Introduction
Malignant biliary strictures are caused by numerous
primary or metastatic disease in intrahepatic, extrahepatic or
hilar locations (1). Surgical
resection is the best, and potentially curative, therapy available.
Regrettably, between 70 and 90% of patients possess malignant
biliary strictures that are unresectable at diagnosis; thus,
biliary decompression becomes the primary goal of intervention
(2). Compared with surgical
intervention, stent insertion offers shorter hospitalization, lower
overall cost and lower morbidity (2). Therefore, endoscopic stenting of the
biliary tract has become the standard therapeutic technique for
patients with a life expectancy of >3-6 months (3,4).
Previous studies have demonstrated the superiority
of self-expanding metal stents (SEMS) over plastic stents for
maintaining biliary drainage (5–7).
However, SEMS can become occluded due to epithelial hyperplasia,
tumor ingrowth or overgrowth, biofilm deposition and sludge
formation (8). Studies have
demonstrated that the median SEMS patency is 120 days (5,9);
however, once biliary stricture occurs, it may lead to morbidity
and mortality. Therefore, long-term patency of SEMS remains an
unresolved issue.
A number of methods, including covered nitinol SEMS
and endoscopic photodynamic therapy (PDT), have been proposed for
increasing the duration of stent patency in malignant biliary
strictures (10–12). In 2010, Gao et al (13) reviewed 20 relevant studies and
concluded that PDT offered considerable benefit for the survival
and quality of life of patients with unresectable
cholangiocarcinoma. However, the most common adverse events of PDT
are cholangitis (27.5%) and phototoxicity (10.2%) (13). In addition, the management of
patients treated with PDT remains expensive and time consuming.
Radiofrequency ablation (RFA), which can be
performed either percutaneously or intraoperatively, has been used
to achieve localized tumor necrosis in solid neoplasms for a number
of years (14,15). The technique delivers a high quantity
of thermal energy to the target tissue, with a curative or
palliative intent. Preliminary animal studies (16,17) and
human clinical studies (18,19) have investigated a promising
endoscopic therapy for patients with malignant biliary strictures.
The aim of the present study was to evaluate the feasibility and
safety of endoscopic RFA for the treatment of malignant biliary
strictures.
Materials and methods
Ethics statement
The present study was approved by the Medical Center
for Digestive Diseases, the Second Affiliated Hospital of Nanjing
Medical University (Nanjing, China). In addition, institutional
review board approval was obtained for this retrospective study.
Each patient was fully informed of the details and complications of
the procedure, and written informed consent was obtained.
Patients
A total of 12 patients (6 male and 6 female; median
age, 68 years; age range, 46–82 years) with unresectable malignant
biliary strictures were referred to the Medical Center for
Digestive Diseases and underwent endoscopic RFA between December
2011 and October 2013. The majority (10/12) of the patients were
referred to the Center due to jaundice, whereas the remaining
patients complained of upper abdominal discomfort. Prior to the RFA
procedure, all biliary strictures were confirmed by magnetic
resonance cholangiopancreatography, computer tomography or
endoscopic retrograde cholangiopancreatography (ERCP).
The following variables were measured: Patient
demographics, procedure-related complications, duration of hospital
stay, adverse events within 30 days post-intervention, stent
patency following the last electively performed RFA procedure in
each patient, and 30- and 90-day mortality rates. Stent patency and
patient survival time were assessed using Kaplan-Meier statistical
analysis.
