Introduction
Atherosclerosis (AS) and its complications are the
leading cause of mortality worldwide. The proliferation of medial
smooth muscle cells (SMCs) plays a key role in the pathogenesis of
AS. Macrophages from intimal SMCs accumulate in the subendothelial
region, resulting in the formation and rupture of plaques, and
consequent vascular occlusion and clinical events (1,2).
Previously, a serum-derived factor in platelets was
found to be able to stimulate the growth of SMCs; platelet-derived
growth factor (PDGF) was subsequently purified. PDGF can accumulate
at injury sites and proliferate in the extracellular matrix,
expanding the area of injury and/or leading to further damage. PDGF
plays important roles in different stages during the development
and progression of various diseases. The findings of a number of
studies have supported the hypothesis that PDGF is associated with
SMC accumulation during the development of AS (1,3).
Lower extremity arterial occlusive disease (LEAOD)
is a manifestation of systemic AS in the limbs. The growth of
atherosclerotic material and secondary thrombosis can lead to
stenosis or occlusion of the arterial lumen, which is clinically
manifested as a limb blood circulation disorder that may predispose
patients to ulcers or gangrene. While the majority of previous
studies have been performed on animals or cells in vitro
(4–8), the current study was conducted using
the diseased arteries of LEAOD patients. The expression levels of
PDGF-A and PDGF-B were analyzed in the AS plaques of LEAOD patients
in order to investigate their association with the development of
AS.
Materials and methods
Laboratory materials
Arterial specimens were collected from 19 LEAOD
patients (case group; male, 14; female, 5; age, 51–70 years) who
had undergone an above-knee amputation between September 2007 and
September 2010. All the LEAOD cases were classified as Fontaine
stage IV (9), suffering from
various stages of ischemia and necrosis in the limbs. Comorbidities
included hypertension (n=14), hyperlipidemia (n=4), coronary heart
disease (n=11), diabetes (n=11) and uremia (n=1). In addition,
normal artery samples from three patients (male, 2; female, 1; age,
25–52 years) who had undergone an above-knee amputation for reasons
not associated with AS, had no atherosclerosis, tumors or
Takayasu’s ateritis, were used as the control group. Specimen
collection was examined and approved by the Ethics Committee of the
Capital University of Medical Sciences (Beijing, China), and signed
informed consent was obtained from each patient. The specimens were
stored in liquid nitrogen at −80°C during transportation.
Reagents and equipment
Reagents used in the study are listed in Table I, while the equipment used are
listed in Table II. Other
chemical agents, including diethylpyrocarbonate (DEPC)-treated
water, chloroform, anhydrous ethanol, isopropanol and EDTA, were
purchased from Sinopharm Chemical Reagent Co., Ltd., (Shanghai,
China).
 | Table IReagents used in the study. |
Table I
Reagents used in the study.
| Reagent | Supplier |
|---|
| Total RNA extraction
reagent, TRIzol | Invitrogen Life
Technologies, Carlsbad, CA, USA |
| Agarose (1.5%) | BioWest, Barcelona,
Spain |
| 2X Power Taq
PCR MasterMix | BioTeke Corporation,
Beijing, China |
| Primers Sangon | Biotech Co., Ltd.,
Shanghai, China |
| ReverTra
Ace-α-® cDNA kit | Toyobo Co., Ltd.,
Osaka, Japan |
| DEPC water | Beyotime Institute of
Biotechnology, Haimen, China |
| Multi-functional DNA
Gel Extraction kit II (Spin-Column) | BioTeke Corporation,
Beijing, China |
| Realtime PCR Master
Mix | Toyobo Co., Ltd.,
Osaka, Japan |
 | Table IIEquipment used in the study. |
Table II
Equipment used in the study.
| Equipment | Supplier |
|---|
| Ultraviolet
spectrophotometer | BioVision, Milpitas,
CA, USA |
| Low-temperature
refrigerator | Sanyo Electric Co.,
Ltd., Moriguchi, Japan |
| Low-temperature
high-speed centrifuge | Beckman Coulter,
Inc., Brea, CA, USA |
| Electronic
balance | Cany Precision
Instrument Co., Ltd., Shanghai, China |
| ABI 7500 Real-Time
PCR System | Applied Biosystems
Life Technologies, Carlsbad, CA, USA |
Specimen treatment
Following adventitia of the cryopreserved artery,
the internal median layers, weighing 0.20 g, were harvested from
the case and control groups.
