Introduction
Polycythemia vera (PV) is a myeloproliferative
neoplasm (MPN) of unknown etiology that involves the clonal
proliferation of erythrocytes. PV typically manifests as blood
circulation disorder, hypertension and cerebral infarction. Its
pathogenesis is unknown and may be associated with JAK2 mutation
(1). The pathological lesions of PV
are predominantly involved bone marrow, spleen and liver (2). PV associated with renal disease is
clinically rare. Only 23 cases have been reported in the
literature, and in the majority of these cases it was considered
that PV-induced glomerular hyperperfusion and hyperfiltration were
associated with the occurrence of nephropathy (3).
A rare case of PV associated with immunoglobulin
(Ig) A nephropathy (IgAN) and an elevated serum creatinine (SCr)
level but normal glomerular filtration rate is described in the
present case report. In addition, the previous case reports of PV
associated with renal disease are reviewed and the clinical
features are summarized.
Case report
A 57-year-old Chinese man with a 6-year history of
PV associated with protein-and hematuria was admitted to the
Department of Nephrology of Navy General Hospital (Beijing, China)
on November 21, 2011 with week-long symptom exacerbation. Six years
prior to this, the Department of Hematology used a bone marrow
aspiration to diagnose this patient with PV and observed
significantly increased leukocyte, erythrocyte and platelet counts.
The urinalysis performed at that time showed a 1,200 mg 24 h urine
protein content (24-h UR) and 10–15 erythrocytes/high-power field
(hp). The patient also displayed hypertension and hyperuricemia.
The patient was treated with hydroxyurea (HU), regular phlebotomies
(RPs) and interferon (IFN) therapy, and his leukocyte, erythrocyte
and platelet counts returned to the normal range. The 24-h UR was
300–500 mg at follow-up. One week prior to admission, the patient
complained of a sore throat. The patient's past medical history
included an enlarged liver, widening of the portal vein and
splenomegaly. The physical exam showed a blood pressure of 148/102
mmHg, facial flushing, conjunctival hyperemia and a dark red color
on the forehead, tip of the nose, lips, thenar muscles and fingers.
No throat congestion or edema was detected. The spleen was palpable
below the rib ridge.
Serum analysis revealed a leukocyte count of
19.25×109/l, hemoglobin content of 207 g/l and platelet
count of 735×109/l. The urea nitrogen and SCr levels
were 14.89 and 1.22 mg/dl, respectively. The urine erythrocyte
count was 8–10/hp, and the 24-h UR was 2,796 mg. The liver
function, serum lipid profile, uric acid, electrolytes, glucose,
IgM, IgA, IgG and complement 3 (C3) and C4 serum levels were
normal. Coagulation and the levels of anti-streptolysin O and
high-sensitivity C-reactive protein were also normal. Several
antibodies, such as anti-myeloperoxidase, proteinase 3 (PR3),
double-stranded DNA (dsDNA), nucleosome and Sjögren's syndrome type
A (SS-A) and type B (SS-B) antibodies, tested negative and excluded
an autoimmune disorder or systemic disease. The urine osmolality
was 609.00 mOsm/kg. An ultrasonography revealed increased renal
parenchymal echogenicity of both kidneys. The maximal dimensions of
the right and left kidneys were determined to be 12.4 and 12.1 cm,
respectively. A renal radioisotope test was conducted to measure
the glomerular filtration rate (GFR) and was 41.2 ml/min in the
left kidney and 51.9 ml/min in the right kidney. The total GFR was
93.3 ml/min.
A renal biopsy was performed, after informed consent
was obtained from the patient. The specimens were embedded in
paraffin, sliced to 3-mm thickness and then stained with
hematoxylin and eosin (H&E), periodic acid-Schiff (PAS),
periodic acid silver methenamine (PASM) and Masson stains. Three
pieces of renal tissue and a total of 16–20 glomeruli were then
examined using light microscopy (LM). This analysis revealed 3
glomeruli with global sclerosis, and the remainder had mild diffuse
hyperplasia of the mesangium and mesangial matrix. The glomerular
capillaries were not dilated. No micro-thrombosis was observed. The
renal tubular epithelial cells showed mild vacuolar degeneration.
