Introduction
Antiphospholipid syndrome (APS) is a systemic
autoimmune disorder characterized by recurrent arterial and/or
venous thrombosis, pregnancy morbidity, thrombocytopenia and
elevated titers of antiphospholipid antibodies (aPLs) (1,2). aPLs
are a family of antibodies that target phospholipid-binding
proteins, including lupus anticoagulant, anticardiolipin (aCL), and
anti-β2-glycoprotein I (anti-β2-GPI) (2,3). APS
displays a variety of clinical manifestations which are recognized
to be associated with the presence of these aPLs (4). Thrombus formation is a key event in APS
(5). A number of studies have been
conducted in order to identify the pathogenesis of aPL-induced
thrombosis, and it has been suggested that activation of monocytes
(6,7), platelets (6) and endothelial cells (8) contribute to the thrombotic process. In
particular, the procoagulant activity of monocytes and deregulated
tissue factor expression were associated with an increased risk of
thrombus formation. Although the association between monocytes and
the development of thrombotic complications in patients with APS
has been well-established (9), the
mechanisms underlying aPL action on monocytes remain largely
unknown.
Pregnant women with APS are at a high risk of
complications, mainly due to thromboses in the placenta which can
lead to miscarriages (10,11). The treatment of women with APS is
predominantly focused on the prevention of further thrombotic
events and subsequent fetal loss (12). Treatment strategies for patients with
APS are individualized and usually require lifelong anticoagulant
therapy (13,14). In the case of catastrophic aPL
syndrome, a rare life-threatening variant of APS, plasma exchange
(PE) therapy is recommended (15).
Therapeutic PE (also termed plasmapheresis) is an extracorporeal
blood purification method. The procedure includes the separation of
blood plasma from cellular components, which are subsequently
returned back to the patient, and the plasma is discarded and
replaced with substitution fluids. This approach allows active and
rapid body cleaning, and the elimination of circulating pathogenic
autoantibodies, immunoglobulins, immune complexes and toxins
(16–18).
Automated plasmapheresis is routinely used to
collect plasma products from healthy donors for medical use
(19,20). The results of previous studies have
suggested that PE in combination with conventional methods of
treatment appears to be effective in managing a range of
neurological, hematological, renal, metabolic, infectious and
immunological diseases (21,22). Furthermore, a previous study
demonstrated that PE is able to prevent the deleterious effects on
both mother and fetus in women with familial hyperlipidemia or
systemic lupus erythematosus (23).
Limited data is available regarding the effect of PE on pregnancy
outcome in patients with APS. However, a number of clinical studies
have reported promising results of PE therapy in APS, and
recommended PE as an additional treatment modality during high-risk
pregnancies in women with APS (24,25). The
use of PE was effective in reducing the titers of aCL and
anti-β2-GPI antibodies (24,25). Regardless of these data, few studies
have investigated the immunological changes that occur following PE
in APS. In consideration of the previous results indicating the
involvement of monocytes in thrombotic complications, the present
study aimed to investigate the effects of PE on the phenotype of
peripheral blood monocytes in women with APS who experienced
recurrent pregnancy losses. Monocyte transcriptional activity and
the mRNA expression levels of 11 genes associated with signaling
and recognition processes occurring in immune and inflammatory
responses were investigated.
Materials and methods
Patients
A total of 11 women suffering from recurrent
miscarriages and APS (mean age, 30±5.6 years) were selected for the
present study. The patients with APS were classified at the
Institute of Perinatology, Obstetrics and Gynecology according to
the following criteria: i) Women with a history of two unexplained
consecutive spontaneous pregnancy losses in the first and second
trimesters; and ii) the presence of anti-cardiolipin antibodies
[(immunoglobulin (Ig)G≥40 GPL units] and/or anti-β2-GPI (IgG≥40 GPL
units) and/or positive lupus anticoagulant antibodies present in
the plasma at two separate time points. None of the women met the
criteria for systemic lupus erythematosus or any concomitant
systemic autoimmune disease. Blood samples were obtained from the
patients prior to and following PE therapy. All patients received a
course of four PE sessions on alternating days. The first blood
sampling was performed immediately prior to the PE procedure. The
second blood sampling was obtained on day 3 following the last PE
session. Nine healthy women (mean age, 29±8.5 years) without a
positive family history of APS, autoimmune diseases, or thrombosis
were enrolled as a control group at the Viola Blood Center and
Diagnostics (Yerevan, Armenia). All participants of the present
study provided written-informed consent. The present study was
approved by the ethics committee of the Institute of Molecular
Biology of the NAS RA (approval no. IRB IORG0003427).