Total RNA extraction (TRIzol method)
TRIzol reagent (1 ml) was added to the harvested
arterial specimens. The specimens were ground, homogenized for 15
sec and incubated at room temperature for 5 min. Next, 0.2 ml
chloroform was added and the tubes were shaken vigorously for 15–30
sec by hand, followed by incubation at room temperature for 5 min.
Subsequently, the samples were centrifuged at 12,000 × g for 15 min
at 4°C. The upper aqueous phase, containing the total RNA, was
carefully removed and transferred to a new 1.5-ml centrifuge tube.
Isopropyl alcohol (0.5 ml) was added and the samples were incubated
at room temperature for 10 min following thorough mixing. Next, the
samples were centrifuged at 12,000 × g for 8 min at 4°C. The
supernatant was carefully removed, and 1 ml DEPC-treated water
(75%) was added slowly along the tube sides. The samples were mixed
thoroughly and incubated for 30 min. Centrifugation (8,000 g) at
4°C for 5 min was performed to discard the supernatant, after which
the samples were air-dried at room temperature for 5–10 min. The
pellet was dissolved in 20 μl DEPC-treated water. For
electrophoresis, the samples (5 μl) were added to a 1.5% agarose
gel (10 V/cm) and were run for ~20 min. The gels were analyzed
using a UV spectrophotometer. Subsequently, RNA was
reverse-transcribed to provide cDNA.
cDNA synthesis
The reaction solution shown in Table III was prepared in a 0.2-ml
centrifuge tube. The reaction was stopped by heating at 65°C for 5
min, followed by cooling on ice. The materials listed in Table IV were added in the indicated
order. Reverse transcription was subsequently performed, according
to the conditions listed in Table
V. cDNA samples were stored at −20°C until required for further
use.
 | Table IIIReaction solution in the centrifuge
tube. |
Table III
Reaction solution in the centrifuge
tube.
| Component of reaction
solution | Dosage (μl) |
|---|
| Random primer (25
pmol/μl) | 1 |
| RNA | 2 |
|
DEPC-dH2O | ≤12 |
 | Table IVMaterials added to the centrifuge
tube. |
Table IV
Materials added to the centrifuge
tube.
| Component | Dosage (μl) |
|---|
| Reaction solution
(Table III) | 12 |
| 5X reverse
transcription buffer | 4 |
| dNTP mixture (10
mmol/l each) | 2 |
| RNase Inhibitor (10
U/μl) | 1 |
| ReverTra Ace | 1 |
 | Table VConditions for reverse
transcription. |
Table V
Conditions for reverse
transcription.
| Temperature (°C) | Time (min) |
|---|
| 30 | 10 |
| 42 | 20 |
| 99 | 5 |
| 4 | 5 |
Quantitative polymerase chain reaction
(PCR)
Primers for PDGF-A and PDGF-B used in the reaction
system are listed in Tables VI
and VII, respectively. The
target genes and the internal control (β-actin) were amplified
separately. The reaction system is shown in Table VIII. The reaction conditions
were as follows: 95°C for 3 min, 40 cycles: 95°C for 15 sec, 60°C
for 15 sec; 72°C extension for 3 min. Following the reaction,
melting-curve analysis was performed to verify the product
specificity.
 | Table VIPrimers of PDGF-A in the reaction
system. |
Table VI
Primers of PDGF-A in the reaction
system.
| Primer | Length (bp) | Position | Tm (°C) | GC content (%) | Sequence |
|---|
| Forward | 20 | 947–966 | 59 | 55 |
GCAGTCAGATCCACAGCATC |
| Reverse | 22 | 1001–1022 | 60 | 45 |
TCCAAAGAATCCTCACTCCCTA |
 | Table VIIPrimers of PDGF-B in the reaction
system. |
Table VII
Primers of PDGF-B in the reaction
system.
| Primer | Length (bp) | Position | Tm (°C) | GC content (%) | Sequence |
|---|
| Left | 20 | 1515–1534 | 60 | 55 |
CTGGCATGCAAGTGTGAGAC |
| Right | 19 | 1603–1621 | 60 | 53 |
CGAATGGTCACCCGAGTTT |
 | Table VIIIReaction system used for the
amplifications of the target genes and the internal control
(β-actin). |
Table VIII
Reaction system used for the
amplifications of the target genes and the internal control
(β-actin).
| Component | Dosage (μl) |
|---|
| Sterile
double-distilled water | 23 |
| SYBR®
Green Realtime PCR Master Mix | 25 |
| Upstream primers
(10 μM) | 0.5 |
| Downstream primers
(10 μM) | 0.5 |
| cDNA | 1 |
| Total volume | 50 |
When a fluorescent dye is bound to the
double-stranded DNA, a particular sequence cannot be identified,
potentially leading to false-positive results. Therefore, for
product identification, melting-curve analysis was performed to
eliminate the impact of the primer dimers. For each sample, a
single peak on the melting-curve indicated a single PCR
product.