Frozen sections were stained for immunofluorescence (IF), and
rabbit anti-human IgG, IgA, IgM, C3 and C1q were used for direct IF
staining. IF showed 2–3 glomeruli with granular depositions of IgA
(4+; Fig. 1) and IgM+ in the
mesangium but negative results for IgG, C1q, C3 and fibronectin.
These results generated a diagnosis of IgAN (Lee classification
I–II) (4).
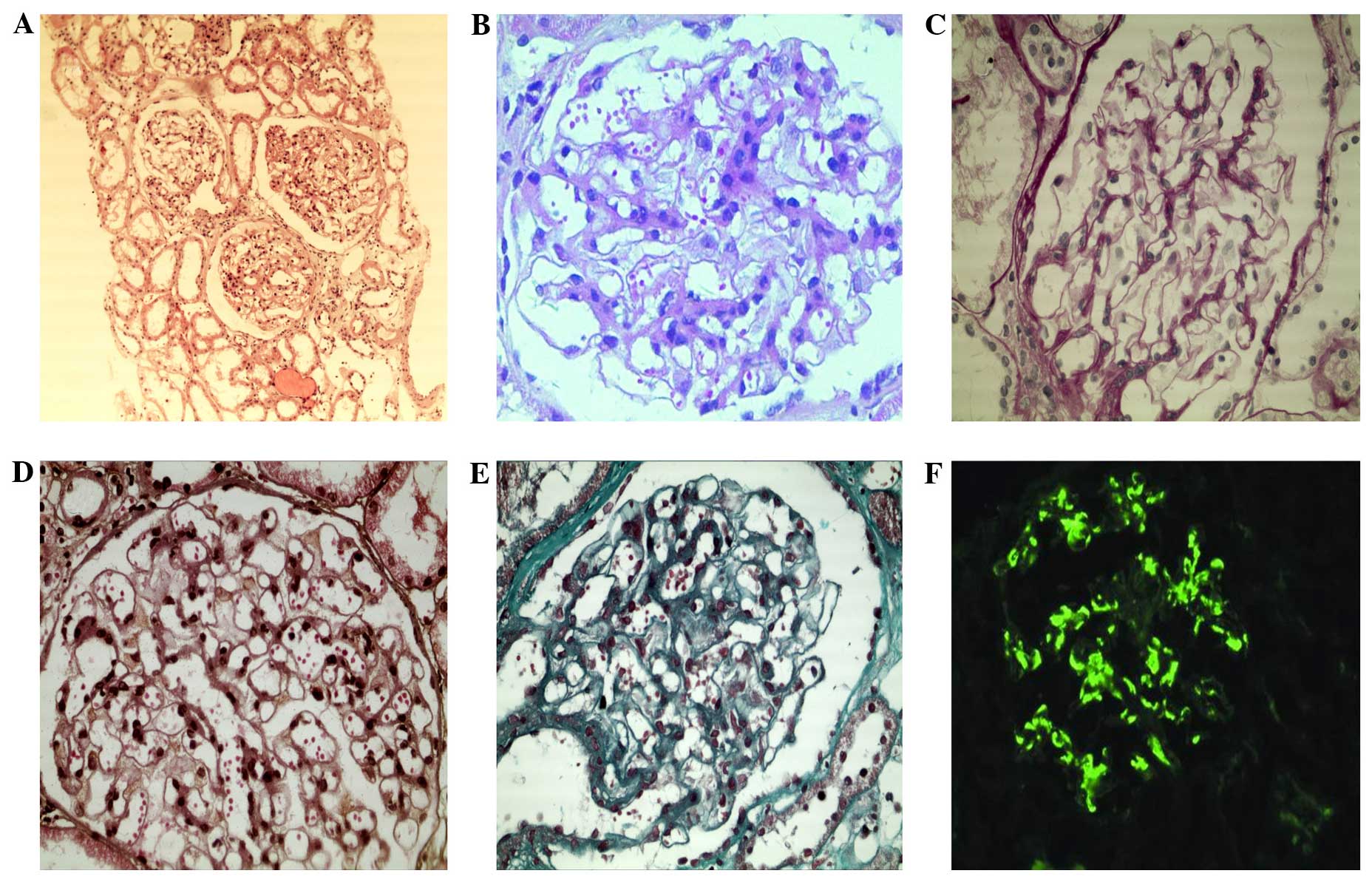 | Figure 1.Light-microscopy and
immunofluorescence microscopy of glomeruli. (A) H&E staining
(magnification, x200); (b) H&E staining (magnification,
x400);(C) PAS staining (magnification, x400); (D) PASM staining
(magnification, x400); (E) Masson staining (magnification, x400).
