Introduction
Researchers proposed the concept of osseointegration
in the 1960s, which laid the theoretical foundation for prostheses
implantation (1). Osseointegration
is the close connection between implant and bone in the body
without soft tissue interval, and is a prerequisite for prosthesis
stability, secure fixation and for the prosthesis to bear certain
pressure (2). There are numerous
factors that affect osseointegration, including biocompatibility,
shape design, material characteristics, patient bone conditions,
the application of bioactive molecules and surgical techniques
(2). Among these, material shape
design and characterization are the key factors that impact
bone-implant binding capacity and strength (2).
Currently, the surfaces of artificial joint
prostheses are usually treated with rough surfaces and
hydroxyapatite coating in order to provide good integration of the
bone and implant (3,4). However, this treatment limits the depth
and scope on the combination of bone and implants (5–7). A
porous implant design is speculated to an effective way to improve
bone ingrowth into prostheses and enhance the combination of the
range between bone and prosthetic surface so that aseptic loosening
may be prevented and long-term stability of artificial joints can
be obtained (8).
However, at present although several porous
materials have been used, they exhibit a range of disadvantages.
Titanium (Ti) has been extensively used for its excellent physical
properties and biocompatibility (9).
Though Ti has a porosity of 70%, which exhibited a plateau stress
of 53 MPa and an elastic modulus of 3.4 GPa (10), its compressive strength is still
lower compared with cortical bone. Certain other elements, such as
aluminum, nickel, iron, vanadium and chromidium, can generate
adverse biological effects due to the release of metal ions
(11). By contrast, Ti, niobium (Nb)
and tantalum (Ta) are believed to be nontoxic metals with good
biocompatibility (12). Compared
with Ta, Ti and Nb are lightweight and inexpensive (13). There is limited prior research on
porous Ti-Nb binary alloys. In our previous studies, we have
investigated the biocompatibility of porous Ti-25Nb in vitro
(13). This study aims to
investigate the characteristics of porous Ti-25Nb in vivo
and observe the binding capacity and strength of bone-implant
interface, thus providing further theoretical basis for the
clinical application of porous Ti-25Nb alloy.
Materials and methods
Ethical approval
All animal experiments were performed according to
protocols approved by the Institutional Animal Care and Use
Committee of Central South University (Changsha, China).
Design of the implanted prosthesis
sample for the rabbit model
A New Zealand white rabbit, provided by the
Experimental Animal Center of the Third Xiangya Hospital of Central
South University, weighing ~2.4 kg was euthanized, a full-length
femur was exposed after careful dissection, and then the
anteroposterior and lateral X-rays were obtained. The femur was cut
in the sagittal plane and coronal plane, respectively, so as to
observe the morphology of the medullary cavity. The prosthesis
specimen was designed accordingly.
Preparation of porous Ti-25Nb
prosthesis samples
Samples with different porosities were prepared
using powder metallurgy. The details of the process were described
in our previous study (9).
In vivo experiment
A total of 36 healthy New Zealand rabbits (age, 4–6
months; weight, 2.2–2.6 kg) were divided into three groups (n=12
per group). Dense specimens (<2% porosity), specimens with 40%
porosity and specimens with 70% porosity were tested in each of the
three groups, respectively.
Surgical procedures
After the rabbits were anesthetized using
pentobarbital (1 ml/kg; Sigma-Aldrich, St. Louis, MO, USA), skin
preparation and sterilization were performed on the lateral sides
of the left hips. A 3-cm straight incision was made on the skin of
the greater trochanter; the subcutaneous tissues and deep fascia
were cut layer by layer. Then, the gluteus maximus and the greater
trochanter were exposed. Parts of muscles attached to the great
trochanter were stripped, and the bone of the greater trochanter
and the top of the femoral neck were broken away. The medial part
of the femoral neck were carefully protected. Reamed into the
medullary cavity, gradually enlarged it, and drilled into 2 cm
depth using a drill 4–6 mm in diameter. The medullary cavity was
cleaned using normal saline and specimens were implanted into the
cavities along the longitudinal axis of the femur. The wound was
flushed with hydrogen peroxide and saline and sutured layer by
layer. Postoperatively, intramuscular injection of penicillin was
performed to prevent infection, for three days.
Postoperative X-rays
Rabbits underwent anteroposterior and lateral X-rays
at two, four, and eight-week time points postoperatively.
Sample extraction and preparation for
tests
Three rabbits in each group were sacrificed using 3%
pentobarbital overdose (4 ml/kg) at two, four and eight-week time
points postoperatively. The bilateral femurs and soft tissue on
them were all removed. Then they were immersed in formalin solution
as saved backup. Two of the specimens in each group underwent
scanning electron microscope (SEM) and energy dispersive X-ray
spectroscopy (EDS) analysis, with the rest for pulling-out
test.
SEM of bone-implant interface and EDS
analysis
The anterior and posterior femoral cortical bones
were cut along the sagittal plane longitudinally, exposing the
bone-implant interface. The specimens, which were to be observed by
SEM and EDS analysis, were dried. EDS elemental analysis was
performed to characterize the calcium (Ca), Ti, Nb and phosphorus
(P) content of the samples.
Pulling-out test
The samples that contained femurs with porous
Ti-25Nb specimens implanted were fixed into the mixture by mixing
the denture powder (Fig. 1A)
(denture base polymer; Shanghai Beiqiong Tooth Material Co., Ltd.,
Shanghai, China) with self-curing denture water (methyl
methacrylate) in the mold. Care was taken to avoid the denture
powder infiltrating into the interface of exposed prosthesis and
bone, which may increase the pulling-out strength and affect the
accuracy of the observed data. A steel wire was crossed into a hole
pre-prepared on the prosthesis for pulling out (Fig. 1B), and it was fixed into the clamp of
the mechanical testing machine (3369 Dual-Column Universal Testing
System; Instron, Norwood, MA, USA). The distal ends of the
specimens were also fixed. The machine's continual displacement of
was set to 2 mm/min and underwent pretension for this test. The
pulling strength and displacement were recorded automatically using
a computer.