For quantitative analysis, the target gene mRNA
expression was calculated using the following formula: 2−(Ct
of cytokine − Ct of β−actin)×103, where Ct
represented the cycle threshold (fractional cycle number at which
the amount of amplified copies reaches a fixed threshold). Fewer
cycles are required to reach exponential amplification when the
starting copy number of the target is high; thus, the Ct value is
smaller. The cytokines used in the current study were PDGF-A and
PDGF-B.
Statistical analysis
Ct values of PDGF-A and PDGF-B were used as the
statistical parameters. Statistical analysis was performed using
SPSS version 16.0 software (SPSS, Inc., Chicago, IL, USA). Data in
the case and control groups were analyzed using the t-test or the
t′-test based on homegeniety of variance. In the case group,
pairwise correlations were investigated using linear correlation
analysis.
Results
Ct values of the cytokines
The Ct values of PDGF-A and PDGF-B in the case and
control groups are summarized in Tables IX and X, respectively. The homogeneity tests
revealed that P>0.05 in each group.
 | Table IXCt values of arterial PDGF-A in the
case and control groups. |
Table IX
Ct values of arterial PDGF-A in the
case and control groups.
| Group | Ct value | P-value |
|---|
| Case | 34.38±5.80 | 0.001 |
| Control | 21.94±1.05 | |
 | Table XCt values of arterial PDGF-B in the
case and control groups. |
Table X
Ct values of arterial PDGF-B in the
case and control groups.
| Group | Ct value | P-value |
|---|
| Case | 33.95±5.92 | 0.012 |
| Control | 24.15±3.12 | |
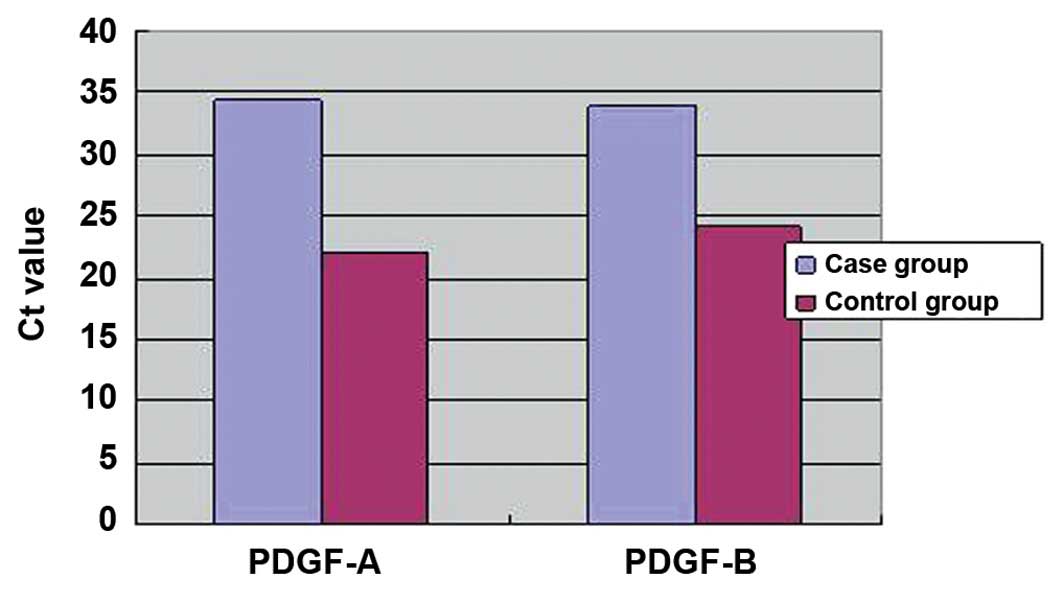
Comparison of the Ct values
Comparisons of the Ct values for PDGF-A and PDGF-B
between the case and control groups are shown in Fig. 1. A statistically significant
difference was observed for the PDGF-A Ct values between the case
and control groups (t′=8.51, P=0.000). In addition, the Ct value of
PDGF-B was found to be significantly different between the two
groups (t′=2.77, P=0.012). Therefore, the expression levels of
PDGF-A and PDGF-B were found to be significantly higher in the case
group when compared with the control group.