PE therapy
The principle of plasmapheresis is to remove the
plasma with retransfusion of the blood cells. The procedure is
multistage and involves taking blood from a patient and placing it
into a disposable plastic bag blood collection system (Baxter
International, Inc., Deerfield, IL, USA). Separation of the plasma
from the blood cells was achieved by centrifugation at 1,900 × g
for 15 min at room temperature. The plasma was discarded and
replaced with Ringer's solution (Liqvor Pharmaceuticals, Yerevan,
Armenia), and the blood is then returned.
Preparation of peripheral/circulating
monocytes
Fresh venous blood samples were obtained and placed
into Vacutainer tubes containing ethylenediaminetetraacetic acid
(BD Biosciences, Franklin Lakes, NJ, USA). Peripheral blood
mononuclear cells (PBMCs) were isolated using Ficoll density
gradient centrifugation (Life Science Sweden, Sweden) according to
the manufacturer's protocol. Monocytes were isolated from
peripheral blood mononuclear cell (PBMCs) by plastic adherence.
Briefly, 1×106 PBMCs were distributed in plastic tissue
culture flasks (Corning, Inc., Oneonta, NY, USA) and allowed to
adhere at 37°C for 2 h in 2 ml RPMI-1640 medium (Corning, Inc.)
supplemented with 10% fetal bovine serum (Sigma-Aldrich, St. Louis,
MO, USA). Non-adherent cells were removed and the adherent cells
were washed twice with phosphate-buffered saline. Following washing
>75% of the cells were identified as monocytes, as assessed by
FACS Calibur flow cytometry (BD Biosciences) using
R-phycoerythrin-conjugated anti-human CD14 IgG antibody (BioLegend,
Inc., London, UK) (26).
Monocytes (1×106 cells/ml) were cultured
separately at 37°C for 4 h in RPMI-1640 medium supplemented with
10% fetal bovine serum, 2 mmol/l glutamine (Sigma-Aldrich), 5 mM
HEPES (Sigma-Aldrich) in the absence or presence of 10 ng/ml
lipopolysaccharide (LPS; Escherichia coli 026:B6;
Sigma-Aldrich) or 10 ng/ml LPS + 100 µM ATPγ-S (Sigma-Aldrich) in a
total volume of 1 ml. Following the culture period or directly
following isolation, the cells were washed once with cold PBS, and
stored in 150 µl RNAlater (Qiagen GmbH, Hilden, Germany) at −20°C
until use.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was isolated from monocytes samples using
a High Pure miRNA Isolation kit (cat. no. 05080576001; Roche
Diagnostics, Basel, Switzerland) according to the manufacturer's
protocol. All samples were treated with RNaseOUT Recombinant
Ribonuclease Inhibitor (Invitrogen; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) and maintained at −80°C. cDNA synthesis and qPCR
were performed as described previously (27). Briefly, cDNA synthesis was performed
using a Transcriptor First Stand cDNA Synthesis kit (Roche
Diagnostics) and stored at −20°C prior to further use. RT-qPCR was
performed using gene-specific primers designed using the Universal
Probe Library system (Roche Diagnostics). The primer and probes
sequences for the investigated genes (11 in total) are listed in
Table I. Each qPCR reaction was
performed in triplicate along with a negative template control (no
cDNA), negative RT control (no reverse transcription) and positive
template control (calibrator) in quadruplicates. Target gene
expression levels were normalized against the reference gene
ribosomal protein L32. Human universal reference RNA (Agilent
Technologies, Inc., Santa Clara, CA, USA) was used as a calibrator
at the concentration of 1.25 ng RNA/reaction in quadruplicates.
qPCR reactions were performed using the Rotor-Gene 3000 system
(Corbett Research, Mortlake, Australia) in a 0.1 ml strip tubes
(Qiagen GmbH) and a final volume of 20 µl. The relative expression
levels were calculated using the second derivative method
(Rotor-Gene software 6.1.71; Corbett Research) (28).
 | Table I.Description of investigated genes and
primers used in the present study. |
Table I.
Description of investigated genes and
primers used in the present study.
