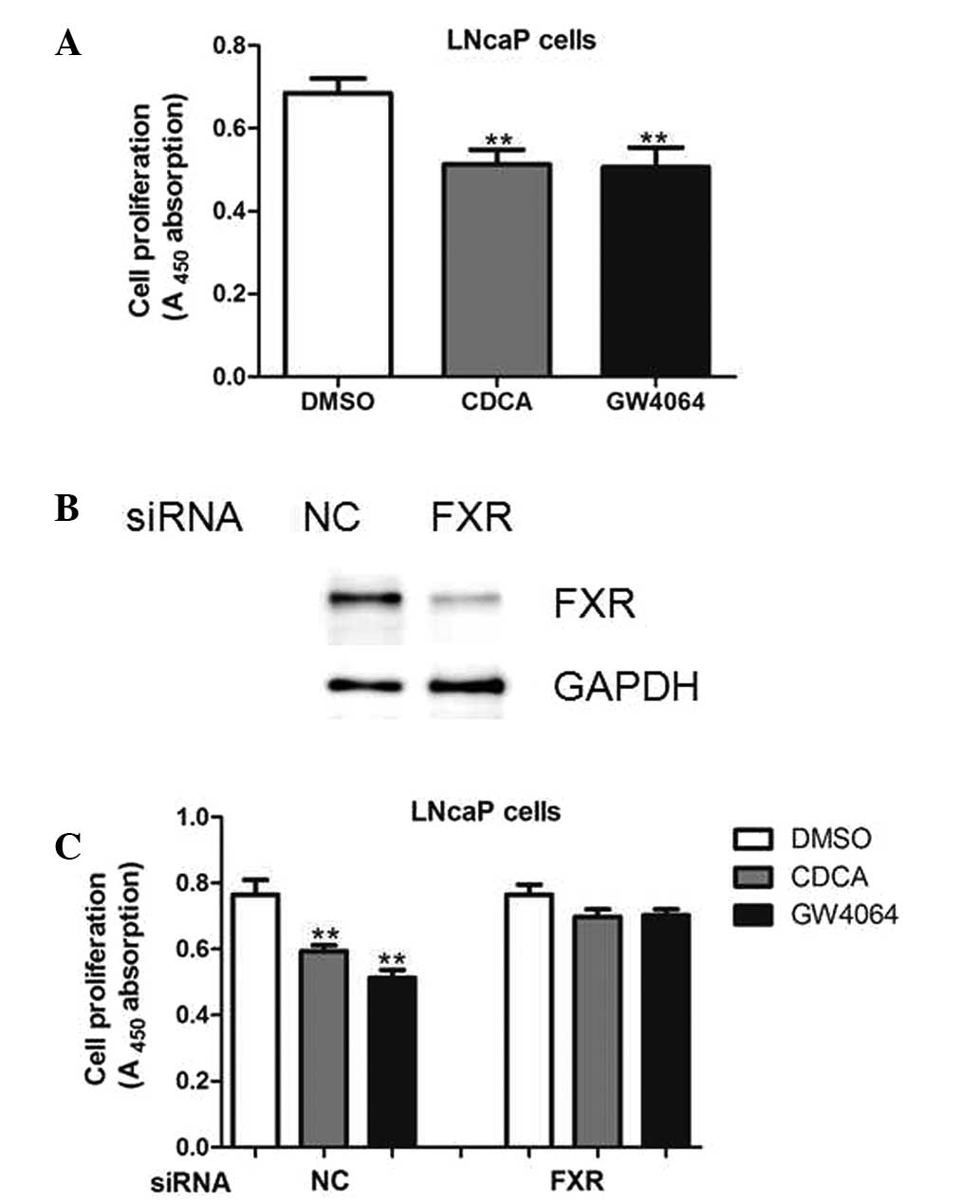
To evaluate the effects of FXR on prostate cancer
cell growth, LNcaP cells were treated with the FXR agonists, CDCA
and GW4064. As shown in Fig. 1,
CDCA and GW4064 decreased the proliferative ability of LNcaP cells
(Fig. 1). Next, endogenous FXR
expression was silenced using specific small interfering RNA oligos
in the LNcaP cells (Fig. 1B). As
expected, CDCA and GW4064 were unable to exert antiproliferative
roles in the presence of siRNA oligos targeting FXR (Fig. 1C), indicating that the
antiproliferative roles of the two compounds were dependent on FXR
expression.