ERCP procedure
ERCP was performed using a duodenoscope (TJF 260 V;
Olympus Corporation, Tokyo, Japan) and a Habib™ EndoHBP catheter
(EMcision, London, UK). The catheter had an 8 F bipolar probe and
two ring electrodes (8 mm apart), with the distal electrode 5 mm
from the leading edge, providing local coagulative necrosis over a
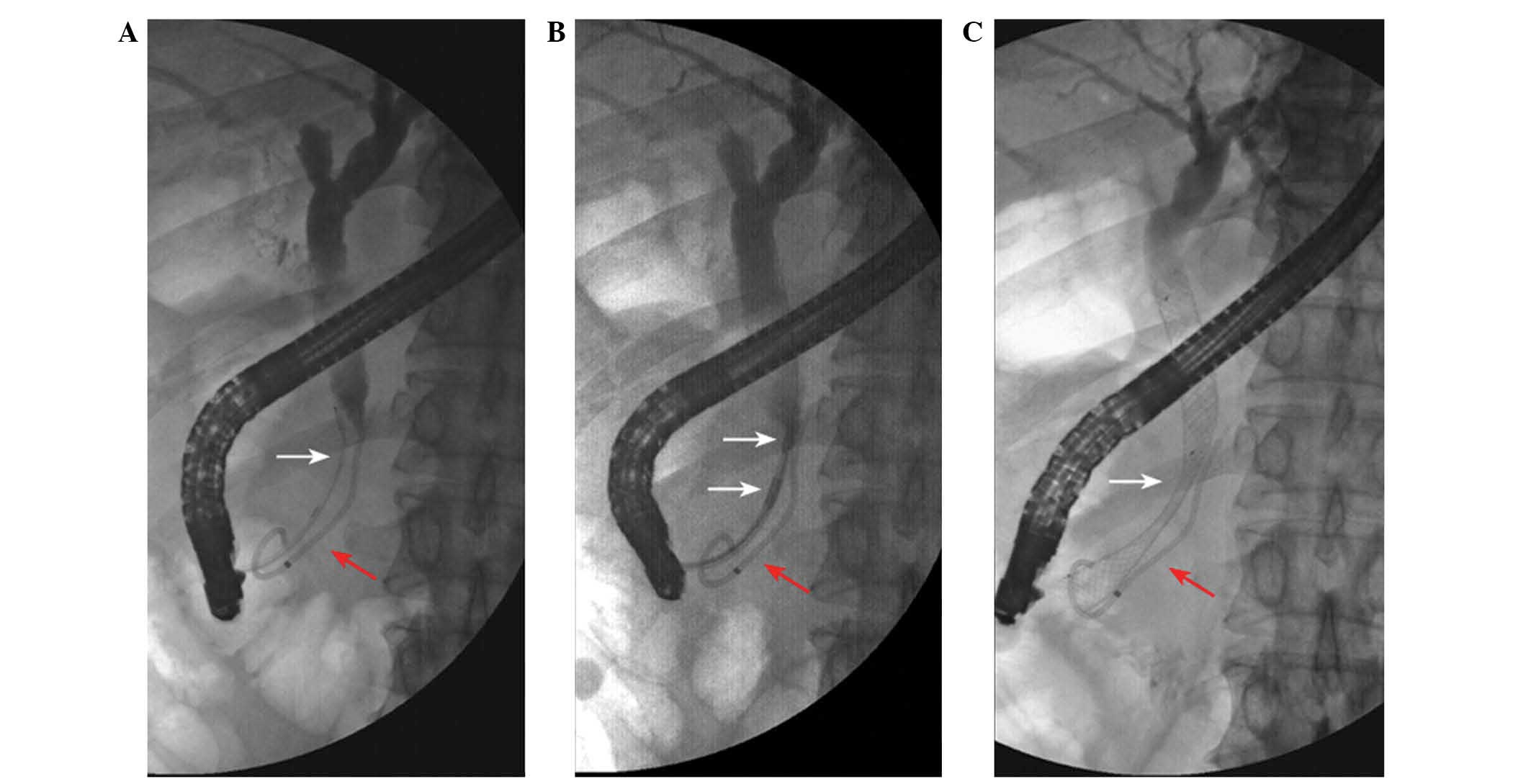
length of 2.5 cm (Fig. 1). The
catheter is compatible with standard side-viewing endoscopes (3.2
mm working channel), and can be passed over 0.035 inch guidewires.
Ablation was performed using an RFA generator (1500 RF generator;
RITA Medical Systems, Inc., Fremont, CA, USA), which delivered
electrical energy at 400 kHz and was set at 7–10 W for 90–120
sec.
Following cannulation of the common bile duct, a
cholangiography was performed to assess the length, diameter and
location of the biliary stricture. An RF catheter was placed
through the biliary stricture under fluoroscopic guidance.