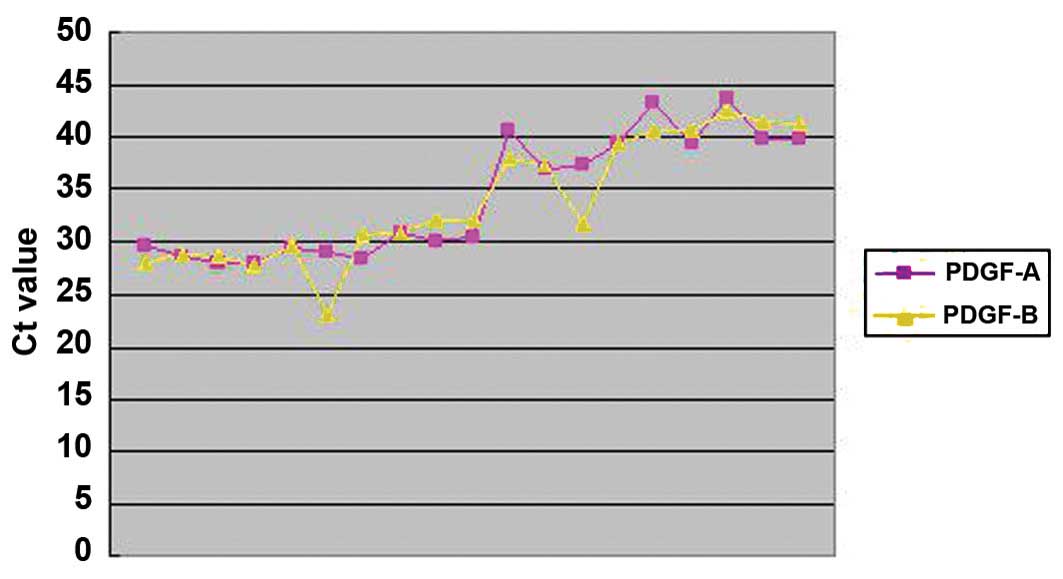
Correlation analysis
The correlation between the expression levels of
PDGF-A and PDGF-B in the case group is shown in Fig. 2. The Ct value of PDGF-A was
significantly correlated with the Ct value of PDGF-B (r=0.918,
P=0.000), indicating that the expression of PDGF-A in the vascular
wall correlates with the expression of PDGF-B.
Discussion
The PDGF family includes a number of PDGFs and PDGF
receptors (PDGFRs). Mitogenic PDGF is widely distributed throughout
the body and produced in platelets; however, the growth factor can
also be produced in SMCs, macrophages and endothelial cells (ECs)
following vascular injury (10).
The PDGF family consists of disulfide-bonded homodimers or
heterodimers with four possible subunits (PDGF-A, PDGF-B, PDGF-C
and PDGF-D). The A-chain is typically synthesized and secreted by
SMCs that are proliferated following intimal injury, whereas the
B-chain is produced mainly by macrophages and ECs (1). After production, PDGF activates the
PDGFR via autocrine and/or paracrine signaling. Two distinct
subunits, α and β, dimerize to form three types of PDGFR (PDGFR-αα,
-αβ and -ββ).
As a key mediator of inflammation, PDGF can induce
the migration and proliferation of SMCs in the intima, which is an
important mechanism in the development of AS. PDGF exhibits a
number of biological characteristics. Firstly, PDGF has a mitogenic
effect, whereby PDGF facilitates DNA synthesis and enables cell
lysis and proliferation by adjusting the regeneration of
extracellular matrix, which stimulates the division and
proliferation of vascular SMCs (VSMCs) (11). Wang et al (12) demonstrated that PDGF-B has stronger
proproliferative and mitogenic effects compared with PDGF-A.
Secondly, PDGF exhibits chemotactic activity by contributing to SMC
and fibroblast accumulation at injured sites, which is subsequently
followed by the proliferative response. This chemotactic activity
is particularly important for injury repair. Finally, PDGF exhibits
a vasoconstrictive effect (13).
