Introduction
Exfoliative dermatitis, also known as erythroderma,
is an uncommon but serious skin disorder which results in
generalized eruption of the skin. It is usually caused by a variety
of underlying dermatoses, drug reactions and malignancies,
including lymphoma, leukemia and solid tumors (1–8).
Previous studies have demonstrated that therapeutic agents
including sulfonamides, captopril, phenobarbital, all-transretinoic
acid and anti-tubercular drugs, such as rifampicin, isoniazid,
ethambutol and pyrazinamide, may induce exfoliative dermatitis
(3–8).
Protein pump inhibitors (PPIs) are commonly-used
prescription drugs, which can inhibit gastric acid secretion by
blocking the gastric H+/K+-ATPase that is
necessary for the final step of this process. PPIs are widely
administered for the treatment of numerous acid-associated
diseases, including peptic ulcer, gastroesophageal reflux disease
and Zollinger-Ellison syndrome (9,10). In
addition, PPIs have an important role in eradicating infections
caused by Helicobacter pylori, controlling gastrointestinal
bleeding and preventing nonsteroidal anti-inflammatory drug-induced
gastric mucosal injuries. Furthermore, Ramirez et al
(11) suggested that the long-term
use of PPIs may be considered a major risk factor in the occurence
of anaphylaxis with common PPI-induced anaphylactic symptoms
including skin rashes, erythema and systemic pruritus, and rare
symptoms including urticaria, herpes and skin allergic edema. In
severe cases, acute diffuse epidermal necrolysis and anaphylactic
shock have also been reported (12).
Case report
On September 29th 2014, a 74-year-old
female patient was admitted to Hubei General Hospital (Hubei,
China) following a road accident with pains in the chest, abdomen,
waist, back, nose, left wrist and lower limbs. A cranial computed
tomography (CT) scan did not detect any abnormalities, whereas a
nasal CT scan suggested a right nasal passage bone fracture and a
chest CT scan detected numerous bilateral rib fractures, bilateral
small pneumothoraces, traumatic wet lungs and compression fractures
of the T12 and L1 vertebral bodies. Prior to the accident, the
patient had been healthy, without a history of food or drug
allergies.
A physical examination of the patient revealed the
following: Body temperature, 36.5°C; pulse rate, 100 bpm;
respiratory rate, 21 breaths per minute; blood pressure, 100/60
mmHg; and body weight, 53 kg. The patient had perirhinal bruises,
minor bleeding and local tenderness. Rough breathing sounds and
diffuse moist rates were detected for both lungs. No spinal
deformities were detected; however, movement of the thoracic and
lumbar vertebrae was markedly restricted. In addition, the lower
thoracic and lumbar vertebrae exhibited tenderness and percussive
pain, which radiated to the lower limbs. The bilateral patellar
tendon reflex and Achilles tendon reflex were significantly
decreased. The lower limbs exhibited hypoesthesia, and the left
side was particularly severe. Furthermore, the muscle strength of
the lower limbs was significantly reduced, with grade 2 on the left
iliopsoas, quadriceps femoris and extensor pollicis muscle, and
grade 2+ on the right iliopsoas, quadriceps femoris and extensor
pollicis muscle. The muscle tension was normal and the dorsalis
pedis artery pulse was palpated. The diagnosis at admission
included: i) Chest and waist trauma, including compression
fractures of the T12 and L1 vertebral bodies and incomplete
paraplegia; ii) chest trauma, including multiple rib fractures,
bilateral small pneumothoraces and traumatic wet lungs; iii) a
nasal bone fracture; and iv) a left wrist trauma.
On October 5th, the patient was
transferred from the Department of Orthopedics to the Department of
Thoracic Surgery, and a daily intravenous administration of 30 mg
lansoprazole was initiated in order to prevent the development of a
stress ulcer. On October 8th, the patient complained of
abdominal pain, and a gastric tube was inserted for
gastrointestinal decompression, after which blood was detected in
the drained gastric fluid. Subsequently, the patient was diagnosed
with gastrointestinal bleeding and was treated with the following
hemostatic therapies, daily intravenous injections of 2 units
hemocoagulase agkistrodon and 1,000 units thrombin lyophilized
powder, administered via the nasal cavity every 4 h. A subsequent
gastroscopy detected duodenal bulb bleeding, which was associated
with the formation of a blood clot. As a result of the
gastrointestinal bleeding, the once daily intravenous drip of 30 mg
lansoprazole was increased to twice daily.