Following RFA, a plastic stent (Flextent; Changzhou New District
Garson Medical Stent Apparatus Co., Ltd., Changzhou, China) or SEMS
(Wallstent; Boston Scientific, Ltd., Marlborough, MA, USA) was
applied. Subsequently, a cholangiogram was obtained to confirm the
correct positioning of the endoscopic stent and the patency of the
biliary tract (Fig. 2).
RFA energy can be delivered repetitively at
different tumor sites within one procedure, according to the
stricture size. It is recommended that the probe be kept stable for
1 min following the delivery of RFA energy in order to avoid the
adhesion of tissue to the electrodes (18). Each patient was followed-up regularly
following the procedure, and no patients were lost to follow-up.
The mean follow-up period was 10.8 months (range, 6–19 months).
Statistical analysis
SPSS version 17.0 (SPSS, Inc., Chicago, IL, USA) was
used to perform data analysis. Continuous variables were compared
using t-test. Stent patency and patient survival time were assessed
using Kaplan-Meier statistics.
Results
Patient demographics
A total of 12 patients underwent 20 RFA procedures
for the treatment of malignant biliary strictures. Among them, nine
patients had unresectable hilar cholangiocarcinoma (Bismuth
classification type I, n=5; type IIIa, n=1; and type IV, n=3)
(20), one patient had primary liver
cancer with hepatectomy, one patient had gastric cancer with
gastrectomy and one patient had congenital choledochal cysts with
high grade intraepithelial neoplasia. Patient demographics are
shown in Table I.
 | Table I.Patient demographics. |
Table I.
Patient demographics.
| Parameter | Variable |
|---|
| Number of
patients | 12 |
| Median age (range),
years | 68 (46–82) |
| Gender
(male/female) | 6/6 |
| Disease |
|
|
Cholangiocarcinoma | 9 |
| Bismuth
type I | 5 |
| Bismuth
type IIIa | 1 |
| Bismuth
type IV | 3 |
| Primary liver
cancer | 1 |
| Gastric cancer | 1 |
| Congenital
choledochal cysts | 1 |
RFA procedures
Endoscopic RFA was performed on the patient
diagnosed with congenital choledochal cysts with high grade
intraepithelial neoplasia. The remaining patients received plastic
stents or SEMSs following endoscopic RFA. A total of seven patients
underwent plastic stent insertion prior to endoscopic RFA, two of
whom received SEMSs following endoscopic RFA; the remaining
patients underwent plastic stent exchange. In one patient, RFA was
performed inside the stent to reestablish patency of the occluded
SEMS. Two patients with Bismuth IV hilar cholangiocarcinoma
underwent immunological cytotherapy following endoscopic RFA. All
20 RFA procedures were performed without any technical problems. A
total of 16 RFAs were performed using ERCP and four were performed
using a percutaneous choledochoscope-assisted approach. A total of
14 RFA procedures were performed at 10 W for 90 sec, whereas the
remaining six RFA procedures were performed at 7 W for 120 sec.
The majority of patients (10/12 patients) underwent
only one RFA session. Repetitive RFA sessions were electively
performed in two patients, one of whom had been diagnosed with
primary liver cancer, and the other had been diagnosed with
congenital choledochal cysts (6 and 4 times, respectively).
Following the RFA procedure, SEMSs were placed in
four patients, and plastic stents were placed in seven patients. No
stent was placed in the patient with congenital choledochal cysts.
The mean hospital stay following the RFA procedure was 8.9 days
(range, 2–22 days). No severe complications, such as hemorrhage,
bile leak, sepsis, biliary stricture and iatrogentic thermal injury
to adjacent structures occurred prior to or following the
procedure.
Biochemical parameters
Postoperative white blood cell (WBC) counts were
elevated in four patients, two of whom experienced fever (38.2 and
38.9°C, respectively). Serum amylase levels were elevated in five
patients, one of whom was diagnosed with post-ERCP pancreatitis.
All of the patients were managed with conventional therapy.