Statistical analysis
Statistical analyses were performed by SPSS version
20.0 (IBM SPSS, Amronk, NY, USA). Variance analysis was used to
compare the differences between groups at the same time point.
Values of P<0.05 were considered significant.
Results
Design of the implanted
prosthesis
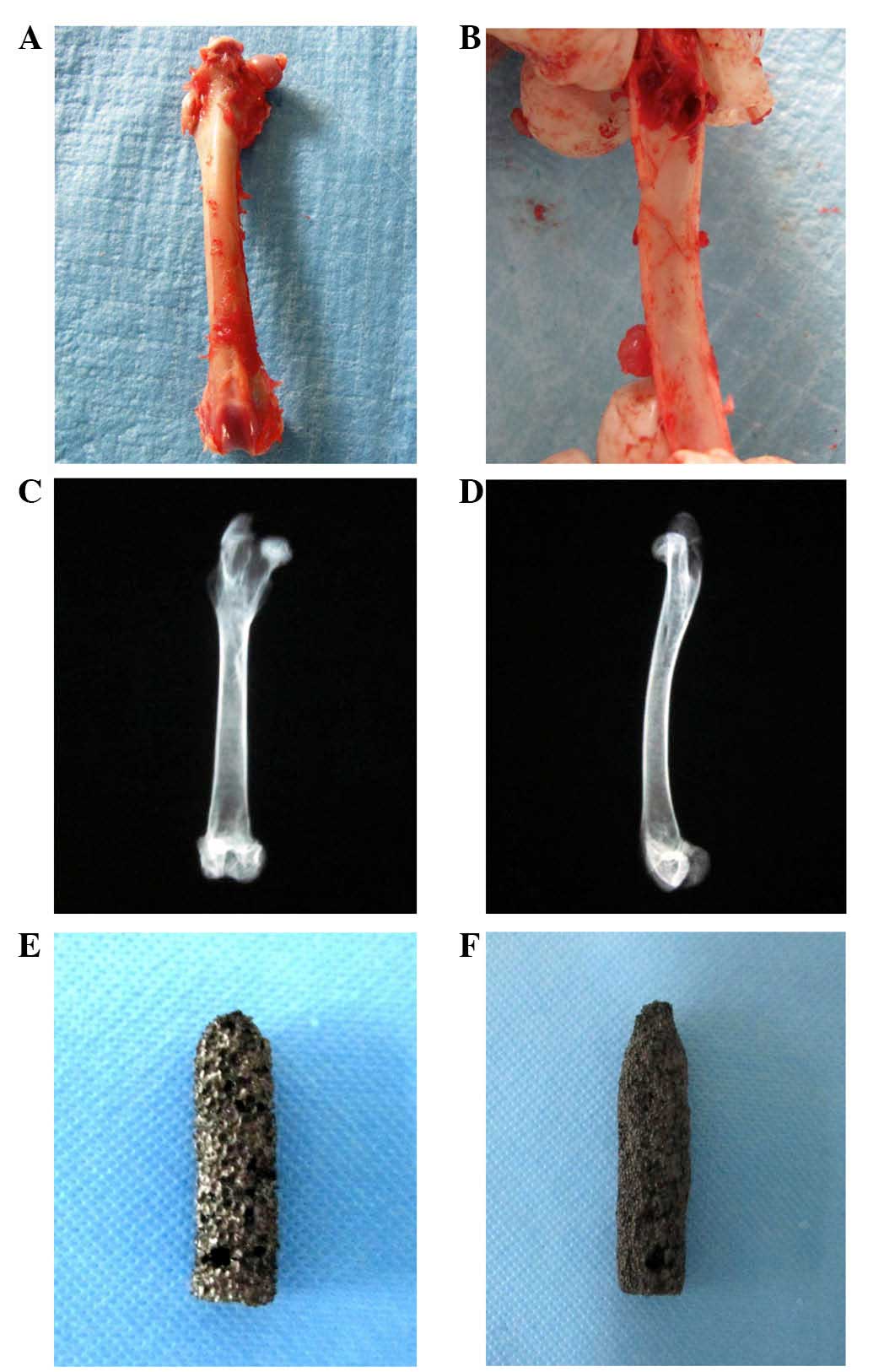
After dissecting the tissues around the femur, the
morphology and the inner diameter of the medullary cavity were
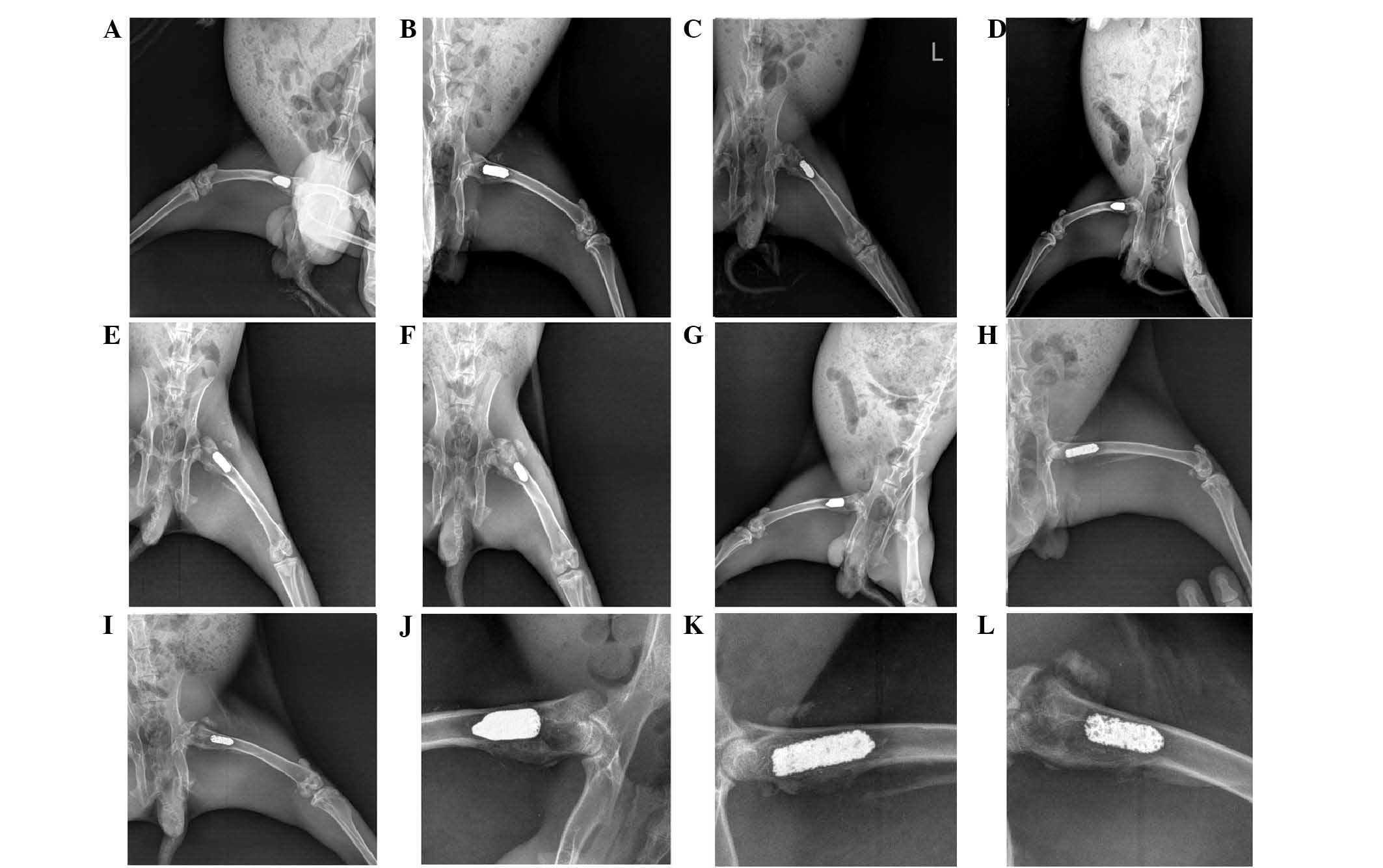
observed (Fig. 2A and B). The X-ray
was obtained for the whole-length femur (Fig. 2C and D). It showed that the cortex of