(A–E) Light-microscopy shows mild mesangial proliferation. (F)
Immunofluorescence microscopy reveals intense granular IgA
deposition in the mesangial area (magnification, x400). H&E,
hematoxylin and eosin; PAS, periodic acid-Schiff; PASM, periodic
acid silver methenamine; IgA, immunoglobulin A. |
The patient was administered amlodipine besylate (5
mg), valsartan capsules (160 mg), an enteric-coated aspirin tablet
(100 mg) and HU (0.5 g) daily. An outpatient follow-up was
conducted once every 2 weeks. Multiple follow-up examinations
yielded the following results: The 24-h UR was positively
correlated with the red blood cell count, hemoglobin level, and
white blood cell and platelet counts. In addition, the blood
pressure of the patient was in the range of 120–140/70–95 mmHg.
Discussion
To the best of our knowledge, only 23 cases of PV
associated with renal disease are reported in the current
literature. Among these cases, 21 were diagnosed by renal biopsies.
The average age of the patient at the time of the renal biopsy was
53.43 years. The male-to-female ratio was 1.63:1. The clinical
manifestations included 14 cases (66.7%) of nephrotic syndrome (NS;
24-h UR ≥3.5 g/l), 6 cases (28.6%) of mild to moderate proteinuria
(24-h UR <3.5 g/l) and 1 case without information. Eight cases
(38.1%) presented as hematuria positive, 12 cases (57.1%) were
negative and 1 case was without information. Among those patients,
there were 6 cases (28.6%) with normal renal function or mild renal
insufficiency (defined as SCr ≤1.5 mg/dl in males and 1.2 mg/dl in
females, GFR ≥70 ml/min), 8 patients (38.1%) with moderate renal
insufficiency (SCr ≤1.5/1.2–2.8 mg/dl, GFR 30–60 ml/min), 4 cases
(19.0%) with severe renal insufficiency (SCr ≥2.8 mg/dl, GFR ≤30
ml/min) and 3 cases without information. Hypertension was observed
in all cases. A hypertension crisis occurred in 1 case (5). With respect to patient follow-up, 5
cases (23.8%) progressed to end stage renal disease (ESRD) and
required routine dialysis. The remaining patients (76.2%) continued
to show mild proteinuria and renal insufficiency.
The histological results for these cases of PV
associated with renal disease included 8 cases (38.1%) of IgAN
(5–11), 10 cases (47.6%) of focal segmental
glomerulosclerosis (FSGS) (3,12–19),
2 cases (9.5%) of membranoproliferative glomerulonephritis (MPGN)
(20,21) and 1 case (4.8%) of rapidly
progressive glomerulonephritis (RPGN).
Eight cases of PV associated with IgAN have been
reported in the literature (5–11). All 8
patients were male with an average age of 47.87 years. Six cases
(75%) presented NS, and 1 case (12.5%) presented moderate
proteinuria. Five cases (62.5%) were positive for uric
erythrocytes, and 2 cases (25%) were negative. The numbers of cases
of normal, moderate and severe renal dysfunction were 2 (25%), 4
(50%) and 1 (12.5%), respectively. All 8 patients presented with
hypertension, including 1 case with a hypertension crisis that was
diagnosed histopathologically as mesangial proliferative
glomerulonephritis (MsPGN), global glomerulosclerosis and
necrotizing arteritis (5). Two cases
also presented with hyperuricemia (8,9) and 1
case with cerebral infarction (5).