The median length of the strictures treated was 17.5
mm. The mean stricture length treated was 18 mm (SD, 9.5 mm; range,
8–35 mm). The mean stricture diameter prior to RFA was 5.3 mm (SD,
0.9 mm; range 5–8 mm), while the mean diameter following RFA was
12.6 mm (SD, 3.1 mm; range 8–15 mm). The stricture diameters prior
to and following RFA were compared using a paired t-test. There was
a significant increase of 7.3 mm (t=8.6, P≤0.001) in the bile duct
diameter following RFA compared with prior to RFA.
The mean bilirubin expression level prior to RFA was
297.5 µmol/l (SD, 210.5 µmol/l; range 74.6–647.3 µmol/l), while the
mean bilirubin expression level following RFA was 134.3 µmol/l (SD,
86.4 µmol/l; range 31.2–225.2 µmol/l). There was a significant
decrease in the expression level of bilirubin (163.2 µmol/l)
following RFA (t=3.3, P=0.011; Table
II) compared with prior to RFA.
 | Table II.Procedure details and biochemical
parameters. |
Table II.
Procedure details and biochemical
parameters.
| Parameter | Variable |
|---|
| Length of stricture,
mm |
18±9.5a |
| Median number of
ablations (range) | 1 (1–6) |
| Preablation diameter,
mm |
5.3±0.9a |
| Postablation
diameter, mm | 12.6±3.1a |
| Preablation
bilirubin, µmol/l |
297.5±210.5a |
| Postablation
bilirubin, µmol/l |
134.3±86.4a |
Each patient was followed-up regularly following the
procedure (mean, 10.8 months; range, 6–19 months), and no patients
were lost to follow-up. At the end of follow-up, three patients
were alive, two of which had SEMS patency, and the third with
plastic stent patency.