the lower femoral neck was thick, while the cortex in the femur
shaft was loose. According to these characteristics, the prosthesis
was designed for a cylindrical shape, with a diameter of 7 mm and a
height of 25 mm. Its distal end turns out to be cone-shaped
(Fig. 2E and F). A hole with a
diameter of 1.5 mm was designed on the top part of the specimen so
as to be prepared for the pulling-out test (Fig. 2E and F).
Observations for surgical procedures
and postoperative conditions
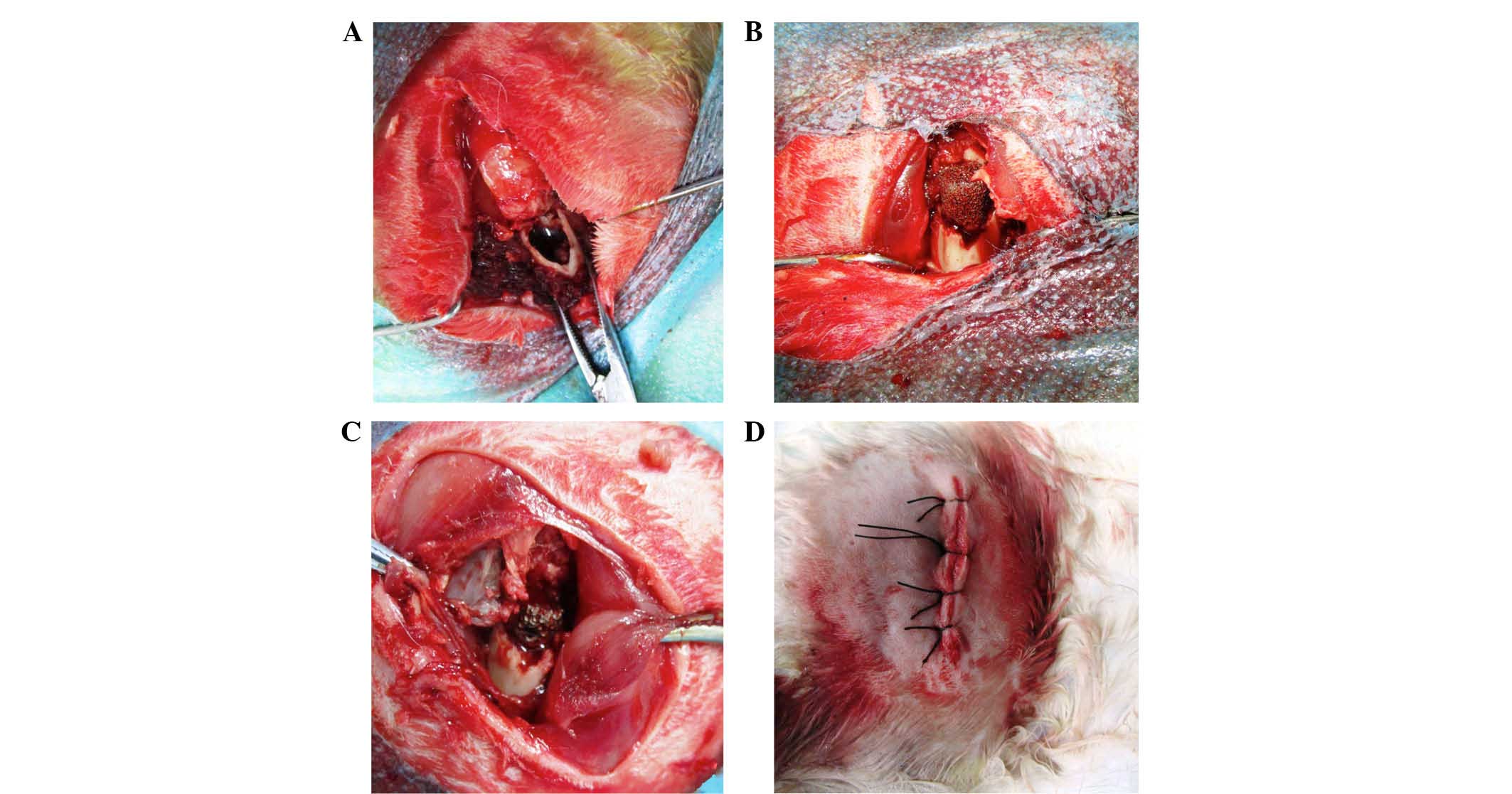
The process of the surgical procedure of the
specimen implantation was observed (Fig.