FXR overexpression represses LNcaP cell
proliferation
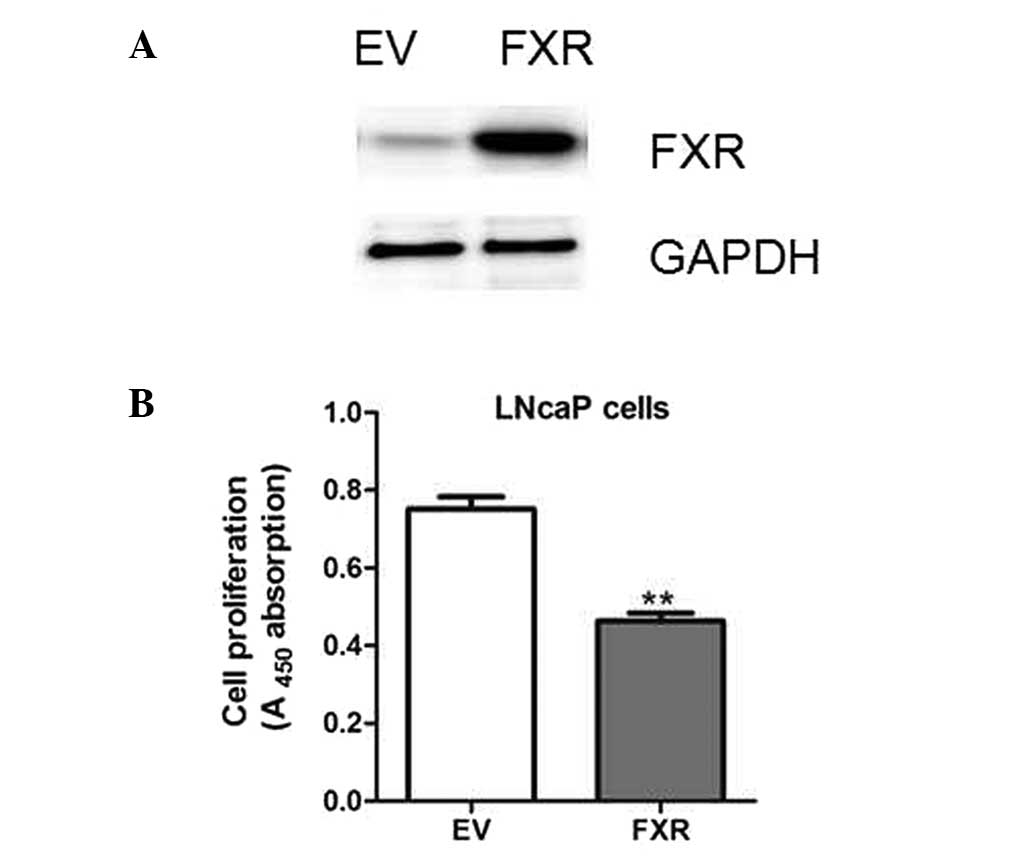
To further determine the potential functions of the
FXR, LNcaP cells were transfected with plasmids encoding FXR cDNA
or an empty vector (Fig. 2A). As a
result, FXR overexpression resulted in decreased cell
proliferation, as measured by BrdU analysis (Fig. 2B). Therefore, the results indicated
that FXR may be a tumor suppressor in prostate cancer cells.
FXR upregulates the expression levels of
the PTEN tumor suppressor
As FXR was shown to inhibit cell proliferation, the
effects of the receptor on the expression of the genes associated
with cell proliferation were investigated. Results from
quantitative PCR analysis indicated that PTEN was highly
upregulated following CDCA or GW4064 treatment, while other genes,
including p53, FOXO1, E2F1 and RB1, remained unchanged (Fig. 3A). In addition, the upregulation of
PTEN was confirmed by western blot analysis (Fig. 3B). Consistently, a reduction in the
level of Akt phosphorylation was observed in the LNcaP cells
treated with CDCA or GW4064 (Fig.
3B). Furthermore, PTEN expression was upregulated in the LNcaP
cells transfected with FXR when compared with the cells transfected
with the empty vectors (Fig. 3C and
D).
