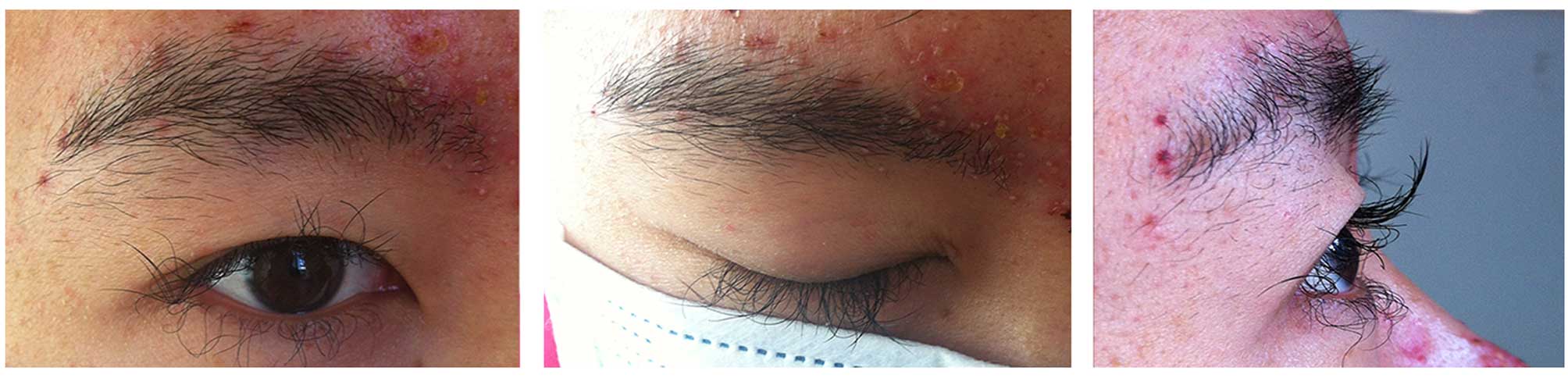
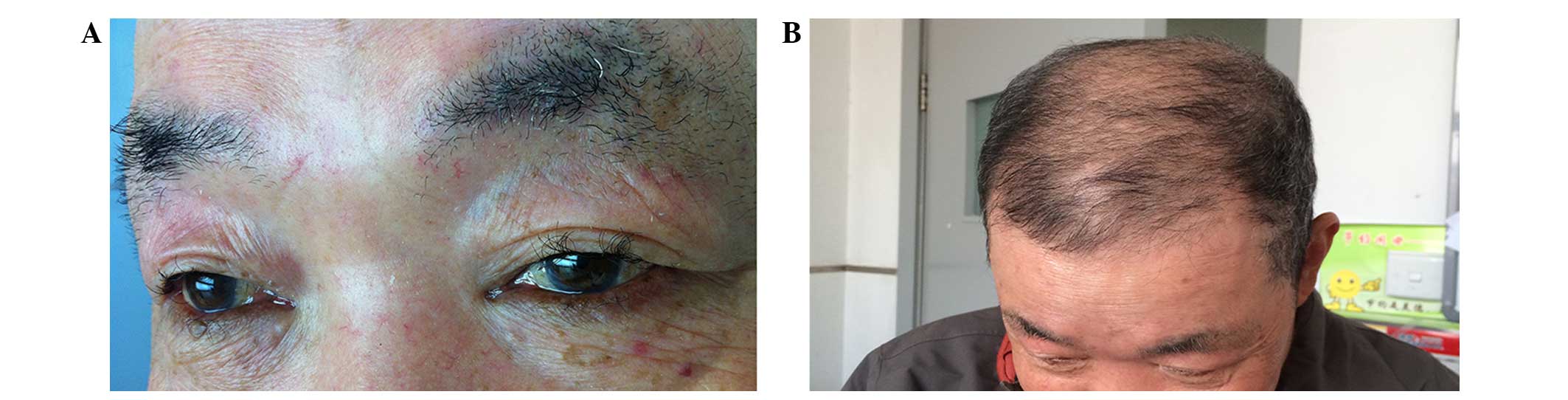
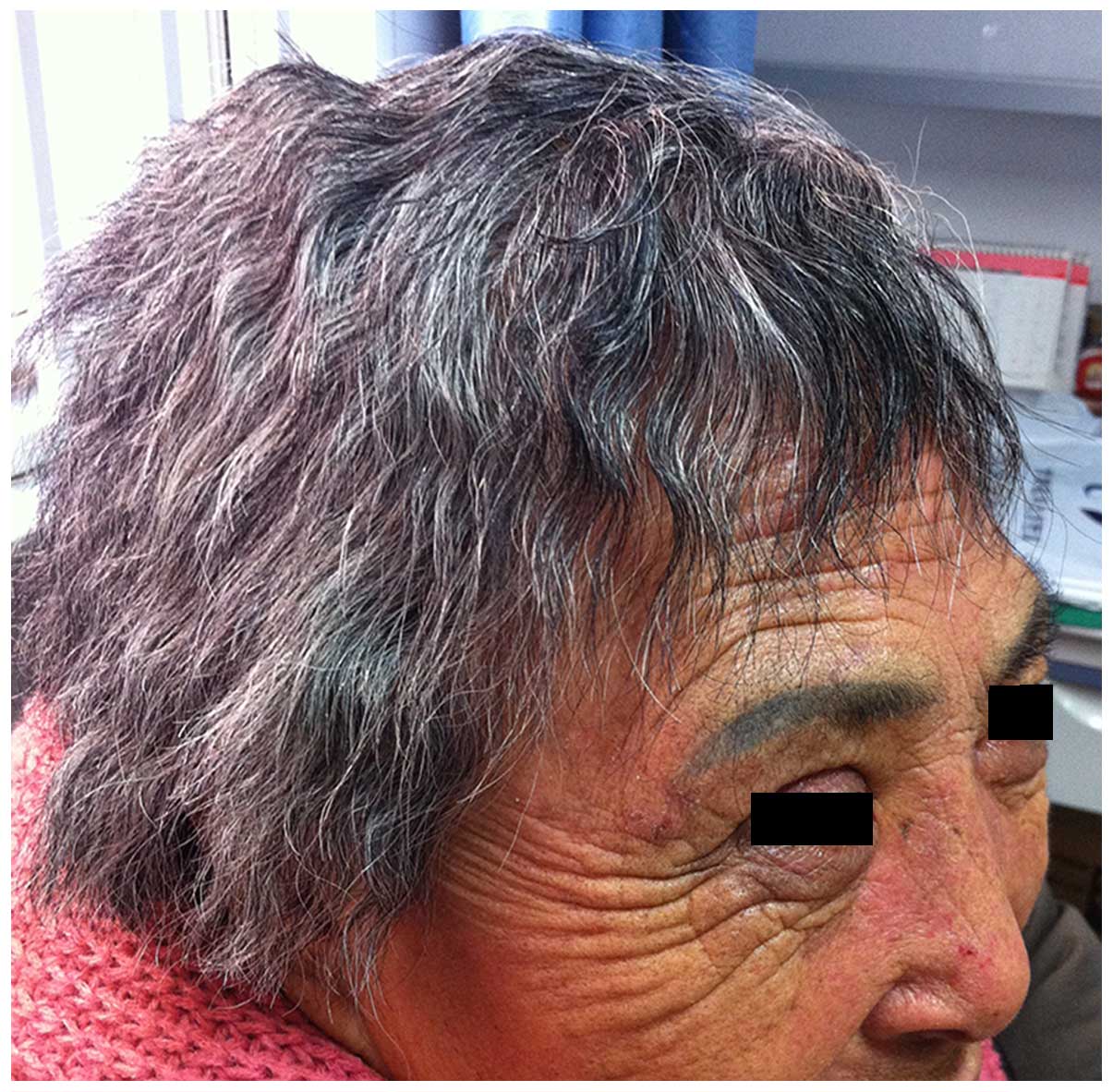
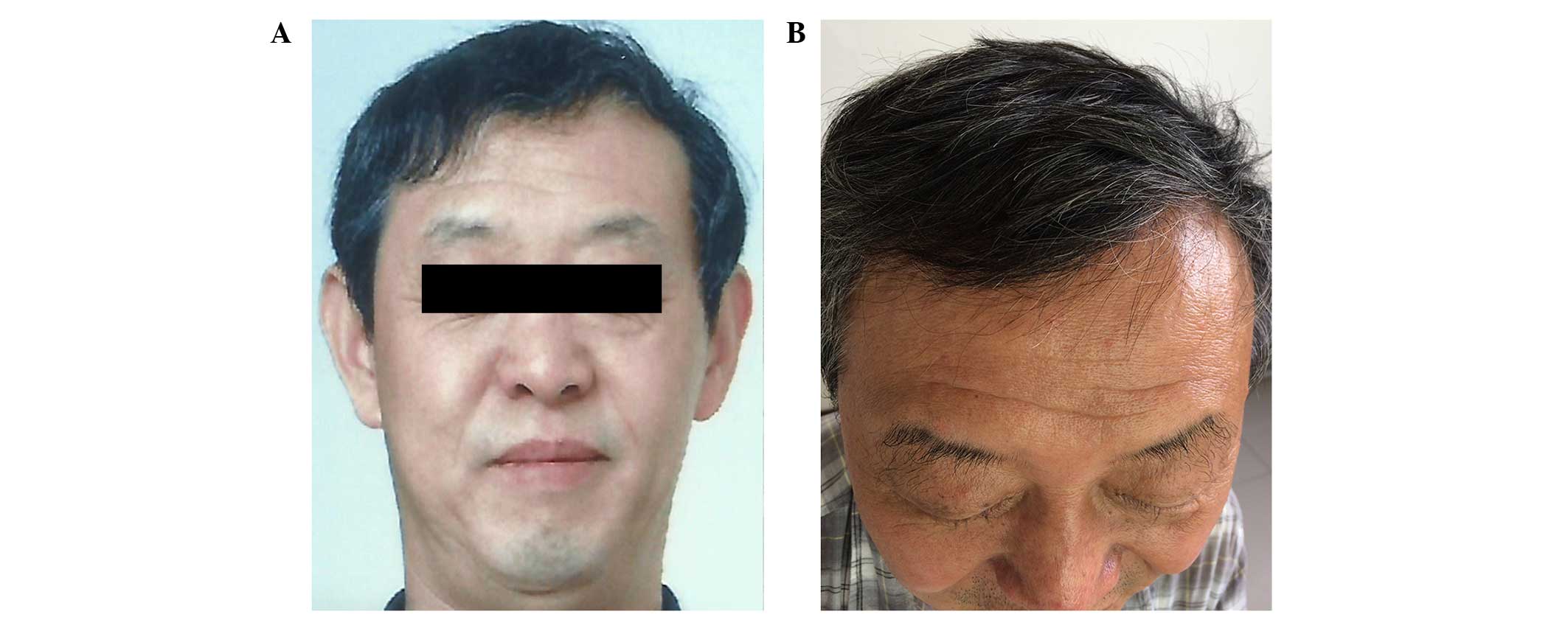
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ramalingam S and Belani C: Systemic
chemotherapy for advanced non-small cell lung cancer: Recent
advances and future directions. Oncologist. 13(Suppl 1): 5–13.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Blackhall FH, Shepherd FA and Albain KS:
Improving survival and reducing toxicity with chemotherapy in
advanced non-small cell lung cancer: A realistic goal? Treat Respir
Med. 4:71–84. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mok TS, Wu YL, Thongprasert S, Yang CH,
Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, et
al: Gefitinib or carboplatin-paclitaxel in pulmonary
adenocarcinoma. N Engl J Med. 361:947–957. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Han JY, Park K, Kim SW, Lee DH, Kim HY,
Kim HT, Ahn MJ, Yun T, Ahn JS, Suh C, et al: First-SIGNAL:
First-line single-agent iressa versus gemcitabine and cisplatin
trial in never-smokers with adenocarcinoma of the lung. J Clin
Oncol. 30:1122–1128. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu YL, Zhou C, Liam CK, Wu G, Liu X, Zhong
Z, Lu S, Cheng Y, Han B, Chen L, et al: First-line erlotinib versus
gemcitabine/cisplatin in patients with advanced EGFR
mutation-positive non-small-cell lung cancer: Analyses from the
phase III, randomized, open-label, ENSURE study. Ann Oncol.