Mortality rates
Marking the first RFA procedure of each patient as
day 1, the 30- and 90-day mortality were 0 and 8.3%, respectively.
One patient succumbed to cachexia on day 40.
Stent patency
In each patient, stent patency was assessed from the
time at which the final elective RFA procedure was performed. All
stents were patent on day 30. One patient succumbed to mortality
due to cachexia on day 40 with SEMS patency. One patient exhibited
plastic stent occlusion on day 40. The second stent occlusion
occurred in a patient with a plastic stent on day 95.
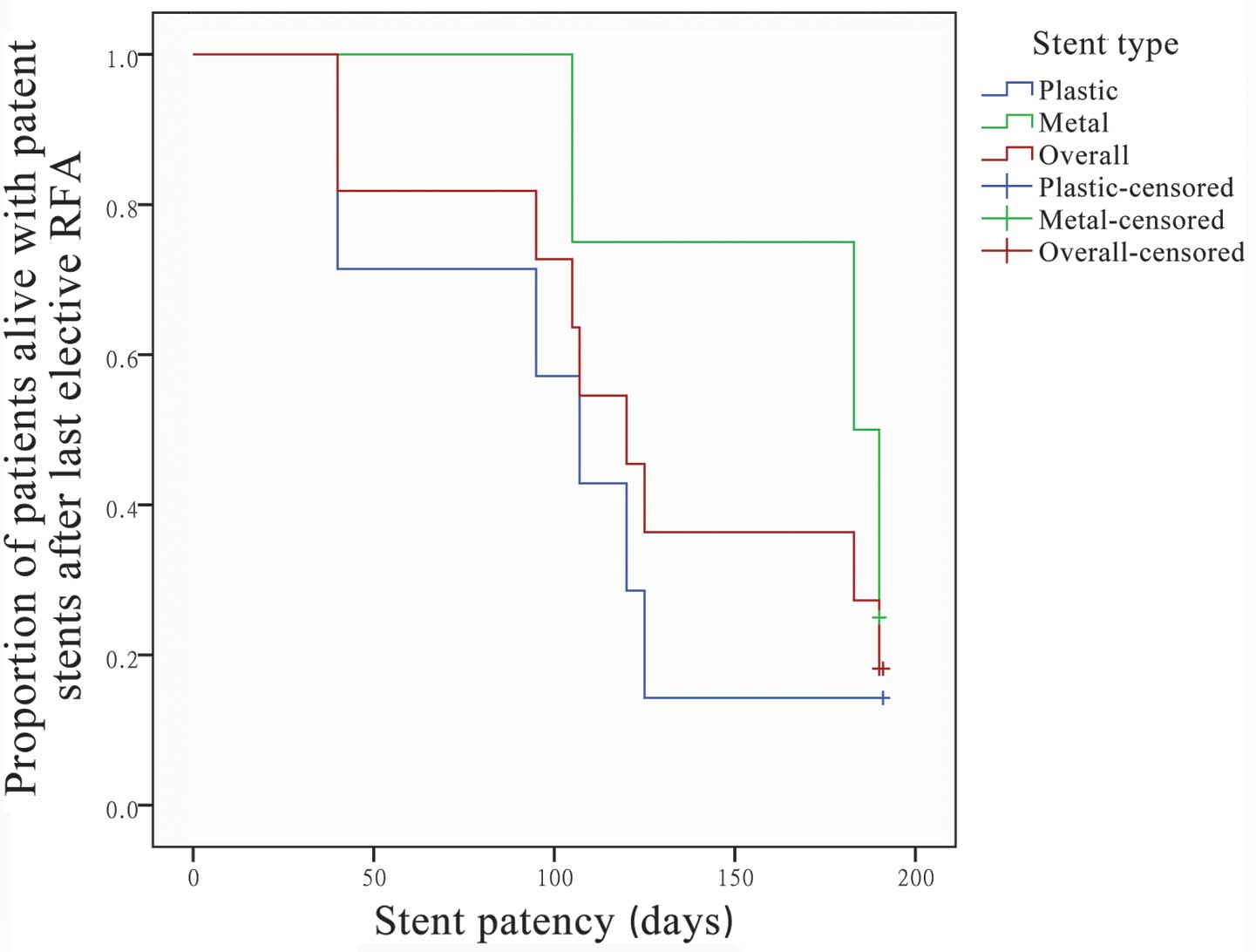
Of the patients treated with SEMS (n=4), the median
stent patency was 125 days (95% confidence interval (CI), 0–252.2
days); two patients succumbed to mortality on days 40 and 125 with
patent stents. Of the patients treated with plastic stents (n=7),
the median stent patency was 107 days (95% CI, 101.9–112.1 days).
No significant difference was detected between the metal and
plastic stents (P=0.84). Of the 11 patients with stents inserted
following RFA, the median stent patency was 125 days (95% CI,
94.7–155.3 days) and the mean stent patency was 131 days (95% CI,
97.3–164.7 days) (Fig. 3).
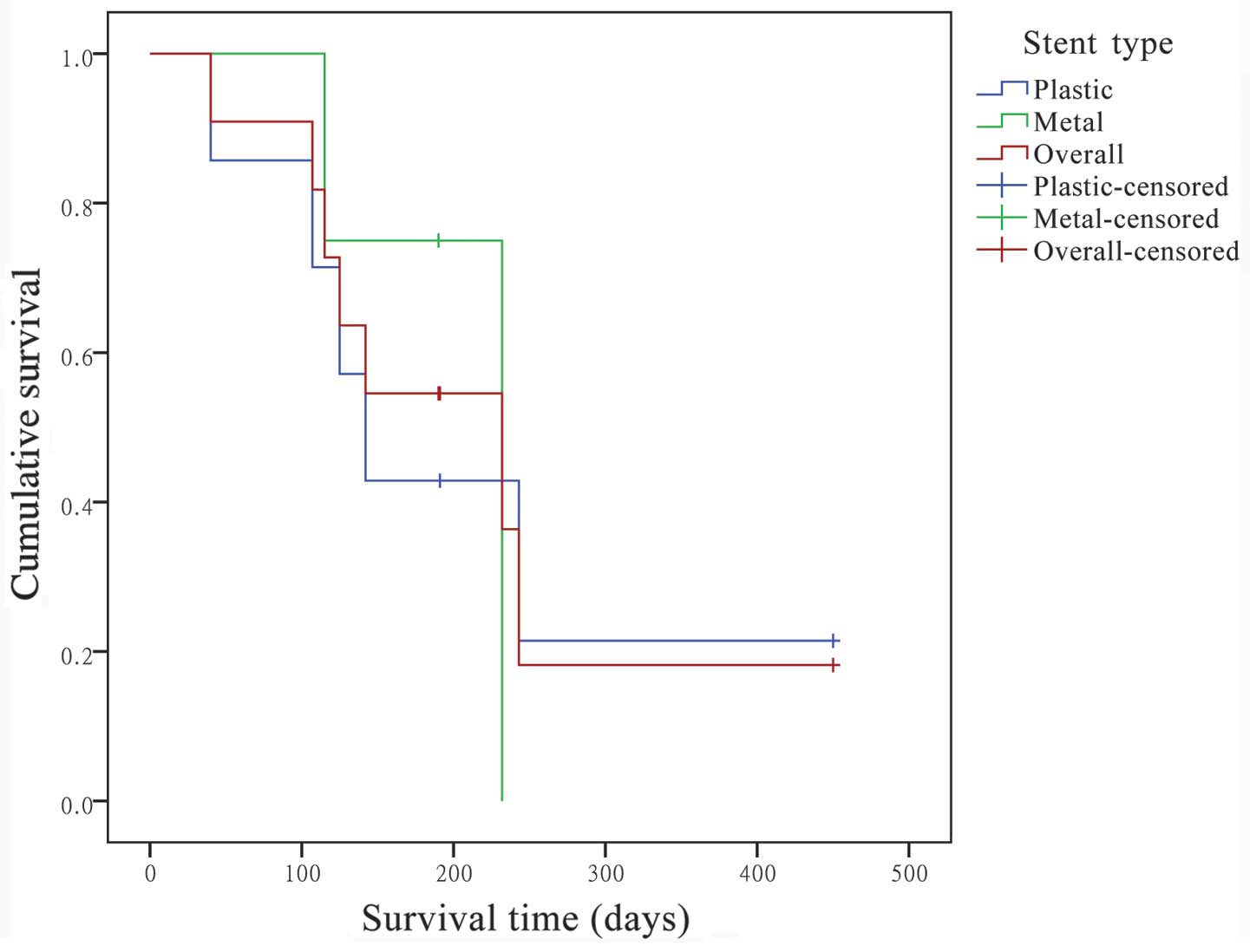
Kaplan-Meier survival curve analysis showed that the
extrapolated median survival following the first RFA was 232.0 days
(95% CI, 32.0–432.0 days), and the extrapolated mean survival was
304.7 days (95% CI, 151.3–458.1 days) (Fig. 4).
Discussion
RFA, which can be performed percutaneously or
intraoperatively, is used to achieve localized tumor necrosis in
primary and secondary hepatic tumors (21,22).
During RFA, a high quantity of thermal energy is delivered to
target tissue, which may prolong the duration of stent patency
(23). RFA has demonstrated
promising results for treating malignant biliary strictures in
previous studies (18,19).
In the present study, 20 RFA procedures were
successfully performed in 100% of the patients, and there were no
difficulties introducing other catheters following stent insertion.
The present results demonstrated that the median stent patency of
the 11 patients with stents inserted following RFA was 125 days and
the mean stent patency was 131 days, which is consistent with the
results of a study by Loew et al (9). However, the present results showed that
the median duration of stent patency in four patients with SEMS was
125 days, and that of the seven patients with plastic stents was
107 days. There was no significant difference between the two
groups. Previous studies have shown that the duration of stent
patency of SEMS was longer compared with that of plastic stents
(5–7). Therefore, the present results are
inconsistent with the findings of previous studies, which may be
due to the relatively small study population investigated in the
present study and another one patient experienced post-ERCP
pancreatitis. Each patient was managed with conventional therapy.
Elevated WBC counts were detected in four patients, two of whom had
a fever, which could be regarded as a common reaction following the
procedure.