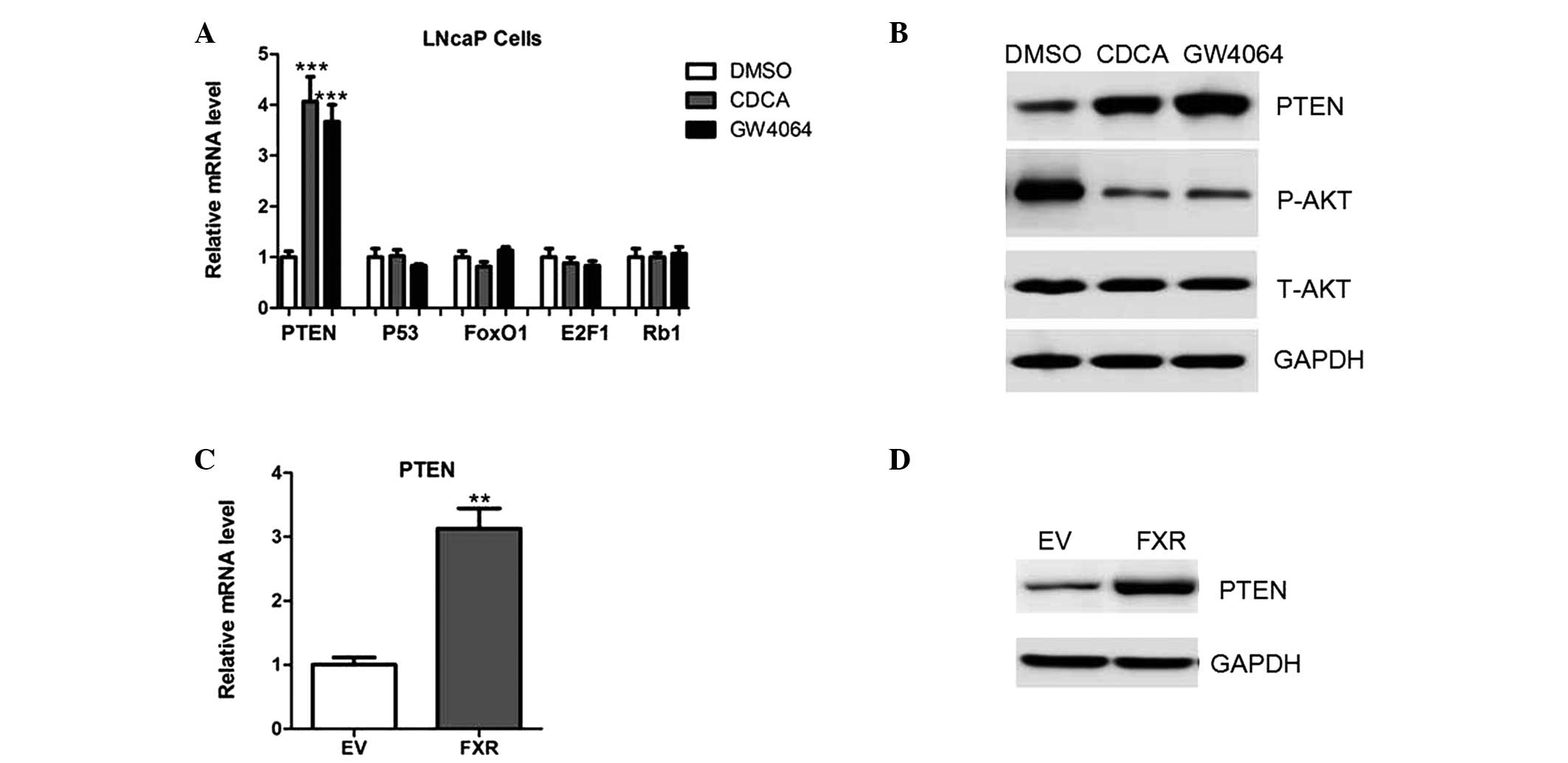 | Figure 3FXR upregulates the expression of
PTEN. (A) mRNA expression levels of PTEN, p53, FOXO1, E2F1 and RB1
in LNcaP cells treated with a vehicle control (DMSO), CDCA or
GW4064. (B) Protein expression levels of PTEN, phosphorylated and
total Akt in LNcaP cells treated with a vehicle control (DMSO),
CDCA or GW4064. (C) mRNA and (D) protein expression levels of PTEN
in LNcaP cells transfected with plasmids expressing an empty vector
or FXR. FXR, farnesoid X receptor; DMSO, dimethyl sulfoxide; CDCA,
chenodeoxycholic acid; EV, empty vector; P-AKT, phosphorylated Akt;
T-AKT, total Akt. |
FXR expression levels are decreased in
prostate cancer tissues
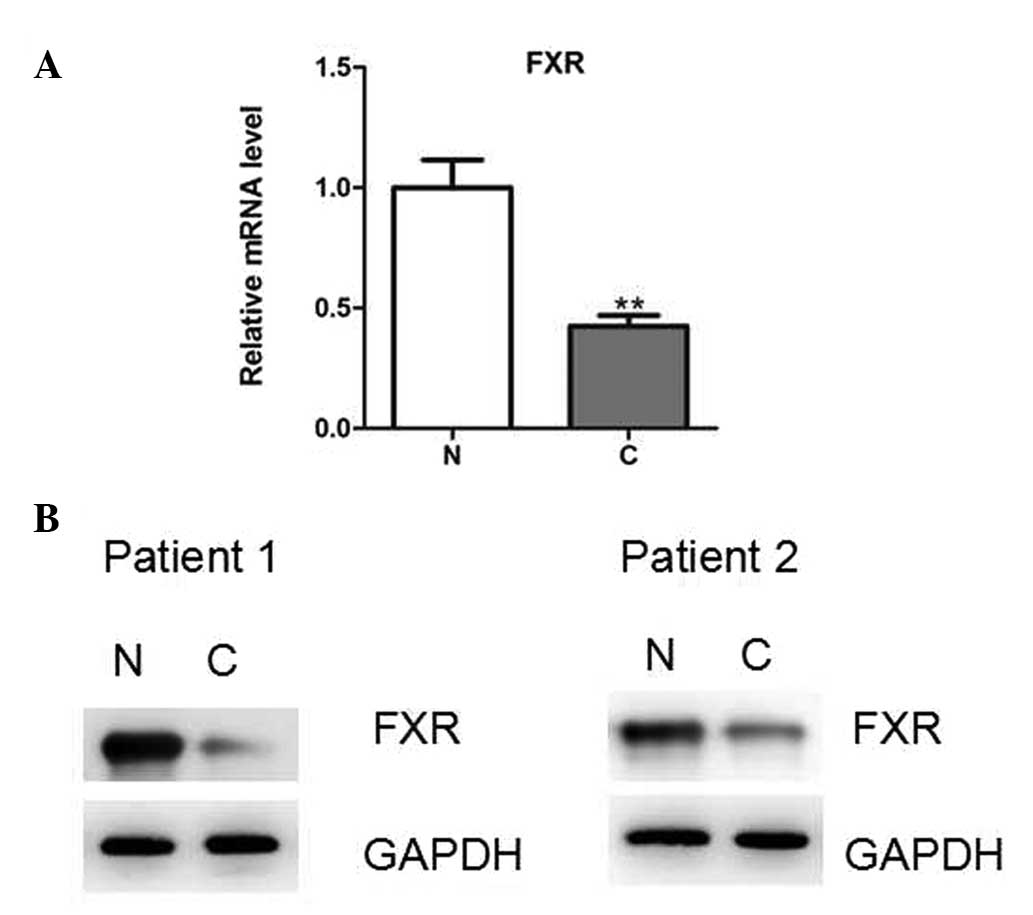
Finally, whether FXR was differentially expressed in
human prostate cancer tissues was investigated. The mRNA and
protein expression levels were determined using quantitative PCR
and western blot analysis, respectively, in human prostate cancer
tissues and pair-matched adjacent normal tissues. The results
demonstrated that FXR expression was significantly decreased in the
prostate cancer tissues (Fig. 4A and
B).
Discussion
In the present study, FXR activation or
overexpression was demonstrated to inhibit cell proliferation in
LNcaP cells. In addition, FXR expression was downregulated in
prostate cancer tissues. Therefore, to the best of our knowledge,
the present study, for the first time, identified that FXR may be a