Pathological observations of the LM analyses revealed 5 cases
(62.5%) of MsPGN, 1 case (12.5%) of sclerosing glomuleronephritis
(SGN) (7) and 1 case that was
initially diagnosed as MPGN following the initial renal biopsy but
was determined to be crescentic glomerulonephritis (CGN) with a
second biopsy, which was performed after the patient experienced a
rapid deterioration in renal function (11). Five cases (62.5%) exhibited
glomerular crescent formation. The IF analyses revealed IgA
deposition in the mesangium and glomerular capillary walls in all
cases. Three cases (37.5%) presented with IgA deposition only. The
numbers of cases associated with depositions of C3, IgM or IgG were
5 (62.5%), 3 (37.5%), and 1 (12.5%), respectively. Electron
microscopy (EM) was only performed in 2 of the 8 cases (5,8), and
revealed electron-dense deposits in the mesangial region and
platelets attached to the glomerular capillary wall. For treatment,
2 cases underwent steroid therapy, 2 cases received RP and 1 case
received mycophenolate mofetil along with anti-hypertensive,
anticoagulant, anti-platelet and bone marrow suppression therapies
(HU, busulfan and ranimustine). The prognosis of the patients was
evaluated during the follow-ups. One patient (12.5%) with
pathological features of SGN and crescent formations had a poor
prognosis and required hemodialysis because of ESRD (7). No prognosis was reported in 3 cases. In
the remaining 3 cases (37.5%), proteinuria and renal function were
effectively controlled (Table
I).
 | Table I.Selected features of patients with PV
associated with IgAN. |
Table I.
Selected features of patients with PV
associated with IgAN.
| Patient no. | Author (ref.),
year | Age, years | Gender | PV duration | Renal biopsy LM; IF;
EM | Pro; urinary RBC;
Renal function | Clinical type | Symptoms | Therapy | Outcome Pro; SCr |
|---|
| 1 | Kim (6), 1994 | 56 | M | NR | NR; IgAN; NR | 7.8 g/day; Neg.; SCr
1.3 mg/dl | NS | HBP | NR | NR |
| 2 | Kasuno (5), 1997 | 35 | M | 5 years | MsPGN, NA, Cre; IgA,
C3; EDD-GMA, PLT-CAP | 4 g/day; 20–40/hp;
CCr 34.2 ml/min, SCr 1.6 mg/dl | NS | HBP crisis | ACG, PSL | 1.5 g/day; 1.2
mg/dl |
| 3 | Kasuno (5), 1997 | 51 | M | SI | MsPGN, Cre; IgA, C3;
NR | 2.8 g/day; 30–40/hp;
CCr 62.9 ml/min SCr 1 mg/dl | GN | CI | BUS, RP | NR |
| 4 | Kwon (7), 1999 | 32 | M | NR | SGN, Cre; IgA;
NR | 5.3 g/day; Neg.; SCr
10.0 mg/dl | NS | HBP | NR | HD |
| 5 | Chung (8), 2002 | 46 | M | SI | MsPGN; IgA, IgM, C3;
EDD-GMA | 9.14 g/day; 10–20/hp;
GFR 34.09 ml/min, SCr 2.7 mg/dl | NS, CRF | HBP, HUA | HU, ACEI, CCB | 2.7 g/day |
| 6 | Yaguchi (9), 2005 | 55 | M | 17 years | MsPGN, Cre; IgA, IgM,
C3; NR | 6.9 g/day; 664/µl;
SCr 2.5 mg/dl | NS, CRF | HBP, HUA | PSL | 3.4 g/day |
| 7 | Tian (10), 2011 | 55 | M | NR | MsPGN, Cre; IgA, IgM,
IgG, C3; NR | 13.43 g/day; 202.3/µl
SCr 1.15 mg/day | NS, CRF | HBP | MMF, ARB, CCB,
RP | 1.5 g/day; 56.5/µl,
0.9 mg/dl |
| 8 | Kanauchi (11), 1994 | 53 | M | NR | MPGNa; CGN; IgA; NRb | NR | RPGN | HBP | MCNU | NR |
Ten cases of PV associated with FSGS have been
reported (3,12–19). The
