|
1
|
Gao HM and Hong JS: Gene-environment
interactions: key to unraveling the mystery of Parkinson’s disease.
Prog Neurobiol. 94:1–19. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Venkateshappa C, Harish G, Mythri RB, et
al: Increased oxidative damage and decreased antioxidant function
in aging human substantia nigra compared to striatum: implications
for Parkinson’s disease. Neurochem Res. 37:358–369. 2012.
View Article : Google Scholar
|
|
3
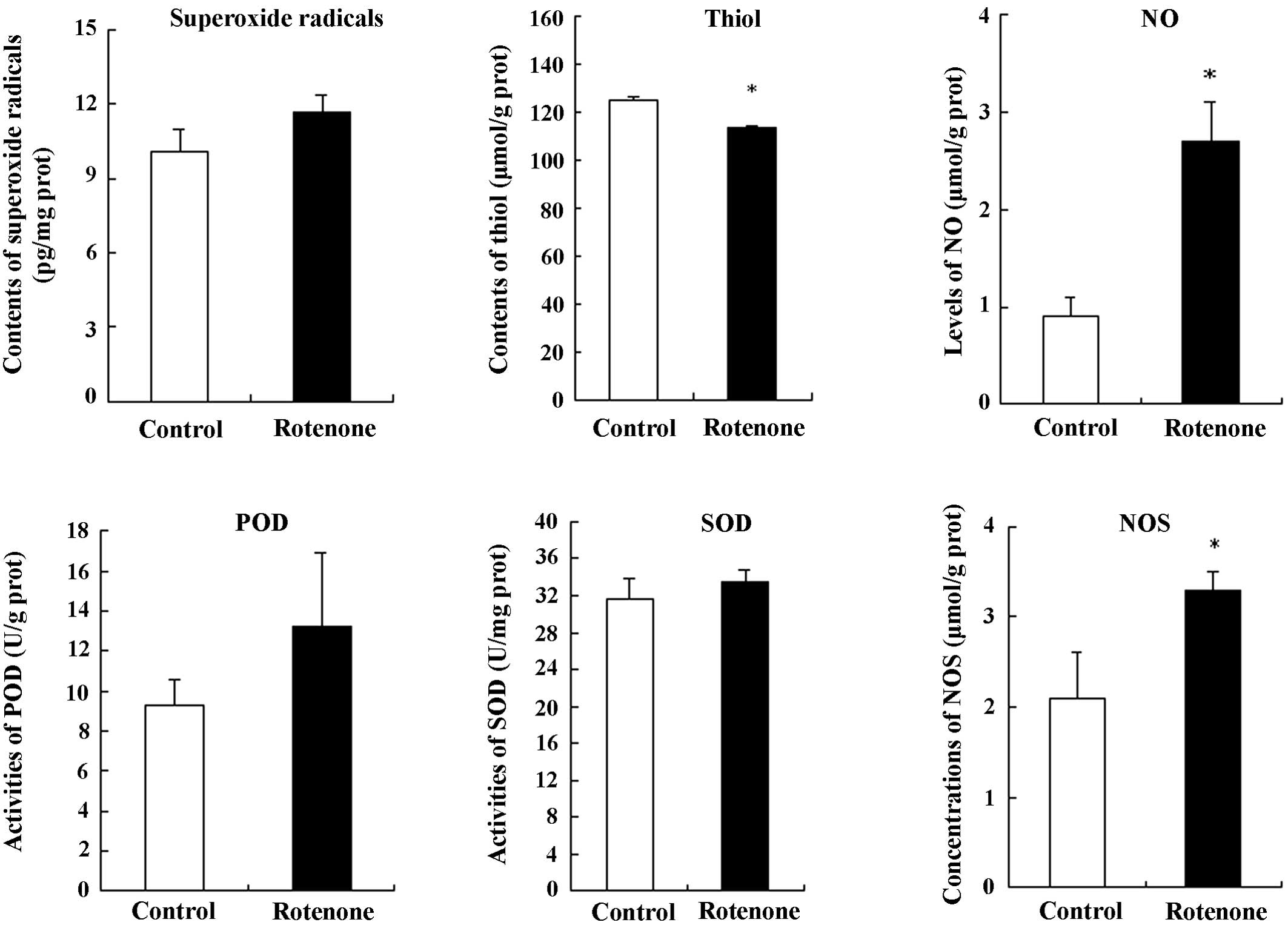
|
Fearnley JM and Lees AJ: Ageing and
Parkinson’s disease: substantia nigra regional selectivity. Brain.