On October 11th, the volume of gastric
drainage significantly increased, and thus hemocoagulase
agkistrodon and thrombin lyophilized powder injections were
administered at the same doses as previously on October
8th, in order to attenuate the bleeding. In addition,
the intravenous drip administering 30 mg lansoprazole was replaced
with an 80 mg esomeprazole intravenous drip twice daily, in order
to inhibit acid secretion.
On October 13th, the gastrointestinal
bleeding was controlled; however, the patient presented with
paradoxical respiration, which progressively deteriorated.
Subsequently, the patient was transferred to an operating room for
emergency open reduction and internal fixation of numerous rib
fractures and the sternal fracture. The surgery was successful;
however, the patient remained critically ill, and thus was
transferred to the intensive care unit (ICU) for continued
treatment.
In the ICU, the patient once again suffered from
gastrointestinal bleeding and subsequent acute kidney injury (AKI);
therefore PPIs were administered in order to inhibit acid
secretion. An intravenous drip administering 40 mg esomeprazole
sodium twice daily was administered from October 14th to
15th prior to continuous pumping of 200 mg esomeprazole
from October 15th to 17th. This regimen was
subsequently repeated; an intravenous drip administering 40 mg
esomeprazole sodium twice daily was administered from October
18th to 26th prior to continuous pumping of
200 mg esomeprazole from October 26th to
28th. On October 28th, the gastrointestinal
bleeding was markedly attenuated and the continuous pumping of 200
mg esomeprazole was replaced with once daily nasal administration
of 40 mg omeprazole enteric-coated capsules. The patient presented
with systemic edema and a significant decrease in the volume of
urine on October 19th; however both were markedly
improved following treatment with hemocoagulase agkistrodon and
thrombin lyophilized powder.
On the night of October 31st, minor skin
rashes were detected on the chest, right lower limb and foot of the
patient. In addition, the patient lost the ability to autonomously
expectorate, due to the overproduction of sputum. Therefore, a
tracheotomy was conducted, and the patient was transferred to the
Department of Thoracic Surgery on November 1st, after
which treatment with omeprazole was terminated and 30 mg
lansoprazole was intravenously administered once daily via a
drip.
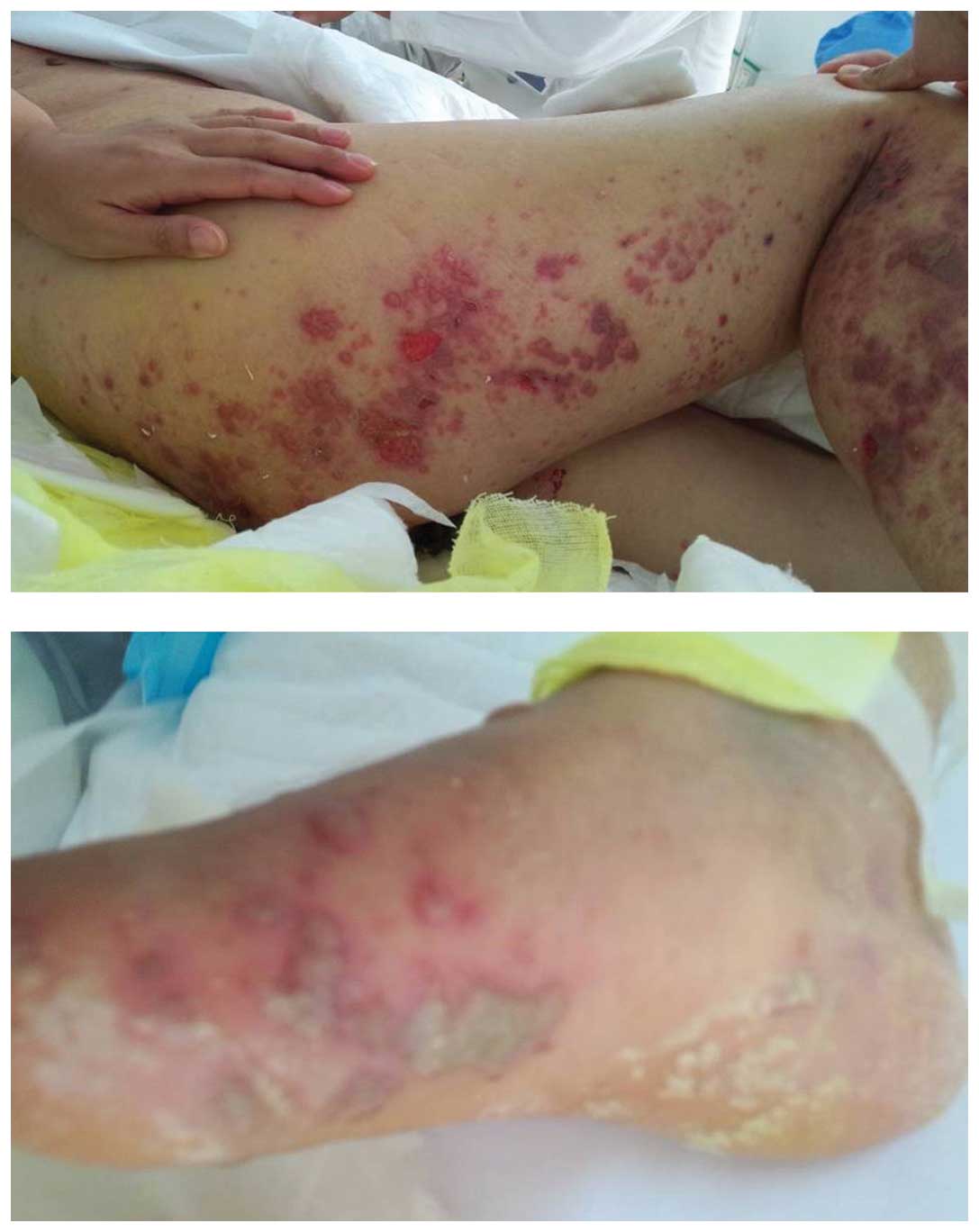
On November 3rd, the severity of the skin
rashes on the chest and right lower limb and foot increased
significantly, with the skin rash on the right lower limb merging
with that on the right foot (Fig.
1). In addition, the routine blood examination revealed an
elevated white blood cell count. On November 4th, the
skin rashes ulcerated and tracheal hemoptysis occurred, after which