26:1883–1889. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang JC, Wu YL, Schuler M, Sebastian M,
Popat S, Yamamoto N, Zhou C, Hu CP, O'Byrne K, Feng J, et al:
Afatinib versus cisplatin-based chemotherapy for EGFR
mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6):
Analysis of overall survival data from two randomised, phase 3
trials. Lancet Oncol. 16:141–151. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhou C, Wu YL, Chen G, Feng J, Liu XQ,
Wang C, Zhang S, Wang J, Zhou S, Ren S, et al: Final overall
survival results from a randomised, phase III study of erlotinib
versus chemotherapy as first-line treatment of EGFR
mutation-positive advanced non-small-cell lung cancer (OPTIMAL,
CTONG-0802). Ann Oncol. 26:1877–1883. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa
K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, et al:
PROFILE 1014 Investigators: First-line crizotinib versus
chemotherapy in ALK-positive lung cancer. N Engl J Med.
371:2167–2177. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lynch TJ, Bell DW, Sordella R,
Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat
SM, Supko JG, Haluska FG, et al: Activating mutations in the
epidermal growth factor receptor underlying responsiveness of
non-small-cell lung cancer to gefitinib. N Engl J Med.
350:2129–2139. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Paez JG, Jänne PA, Lee JC, Tracy S,
Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, et
al: EGFR mutations in lung cancer: Correlation with clinical
response to gefitinib therapy. Science. 304:1497–1500. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sharma SV, Bell DW, Settleman J and Haber
DA: Epidermal growth factor receptor mutations in lung cancer. Nat
Rev Cancer. 7:169–181. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yoshioka H, Mitsudomi T, Morita S, Yatabe
Y, Negoro S, Okamoto I, Seto T, Satouchi M, Tada H, Hirashima T, et
al: Final overall survival results of WJTOG 3405, a randomized
phase 3 trial comparing gefitinib (G) with cisplatin plus docetaxel
(CD) as the first-line treatment for patients with non-small cell
lung cancer (NSCLC) harboring mutations of the epidermal growth
factor receptor (EGFR). 2014 ASCO Annual Meeting Abstract Number:
8117. J Clin Oncol. 32(Suppl; abstr 8117): 5s2014.
|
|
14
|
Shah NT, Kris MG, Pao W, Tyson LB, Pizzo
BM, Heinemann MH, Ben-Porat L, Sachs DL, Heelan RT and Miller VA:
Practical management of patients with non-small-cell lung cancer
treated with gefitinib. J Clin Oncol. 23:165–174. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Alexandrescu DT, Kauffman CL and Dasanu
CA: The cutaneous epidermal growth factor network: Can it be
translated clinically to stimulate hair growth? Dermatol Online J.
15:12009.
|
|
16
|
Pinquié F, de Chabot G, Urban T and
Hureaux J: Maintenance Treatment by Erlotinib and Toxic
Cardiomyopathy: A Case Report. Oncology. 90:176–177. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Macerelli M, Mazzer M, Foltran L,
Cardellino GG and Aprile G: Erlotinib-associated interstitial lung
disease in advanced pancreatic carcinoma: A case report and
literature review. Tumori. 101:e122–e127. 2015.PubMed/NCBI
|
|
18
|
Celik T and Kosker M: Ocular side effects
and trichomegaly of eyelashes induced by erlotinib: A case report
and review of the literature. Cont Lens Anterior Eye. 38:59–60.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hirsch FR, Varella-Garcia M and Cappuzzo
F: Predictive value of EGFR and HER2 overexpression in advanced
non-small-cell lung cancer. Oncogene. 28(Suppl 1): S32–S37. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Oliver GL and McFarlane DC: Congenital
trichomegaly: With associated pigmentary degeneration of the
retina, dwarfism, and mental retardation. Arch Ophthalmol.