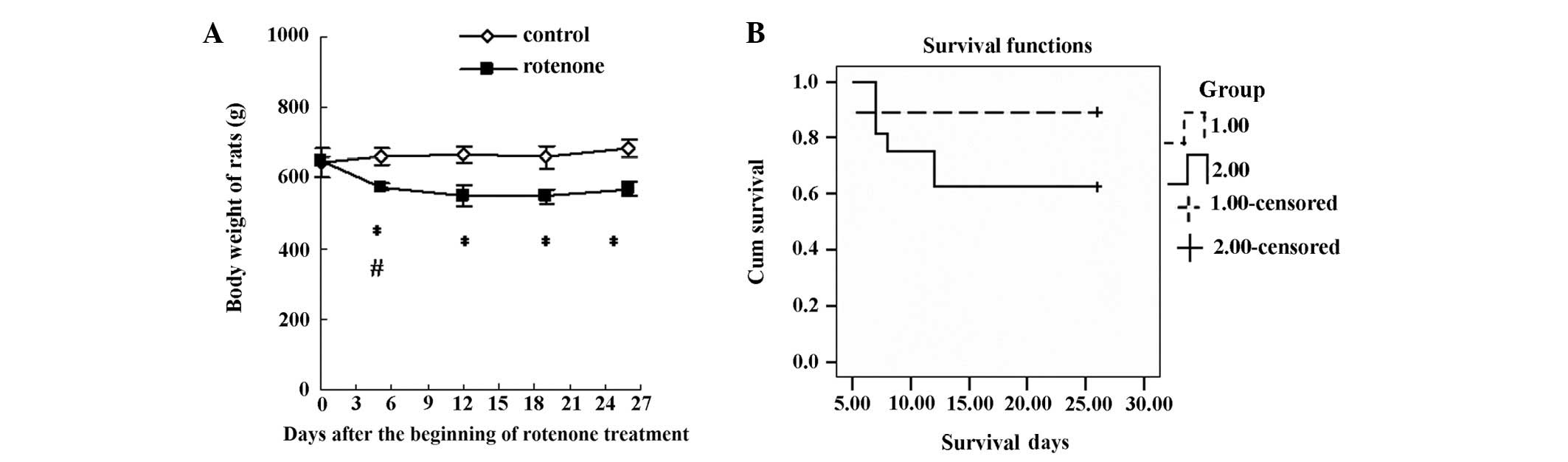
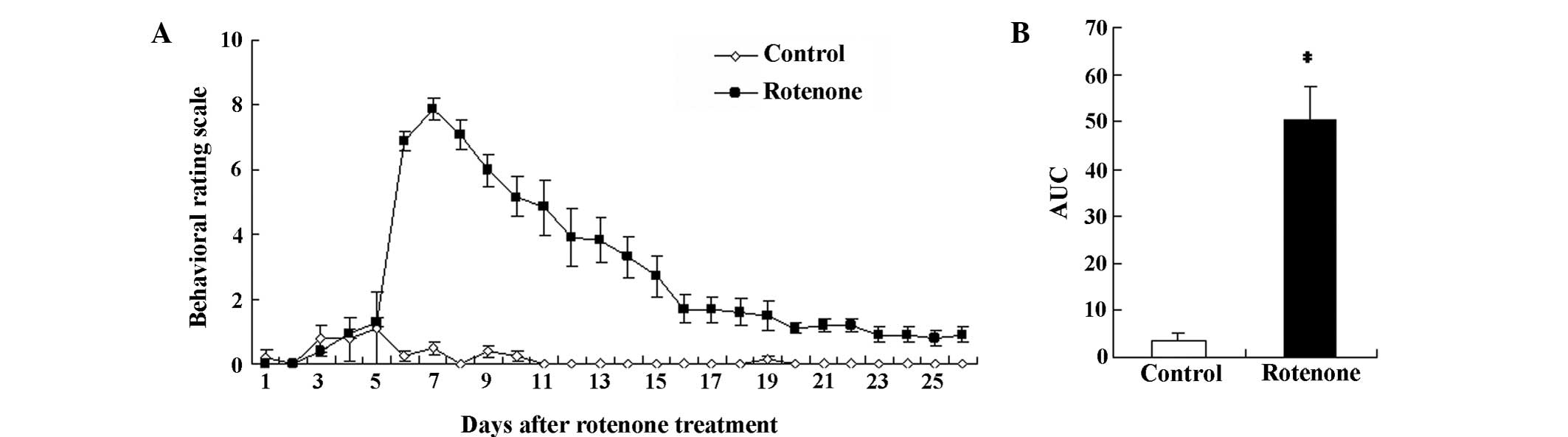
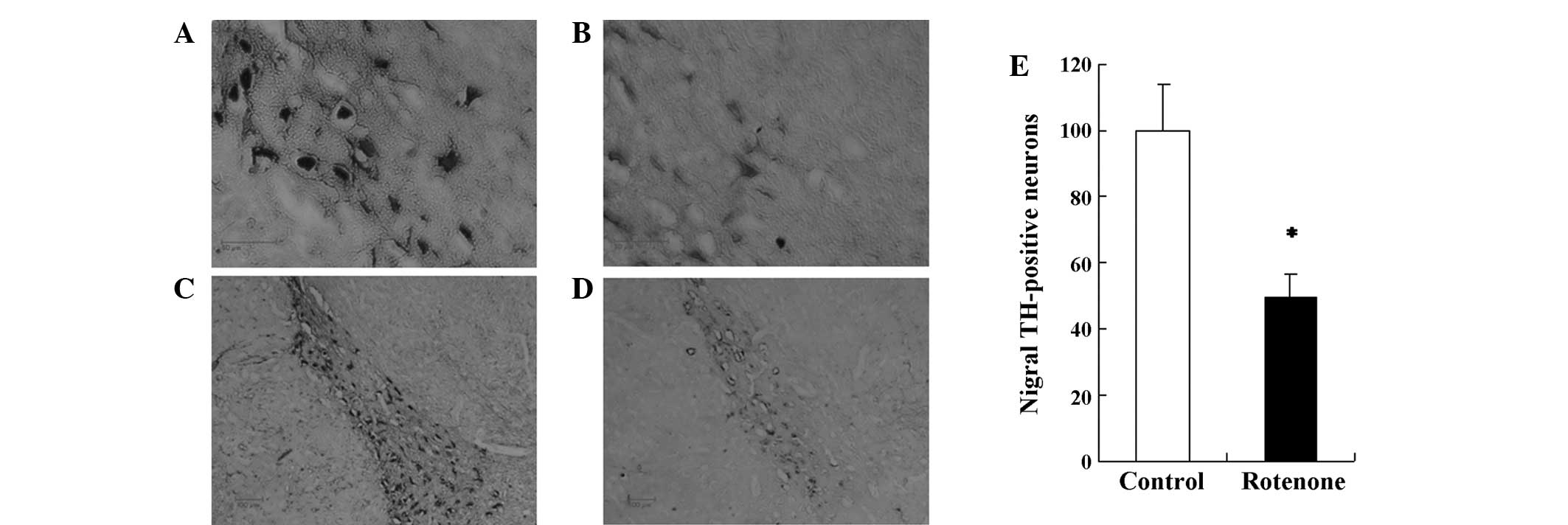
114:2283–2301. 1991. View Article : Google Scholar
|
|
4
|
Marsden CD: Basal ganglia disease. Lancet.
2:1141–1147. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Collier TJ, Kanaan NM and Kordower JH:
Ageing as a primary risk factor for Parkinson’s disease: evidence
from studies of non-human primate. Nat Rev Neurosci. 12:359–366.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Olanow CW, Stern MB and Sethi K: The
scientific and clinical basis for the treatment of Parkinson
disease. Neurology. 72(21 Suppl 4): S1–S136. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Coppedè F: Genetics and epigenetics of
Parkinson’s disease. Scientific World Journal. 2012:4898302012.
View Article : Google Scholar
|
|
8
|
Jung J, Na C and Huh Y: Alterations in
nitric oxide synthase in the aged CNS. Oxid Med Cell Longev.
2012:7189762012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hwang SJ and Huh Y: Age-related changes in
nitric oxide synthase in the lateral geniculate nucleus of rats. J
Mol Histol. 41:129–135. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gupta N, Jing Y, Collie ND, Zhang H and
Liu P: Ageing alters behavioural function and brain arginine
metabolism in male Sprague-Dawley rats. Neuroscience. 226:178–196.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bustamante J, Czerniczyniec A, Cymeryng C
and Lores-Arnaiz S: Age related changes from youth to adulthood in
rat brain cortex: nitric oxide synthase and mitochondrial
respiratory function. Neurochem Res. 33:1216–1223. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hancock DB, Martin ER, Vance JM and Scott
WK: Nitric oxide synthase genes and their interactions with
environmental factors in Parkinson’s disease. Neurogenetics.