3). No fractures and side effects were observed during the
operations and for seven days postoperatively.
Observations for gross specimens and
HE staining
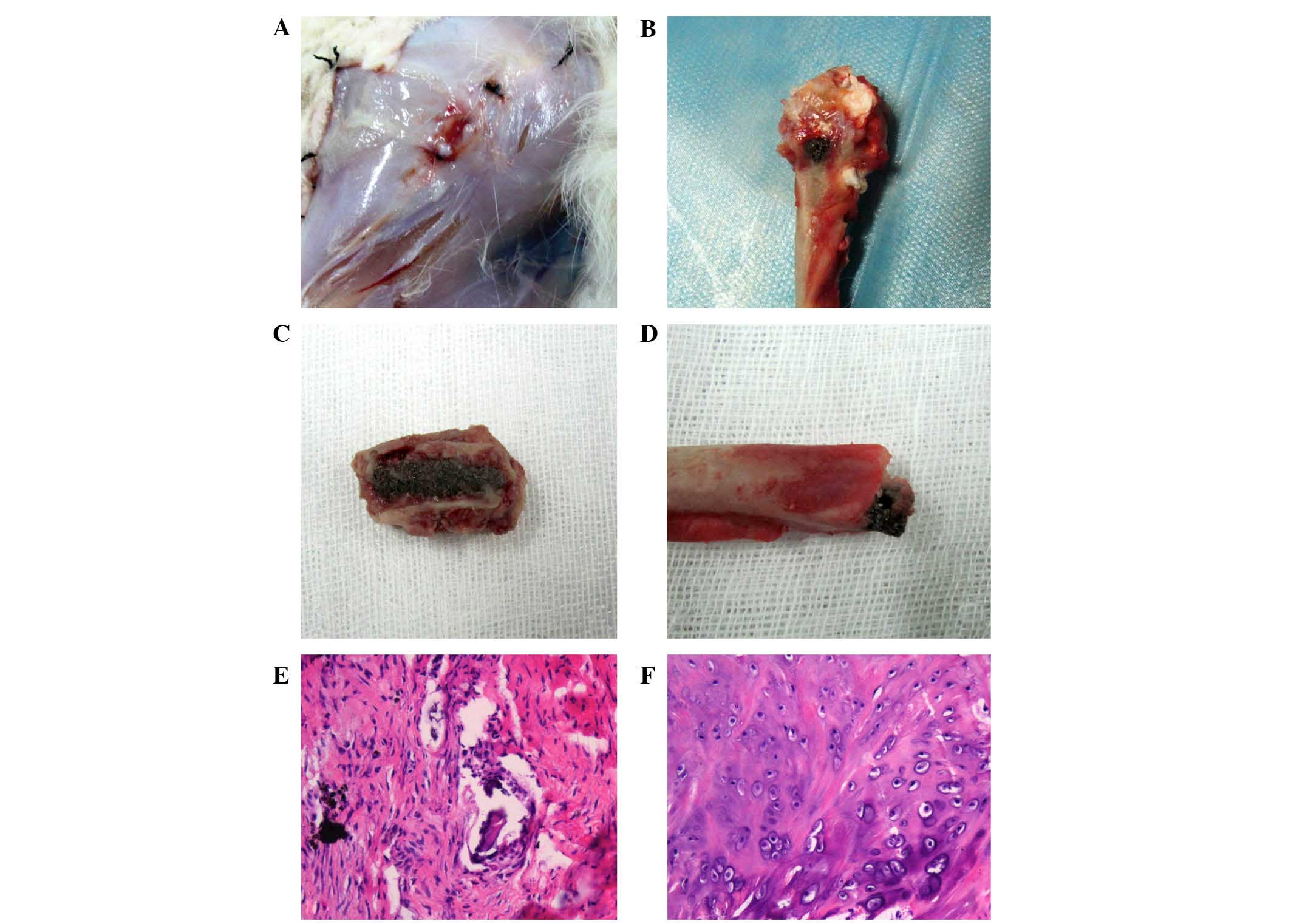
After the rabbits were euthanized, we dissected the
tissues around to expose the implants. The results revealed no
atrophy or noticeable secretions around the femurs (Fig. 4A). No loosening was observed in all
the specimens (Fig. 4B-D). While the
top part of the implant was exposed, a close bonding combination
was seen between the bone and implant (Fig. 4D). The soft tissue around the top
part of the implant was sliced for HE staining observation, and Ca
was observed in the soft tissues around, and no obvious
inflammation was seen (Fig. 4F).
X-ray examination
All specimens in each group showed a close
connection with bone tissues. No low-density shadow of bone
absorption was observed in bone tissues (Fig. 5A-K). By eight weeks, more cancellous
bone was observed around the implants.