average age was 55.8 years with a male-to-female ratio of 0.67. Six
cases (60%) presented NS, and 4 cases (40%) presented mild
proteinuria. Three cases (30%) were positive for uric erythrocytes,
and 6 cases (60%) were negative. The numbers of cases of normal or
mild, moderate, or severe renal dysfunction were 3 (30%), 4 (40%)
and 2 (20%), respectively. The pathological LM analyses confirmed
the FSGS diagnosis in all cases. One patient also showed focal
ischemic nephropathy (18). With the
exception of 3 cases without available IF data, negative IF results
were reported in 4 cases (40%), a combination of IgM, C3 and C1q
depositions were observed in 2 cases (20%), and IgA deposition was
observed in 1 case. EM results were reported in 3 of 10 cases, in
which sclerosis, swelling segmental vacuolar formation of the
podocyte foot processes, and electron-dense material in the
mesangial area and partial effacement of the podocyte foot process
were observed. With respect to treatment, 5 patients received RP, 7
patients received HU and 5 patients received steroid and
immunosuppressive therapy. The prognosis of these patients was
worse than that of the patients with IgAN. Four patients (40%)
progressed to ESRD and required dialysis. In 6 patients (60%),
proteinuria and renal function were effectively controlled (see
Table II).
 | Table II.Selected features of patients with PV
associated with FSGS. |
Table II.
Selected features of patients with PV
associated with FSGS.
| Patient no. | Author (ref.),
year | Age, years | Gender | PV duration | Renal biopsy LM; IF;
EM | Pro; urinary RBC;
Renal function | Clinical type | Symptoms | Therapy | Outcome Pro, SCr |
|---|
| 1 | Sharma (12), 1995 | 40 | F | 2
years | FSGS; NR; NR | 2 g/day; Neg; SCr 2
mg/dl | GN, CRF | HBP | RP, MLS | 0.24 g/day, 1.4
mg/dl |
| 2 | Au (13), 1999 | 28 | M | 20 years | FSGS; Neg.; GS | 2.5 g/day; Neg; SCr
3.6 mg/dl | GN, CRF | HBP | HU, RP | PD |
| 3 | Au (13), 1999 | 41 | F | 14 years | FSGS; IgM, C3, C1q;
NR | 5.4 g/day; Neg; CCr
60 ml/min | NS | HBP | HU, RP | PD |
| 4 | Kosch (14), 2000 | 52 | M | 4
years | FSGS; Neg; EPE | 4 g/day; Neg; SCr 1.1
mg/dl | NS | HBP | RP | 0.4 g/day, NOR |
| 5 | Ulusoy (15), 2010 | 56 | M | 10 years | FSGS; Neg; NR | 4–5.97 g/day; Neg;
SCr 1.9 mg/dl | NS, CRF | HBP | RP, IFN, ACEI,
PSL | 1.26 g/day; 1.4
mg/dl |
| 6 | Okuyama (3), 2007 | 69 | F | 3
years | FSGS; IgM, C3, C1q,
Fn; NR | 8.3 g/day; Neg; SCr
1 mg/dl | NS, CRF | HBP | HU, PSL MLS | 2.1 g/day; 0.53
mg/dl |
| 7 | Martín (16), 2010 | 83 | F | 4
years | FSGS; NR; NR | 4.2–8.4 g/day;
6,750/µl SCr1.1–3.6 mg/dl | GN + NS CRF | NR | HU, PSL | HD |
| 8 | Iyoda (17), 2005 | 66 | F | 3
years | FSGS; NR; NR | 9.6 g/day; NR;
NR | NS, CRF | HBP | HU, PSL | HD |
| 9 | Cai (18), 2011 | 78 | M | 4
years | FSGS, ISCH KI; Neg;
NR | 2–3.5 g/day; +; SCr
2.11 mg/dl eGFR 34.53 ml/min | GN + NS CRF | HBP CI | HU | 1.72 g/d |
| 10 | Dai (19) 2008 | 45 | F | SI | FSGS; IgA, IgM;
EDD-GMA, EPE | 0.714 g/day; ++;
SCr 0.51 mg/dl | GN | HBP | HU, ARB, APD | 0.228 g/day |
Three cases of PV associated with other pathological
manifestations of renal disease have been reported.