a large volume of bloody mucinous sputum was suctioned off.
Following a consultation between the Departments of Pharmacy,
Dermatology and Nephrology, and the ICU, the patient was diagnosed
with exfoliative dermatitis and treatment with lansoprazole was
terminated. Anti-infection treatment, which comprised of
intravenous administration of 4.5 g piperacillin sodium and
tazobactam sodium three times a day and 200 mg teicoplanin twice
daily; and symptomatic treatment, in the form of daily
intramuscular injection of 20 mg diphenhydramine hydrochloride,
were administered instead.
On November 5th, the sputum was yellow
and white in appearance, and lacked traces of blood. On November
6th, the rashes on the chest and right lower limb and
foot were markedly improved and the blisters were healed; however,
the relatives of the patient subsequently decided to terminate all
treatments and the patient succumbed to her traumas,
gastrointestinal hemorrhage and infection-induced multiple organ
(lung, heart and kidney) failure at 1:25 PM on November
10th. Written informed consent was obtained from the
patient's family.
Discussion
The overuse of PPIs is a universal phenomenon in
clinical practice (13–15), which may be due to a lack of clear
and unified withdrawal instructions. Long-term treatment of a
patient with a high-dose of PPIs has previously been associated
with liver and renal dysfunction, gastrointestinal reactions, bone
fractures, hypomagnesemia, malabsorption of vitamin C, B12 and
iron, Clostridium difficile infections, and an increased
incidence of pneumonia (16). In the
present case, an elderly patient presented with bullous exfoliative
dermatitis following long-term treatment with PPIs. To the best of
our knowledge, this is the first report of PPI-induced exfoliative
dermatitis.
Exfoliative dermatitis is a severe eruption of the
skin associated with numerous drugs, including anti-tubercular
drugs, such as rifampicin, isoniazid, ethambutol and pyrazinamide,
sulfonamides, captopril, phenobarbital and all-transretinoic acid
(3–8); however, to the best of our knowledge,
PPI-induced exfoliative dermatitis has not previously been
reported. The clinical symptoms of exfoliative dermatitis include:
Systemic skin diffuse redness; desquamation (more severe on the
face, hands and feet); blisters; erosion, exudation and scab
formation on epidermal folds and mucosae; and pruritus and
thermalgia, leading to systemic skin necrosis and generalized
leaf-like desquamation. In addition, exfoliative dermatitis has
previously been associated with severe systemic symptoms in some
patients, including fever, shivering, and nausea and vomiting
(1). In the case of patients with
severe exfoliative dermatitis, a delay in the initiation of
treatments is associated with life-threatening water and
electrolyte disorders and the occurrence of secondary
infections.