9:249–262. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Aquilano K, Baldelli S, Rotilio G and
Ciriolo MR: Role of nitric oxide synthases in Parkinson’s disease:
a review on the antioxidant and anti-inflammatory activity of
polyphenols. Neurochem Res. 33:2416–2426. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Singh S, Das T, Ravindran A, et al:
Involvement of nitric oxide in neurodegeneration: a study on the
experimental models of Parkinson’s disease. Redox Rep. 10:103–109.
2005. View Article : Google Scholar
|
|
15
|
Tsuboi Y: Environmental-genetic
interactions in the pathogenesis of Parkinson’s disease. Exp
Neurobiol. 21:123–128. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
van der Mark M, Vermeulen R, Nijssen PC,
Mulleners WM, Sas AM, et al: Occupational exposure to pesticides
and endotoxin and Parkinson disease in the Netherlands. Occup
Environ Med. 71:757–764. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dhillon AS, Tarbutton GL, Levin JL, et al:
Pesticide/environmental exposures and Parkinson’s disease in East
Texas. J Agromedicine. 13:37–48. 2008. View Article : Google Scholar
|
|
18
|
He Y, Imam SZ, Dong Z, et al: Role of
nitric oxide in rotenone-induced nigro-striatal injury. J
Neurochem. 86:1338–1345. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bashkatova V, Alam M, Vanin A and Schmidt
WJ: Chronic administration of rotenone increases levels of nitric
oxide and lipid peroxidation products in rat brain. Exp Neurol.
186:235–241. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cannon JR, Tapias V, Na HM, et al: A
highly reproducible rotenone model of Parkinson’s disease.
Neurobiol Dis. 34:279–290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen X, Zhao Z, Zhao Z and Mu Y:
Correlation study between behavioral alteration and nigral
pathological injury in rotenone-reduced rats of Parkinson’s
disease. Zhongguo Shen Jing Jing Shen Ji Bing Za Zhi. 34:232–234.
2008.
|
|
22
|
Li J, Hou Y, Zhang S, et al: Excess iron
undermined bone load-bearing capacity through tumor necrosis
factor-α-dependent osteoclastic activation in mice. Biomed Rep.
1:85–88. 2013.PubMed/NCBI
|
|
23
|
Robyt JF, Ackerman RJ and Chittenden CG:
Reaction of protein disulfide groups with Ellman’s reagent: a case
study of the number of sulfhydryl and disulfide groups in
Aspergillus oryzae -amylase, papain, and lysozyme. Arch Biochem
Biophys. 147:262–269. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Drolet RE, Cannon JR, Montero L and
Greenamyre JT: Chronic rotenone exposure reproduces Parkinson’s
disease gastrointestinal neuropathology. Neurobiol Dis. 36:96–102.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Olanow CW and Tatton WG: Etiology and
pathogenesis of Parkinson’s disease. Annu Rev Neurosci. 22:123–144.
1999. View Article : Google Scholar
|
|
26
|
Beal MF: Excitotoxicity and nitric oxide
in Parkinson’s disease pathogenesis. Ann Neurol. 44:S110–S114.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jenner P and Olanow CW: Oxidative stress
and the pathogenesis of Parkinson’s disease. Neurology.
47:S161–S170. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Taetzsch T and Block ML: Pesticides,
microglial NOX2, and Parkinson’s disease. J Biochem Mol Toxicol.
27:137–149. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wu YN and Johnson SW: Dopamine oxidation
facilitates rotenone-dependent potentiation of N-methyl-D-aspartate
currents in rat substantia nigra dopamine neurons. Neuroscience.
195:138–144. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Joniec I, Ciesielska A,
Kurkowska-Jastrzebska I, et al: Age- and sex-differences in the
nitric oxide synthase expression and dopamine concentration in the
murine model of Parkinson’s disease induced by
1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Brain Res. 1261:7–19.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bai Q, He J, Qiu J, et al: Rotenone
induces KATP channel opening in PC12 cells in association with the
expression of tyrosine hydroxylase. Oncol Rep. 28:1376–1384.
