Introduction
Carotid artery stenosis refers to the stenosis of
the lumen owing to carotid atherosclerotic plaque, arteritis, or
fibromuscular dysplasia. Its incidence is high, accounting for 10%
of the population >60 years old. Carotid artery stenosis is
among the leading causes of ischemic stroke and patients usually
arrive at neurological clinics with stroke symptoms of hemiplegia
and homonymous hemianopia (1).
Targeted surgery treatments include carotid endarterectomy (CEA)
and CAS. Two types of surgery for the treatment of carotid artery
stenosis remain controversial. However, CAS has increasingly become
accepted as a mainstay therapy for the treatment of stenosis of the
carotid artery bifurcation lesions due to the reduced probability
of non-stroke complications and mortality due to CAS trauma
(2). However, the resultant shedding
carotid plaque may cause retinal artery embolism, resulting in the
severe loss of visual function in patients after CAS procedure. The
present study describes a case of branch retinal artery occlusion
following CAS, reinforcing the necessity for ophthalmic evaluation
following the surgery.
Case report
A 70 year old male presented to the Department of
Neurosurgery at Xuanwu Hospital of Capital Medical University
(Beijing, China) in April 2014 after having suffered from
intermittent dizziness for 6 months. The patient had suffered from
hypertension for 20 years and diabetes mellitus for 15 years. An
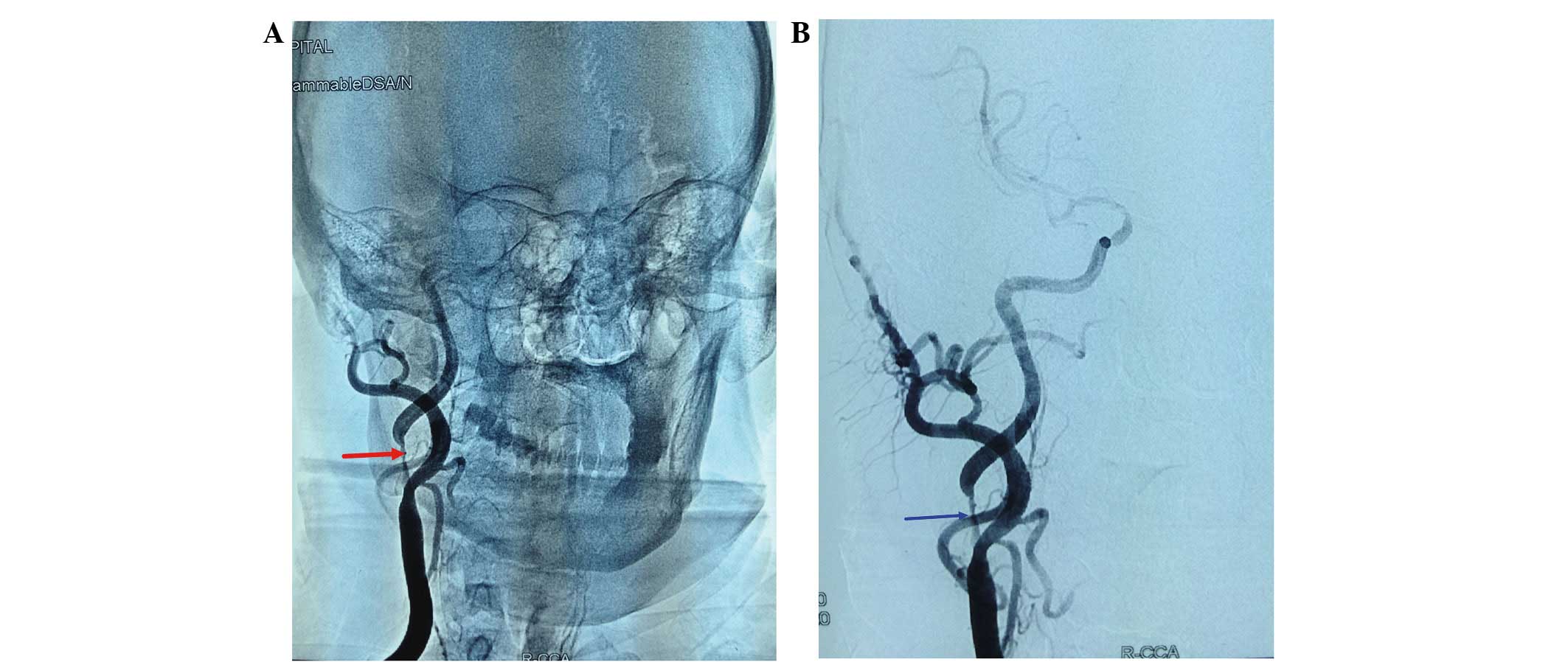
intracarotid angiography revealed severe (99%) right carotid artery
stenosis. Percutaneous transfemoral carotid stenting was performed
(Fig. 1). The patient complained of
blurred vision and scotoma in the superior visual field of the
right eye 8 h after the procedure. The best corrected visual acuity
of the right eye was 0.2 and 0.8 in the left eye. The intraocular
pressure was 11 and 12 mmHg in the right and left eye,
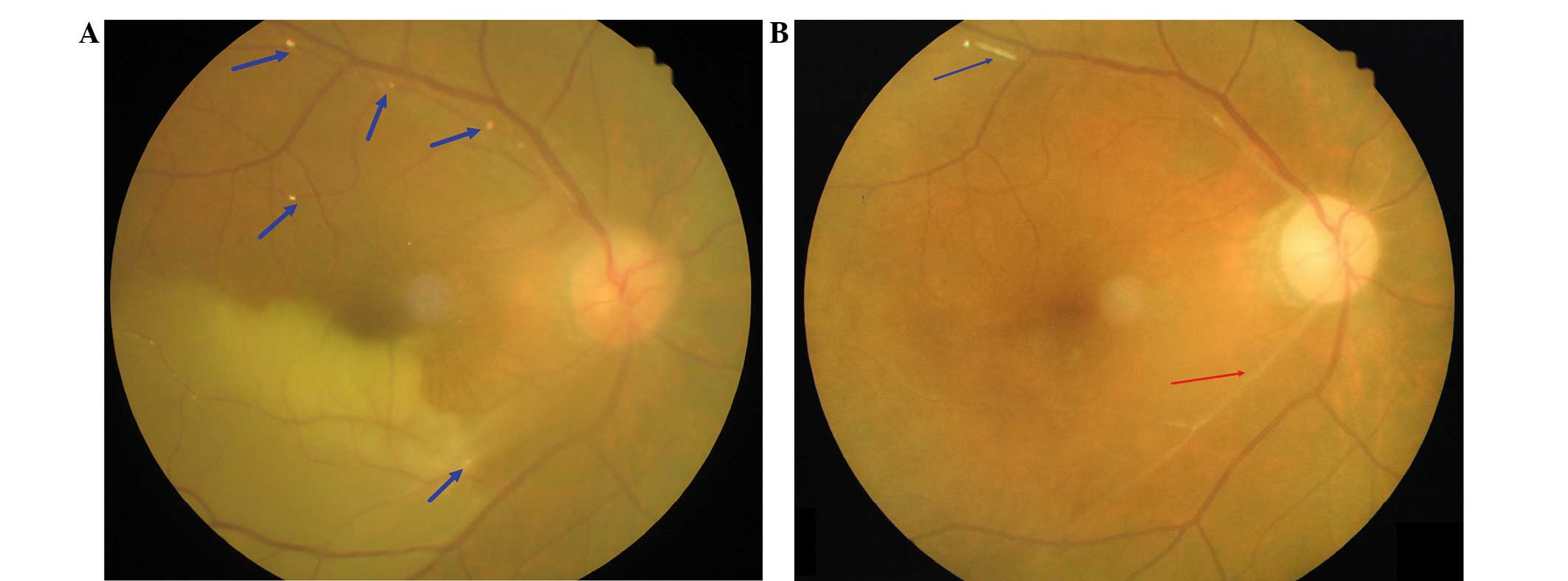
respectively. Fundus photography revealed retinal whitening and
edema in the inferior quadrant of the right eye with partial
involvement of the fovea (Fig. 2A).