| Gene | Gene name (full) | GenBank accession
no.a | Sense and antisense
primers | LNA
probeb |
|---|
| RPL32c | Ribosomal protein
L32 | NM_000994.3 | F,
5′-GAAGTTCCTGGTCCACAACG-3′ | #17 |
|
|
|
| R,
5′-GCGATCTCGGCACAGTAAG-3′ |
|
| IL-1βc | Interleukin 1β | NM_000576.2 | F,
5′-TACCTGTCCTGCGTGTTGAA-3′ | #78 |
|
|
|
| R,
5′-TCTTTGGGTAATTTTTGGGATCT-3′ |
|
| NLRP3c | Nod-like receptor
family, pyrin domain containing 3 | NM_001243133.1 | F,
5′-TGTCCTCCCAAGCTCCTCT-3′ | #27 |
|
|
|
| R,
5′-AAGCAGCACTCATGCGAGA-3′ |
|
| CCL2c | Chemokine (C-C
motif) ligand 2 | NM_002982.3 | F,
5′-AGTCTCTGCCGCCCTTCT-3′ | #40 |
|
|
|
| R,
5′-GTGACTGGGGCATTGATTG-3′ |
|
| TNF-α c | Tumor necrosis
factor-α | NM_000594.3 | F,
5′-CAGCCTCTTCTCCTTCCT-3′ | #29 |
|
|
|
| R,
5′-GCCAGAGGGCTGATTAGA-3′ |
|
| TLR2d | Toll-like receptor
2 | NM_003264.3 | F,
5′-CGTTCTCTCAGGTGACTG-3′ | #14 |
|
|
|
| R,
5′-CCTTTGGATCCTGCTTGC-3′ |
|
| IL-6d | Interleukin 6 | NM_000600.3 | F,
5′-GAAGCTCTATCTCGCCTCCA-3′ | #7 |
|
|
|
| R,
5′-AGCAGGCAACACCAGGAG-3′ |
|
| CXCL10d | Chemokine (C-X-C
motif) ligand 10 | NM_001565.3 | F,
5′-GAAAGCAGTTAGCAAGGAAA-3′ | #34 |
|
|
|
| R,
5′-GACATATACTCCATGTAGGGA-3′ |
|
| STAT3d | Signal transducer
and activator of transcription 3 | NM_003150.3 | F,
5′-TGATGCAGTTTGGAAATAATG-3′ | #18 |
|
|
|
| R,
5′-CATGTCAAAGGTGAGGGACTC-3′ |
|
| IL-23Ad | Interleukin 23,
α-subunit p19 | NM_016584.2 | F,
5′-GTTCCCCATATCCAGTGTGG-3′ | #76 |
|
|
|
| R,
5′-TCCTTTGCAAGCAGAACTGA-3′ |
|
| P2X7e | Purinergic receptor
P2X, ligand-gated ion channel 7 | NM_002562.5 | Hs00175721_m1 |
|
| NF-κB
p65e | Homo sapiens v-rel
avian reticuloendotheliosis viral oncogene homolog A | NM_003998.3 | Hs00153294_m1 |
|
Statistical analysis
The data are presented as means ± standard
deviation. Statistical analyses were performed using Graph Pad
Prism software, version 5.01 (GraphPad Software, Inc., La Jolla,
CA, USA). Data were compared using paired (prior to PE therapy, vs.
following PE therapy) and unpaired (all other comparisons)
Student's t-tests or a Mann-Whitney U test if the data were not
normally distributed. A P-value of <0.05 was considered to
indicate a statistically significant difference.
Results
Subheading
Evidence for the clinical benefit of PE has been
limited and the majority of previous investigations into the
effectiveness of PE have been conducted in high-risk pregnancies in
women with APS (24,25). However, it is not known whether the
procedure itself affects the functional activity of monocytes.
Therefore, the present study aimed to investigate the impact of PE
therapy on gene expression by measuring the mRNA expression levels
in the monocytes from women with APS who had experienced more than
two pregnancy losses.
Baseline gene expression
Initially, the baseline gene expression levels of 11
genes [interleukin (IL)-1β, IL-6, IL-23, tumor necrosis factor-α
(TNF-α), chemokine (C-C motif) ligand 2 (CCL2), C-X-C motif
chemokine 10 (CXCL10), Nod-like receptor family, pyrin domain
containing 3, purinergic receptor P2X, ligand-gated ion channel 7
(P2X7), nuclear factor (NF)-κB, toll-like receptor 2 (TLR2) and
signal transducer and activator of transcription 3 (STAT3)] in
monocytes were quantified in women with APS and compared with the
expression levels in monocytes from healthy control women (Fig. 1). The results demonstrated that the
baseline mRNA expression of six investigated genes were
downregulated [healthy controls, vs. individuals with APS prior to
PE (mean ± standard deviation)], specifically IL-1β; (1.690±0.467,
vs. 0.394±0.09, P=0.002), IL-6 (0.107±0.046, vs. 0.004±0.001,
P=0.023), IL-23 (0.125±0.068, vs. 0.017±0.005, P=0.039), CCL2
(0.076±0.027, vs. 0.010±0.002, P=0.028), TLR2 (0.107±0.033, vs.
0.029±0.016, P=0.045) and STAT3 (0.164±0.072, vs. 0.027±0.017,
P=0.021); and one gene, CXCL10, was upregulated (0.203±0.161, vs.