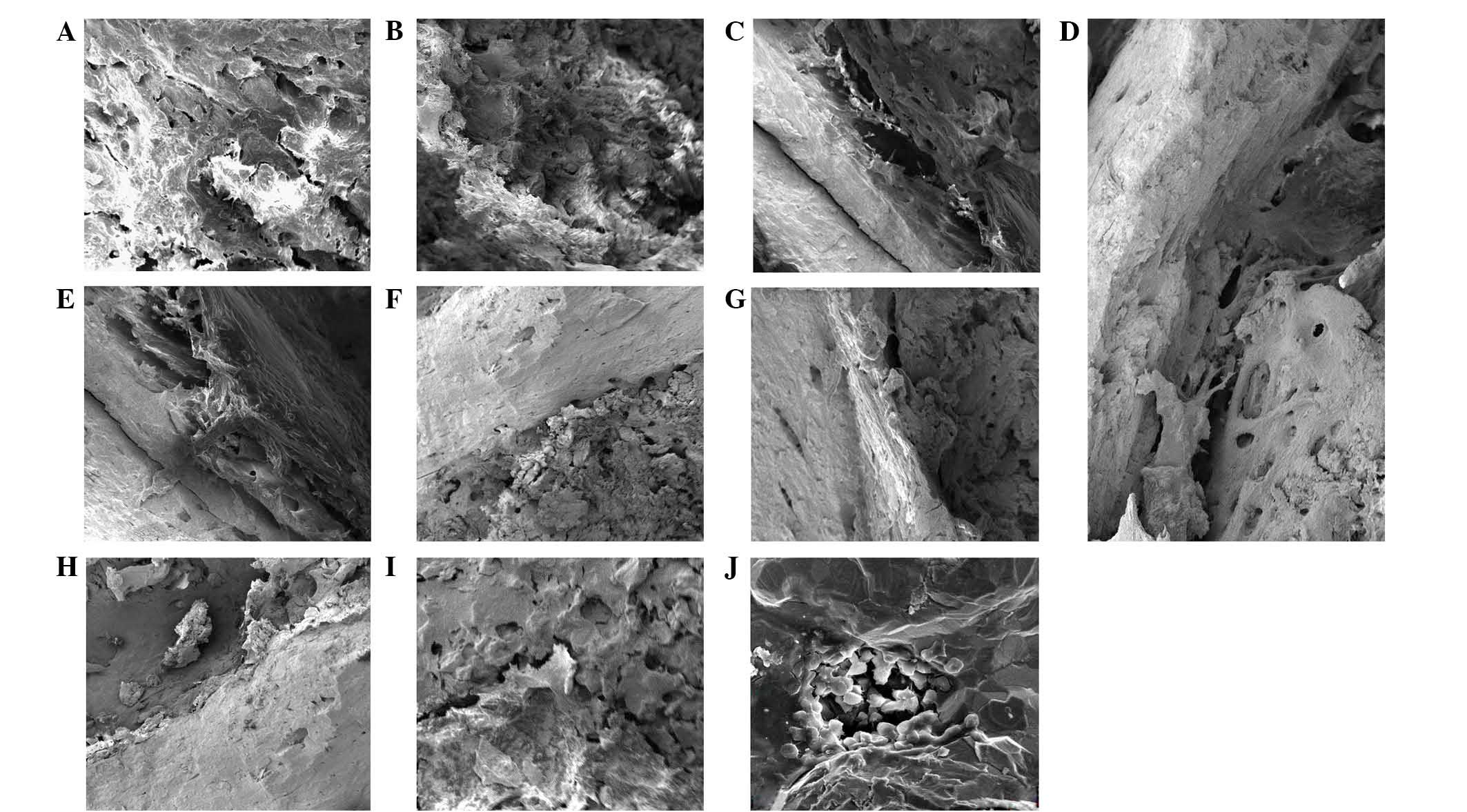
SEM observation
SEM showed Ca deposits on the surface of the
materials (Fig. 6A and B). With
regard to the bonding condition on the interface of bone and
implant, gaps remained between them by two weeks (Fig. 6C and D). A close bonding was
observed, with tissue ingrowth of the pores at the time point of
four weeks (Fig. 6E and F). While it
showed Ca deposits on the surface of the specimens, and more inside
the pores by eight weeks, the interface between had no obvious
boundaries (Fig. 6G and H). The
sectional SEM showed osseointegration between bone and implant;
bone ingrowth could be seen deeply inside the pores (Fig. 6I and J).
EDS analysis
By eight weeks, semiquantitative EDS analysis on the
interface of a specimen and bone showed that Ca, Ti and Nb were
3.24, 23.26 and 3.53% (Fig. 7A),
respectively, while inside the pores, 9.12% for Ca, 21.90% for Ti,
3.83% for Nb, and 4.37% for P was detected (Fig. 7B).