In the present case, an elderly female patient was
treated with PPIs for 28 days (between October 6th and
November 2nd), and a skin rash was detected on October
31st, which markedly spread and ulcerated within 3 days
of the initial presentation. Following a combined consultation
between the Departments of Nephrology, Dermatology and Pharmacy,
and the ICU, treatment of the patient with 30 mg lansoprazole
injection was terminated for 3 days, after which the skin rashes
markedly improved, thus suggesting that a temporal relationship
existed between the exfoliative dermatitis and PPI treatment. In
addition, the patient exhibited hemoptysis as the skin rashes
increased in severity; however, following termination of
lansoprazole treatment and administration of hemostatic drugs, the
hemoptysis was markedly attenuated and the volume of bloody sputum
was significantly decreased. On November 5th, sputum
extracted from the patient was yellow and white in appearance,
indicating respiratory mucosal injuries in the patient. The
symptoms of the patient were consistent with exfoliative
dermatitis, and a temporal relationship between this skin eruption
and treatment with PPIs was again indicated.
The authors of the present case hypothesize that the
occurrence of exfoliative dermatitis in the patient was closely
associated with an accumulation of PPIs. Throughout the duration of
the treatment, lansoprazole and esomeprazole were the predominant
PPIs used to treat the patient. Lansoprazole is metabolized by
cytochrome P450 (CYP)-2C19 and CYP3A4 into 5-hydroxy lansoprazole
and sulfonic lansoprazole, which are excreted with bile and via
defecation (17,18). Esomeprazole (~73%) is metabolized by
CYP2C19 into hydroxy and demethylic metabolites, which lack the
ability to inhibit acid secretion, and ~27% is metabolized by
CYP3A4 into esomeprazole sulfone, which is responsible for the
acid-inhibiting activity of esomeprazole (19). As a result, ~80% inactive and <1%
active metabolites are excreted in bile, and the remainder in
feces. Lansoprazole and esomeprazole share various traits: Firstly,
the major metabolites of both (sulfonic lansoprazole and
esomeprazole sulfone for lansoprazole and esomeprazole,
respectively) are metabolized by CYP3A4 in the liver; and secondly,
the metabolites of both drugs are excreted through bile rather than
via the kidneys. Previous studies have suggested that renal failure
does not affect the secretion of metabolites; however, AKI may
significantly reduce the activity of CYP3A4 (20,21).
Therefore, in the present case, AKI may have significantly
decreased the metabolism of lansoprazole and esomeprazole in the
liver, leading to the accumulation of PPIs. In addition, the
patient was hypoxic and ischemic following the severe traffic
accident, which may have reduced the metabolism of lansoprazole and
esomeprazole in the liver and obstructed the secretion of the
metabolites, leading to their accumulation.
In the present case, the clinician did not respond
to the small number of spot-like skin rashes on the patient on the
night of October 31st, and the rashes rapidly progressed
to a severe eruption within 3 days. Of the PPI-induced skin
anaphylactic reactions, papula (43.8%) is the most commonly
observed (12). To the best of our
knowledge, the present case is the first to report the occurrence
of PPI-induced exfoliative dermatitis, and aims to draw attention
to the risk of PPI-induced anaphylaxis during clinical practice,
particularly in high-risk patients, including the elderly and those
with liver and renal dysfunction, or a history of allergies. The
present case suggests that treatment of a patient with PPI should
be terminated upon detection of an adverse reaction, in order to
prevent the occurrence of subsequent severe adverse reactions and
events.
References
|
1
|
Sehgal VN, Srivastava G and Sardana K:
Erythroderma/exfoliative dermatitis. A synopsis. Int J Dermatol.