0.960±0.258, P=0.03). P2X7, NF-κB and TNF-α mRNA expression did not
differ between APS and healthy control monocytes (Fig. 1).
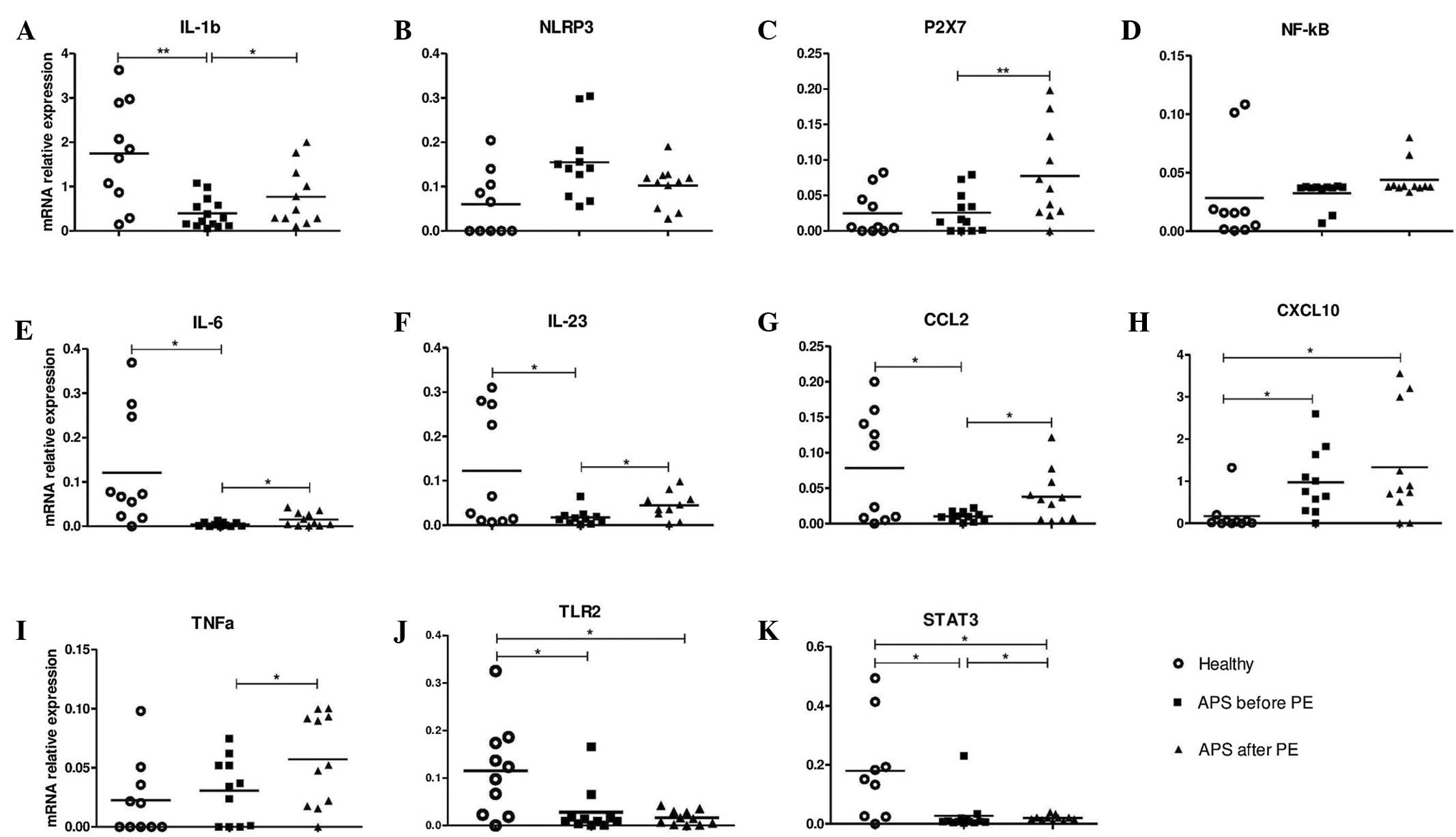 | Figure 1.Relative mRNA expression levels of
candidate genes in monocytes isolated from healthy subjects,
patients with APS prior to PE therapy, and patients with APS on the
third day following PE therapy. The mRNA expression levels of (A)
IL-1b, (B) NLRP3, (C) P2X7, (D) NF-κB, (E) IL-6, (F) IL-23, (G)
CCL2, (H) CXCL10, (I) TNF-α, (J) TLR2 and (K) STAT3 were normalized
against those of RPL32. Horizontal bars represent the mean values
for each group. *P<0.05 and **P<0.01. APS, antiphospholipid
syndrome; PE, plasma exchange; RPL32, Ribosomal protein L32; IL,
interleukin; NLRP3, Nod-like receptor family, pyrin domain
containing 3; P2X7, purinergic receptor P2X, ligand-gated ion
channel 7; NF-κB, nuclear factor-κB; CCL2, chemokine (C-C motif)
ligand 2; CXCL10, chemokine (C-X-C motif) ligand 10; TNF-α, tumor
necrosis factor-α; TLR2, toll-like receptor 2; STAT3, signal
transducer and activator of transcription 3. |
Effect of PE therapy on gene
expression
The expression levels of all investigated genes
prior to and following PE therapy were compared. A number of the
investigated genes were upregulated following plasmapheresis. As
shown in Fig. 1, the mRNA expression
levels (prior to PE, vs. following PE therapy) of IL-1β
(0.394±0.09, vs. 0.718±0.213, P=0.038), IL-6 (0.004±0.001, vs.