2012.PubMed/NCBI
|
|
32
|
Richter F, Hamann M and Richter A: Chronic
rotenone treatment induces behavioral effects but no pathological
signs of parkinsonism in mice. J Neurosci Res. 85:681–691. 2007.
View Article : Google Scholar
|
|
33
|
Coulom H and Birman S: Chronic exposure to
rotenone models sporadic Parkinson’s disease in Drosophila
melanogaster. J Neurosci. 24:10993–10998. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Inden M, Kitamura Y, Takeuchi H, et al:
Neurodegeneration of mouse nigrostriatal dopaminergic system
induced by repeated oral administration of rotenone is prevented by
4-phenylbutyrate, a chemical chaperone. J Neurochem. 101:1491–1504.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pan-Montojo F, Schwarz M, Winkler C, et
al: Environmental toxins trigger PD-like progression via increased
alpha-synuclein release from enteric neurons in mice. Sci Rep.
2:8982012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ebadi M, Sharma SK, et al: Peroxynitrite
in the pathogenesis of Parkinson’s disease and the neuroprotective
role of metallothioneins. Methods Enzymol. 396:276–298. 2005.
View Article : Google Scholar
|
|
37
|
Levecque C, Elbaz A, Clavel J, et al:
Association between Parkinson’s disease and polymorphisms in the
nNOS and iNOS genes in a community-based case-control study. Hum
Mol Genet. 12:79–86. 2003. View Article : Google Scholar
|
|
38
|
Huerta C, Sanchez-Ferrero E, Coto E, et
al: No association between Parkinson’s disease and three
polymorphisms in the eNOS, nNOS, and iNOS genes. Neurosci Lett.
413:202–205. 2007. View Article : Google Scholar
|
|
39
|
Tuncel D, Inanc Tolun F and Toru I: Serum
insulin-like growth factor-1 and nitric oxide levels in Parkinson’s
disease. Mediators Inflamm. 2009:1324642009. View Article : Google Scholar
|
|
40
|
Huskic J, Paperniku A, Husic A, Alendar F
and Mulabegovic N: Significantly reduced salivary nitric oxide
synthesis in patients with Parkinson’s disease. Bosn J Basic Med
Sci. 5:86–89. 2005.
|
|
41
|
Chung KK, Dawson VL and Dawson TM:
S-nitrosylation in Parkinson’s disease and related
neurodegenerative disorders. Methods Enzymol. 396:139–150. 2005.
View Article : Google Scholar
|
|
42
|
Chung KK, Thomas B, Li X, et al:
S-nitrosylation of parkin regulates ubiquitination and compromises
parkin’s protective function. Science. 304:1328–1331. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Uehara T, Nakamura T, Yao D, et al:
S-nitrosylated protein-disulphide isomerase links protein
misfolding to neurodegeneration. Nature. 441:513–517. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Borutaite V and Brown GC: S-nitrosothiol
inhibition of mitochondrial complex I causes a reversible increase
in mitochondrial hydrogen peroxide production. Biochim Biophys
Acta. 1757:562–566. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fang J, Nakamura T, Cho DH, Gu Z and
Lipton SA: S-nitrosylation of peroxiredoxin 2 promotes oxidative
stress-induced neuronal cell death in Parkinson’s disease. Proc
Natl Acad Sci USA. 104:18742–18747. 2007. View Article : Google Scholar
|
|
46
|
Schildknecht S, Gerding HR, Karreman C, et
al: Oxidative and nitrative alpha-synuclein modifications and
proteostatic stress: implications for disease mechanisms and
interventions in synucleinopathies. J Neurochem. 125:491–511. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Nunes C, Barbosa RM, Almeida L and
Laranjinha J: Nitric oxide and DOPAC-induced cell death: from GSH
depletion to mitochondrial energy crisis. Mol Cell Neurosci.
48:94–103. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chinta SJ and Andersen JK: Reversible
inhibition of mitochondrial complex I activity following chronic
dopaminergic glutathione depletion in vitro: implications for
Parkinson’s disease. Free Radic Biol Med. 41:1442–1448. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Di Monte DA, Chan P and Sandy MS:
Glutathione in Parkinson’s disease: a link between oxidative stress
and mitochondrial damage? Ann Neurol. 32(Suppl): S111–S115. 1992.
View Article : Google Scholar
|