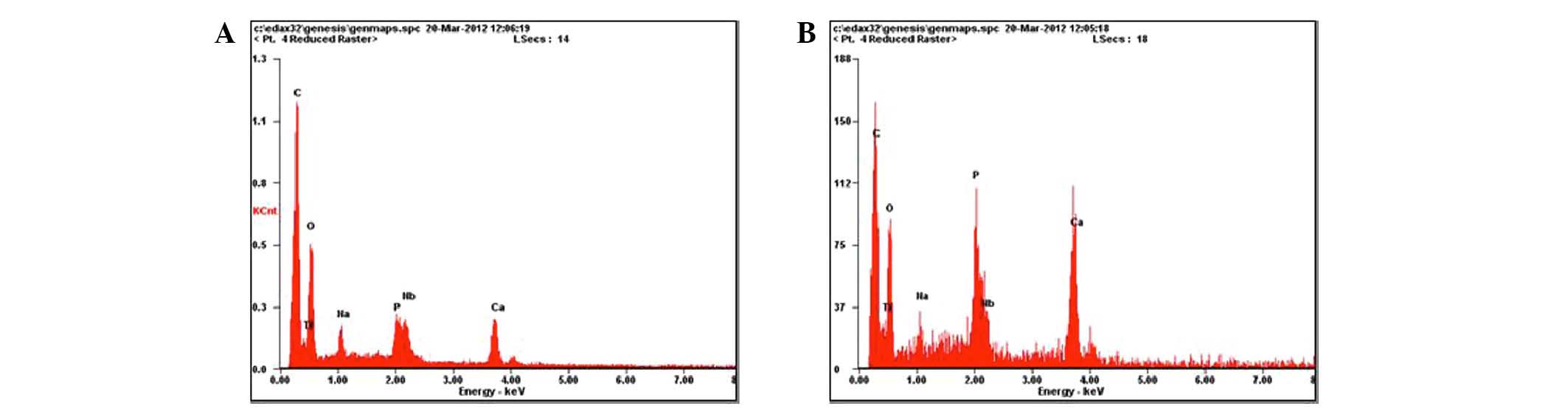 | Figure 7.Energy dispersive X-ray spectroscopy
(EDS) analysis. (A) By eight weeks, semiquantitative EDS analysis
on the interface of a specimen and bone showed that calcium,
titanium and niobium were 3.24, 23.26 and 3.53%, respectively. (B)
Inside the pores, the values for calcium, titanium, niobium and
phosphorus were 9.12, 21.90, 3.83 and 4.37%, respectively. |
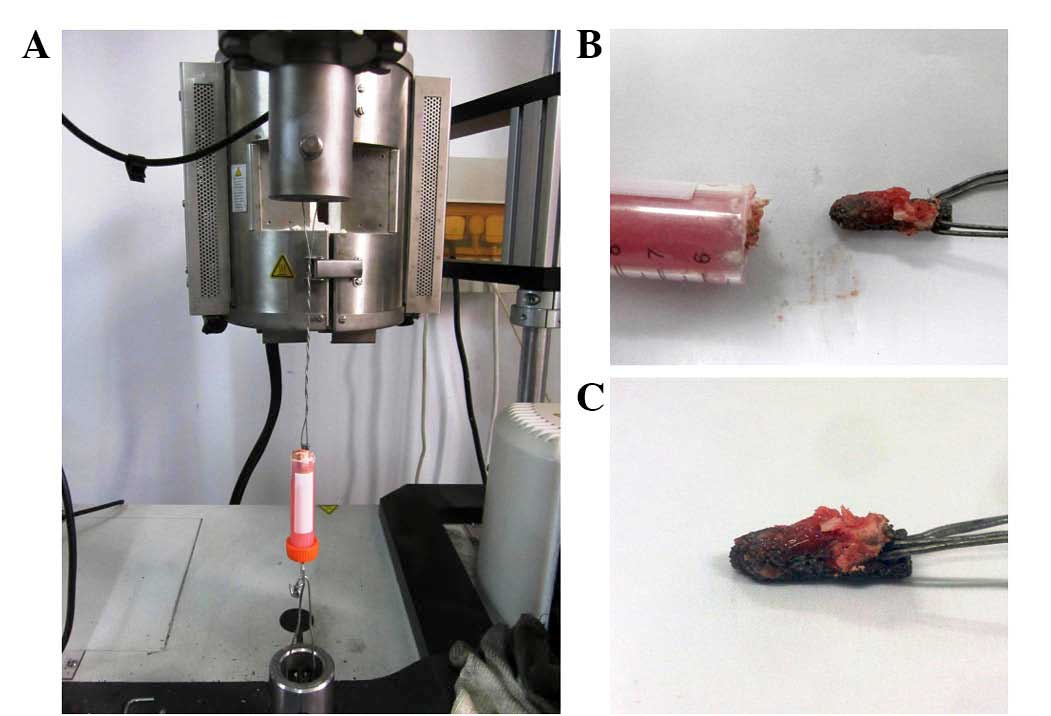
Analysis of pulling-out test
A pulling-out test was conducted using a mechanical
testing machine (Instron) (Fig. 8A).
The upper and distal ends of the specimens were fixed in one
vertical axis so as to avoid shearing force. The data were
collected automatically by the machine, linked to the computer. All
tests were accomplished without any rupture of the prostheses;
however, certain prostheses were well pulled out with part of the
bone adhered (Fig. 8B and C). The
data of the different groups are listed in Table I. At two, four and eight weeks, the
maximum pulling-out force of the dense group was smaller compared
with the other two groups, and the difference was statistically
significant (P<0.05). The pulling-out force in the 70% porosity
group was significantly higher than the 40% porosity group by four
and eight weeks (P<0.05), while there was no significant
difference between the two groups at two weeks (P>0.05). By
eight weeks, the maximum pulling-out force of the 40% and 70%
porosity groups increased significantly compared with at two and
four weeks (P<0.05), while the force between two and four weeks
had no significant difference (P>0.05).
 | Table I.Comparison of pull-out strength in the
three groups. |
Table I.
Comparison of pull-out strength in the
three groups.
|
| Maximal pull-out
force (N) |
|---|
|
|
|
|---|
| Group | 2 weeks | 4 weeks | 8 weeks |
|---|
| Dense |
76.0±11.1 |
80.2±16.0 |
113.8±11.5 |
| 40% porosity |
225.9±19.5 |
260.0±22.8 |
334.6±25.7 |
| 70% porosity |
229.5±25.5 |
280.3±20.8 |
376.1±27.4 |
Discussion
Osseointegration is a phenomenon by which a
prosthesis may make contact with the bone tissue directly and that
is able to withstand the stress of use (1). It is a combinative reaction of bone and
implant, which can be affected by a variety of factors, including
biocompatibility of the implant, morphology design of the implant,
surface structural properties and internal morphology of the
implant, the partial mechanical environment and biological
environment, systematic factors of the body, surgical techniques
and the load supported (2). The
bone-implant interface provides safe and reliable mechanical
transmission between implants and bone tissue, and the situation of
interface formation has a great influence on the long-term
stability. A stable tightly combined interface is not only
conducive to prevent migration of wear particles but also provides
a good foundation for the long-term stability of the prosthesis
(14). With the rate of the
bone-implant interface osseointegration improved, the mechanical
strength of the interface is also substantially improved and the
early osseointegration has a greater impact on bone-implant
interface strength (14).