0.017±0.005, P=0.025), IL-23 (0.017±0.006, vs. 0.037±0.014,
P=0.047), CCL2 (0.010±0.002, vs. 0.047±0.016; P=0.030), P2X7
(0.026±0.008, vs. 0.081±0.026, P=0.029) and TNF-α (0.027±0.008, vs.
0.055±0.016, P=0.015) were increased in APS monocytes following PE
therapy. STAT3 mRNA expression levels (0.027±0.017, vs.
0.022±0.003, P=0.049) were slightly decreased following PE
therapy.
Expression levels
The expression levels (healthy controls, vs. APS
following PE therapy) of TLR2 (0.107±0.034, vs. 0.015±0.005,
P=0.015) and STAT3 (0.164±0.072, vs. 0.022±0.003, P=0.040) were
significantly higher in the healthy monocytes compared with the APS
monocytes following PE therapy. Furthermore, the mRNA expression
levels of CXCL10 were significantly higher in APS cells
(0.204±0.161, vs. 1.266±0.467, P=0.049).
Discussion
There has been considerable evidence that thrombosis
is a common complication of APS, and monocytes are the primary
cellular elements which are involved in the hypercoagulation
observed in this pathology (29).
Furthermore, the proinflammatory state of monocytes was
demonstrated to be associated with predisposition to thrombosis
(30). The present study examined
whether monocytes from patients with APS displayed an altered or
proinflammatory state. Contrary to expectations, low baseline
expression levels of IL-1β, IL-6, IL-23, CCL2, TLR2 and STAT3 were
observed, and only the expression of CXCL10 was upregulated in APS
monocytes. Upregulated CXCL10 expression combined with
downregulated CCL2 baseline expression in APS monocytes is of
particular interest as these two chemokines possess unique roles in
autoimmune diseases (31). CCL2 and
CXCL10 regulate immune responses via the activation and recruitment
of immune cells (32). CCL2/monocyte
chemoattractant protein-1 is a crucial factor for the development
of adaptive T helper cells (Th)2 responses, whereas CXCL10 is a
strong chemoattractant for activated Th1 lymphocytes (33), and a reliable marker of Th1-mediated
autoimmune diseases (34). The
results of the present study demonstrated that a
CCL2-low/CXCL10-high transcriptional profile of circulating APS
monocytes, which may suggest a shift towards Th1 immune response in
APS. A reduction in Th1 cellular immune response and a
corresponding increase in Th2 cells are essential for successful
pregnancy (35). An opposite
tendency was present in women with recurrent miscarriages (36,37). In
addition to a Th1-biased response in women with recurrent
miscarriages, altered PBMC activity, prior to and following
pregnancy loss, was also described (37). Transcriptional patterns of PBMCs that
could account for the pathogenesis of primary APS have been studied
by Bernales et al (38).
Despite a wide diversity in the range of expression profiles, the
authors of the study stated that altered cellular phenotype was
specific for APS (38). The reason
for the low functional activity of APS monocytes is unknown.
Further investigations are required in order to clarify whether the
downregulation of gene expression in monocytes is due to an
APS-associated, inherent feature. There is a possibility that this
transcriptional profile of monocytes is associated with marked
hormonal and immunological changes during pregnancy in addition to
the biological changes that occur during miscarriage (39).