SMCs are the direct cause of vascular stenosis or
occlusion. As a key inflammatory mediator, PDGF-A is synthesized
and secreted by SMCs in the vascular intima during injury, while
PDGF-B is produced mainly by macrophages and ECs (14). PDGF-A and PDGF-B increase the
thickness of the fibrous capsules of plaques (15). Various inflammatory signaling
pathways participate in the development of AS. As the most potent
growth factor of SMCs, PDGF-B induces the synthesis and secretion
of PDGFR-β and SMC progenitor cells, causing intimal thickening
(16,17). Furthermore, PDGF-B induces the
recruitment and differentiation of intimal SMCs and stimulates
angiogenesis (18).
PDGF-AA, PDGF-AB and PDGF-BB are potent promoters of
VSMC proliferation and their migration in the subendocardial
region. The expression level of PDGF can increase by 10–20 times at
6 h following vascular injury. During the development of AS, PDGF
stimulates the medial SMCs to change from a contractile to a
synthetic phenotype, increasing their proliferative capacity.
Following the phenotypic change, the cells obtain stronger
synthesis and secretory functions. After proliferation, the intimal
SMCs express PDGF-A and PDGFR genes and secrete bioactive PDGF-like
molecules. The bioactive PDGF-like molecules can promote the
proliferation and migration of VSMCs and the deposition of the
extracellular matrix (19). In
addition, PDGF-A and PDGF-B are involved in the thickening of the
fibrous capsules of plaques (15),
and may detach from the fat-rich core matrix of thrombi in the
arterial lumen, further exacerbating the clinical symptoms
(14).
A large number of studies (20,21)
have demonstrated increased expression levels of PDGF and PDGFR in
human AS plaques, coronary blood vessels following angioplasty and
the vascular walls of animal models with vascular injury. These
observations indicate that PDGF and PDGFR are involved in the
pathogenesis of AS and the repair of arterial injury.
To date, the majority of studies have been conducted
on human medium-sized arteries or on arteries of animal models;
however, the present study was performed using human lower limb
large arteries. Quantitative analysis of the expression levels of
PDGF-A and PDGF-B in the diseased arteries revealed that the two
cytokines were highly expressed in the case group. Therefore, high
expression levels of PDGF-A and PDGF-B are also present in the AS
plaques of the human lower limb large arteries. Furthermore, a
previous study demonstrated that PDGF can directly stimulate the
expression and enhance the activity of nuclear factor-κB (NF-κB),
which is a key regulator of arteriosclerosis (26). The highly-expressed PDGF may enter
the vascular subendothelium and activate NF-κB in the vascular
SMCs, enhancing cellular proliferation.
Each PDGF subtype exhibits angiogenic activity.
Arterial occlusion resulting from excessive neointimal
proliferation is a sign of restenosis following balloon angioplasty
in AS patients. Inhibition of intraplaque angiogenesis is an
important approach used in the stabilization of AS plaques. A
number of growth factors and cytokines are involved in the process
of restenosis, among which PDGF is considered to be the most
significant, due to the effect of PDGFRβ on the tunica media SMCs.
In addition, various PDGF-induced cell responses, including the
regulatory effect of PDGF on vascular tone, the negative
feedback-driven regulation during platelet activation and the
promotion of neovascularization, are associated with AS diseases.
Previous studies have also investigated the expression of PDGF-A
and PDGF-B in arteries following coronary angioplasty. Over past
two decades, a number of studies have investigated the stent-based
delivery of the PDGFR inhibitor; however, its effectiveness remains
unsatisfactory. Furthermore, inhibition of PDGF expression may
inhibit injury-induced intimal expansion and the formation of
collateral circulation, hampering the improvement of the ischemic
status. Therefore, future studies should utilize the existing
knowledge regarding PDGF and its receptor in order to facilitate
the prevention and treatment of AS and the resulting clinical
events (22,23).
The results of the present study confirmed the
positive linear correlation between highly-expressed PDGF-A and
PDGF-B in the lower limb arteries of LEAOD patients. In addition, a
PDGFR kinase inhibitor-coated stent was found to partially inhibit
vascular restenosis following percutaneous transluminal coronary
angioplasty, also indicating that PDGF-A and PDGF-B are closely
correlated with the development of AS (24,25).
Kozaki et al (27)
demonstrated that the elimination of PDGF-B in circulating cells
and the blockade of two PDGFRs transiently delayed fibrous cap
formation in rats with early AS. However, SMC accumulation at the
injured sites was not prevented, indicating that stenosis or
occlusion of the arterial lumen by AS is a complex process
involving numerous factors.