Based on the literature, the possible pathogenesis
of PV associated with renal disease may occur as follows. First, PV
leads to increases in blood volume and viscosity, thus causing a
passive expansion of the capillaries and intimal injury, which
results in vascular microthrombi, glomerular capillary occlusion
and a reduction in the GFR, thereby leading to tissue ischemia. If
the ischemia persists without relief, it is likely to result in
chronic renal damage. Second, PV is often associated with
hypertension and hyperuricemia, which affect renal
microcirculation. In addition, thrombocytosis and the abnormal
activation of megakaryocytes might be critical factors for
glomerular sclerosis. Cytokines and growth factors also play
important roles. Au et al observed that increases in the
number of megakaryocytes correlated with dysfunction and the
overproduction of platelet-derived growth factor (PDGF) and
transforming growth factor (TGF)-β in patients with PV (13). The degranulation of the
platelet-induced sustained release of PDGF and other cytokines
induces a rapid progression to crescentic glomerulonephritis and
glomerulosclerosis. Nishi et al observed that in patients
with PV associated with MPGN, platelets, macrophages and fibers
could attach to the glomerular capillary wall and induce the
upregulation of thrombomodulin in the capillaries and the PDGF
receptor in mesangial cells (20).
PDGF can subsequently stimulate the proliferation of glomerular
mesangial cells and the formation of extracellular matrix. TGF-β
induces collagen and fibrinogen synthesis via mesangial cells, thus
leading to mesangial sclerosis and podocyte apoptosis. These
processes result in FSGS-like lesions. In addition, the medications
used for PV treatment can cause renal impairment. Ravandi-Kashani
et al reported that patients presented with thrombotic
microangiopathy and RF following IFN therapy and subsequently
required hemodialysis (23). Read
et al observed that HU could induce dose-dependent minimal
change nephrotic syndrome (MCNS) in Wistar rats. However, no
similar results have been reported for humans (24).
The case reported in the present study is the ninth
case of PV associated with IgAN. The patient was male, which is
consistent with previous studies. More clinical evidence is
required to conclude whether there are gender differences in the
incidence of this disease. The protein- and hematuria were well
controlled for >6 years with PV treatment. The patient's serum
Ig and complement levels were normal. Both kidneys were relatively
large and the SCr level was higher than normal, but GFR was within
the normal range. The following points require consideration. i)
The patient had no manifestations of tubular-interstitial damage,
such as the increased output of nocturia and hypotonic urine, and
it may be reasoned that the kidney damage caused by hypertensive
renal arteriosclerosis and hyperuricemia was insufficient to
explain the elevated SCr, and the glomerular lesions were the main
diagnosis. ii) Renal hemodynamic changes could result in increased
GFR in the early stage of diabetic nephropathy, and the GFR can
gradually return to normal and then decrease to below normal in
stage III. The patient might have experienced early stage
hyperfiltration and hyperperfusion, followed by a reduction in the
GFR to the normal range when the disease progressed to the elevated
SCr stage. However, diabetic nephropathy is a glomerulosclerosis
caused by the thickening of the glomerular capillary basement
membrane and mesangial dilation, and IF mainly showed an IgG
deposit. For this patient, only a mild diffuse hyperplasia of the
mesangial cells and matrix was observed, and the pathological
manifestation was mild, which was inconsistent with the elevated
SCr and significantly different from renal damage caused by
hyperfiltration in diabetic nephropathy. iii) No micro-thrombosis
or platelet aggregation was observed in the renal tissues, and the
coagulation tests did not show a high-coagulation or high-viscosity
state. Therefore, renal hemodynamic changes were believed to play a
major role in this patient. iv) Makdassy et al studied the
pathogenesis of IgAN in 322 patients and observed its association
with other diseases in 57.8% of cases, among which it was
associated with hematopathy in 3.7%, and generally associated with
PV (25). The main mechanism might
be the increased formation of IgA immune complex and reduced
clearance. The patient experienced a sore throat 1 week prior to
hospitalization; therefore, the cause of the IgAN may be
questioned. Possibly, the IgAN was combined with the 6-year PV
diagnosis, or the IgAN was induced by the excessive activation of B
cells and their differentiation toward IgA-producing plasma cells
due to the recent immune regulation dysfunction of the palatine
tonsils. We are certain that PDGF is associated with IgAN activity,
which stimulates mesangial cell proliferation and is involved in
the formation and development of IgAN nephropathy. In patients with
PV, PDGF expression increases and significantly promotes the
progress of IgAN. v) Finally, in some patients with secondary IgAN,
IgAN could be improved or alleviated following the elimination of
the cause. For the patient of the present study, the 24-h UR was
positively correlated with the blood cell counts, which was
consistent with this characteristic.