Notably, the present study indicated the
immunomodulatory effect of plasmapheresis on APS monocytes. PE
therapy resulted in increased mRNA expression levels in six of the
11 investigated genes, namely IL-1β, IL-6 IL-23, TNF-α, CCL2 and
P2X7. Despite the fact that PE therapy increased the expression
levels of a wide range of mediators, their expression levels did
not exceed those in the cells from healthy subjects, and increased
from low baseline expression levels to normal, physiological
ranges. Although the exact mechanisms underlying this effect have
yet to be elucidated, they likely involve multiple processes. In
addition to the removal of IGs, increasing evidence suggests an
immunomodulatory effect of PE. These include Th1/Th2 balance with a
shift to Th2 cell response (40), as
well as the suppression of IL-2 and interferon-γ production
(41). Notably, PE therapy in the
present study significantly increased CCL2 expression, whereas
CXCL10 mRNA expression levels were not affected by the treatment
procedure. The observed effect may be viewed as beneficial for
Th1/Th2 balance restoration in APS patients following pregnancy
loss.
It is well known that ex vivo manipulation of
immune cells may induce their activation, and it is difficult to
ensure that in vitro conditions will be the same as
physiological conditions. In particular, the cell isolation
procedure may affect gene expression and protein secretion by
monocytes (42,43). In lung epithelial cells,
centrifugation affects proliferation and expression of certain
cytokines such as IL-1β (44). PE
therapy involves a number of steps: Separation of blood components
by centrifugation, erythrocytes washing and restoration of blood
volume with replacement fluids. It may be hypothesized that ex
vivo manipulation of blood cells during PE may have temporary
and reversible effects on non-selective activation of blood cells.
The half-life of circulating monocytes was estimated to be ≥3 days
(45), and PE-activated circulating
monocytes will be replenished with bone marrow-derived cells. At
present, it is difficult to determine whether the observed changes
in the expression levels of the candidate genes are associated with
the unwanted side-effect of experimental manipulations or whether
they indicate the positive effect of PE on functional recovery in
APS monocytes. This limitation should be addressed in future
studies of the impact of PE treatment on the immune system.
The therapeutic rationale for PE during APS has yet
to be fully elucidated. Although PE is not routinely used in APS
management, this treatment modality serves as a promising tool for
novel and optimized treatment strategies. The present preliminary
study demonstrated the immunomodulatory effect of plasmapheresis on
monocytes obtained from APS patients. In a more general context,
these observations should be considered with caution, as the
activating effect of PE on immune cells may have dual consequences
including adverse ones. In the case of APS, normalizing monocyte
activity following PE is likely to be beneficial; however,
treatment plans for PE therapy in other pathological conditions
should be examined on a case-by-case basis with consideration of
possible unwanted side-effects.
Acknowledgements
The authors of the present study are grateful to Dr
Armen Blbulyan (Institute of Perinatology, Obstetrics and
Gynecology, Yerevan, Armenia) for assistance with blood sampling.
The present study was supported by a grant from the Czech
government (grant no. CZ.1.07/2.3.00/30.00041; visegrad fund no.
51300184) and the State Committee Science MES RA (research project
no. SCS13-1F236).
References
|
1
|
de Jesus GR, Agmon-Levin N, Andrade CA,
Andreoli L, Chighizola CB, Porter TF, Salmon J, Silver RM, Tincani
A and Branch DW: 14th International congress on antiphospholipid
antibodies task force report on obstetric antiphospholipid
syndrome. Autoimmun Rev. 13:795–813. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Miyakis S, Lockshin MD, Atsumi T, Branch
DW, Brey RL, Cervera R, Derksen RH, DE Groot PG, Koike T, Meroni
PL, et al: International consensus statement on an update of the
classification criteria for definite antiphospholipid syndrome
(APS). J Thromb Haemost. 4:295–306. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gris JC and Bouvier S: Antiphospholipid
syndrome: Looking for a refocusing. Thromb Res 131 Suppl.
1:S28–S31. 2013. View Article : Google Scholar
|
|
4
|
Galli M, Borrelli G, Jacobsen EM, Marfisi
RM, Finazzi G, Marchioli R, Wisloff F, Marziali S, Morboeuf O and
Barbui T: Clinical significance of different antiphospholipid
antibodies in the WAPS (warfarin in the antiphospholipid syndrome)
study. Blood. 110:1178–1183. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Khamashta MA and Asherson RA: Hughes
syndrome: Antiphospholipid antibodies move closer to thrombosis in
1994. Br J Rheumatol. 34:493–494. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Reverter JC, Tàssies D, Font J, Khamashta
MA, Ichikawa K, Cervera R, Escolar G, Hughes GR, Ingelmo M and
Ordinas A: Effects of human monoclonal anticardiolipin antibodies
on platelet function and on tissue factor expression on monocytes.
Arthritis Rheum. 41:1420–1427. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kornberg A, Blank M, Kaufman S and
Shoenfeld Y: Induction of tissue factor-like activity in monocytes
by anti-cardiolipin antibodies. J Immunol. 153:1328–1332.