These results suggested that RFA treatment of
malignant biliary strictures in the present study was safe and
effective. However, endoscopic RFA may induce iatrogenic thermal
injury to adjacent structures, and the iatrogenic thermal injury
may lead to perforation of involved or intact bile ducts, or vessel
injury (21,24). Dolak et al (19) have previously described a case of
partial liver infarction in a 49-year-old patient with Bismuth IV
hilar cholangiocarcinoma following an RFA procedure. The authors
hypothesized that this event was caused by thermal injury to a
segmental branch of a liver artery. Finally, the patient was
managed conservatively and presented with a favorable course. The
authors recommended pre-interventional imaging to accurately assess
the tumor surroundings, in particular the vascular and biliary
structures, particularly for the treatment of proximal
strictures.
Tal et al (24) described three cases of hemobilia that
occurred within 4–6 weeks of an RFA procedure. Two patients
succumbed to hemorrhagic shock, and the surviving patient was
managed with immediate SEMS insertion into the bleeding bile duct.
The authors hypothesized the hemobilia may be caused by the
necrotic effect induced by RFA. Possible preemptive strategies to
avoid biliary bleeding complications include pre-interventional
assessment with intraductal ultrasound to exclude large blood
vessels in the surrounding tissue. Inserting a SEMS immediately
after the RFA procedure may be an effective method for the
prevention of late bleeding complications.
Notably, in the present study, one patient with
congenital choledochal cysts and high grade intraepithelial
neoplasia underwent the RFA procedure four times between March 2013
and October 2013. The patient was a 47-year-old male with type IV-A
congenital choledochal cysts who underwent cyst excision of the
dilated extrahepatic bile duct with Roux-en-Y hepaticojejunostomy
prior to the RFA procedure. A T-tube was inserted into the common
hepatic duct during the operation, and the RFA procedure was
performed using a choledochoscopic approach. The patient attended
regular follow-ups, tolerated the procedure well and experienced
good palliation of his symptoms (25).
In conclusion, the present study demonstrated that
RFA is an efficient and safe treatment strategy for the palliation
of unresectable malignant biliary strictures. However, the study
was a retrospective analysis with a small population, which are
notable limitations. A large-scale, prospective multicenter trial
with a long-term follow-up evaluation period is required in order
to further quantify the benefits of RFA on stent patency and
survival rates.
References
|
1
|
Webb K and Saunders M: Endoscopic
management of malignant bile duct strictures. Gastrointest Endosc
Clin N Am. 23:313–331. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Moss AC, Morris E, Leyden J and MacMathuna
P: Malignant distal biliary obstruction: A systematic review and
meta-analysis of endoscopic and surgical bypass results. Cancer
Treat Rev. 33:213–221. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shepherd HA, Royle G, Ross AP, Diba A,
Arthur M and Colin-Jones D: Endoscopic biliary endoprosthesis in
the palliation of malignant obstruction of the distal common bile
duct: A randomized trial. Br J Surg. 75:1166–1168. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Andersen JR, Sorensen SM, Kruse A,
Rokkjaer M and Matzen P: Randomised trial of endoscopic
endoprosthesis versus operative bypass in malignant obstructive
jaundice. Gut. 30:1132–1135. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kaassis M, Boyer J, Dumas R, Ponchon T,
Coumaros D, Delcenserie R, Canard JM, Fritsch J, Rey JF and Burtin
P: Plastic or metal stents for malignant stricture of the common
bile duct? Results of a randomized prospective study. Gastrointest
Endosc. 57:178–182. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Soderlund C and Linder S: Covered metal
versus plastic stents for malignant common bile duct stenosis: A
prospective, randomized, controlled trial. Gastrointest Endosc.
63:986–995. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hong W, Sun X and Zhu Q: Endoscopic
stenting for malignant hilar biliary obstruction: Should it be
metal or plastic and unilateral or bilateral? Eur J Gastroenterol
Hepatol. 25:1105–1112. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Donelli G, Guaglianone E, Di Rosa R,
Fiocca F and Basoli A: Plastic biliary stent occlusion: factors
involved and possible preventive approaches. Clin Med Res. 5:53–60.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Loew BJ, Howell DA, Sanders MK, Desilets
DJ, Kortan PP, May GR, Shah RJ, Chen YK, Parsons WG, Hawes RH, et
al: Comparative performance of uncoated, self-expanding metal
biliary stents of different designs in 2 diameters: Final results
of an international multicenter, randomized, controlled trial.