Currently, whether renal disease is a complication
or comorbidity of PV remains unclear. More clinical evidence is
required to confirm a causal relationship.
The comparison of the two main pathological types of
PV combined with renal disease showed the following
characteristics: i) IgAN mainly occurred in males. There was no
detectable gender difference in the FSGS incidence and the age of
onset for FSGS was older than for IgAN. ii) NS was the predominant
clinical manifestation of both IgAN and FSGS, IgAN was often
accompanied with hematuria and mild-to-moderate renal
insufficiency. FSGS progressed relatively faster.
In summary, the present study reports a rare
clinical case of IgAN in a patient with PV. Although PV associated
with renal disease is rarely observed in clinical practice, there
are a variety of pathological types, which should be considered by
clinicians during the diagnosis and treatment of PV. More clinical
studies are necessary to further characterize the clinical and
histopathological features and incidence of this disease to develop
the most appropriate treatment strategies.
References
|
1
|
Godfrey AL, Chen E, Pagano F, Silber Y,
Campbell PJ and Green AR: Clonal analyses reveal associations of
JAK2V617F homozygosity with hematologic features, age and gender in
polycythemia vera and essential thrombocythemia. Haematologica.
98:718–721. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kuriakose E, Vandris K, Wang YL, Chow W,
Jones AV, Christos P, Cross NC and Silver RT: Decrease in JAK2
V617F allele burden is not a prerequisite to clinical response in
patients with polycythemia vera. Haematologica. 97:538–542. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Okuyama S, Hamai K, Fujishima M, Ohtani H,
Komatsuda A, Sawada K and Wakui H: Focal segmental
glomerulosclerosis associated with polycythemia vera: report of a
case and review of the literature. Clin Nephrol. 68:412–415. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee SM, Rao VM, Franklin WA, Schiffer MS,
Aronson AJ, Spargo BH and Katz AI: IgA nephropathy: Morphologic
predictors of progressive renal disease. Human Pathol. 13:314–322.
1982. View Article : Google Scholar
|
|
5
|
Kasuno K, Ono T, Kamata T, Kawamura T,
Suyama T, Oyama A, Matsushima H, Kuwahara T, Muso E and Sasayama S:
IgA nephropathy associated with polycythaemia vera: Accelerated
course. Nephrol Dial Transplant. 12:212–215. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim MY: A case report of polycythemia vera
associated with IgA nephropathy. Korean J Nephrol. 13:977–981.
1994.
|
|
7
|
Kwon MY: A case report of chronic renal
failure caused by IgA nephropathy combined with polycythaemia vera.