1994.PubMed/NCBI
|
|
8
|
Branch DW and Rodgers GM: Induction of
endothelial cell tissue factor activity by sera from patients with
antiphospholipid syndrome: A possible mechanism of thrombosis. Am J
Obstet Gynecol. 168:206–210. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
López-Pedrera C, Cuadrado MJ, Herández V,
Buendïa P, Aguirre MA, Barbarroja N, Torres LA, Villalba JM,
Velasco F and Khamashta M: Proteomic analysis in monocytes of
antiphospholipid syndrome patients: Deregulation of proteins
related to the development of thrombosis. Arthritis Rheum.
58:2835–2844. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ruiz-Irastorza G and Khamashta MA:
Management of thrombosis in antiphospholipid syndrome and systemic
lupus erythematosus in pregnancy. Ann N Y Acad Sci. 1051:606–612.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Di Prima FA, Valenti O, Hyseni E, Giorgio
E, Faraci M, Renda E, De Domenico R and Monte S: Antiphospholipid
syndrome during pregnancy: The state of the art. J Prenat Med.
5:41–53. 2011.PubMed/NCBI
|
|
12
|
Meroni PL, Moia M, Derksen RH, Tincani A,
McIntyre JA, Arnout JM, Koike T, Piette JC, Khamashta MA and
Shoenfeld Y: Venous thromboembolism in the antiphospholipid
syndrome: Management guidelines for secondary prophylaxis. Lupus.
12:504–507. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Erkan D, Ortel TL and Lockshin MD:
Warfarin in antiphospholipid syndrome-time to explore new horizons.
J Rheumatol. 32:208–212. 2005.PubMed/NCBI
|
|
14
|
Tincani A, Branch W, Levy RA, Piette JC,
Carp H, Rai RS, Khamashta M and Shoenfeld Y: Treatment of pregnant
patients with antiphospholipid syndrome. Lupus. 12:524–529. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Marson P, Bagatella P, Bortolati M, Tison
T, De Silvestro G, Fabris F, Pengo V and Ruffatti A: Plasma
exchange for the management of the catastrophic antiphospholipid
syndrome: Importance of the type of fluid replacement. J Intern
Med. 264:201–203. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ibrahim RB and Balogun RA: Medications in
patients treated with therapeutic plasma exchange: Prescription
dosage, timing and drug overdose. Semin Dial. 25:176–189. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ibrahim RB and Balogun RA: Medications and
therapeutic apheresis procedures: Are we doing our best? J Clin
Apher. 28:73–77. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kaplan AA: Therapeutic apheresis for renal
disorders. Therap Apher. 3:25–30. 1999. View Article : Google Scholar
|
|
19
|
Bove LL, Bednall T, Masser B and Buzza M:
Understanding the plasmapheresis donor in a voluntary,
nonremunerated environment. Transfusion. 51:2411–2424. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Crocco I, Franchini M, Garozzo G, Gandini
AR, Gandini G, Bonomo P and Aprili G: Adverse reactions in blood
and apheresis donors: Experience from two Italian transfusion
centres. Blood Transfus. 7:35–38. 2009.PubMed/NCBI
|
|
21
|
Nguyen TC, Kiss JE, Goldman JR and
Carcillo JA: The role of plasmapheresis in critical illness. Crit
Care Clin. 28:453–468. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nakanishi T, Suzuki N, Kuragano T,
Nagasawa Y and Hasuike Y: Current topics in therapeutic
plasmapheresis. Clin Exp Nephrol. 18:41–49. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dittrich E, Schmaldienst S, Langer M,
Jansen M, Hörl WH and Derfler K: Immunoadsorption and plasma
exchange in pregnancy. Kidney Blood Press Res. 25:232–239. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bortolati M, Marson P, Chiarelli S, Tison
T, Facchinetti M, Gervasi MT, De Silvestro G and Ruffatti A: Case
reports of the use of immunoadsorption or plasma exchange in
high-risk pregnancies of women with antiphospholipid syndrome. Ther
Apher Dial. 13:157–160. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bontadi A, Ruffatti A, Marson P, Tison T,
Tonello M, Hoxha A, De Silvestro G and Punzi L: Plasma exchange and
immunoadsorption effectively remove antiphospholipid antibodies in
pregnant patients with antiphospholipid syndrome. J Clin Apher.
27:200–204. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ferrer DG, Jaldín-Fincati JR, Amigone JL,
Capra RH, Collino CJ, Albertini RA and Chiabrando GA: Standardized
flow cytometry assay for identification of human monocytic
heterogeneity and LRP1 expression in monocyte subpopulations:
Decreased expression of this receptor in nonclassical monocytes.