tumor suppressor in the progression of prostate cancer. However,
the mechanisms underlying FXR downregulation remain unknown.
Previous studies have demonstrated that glucose, insulin,
proinflammatory cytokines and certain microRNAs are able to
regulate FXR in a variety of tissues or cells (14–16).
Therefore, further research into whether these factors contribute
to the downregulation of FXR expression in prostate cancer should
be performed.
Previous studies have demonstrated that FXR can
protect against tumorigenesis and inhibit cell proliferation in
several cancer types, including hepatocellular carcinoma and colon
cancer (10–12). Through the induction of downstream
target genes, such as SHP, FXR suppresses cell proliferation and
promotes apoptosis (17).
Accordingly, SHP null mice were shown to develop spontaneous liver
tumors, and the expression of SHP was demonstrated to be
downregulated in human cancer tissues (18,19).
In the present study, the results revealed that the
expression of the tumor suppressor gene, PTEN, was upregulated
following FXR activation. In humans, the loss or mutation of PTEN
has been observed in a group of autosomal dominant syndromes, which
are characterized by neurological disorders, multiple hamartomas
and cancer susceptibility (20).
In prostate cancer tissues, aberrant methylation of the PTEN gene
has been observed, which resulted in the inactivation of PTEN and
the hyperactivation of Akt (21).