43:39–47. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rym BM, Mourad M, Bechir Z, Dalenda E,
Faika C, Iadh AM and Amel BO: Erythroderma in adults, A report of
80 cases. Int J Dermatol. 44:731–5. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dua R, Sindhwani G and Rawat J:
Exfoliative dermatitis to all four first line oral anti-tubercular
drugs. Indian J Tuberc. 57:53–6. 2010.PubMed/NCBI
|
|
4
|
Jaisuresh K: Pyrazinamide-induced
exfoliative dermatitis in a patient on hemodialysis: A rare
complication. Case Rep Nephrol. 2013:3872932013.PubMed/NCBI
|
|
5
|
Rabbat JC, Lin MY and Moy JN: Induction of
tolerance in a patient with a history of exfoliative dermatitis to
trimethoprim-sulfamethoxazole. Ann Allergy Asthma Immunol.
109:360–361. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
O'Neill PG, Rajan N, Charlat ML and Bolli
R: Captopril-related exfoliative dermatitis. Tex Med. 85:40–41.
1989.
|
|
7
|
Sawaishi Y, Komatsu K, Takeda O, Tazawa Y,
Takahashi I, Hayasaka K and Takada G: A case of tubulo-interstitial
nephritis with exfoliative dermatitis and hepatitis due to
phenobarbital hypersensitivity. Eur J Pediatr. 151:69–72. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ipek Y, Hulya D and Melih A: Disseminated
exfoliative dermatitis associated with all-transretinoic Acid in
the treatment of acute promyelocyticleukemia. Case Rep Med.
2012:2361742012.PubMed/NCBI
|
|
9
|
Wedemeyer RS and Blume H: Pharmacokinetic
drug interaction profiles of proton pump inhibitors: An update.
Drug Saf. 37:201–211. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Watson C, Zhu L, Guan S, Machen TE and
Forte JG: Reaction of proton pump inhibitors with model peptides
results in novel products. J Pharmacol Sci. 122:213–22. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ramírez E, Cabañas R, Laserna LS, Fiandor
A, Tong H, Prior N, Calderón O, Medrano N, Bobolea I, Frías J and
Quirce S: Proton pump inhibitors are associated with
hypersensitivity reactions to drugs in hospitalized patients, A
nested case-control in a retrospective cohort study. Clin Exp
Allergy. 43:344–352. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chang YS: Hypersensitivity reactions to
proton pump inhibitors. Curr Opin Allergy Clin Immunol. 12:348–353.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hood W, McJunkin B, Warnock A, Girme A,
Smith N and Robinson B: Proton pump inhibitor prescribing and costs
in a large outpatient clinic. W V Med J. 110:16–21. 2014.PubMed/NCBI
|
|
14
|
Mouterde O, Bellaïche M, Dumant C and
Mallet E: Gastroesophageal reflux and proton pump inhibitors,
Panacea or prescription abuse? Arch Pediatr. 17:739–740. 2010.(In
French). View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Akram F, Huang Y, Lim V, Huggan PJ and
Merchant RA: Proton pump inhibitors, Are we still prescribing them
without valid indications? Australas Med J. 7:465–470. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Reimer C and Bytzer P: Adverse events
associated with long-term use of proton pump inhibitors. Ugeskr
Laeger. 174:2289–2293. 2012.(In Danish). PubMed/NCBI
|
|
17
|
Zhang D, Zhang Y, Liu M, Wang X, Yang M,
Han J and Liu H: Pharmacokinetics of lansoprazole and its main
metabolites after single and multiple intravenous doses in healthy
Chinese subjects. Eur J Drug Metab Pharmacokinet. 38:209–215. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Andersson T: Pharmacokinetics metabolism
and interactions of acid pump inhibitors. Focus on omeprazole,
lansoprazole and pantoprazole. Clin Pharmacokinet. 31:9–28. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zvyaga T, Chang SY, Chen C, Yang Z,
Vuppugalla R, Hurley J, Thorndike D, Wagner A, Chimalakonda A and
Rodrigues AD: Evaluation of six proton pump inhibitors as
inhibitors of various human cytochromes P450: Focus on cytochrome
P450 2C19. Drug Metab Dispos. 40:1698–1711. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kirwan CJ, MacPhee IA, Lee T, Holt DW and
Philips BJ: Acute kidney injury reduces the hepatic metabolism of
midazolam in critically ill patients. Intensive Care Med. 38:76–84.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Philips BJ, Lane K, Dixon J and Macphee I:
The effects of acute renal failure on drug metabolism. Expert Opin
Drug Metab Toxicol. 10:11–23. 2014. View Article : Google Scholar : PubMed/NCBI
|