PDGF-A and PDGF-B play important roles in various
stages of AS. Therefore, future research should focus on blocking
their adverse effects on arteriosclerosis, while preserving their
positive roles in countering arteriosclerosis and improving
ischemia.
The present study had several limitations. Firstly,
the specimens used in the study were obtained from the arterial
vessels of patients with advanced arteriosclerosis; thus, the
samples were unable to fully reflect the associations between
PDGF-A, PDGF-B and arteriosclerosis throughout the disease course.
Furthermore, although the specimens were obtained from LEAOD
patients, the disease course differed markedly among the patients
(between four days and 10 months). Complications included
hypertension, hyperlipidemia and diabetes, which are independent
risk factors of arteriosclerosis. As a result, the expression
levels of PDGF-A and PDGF-B may have differed due to differences in
the severity of the disease. In addition, the storage period of the
arterial vessels differed among the samples, with intervals up to
three years. Since RNA degrades easily, the long storage interval
may have affected the results of the current study. In future
studies, specimens should be harvested, transported and stored in a
more consistent manner. Furthermore, based on the pathological
stages and risk factors of arteriosclerosis, the expression levels
of PDGF-A and PDGF-B in vascular walls should be analyzed during
different stages of arteriosclerosis in order to provide solid
clinical evidence for the prevention and treatment of the
arteriosclerosis obliterans. In concludion, the results of the
present study demonstrated that the levels of PDGF-A and PDGF-B
were increased in the vessel wall of patients with LEAOD.
References
|
1
|
Kher N and Marsh JD: Pathobiology of
atherosclerosis - a brief review. Semin Thromb Hemost. 30:665–672.
2004. View Article : Google Scholar
|
|
2
|
Sun XF, Suo J, Wang Q, et al: Expressions
of hypoxia inducible factor-1α and vascular endothelial growth
factor in vessels and muscle of ischemia lower limbs in patients
with arteriosclerosis obliterans and significances. Jilin Da Xue
Xue Bao (Yi Xue Ban). 35:337–340. 2009.(In Chinese).
|
|
3
|
Caligiuri G, Rudling M, Ollivier V, Jacob
MP, Michel JB, Hansson GK and Nicoletti A: Interleukin-10
deficiency increases atherosclerosis, thrombosis,and low-density
lipoproteins in apolipoprotein E knockout mice. Mol Med. 9:10–17.
2003.PubMed/NCBI
|
|
4
|
Ross R: Atherosclerosis is an inflammatory
disease. Am Heart J. 138:S419–S420. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lawrence T, Gilroy DW, Colville-Nash PR
and Willoughby DA: Possible new role for NF-kappa B in the
resolution of inflammation. Nat Med. 7:1291–1297. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mallat Z, Gojova A, Marchiol-Fournigault
C, Esposito B, Kamate C, Merval R, Fradelizi D and Tedgui A:
Inhibition of transforming growth factor-beta signaling accelerates
atherosclerosis and induces an unstable plaque phenotype in mice.
Circ Res. 89:930–934. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Idel S, Dansky HM and Breslow JL: A20, a
regulator of NFkappaB, maps to an atherosclerosis locus and differs
between parental sensitive C57BL/6J and resistant FVB/N strains.
Proc Natl Acad Sci USA. 100:14235–14240. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee EG, Boone DL, Chai S, Libby SL, Chien
M, Lodolce JP and Ma A: Failure to regulate TNF-induced NF-kappaB
and cell death responses in A20-deficient mice. Science.
289:2350–2354. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Norgren L, Hiatt WR, Dormandy JA, Nehler
MR, Harris KA and Fowkes FG: Inter-Society Consensus for the
Management of Peripheral Arterial Disease (TASC II). J Vasc Surg.
45:S5–S67. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang X, Thomas DP, Zhang X, et al:
Curcumin inhibits platelet-derived growth factor-stimulated
vascular smooth muscle cell function and injury-induced neointima
formation. Arterioscler Thromb Vasc Biol. 26:85–90. 2006.
View Article : Google Scholar
|
|
11
|
Matsui T, Heidaran M, Miki T, et al:
Isolation of a novel receptor cDNA establishes the existence of two
PDGF receptor genes. Science. 243:800–804. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang JJ, Zhang SX, Lu K, et al: Decreased
expression of pigment epithelium-derived factor is involved in the
pathogenesis of diabetic nephropathy. Diabetes. 54:243–250. 2005.