Korean J Nephrol. 18:843–847. 1999.
|
|
8
|
Chung J, Park PG and Song KI: IgA
nephropathy in a patient with polycythemia vera: Clinical
manifestation of chronic renal failure and heavy proteinuria. Am J
Nephrol. 22:397–401. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yaguchi T, Takimoto C, Uchimura H, et al:
IgA nephropathy associated with polycythemia vera. Nippon Naika
Gakkai Zasshi. 94:1612–1614. 2005.(In Japanese). View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tian M, Liu DJ and Li DT: A case report of
IgA nephropathy combined with polycythaemia vera treated by
mycophenolate mofetil. Shiyong Yaowu Yu Linchuang. 14:358–359.
2011.(In Chinese).
|
|
11
|
Kanauchi M, Dohi K, Shiiki H, Fujii Y and
Ishikawa H: Henoch-Schönlein purpura nephritis associated with
polycythemia vera. Intern Med. 33:36–40. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sharma RK, Kohli HS, Arora P, et al: Focal
segmental glomerulosclerosis in a patient with polycythemia rubra
vera. Nephron. 69:3611995. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Au WY, Chan KW, Lui SL, Lam CC and Kwong
YL: Focal segmental glomerulosclerosis and mesangial sclerosis
associated with myeloproliferative disorders. Am J Kidney Dis.
34:889–893. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kosch M, August C, Hausberg M, et al:
Focal sclerosis with tip lesions secondary to polycythaemia vera.
Nephrol Dial Transplant. 15:1710–1711. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ulusoy S, Ozkan G, Sönmez M, et al:
Absence of hypoalbuminemia despite nephrotic proteinuria in focal
segmental glomerulosclerosis secondary to polycythemia vera. Intern
Med. 49:2477–2480. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Martín JS, Suárez LG and Martín FG: Focal
and segmental glomerulosclerosis associated with polycythemia vera.
Nefrologia. 30:138–140. 2010.(In Spanish). PubMed/NCBI
|
|
17
|
Iyoda M, Ito J, Ajiro Y, et al: Focal
segmental glomerulosclerosisin a patient with polycythemia vera.
Nippon Jinzo Gakkai Shi. 47:828–833. 2005.(In Japanese). PubMed/NCBI
|
|
18
|
Cai MS, Wang M, Gan LY and Dong S: Focal
segmental glomerulosclerosis in patient with polycythemia vera: One
case report. Zhonghua Lao Nian Duo Qi Guan Ji Bing Za Zhi.
10:82–83. 2011.(In Chinese).
|
|
19
|
Dai ZY, Li YS and Wu GH: The clinial
pathobiology analysis of polycythemia vera complicating kidney
disease. Zhongguo Zhongxiyi Jiehe Shenbing Zazhi. 9:1004–1005.
2008.(In Chinese).
|
|
20
|
Nishi Y, Namikoshi T, Sasaki T, et al:
Histopathological manifestations of membranoproliferative
glomerulonephritis and glomerular expression of plasmalemmal
vesicle-associated protein-1 in a patient with polycythemia vera.
Clin Nephrol. 74:393–398. 2010.PubMed/NCBI
|
|
21
|
Fernando BK, Ruwanpathirana HS and
Veerasuthen T: A patient with polycythaemia vera associated with
membranoproliferative glomerulonephritis. Ceylon Med J. 56:119–120.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Oymak O, Oymak FS, Patiroglu T, et al:
Polycythemia vera presenting with rapidly progressive
glomerulonephritis and pyoderma gangrenosum. Nephron. 86:346–347.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ravandi-Kashani F, Cortes J, Talpaz M and
Kantarjian HM: Thrombotic microangiopathy associated with
interferon therapy for patients with chronic myelogenous leukemia:
Coincidence or true side effect? Cancer. 85:2583–2588. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Read NG, Astbury PJ, Evans GO, Goodwin DA
and Rowlands A: Nephrotic syndrome associated with N-hydroxyureas,
inhibitors of 5-lipoxygenase. Arch Toxicol. 69:480–490. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Makdassy R, Beaufils M, Merier A, Mignon
F, Moulonguet-Doleris L and Richet G: Pathologic conditions
associated with IgA mesangial nephropathy: Preliminary results.
Contrib Nephrol. 40:292–295. 1984. View Article : Google Scholar : PubMed/NCBI
|