Therefore, the FXR/PTEN signaling pathway may be a novel
pharmaceutical target for the treatment of prostate cancer.
In conclusion, the key observation of the present
study is that FXR inhibits the proliferation of prostate cancer
cell lines via the upregulation of PTEN expression. Understanding
the precise role played by FXR is likely to advance the knowledge
of prostate cancer biology, which may be beneficial for future
treatment.
References
|
1
|
Makishima M, Okamoto AY, Repa JJ, et al:
Identification of a nuclear receptor for bile acids. Science.
284:1362–1365. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Urizar NL, Liverman AB, Dodds DT, et al: A
natural product that lowers cholesterol as an antagonist ligand for
FXR. Science. 296:1703–1706. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hollman DA, Milona A, van Erpecum KJ and
van Mil SW: Anti-inflammatory and metabolic actions of FXR:
insights into molecular mechanisms. Biochim Biophys Acta.
1821:1443–1452. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Matsubara T, Li F and Gonzalez FJ: FXR
signaling in the enterohepatic system. Mol Cell Endocrinol.
368:17–29. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Porez G, Prawitt J, Gross B and Staels B:
Bile acid receptors as targets for the treatment of dyslipidemia
and cardiovascular disease. J Lipid Res. 53:1723–1737. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sinal CJ, Tohkin M, Miyata M, Ward JM,
Lambert G and Gonzalez FJ: Targeted disruption of the nuclear
receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell.
102:731–744. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gardmo C, Tamburro A, Modica S and
Moschetta A: Proteomics for the discovery of nuclear bile acid
receptor FXR targets. Biochim Biophys Acta. 1812:836–841. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kemper JK: Regulation of FXR
transcriptional activity in health and disease: Emerging roles of
FXR cofactors and post-translational modifications. Biochim Biophys
Acta. 1812:842–850. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lu Y, Ma Z, Zhang Z, et al: Yin Yang 1
promotes hepatic steatosis through repression of farnesoid X
receptor in obese mice. Gut. 63:170–178. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang F, Huang X, Yi T, Yen Y, Moore DD and
Huang W: Spontaneous development of liver tumors in the absence of
the bile acid receptor farnesoid X receptor. Cancer Res.
67:863–867. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Modica S, Murzilli S, Salvatore L, Schmidt
DR and Moschetta A: Nuclear bile acid receptor FXR protects against
intestinal tumorigenesis. Cancer Res. 68:9589–9594. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim I, Morimura K, Shah Y, Yang Q, Ward JM
and Gonzalez FJ: Spontaneous hepatocarcinogenesis in farnesoid X
receptor-null mice. Carcinogenesis. 28:940–946. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen Y, Song X, Valanejad L, et al: Bile
salt export pump is dysregulated with altered farnesoid X receptor
isoform expression in patients with hepatocellular carcinoma.
Hepatology. 57:1530–1541. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Duran-Sandoval D, Mautino G, Martin G, et
al: Glucose regulates the expression of the farnesoid X receptor in
liver. Diabetes. 53:890–898. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim MS, Shigenaga J, Moser A, Feingold K
and Grunfeld C: Repression of farnesoid X receptor during the acute
phase response. J Biol Chem. 278:8988–8995. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang Y, Gong W, Dai S, et al:
Downregulation of human farnesoid X receptor by miR-421 promotes
proliferation and migration of hepatocellular carcinoma cells. Mol
Cancer Res. 10:516–522. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ohno T, Shirakami Y, Shimizu M, et al:
Synergistic growth inhibition of human hepatocellular carcinoma
cells by acyclic retinoid and GW4064, a farnesoid X receptor
ligand. Cancer Lett. 323:215–222. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
He N, Park K, Zhang Y, Huang J, Lu S and
Wang L: Epigenetic inhibition of nuclear receptor small heterodimer
partner is associated with and regulates hepatocellular carcinoma
growth. Gastroenterology. 134:793–802. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang Y, Xu P, Park K, Choi Y, Moore DD
and Wang L: Orphan receptor small heterodimer partner suppresses
tumorigenesis by modulating cyclin D1 expression and cellular
proliferation. Hepatology. 48:289–298. 2008. View Article : Google Scholar
|
|
20
|
Hollander MC, Blumenthal GM and Dennis PA:
PTEN loss in the continuum of common cancers, rare syndromes and
mouse models. Nat Rev Cancer. 11:289–301. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Warde N: Prostate cancer: loss of PTEN
promotes progression of prostate cancer in an androgen-independent
manner. Nat Rev Urol. 8:4122011. View Article : Google Scholar : PubMed/NCBI
|