The majority of the currently available Ti and Ti
alloy implants in clinical application are dense (15,16).
Although certain methods, such as surface treatment, can be applied
to increase the contact area, these methods produce certain effects
on bone integration between implants and bone. However, the bone
tissue can only extend to the implant surface field and not the
interior of implants (16).
Biological fixation is not achieved when long-term stability is not
guaranteed (16). Given the
difference in mechanical properties, the dense-type alloy causes
stress shelter after implantation in the body, which eventually
leads to bone resorption. Numerous studies have indicated that
excess bone loss influences the long-term effect of implants and
leads to implant displacement, aseptic loosening, fracture around
the prosthesis and increase in the difficulty of revision surgery
(14–17). To improve the binding capacity
between interface of bone and implant, the concept of
microtechnology was gradually introduced in recent years (18). Although several porous alloys are now
available in the medical field, porous Ti-Nb alloy is a relatively
cost-effective, which is also nontoxic and has good machinability
and mechanical strength compared with other porous alloys (13,18). The
porous Ti-Nb alloy used in the present study has a high strength
with the elastic modulus body close to human cortical bone, which
has been verified in our previous study in vitro (13). The degree of bone ingrowth of porous
materials depends on several factors, including the porosity, the
extent of micromotion and stability between implant and bone, and
whether cortical or cancellous bone is contacted with the implant
and the size of the gap between the implant and bone (19,20).
Furthermore, the porosity, pore size and
bone-implant contact situation have effects on bone ingrowth, in
addition to the spatial structure of the material key factor
affecting bone conduction. Studies have shown that porous implants
must have interconnected voids in order to provide space for the
ingrowth of blood vessels and promote further bone tissue ingrowth
(16,21). Another point is that the porous
communication of the material is conductive to fluid transfer
within the body, which can accelerate its tissue growth in order to
increase the fixing strength (22).
The presently investigated porous Ti-Nb alloys, which have a pore
diameter of 200–500 µm, present with three-dimensional
connectivity, thus can provide more space for ingrowth. The EDS
analysis results in this study showed that Ca and P presented
increased deposits in the 70% porosity specimens, which had more
three-dimensional communication.
The increased surface area and roughness of the
materials could improve the mechanical interlocking of the bone,
thus enhancing the stability of the bone and implant. This
mechanical interlock condition increased the strength of the
pulling-out, while this condition could not be found in dense Ti-Nb
alloys. Bonding strength depends on a combination area of
bone-implant contacting, materials with higher porosity, and pore
size that has a larger surface area; therefore, the capacity to
resist pulling-out is increased (23–25). The
pulling-out test showed that the strength was higher in the 70% and
40% porosity groups compared with the dense group after being
implanted for four weeks. The strength for the 70% porosity group
was significantly higher compared with the 40% porosity and dense
groups after being implanted for eight weeks. This result indicates
that high porosity provided more space for the ingrowth of the bone
tissue and enhanced the mechanical interlock. De Vasconcellos et
al (26) developed a porous Ti
alloy using powder metallurgy technology, which can control the
porosity, pore size, and pore connectivity. The in vivo
experiment of his research also showed that high porosity,
appropriate pore size and connectivity of porous material is good
for bone tissue ingrowth and increase of pull-out strength.
SEM examination of the specimens after being
implanted for four and eight weeks showed that the bone tissue grew
into the inner pores. This was attributed to the properties of the
trabecular bone that grows along the mechanical direction (27), which indicated that pores began
bearing the mechanical load after they were implanted. This
indirectly confirms the young modulus of this material, which is
close to the bone around, so that it can avoid the shielding effect
of stress. A study by Ryan et al (28) that the compressive strength and young
modulus of the porous materials were more matched to bone rather
than dense materials. The mismatch of the mechanical properties
between bone and materials can cause stress shielding, leading to
local bone resorption and low bone regeneration, thus affecting the
growth of bone tissue. Studies have shown that the mechanical
stability of the implants depends on the quantity of bone tissue
present (28,29). The present experimental results
showed that the pull-out force increased with the extension of the
time after implantation. This may be associated with the increase
of mature bone tissue around implants, which is consistent with the
findings of Chen et al (30).
As mentioned earlier, we altered the mechanical properties of the
implant by adding the element of Nb into the Ti alloy, so as to
more closely replicate the mechanical properties of bone. In
addition, the porous Ti-Nb alloy not only improved the contact area
of bone-implant and enhanced bonding strength, but also reduced the
difference in elastic modulus between the bone and the implant.
In conclusion, a porous Ti-25Nb alloy implants
presents good biocompatibility as well as providing a biological
fixation between the bone and implant. A porosity of 70% is more
advantageous to the newborn bone ingrowth, combined with achieving
a more solid bone-implant interface.
Acknowledgements
The present study was supported by the Scientific
Research Project of the First Affiliated Hospital of Hunan Normal
University (grant no. RS201305).
References
|
1
|
Brånemark PI, Adell R, Breine U, Hansson
BO, Lindström J and Ohlsson A: Intra-osseous anchorage of dental
prostheses. I. Experimental studies. Scand J Plast Reconstr Surg.
3:81–100. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zarb GA and Schmitt A: Implant
prosthodontic treatment options for the edentulous patient. J Oral
Rehabil. 22:661–671. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Faghihi S, Zhilyaev AP, Szpunar JA, Azari
F, Vali H and Tabrizian M: Nanostructuring of a titanium material
by high-pressure torsion improves pre-osteoblast attachment. Adv
Mater. 19:1069–1073. 2007. View Article : Google Scholar
|
|
4
|
Rupp F, Scheideler L, Olshanska N, de Wild
M, Wieland M and Geis-Gerstorfer J: Enhancing surface free energy
and hydrophilicity through chemical modifi cation of
microstructured titanium implant surfaces. J Biomed Mater Res A.