Cytometry A. 85:601–610. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Manukyan G, Petrek M, Kriegova E,
Ghazaryan K, Fillerova R, Fillerova R and Boyajyan A: Activated
phenotype of circulating neutrophils in familial mediterranean
fever. Immunobiology. 218:892–898. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pfaffl MW: A new mathematical model for
relative quantification in real-time RT-PCR. Nucleic Acids Res.
29:e452001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cuadrado MJ, López-Pedrera C, Khamashta
MA, Camps MT, Tinahones F, Torres A, Hughes GR and Velasco F:
Thrombosis in primary antiphospholipid syndrome: A pivotal role for
monocyte tissue factor expression. Arthritis Rheum. 40:834–841.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wakefield TW, Strieter RM, Wilke CA,
Kadell AM, Wrobleski SK, Burdick MD, Schmidt R, Kunkel SL and
Greenfield LJ: Venous thrombosis-associated inflammation and
attenuation with neutralizing antibodies to cytokines and adhesion
molecules. Arterioscler Thromb Vasc Biol. 15:258–268. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Godessart N and Kunkel SL: Chemokines in
autoimmune disease. Curr Opin Immunol. 13:670–675. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Oghumu S, Lezama-Dávila CM, Isaac-Márquez
AP and Satoskar AR: Role of chemokines in regulation of immunity
against leishmaniasis. Exp Parasitol. 126:389–396. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gu D, Chen Z, Zhao H, Du W, Xue F, Ge J,
Sui T, Wu H, Liu B, Lu S, Zhang L and Yang R: Th1 (CXCL10) and Th2
(CCL2) chemokine expression in patients with immune
thrombocytopenia. Hum Immunol. 71:586–591. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rotondi M, Rosati A, Buonamano A, Lasagni
L, Lazzeri E, Pradella F, Fossombroni V, Cirami C, Liotta F, La
Villa G, et al: High pretransplant serum levels of CXCL10/IP-10 are
related to increased risk of renal allograft failure. Am J
Transplant. 4:1466–1474. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wegmann TG, Lin H, Guilbert L and Mosmann
TR: Bidirectional cytokine interactions in the maternal-fetal
relationship: Is successful pregnancy a TH2 phenomenon? Immunology
Today. 14:353–356. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lissauer D, Goodyear O, Khanum R, Moss PA
and Kilby MD: Profile of maternal CD4 T-cell effector function
during normal pregnancy and in women with a history of recurrent
miscarriage. Clin Sci (Lond). 126:347–354. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Raghupathy R, Makhseed M, Azizieh F, Omu
A, Gupta M and Farhat R: Cytokine production by maternal
lymphocytes during normal human pregnancy and in unexplained
recurrent spontaneous abortion. Hum Reprod. 15:713–778. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bernales I, Fullaondo A, Marín-Vidalled
MJ, Ucar E, Martínez-Taboada V, López-Hoyos M and Zubiaga AM:
Innate immune response gene expression profiles characterize
primary antiphospholipid syndrome. Genes Immun. 9:38–46. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Laird SM, Tuckerman EM, Cork BA, Linjawi
S, Blakemore AI and Li TC: A review of immune cells and molecules
in women with recurrent miscarriage. Hum Reprod Update. 9:163–174.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Goto H, Matsuo H, Nakane S, Izumoto H,
Fukudome T, Kambara C and Shibuya N: Plasmapheresis affects T
helper type-1/T helper type-2 balance of circulating peripheral
lymphocytes. Ther Apher. 5:494–496. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shariatmadar S, Nassiri M and Vincek V:
Effect of plasma exchange on cytokines measured by multianalyte
bead array in thrombotic thrombocytopenic purpura. Am J Hematol.
79:83–88. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Haskill S, Johnson C, Eierman D, Becker S
and Warren K: Adherence induces selective mRNA expression of
monocyte mediators and proto-oncogenes. J Immunol. 140:1690–1694.
1988.PubMed/NCBI
|
|
43
|
Fuhlbrigge RC, Chaplin DD, Kiely JM and
Unanue ER: Regulation of interleukin 1 gene expression by adherence
and lipopolysaccharide. J Immunol. 138:3799–3802. 1987.PubMed/NCBI
|
|
44
|
Yang J, Hooper WC, Phillips DJ, Tondella
ML and Talkington DF: Centrifugation of human lung epithelial
carcinoma a549 cells up-regulates interleukin-1beta gene
expression. Clin Diagn Lab Immunol. 9:1142–1143. 2002.PubMed/NCBI
|
|
45
|
Yona S and Jung S: Monocytes: subsets,
origins, fates and functions. Curr Opin Hematol. 17:53–59. 2010.
View Article : Google Scholar : PubMed/NCBI
|