Gastrointest Endosc. 70:445–453. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Telford JJ, Carr-Locke DL, Baron TH,
Poneros JM, Bounds BC, Kelsey PB, Schapiro RH, Huang CS,
Lichtenstein DR, Jacobson BC, et al: A randomized trial comparing
uncovered and partially covered self-expandable metal stents in the
palliation of distal malignant biliary obstruction. Gastrointest
Endosc. 72:907–914. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kullman E, Frozanpor F, Söderlund C,
Linder S, Sandström P, Lindhoff-Larsson A, Toth E, Lindell G, Jonas
E, Freedman J, et al: Covered versus uncovered self-expandable
nitinol stents in the palliative treatment of malignant distal
biliary obstruction: results from a randomized, multicenter study.
Gastrointest Endosc. 72:915–923. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rumalla A, Baron TH, Wang KK, Gores GJ,
Stadheim LM and de Groen PC: Endoscopic application of photodynamic
therapy for cholangiocarcinoma. Gastrointest Endosc. 53:500–504.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gao F, Bai Y, Ma SR, Liu F and Li ZS:
Systematic review: Photodynamic therapy for unresectable
cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 17:125–131. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bruix J and Sherman M: Management of
hepatocellular carcinoma: an update. Hepatology. 53:1020–1022.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Siperstein AE, Berber E, Ballem N and
Parikh RT: Survival after radiofrequency ablation of colorectal
liver metastases: 10-Year experience. Ann Surg. 246:559–565. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Itoi T, Isayama H, Sofuni A, Itokawa F,
Tamura M, Watanabe Y, Moriyasu F, Kahaleh M, Habib N, Nagao T, et
al: Evaluation of effects of a novel endoscopically applied
radiofrequency ablation biliary catheter using an ex-vivo pig
liver. J Hepatobiliary Pancreat Sci. 19:543–547. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zacharoulis D, Lazoura O, Sioka E,
Potamianos S, Tzovaras G, Nicholls J, Koukoulis G and Habib N:
Habib EndoHPB: A novel endobiliary radiofrequency ablation device.
An experimental study. J Invest Surg. 26:6–10. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Steel AW, Postgate AJ, Khorsandi S,
Nicholls J, Jiao L, Vlavianos P, Habib N and Westaby D:
Endoscopically applied radiofrequency ablation appears to be safe
in the treatment of malignant biliary obstruction. Gastrointest
Endosc. 73:149–153. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dolak W, Schreiber F, Schwaighofer H,
Gschwantler M, Plieschnegger W, Ziachehabi A, Mayer A, Kramer L,
Kopecky A, Schrutka-Kölbl C, et al: Endoscopic radiofrequency
ablation for malignant biliary obstruction: A nationwide
retrospective study of 84 consecutive applications. Surg Endosc.
28:854–860. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bismuth H, Nakache R and Diamond T:
Management strategies in resection for hilar cholangiocarcinoma.
Ann Surg. 215:31–38. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cho YK, Kim JK, Kim MY, Rhim H and Han JK:
Systemic review of randomized trials for hepatocellular carcinoma
treated with percutaneous ablation therapies. Hepatology.
49:453–459. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Izumi N: Recent advances of radiofrequency
ablation for early hepatocellular carcinoma. J Gastroenterol
Hepatol. 26(Suppl 1): S115–S122. 2011. View Article : Google Scholar
|
|
23
|
Wadsworth CA, Westaby D and Khan SA:
Endoscopic radiofrequency ablation for cholangiocarcinoma. Curr
Opin Gastroenterol. 29:305–311. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tal AO, Vermehren J, Friedrich-Rust M,
Bojunga J, Sarrazin C, Zeuzem S, Trojan J and Albert JG:
Intraductal endoscopic radiofrequency ablation for the treatment of
hilar non-resectable malignant bile duct obstruction. World J
Gastrointest Endosc. 6:13–19. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang F, Li Q, Ge X, Yu H, Nie J and Miao
L: Choledochoscopic radiofrequency ablation for congenital
choledochal cysts. Endoscopy. 46(Suppl 1): E373–E374.
2014.PubMed/NCBI
|