76:323–334. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vora A, Kudrna JC, Harder VS and Mazahery
B: Early failure of a proximally cemented, distally uncemented
total hip arthroplasty. J Arthroplasty. 18:889–896. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Song Y, Beaupre G and Goodman SB:
Osseointegration of total hip arthroplasties: Studies in humans and
animals. J Long Term Eff Med Implants. 9:77–112. 1999.PubMed/NCBI
|
|
7
|
LaPorte DM, Mont MA and Hungerford DS:
Proximally porous-coated ingrowth prostheses: Limits of use.
Orthopedics. 22:1154–1160. 1999.PubMed/NCBI
|
|
8
|
Kujala S, Ryhanen J, Danilov A and
Tuukkanen J: Effect of porosity on the osteointegration and bone
ingrowth of a weight-bearing nickel-titanium bone graft substitute.
Biomaterials. 24:4691–4697. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li XK, Yuan CF, Wang JL, Zhang YQ, Zhang
ZY and Guo Z: The treatment effect of porous titanium alloy rod on
the early stage talar osteonecrosis of sheep. PLoS One.
8:e584592013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wen CE, Yamada Y, Shimojima K, Hosokawa H
and Mabuchi M: Novel titanium foam for bone tissue engineering. J
Mater Res. 17:2633–2639. 2002. View Article : Google Scholar
|
|
11
|
Wapner KL: Implications of metallic
corrosion in total knee arthroplasty. Clin Orthop Relat Res.
271:12–20. 1991.PubMed/NCBI
|
|
12
|
Long M and Rack HJ: Titanium alloys in
total joint replacement-a materials science perspective.
Biomaterials. 19:1621–1639. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xu J, Weng XJ, Wang X, Huang JZ, Zhang C,
Muhammad H, Ma X and Liao QD: Potential use of porous
titanium-niobium alloy in orthopedic implants: Preparation and
experimental study of its biocompatibility in vitro. PLoS One.
8:e792892013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Good V, Ries M, Barrack RL, Widding K,
Hunter G and Heuer D: Reduced wear with oxidized zirconium femoral
heads. J Bone Joint Surg Am. 85:105–110. 2003.PubMed/NCBI
|
|
15
|
Ryan GE, Pandit AS and Apatsidis DP:
Porous titanium scaffolds fabricated using a rapid prototyping and
powder metallurgy technique. Biomaterials. 29:3625–3635. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li C and Zhu Z: Dynamic Young's modulus of
open-porosity titanium measured by the electromagnetic acoustic
resonance method. J Porous Mater. 13:21–26. 2006. View Article : Google Scholar
|
|
17
|
Kröger H, Venesmaa P, Jurvelin J,
Miettinen H, Suomalainen O and Alhava E: Bone density at the
proximal femur after total hip arthroplasty. Clin Orthop Relat Res.
66–74. 1998.
|
|
18
|
Wang X, Li Y, Hodgson PD and Wen C:
Biomimetic modification of porous TiNbZr alloy scaffold for bone
tissue engineering. Tissue Eng Part A. 16:309–316. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cornell CN and Lane JM: Current
understanding of osteoconduction in bone regeneration. Clin Orthop
Relat Res. (355 Suppl): S267–S273. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tisdel CL, Goldberg VM, Parr JA, Bensusan
JS, Staikoff LS and Stevenson S: The influence of a hydroxyapatite
and tricalcium-phosphate coating on bone growth into titanium
fiber-metal implants. J Bone Joint Surg Am. 76:159–171.
1994.PubMed/NCBI
|
|
21
|
van Eeden SP and Ripamonti U: Bone
differentiation in porous hydroxyapatite in baboons is regulated by
the geometry of the substratum: Implications for reconstructive
craniofacial surgery. Plast Reconstr Surg. 93:959–966. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nugroho AW, Leadbeater G and Davies IJ:
Processing of a porous titanium alloy from elemental powders using
a solid state isothermal foaming technique. J Mater Sci Mater Med.
21:3103–3107. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Baril E, Lefebvre LP and Hacking SA:
Direct visualization and quantification of bone growth into porous
titanium implants using micro computed tomography. J Mater Sci
Mater Med. 22:1321–1332. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vasconcellos LM, Leite DO, Oliveira FN,
Carvalho YR and Cairo CA: Evaluation of bone ingrowth into porous
titanium implant: Histomorphometric analysis in rabbits. Braz Oral
Res. 24:399–405. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Faria PE, Carvalho AL, Felipucci DN, Wen
C, Sennerby L and Salata LA: Bone formation following implantation
of titanium sponge rods into humeral osteotomies in dogs: A
histological and histometrical study. Clin Implant Dent Relat Res.
12:72–79. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
de Vasconcellos LM, Oliveira FN, Leite Dde
O, de Vasconcellos LG, do Prado RF, Ramos CJ, Graça ML, Cairo CA
and Carvalho YR: Novel production method of porous surface Ti
samples for biomedical application. J Mater Sci Mater Med.
23:357–364. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Allori AC, Sailon AM, Pan JH and Warren
SM: Biological basis of bone formation, remodeling, and repair-part
III: Biomechanical forces. Tissue Eng Part B Rev. 14:285–293. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ryan G, Pandit A and Apatsidis DP:
Fabrication methods of porous metals for use in orthopaedic
applications. Biomaterials. 27:2651–2670. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Simank HG, Stuber M, Frahm R, Helbig L,
van Lenthe H and Müller R: The influence of surface coatings of
dicalcium phosphate (DCPD) and growth and differentiation factor-5
(GDF-5) on the stability of titanium implants in vivo.
Biomaterials. 27:3988–3994. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen D, Bertollo N, Lau A, Taki N, Nishino
T, Mishima H, Kawamura H and Walsh WR: Osseointegration of porous
titanium implants with and without electrochemically deposited DCPD
coating in an ovine model. J Orthop Surg Res. 6:562011. View Article : Google Scholar : PubMed/NCBI
|