View Article : Google Scholar
|
|
13
|
Grotenderst GR, Chang T, Sepppa HEJ, et
al: Platelet-derived growth factor in a chemottractant for
vassseular smooth muscle cells. J Cell Physiol. 113:261–266. 1982.
View Article : Google Scholar
|
|
14
|
Zhang LY, Zhang DW, Wu XJ, et al:
Expression of platelet-derived growth factor-A in atherosclerosis
plaque of arteriosclerosis obliterans patient. Zhongguo Dong Mai
Ying Hua Za Zhi. 14:61–63. 2006.(In Chinese).
|
|
15
|
Góis J, Higuchi M, Reis M, et al:
Infectious agents, inflammation, and growth factors: how do they
interact in the progression or stabilization of mild human
atherosclerotic lesions? Ann Vasc Surg. 20:638–645. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Armulik A, Abramsson A and Betsholtz C:
Endothelial/pericyte interactions. Circ Res. 97:512–523. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xiao Q, Zeng L, Zhang Z, Hu Y and Xu Q:
Stem cell-derived Sca-1+ progenitors differentiate into
smooth muscle cells, which is mediated by collagen IV-integrin
alpha1/beta1/alphav and PDGF receptor pathways. Am J Physiol Cell
Physiol. 292:C342–C352. 2007. View Article : Google Scholar
|
|
18
|
Zhao B and Gong NQ: The effect of
adenovirus-mediated anti-extracellular signal regulated kinase 2
gene therapy on intimal change in transplant arteriosclerosis.
Zhonghua Shi Yan Wai Ke Za Zhi. 28:514–516. 2011.(In Chinese).
|
|
19
|
Ramírez-Tortosa MC, Mesa MD, Aguilera MC,
et al: Oral administration of a turmeric extract inhibits LDL
oxidation and has hypocholesterolemic effects in rabbits with
experimental atherosclerosis. Atherosclerosis. 14:371–378. 1999.
View Article : Google Scholar
|
|
20
|
Tanizawa S, Ueda M, van der Loos CM, van
der Wal AC and Becker AE: Expression of platelet derived growth
factor B chain and beta receptor in human coronary arteries after
percutaneous transluminal coronary angioplasty: an
immunohistochemical study. Heart. 75:549–556. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ueda M, Becker AE, Kasayuki N, Kojima A,
Morita Y and Tanaka S: In situ detection of platelet-derived growth
factor-A and factor-B chain mRNA in human coronary arteries after
percutaneous transluminal coronary angioplasty. Am J Pathol.
149:831–843. 1996.PubMed/NCBI
|
|
22
|
Kenagy RD, Hart CE, Stetler-Stevenson WG
and Clowes AW: Primate smooth muscle cell migration from aortic
explants is mediated by endogenous platelet-derived growth factor
and basic fibroblast growth factor acting through matrix
metalloproteinases 2 and 9. Circulation. 96:3555–3560. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ferns GA, Raines EW, Sprugel KH, Motani
AS, Reidy MA and Ross R: Inhibition of neointimal smooth muscle
accumulation after angioplasty by an antibody to PDGF. Science.
253:1129–1132. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tanizawa S, Ueda M, van der Loos CM, van
der Wal AC and Becker AE: Expression of platelet derived growth
factor B chain and beta receptor in human coronary arteries after
percutaneous transluminal coronary angioplasty: an
immunohistochemical study. Heart. 75:549–556. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ueda M, Becker AE, Kasayuki N, Kojima A,
Morita Y and Tanaka S: In situ detection of platelet-derived growth
factor-A and factor-B chain mRNA in human coronary arteries after
percutaneous transluminal coronary angioplasty. Am J Pathol.
149:831–843. 1996.PubMed/NCBI
|
|
26
|
Mo B, Liu W, Yang L and Jiao J: Modulation
of expression of platelet-derived growth factor by nuclear
transcription factor-kappab in rats retina. Zhonghua Yan Ke Za Zhi.
43:49–54. 2007.(In Chinese). PubMed/NCBI
|
|
27
|
Kozaki K, Kaminski WE, Tang J, et al:
Blockade of platelet-derived growth factor or its receptors
transiently delays but does not prevent fibrous cap formation in
ApoE null mice. Am J Pathol. 161:1395–1407. 2002. View Article : Google Scholar : PubMed/NCBI
|