74:169–171. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kaplan MH, Sadick NS and Talmor M:
Acquired trichomegaly of the eyelashes: A cutaneous marker of
acquired immunodeficiency syndrome. J Am Acad Dermatol. 25:801–804.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Klutman NE and Hinthorn DR: Excessive
growth of eyelashes in a patient with AIDS being treated with
zidovudine. N Engl J Med. 324:18961991. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Carser JE and Summers YJ: Trichomegaly of
the eyelashes after treatment with erlotinib in non-small cell lung
cancer. J Thorac Oncol. 1:1040–1041. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lane K and Goldstein SM:
Erlotinib-associated trichomegaly. Ophthal Plast Reconstr Surg.
23:65–66. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Braiteh F, Kurzrock R and Johnson FM:
Trichomegaly of the eyelashes after lung cancer treatment with the
epidermal growth factor receptor inhibitor erlotinib. J Clin Oncol.
26:3460–3462. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang SB, Lei KJ, Liu JP and Jia YM:
Eyelash trichomegaly following treatment with erlotinib in a
non-small cell lung cancer patient: A case report and literature
review. Oncol Lett. 10:954–956. 2015.PubMed/NCBI
|
|
27
|
Pascual JC, Bañuls J, Belinchon I, Blanes
M and Massuti B: Trichomegaly following treatment with gefitinib
(ZD1839). Br J Dermatol. 151:1111–1112. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bouché O, Brixi-Benmansour H, Bertin A,
Perceau G and Lagarde S: Trichomegaly of the eyelashes following
treatment with cetuximab. Ann Oncol. 16:1711–1712. 2005. View Article : Google Scholar
|
|
29
|
Price TJ and Nott L: Unusual hair changes
with prolonged erlotinib exposure. Intern Med J. 38:8072008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Becker A, van Wijk A, Smit EF and Postmus
PE: Side-effects of long-term administration of erlotinib in
patients with non-small cell lung cancer. J Thorac Oncol.
5:1477–1480. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Naidoo J, Sima CS, Rodriguez K, Busby N,
Nafa K, Ladanyi M, Riely GJ, Kris MG, Arcila ME and Yu HA:
Epidermal growth factor receptor exon 20 insertions in advanced
lung adenocarcinomas: Clinical outcomes and response to erlotinib.
Cancer. 121:3212–3220. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yang M, Xu X, Cai J, Ning J, Wery JP and
Li QX: NSCLC harboring EGFR exon-20 insertions after the regulatory
C-helix of kinase domain responds poorly to known EGFR inhibitors.
Int J Cancer. 139:171–176, Epub ahead of print. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Roé E, García Muret MP, Marcuello E,
Capdevila J, Pallarés C and Alomar A: Description and management of
cutaneous side effects during cetuximab or erlotinib treatments: A
prospective study of 30 patients. J Am Acad Dermatol. 55:429–437.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Shepherd FA, Pereira Rodrigues J, Ciuleanu
T, Tan EH, Hirsh V, Thongprasert S, Campos D, Maoleekoonpiroj S,
Smylie M, Martins R, et al: National Cancer Institute of Canada
Clinical Trials Group: Erlotinib in previously treated
non-small-cell lung cancer. N Engl J Med. 353:123–132. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shi Y, Zhang L, Liu X, Zhou C, Zhang L,
Zhang S, Wang D, Li Q, Qin S, et al: Icotinib versus gefitinib in
previously treated advanced non-small-cell lung cancer (ICOGEN): A
randomised, double-blind phase 3 non-inferiority trial. Lancet
Oncol. 14:953–961. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zheng H, Wang Q, Shi H, Zhang H, Hu F and
Li B: Favorable response to icotinib in a lung cancer patient with
a special mutation at exon 19 of epidermal growth factor receptor.
Thorac Cancer. 5:358–361. 2014. View Article : Google Scholar : PubMed/NCBI
|