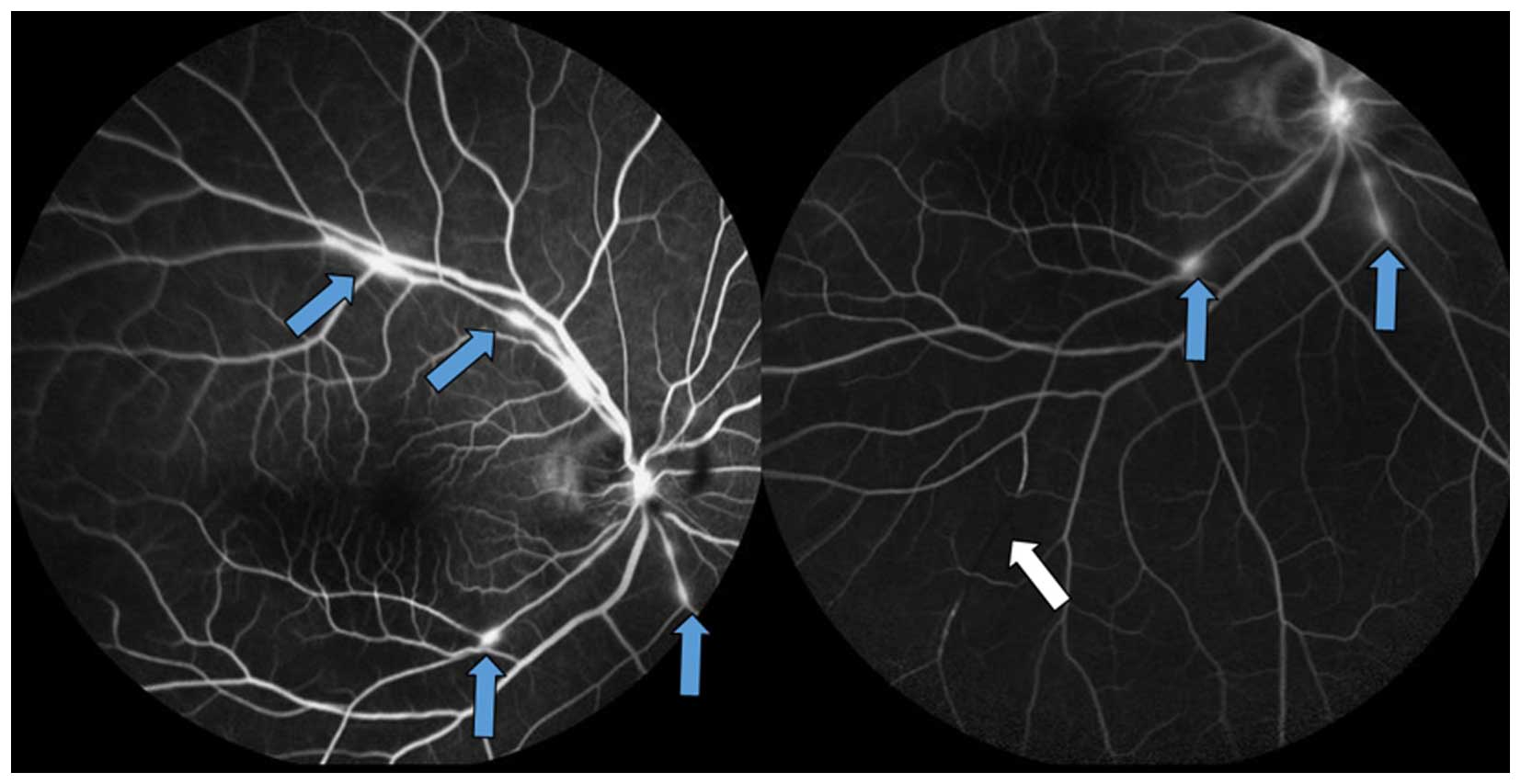
Multiple intra-arterial emboli were discovered and confirmed by
fundus fluorescent angiography, fluorescein angiogram indicated
delayed filling and emboli and perivascular leaking of the right
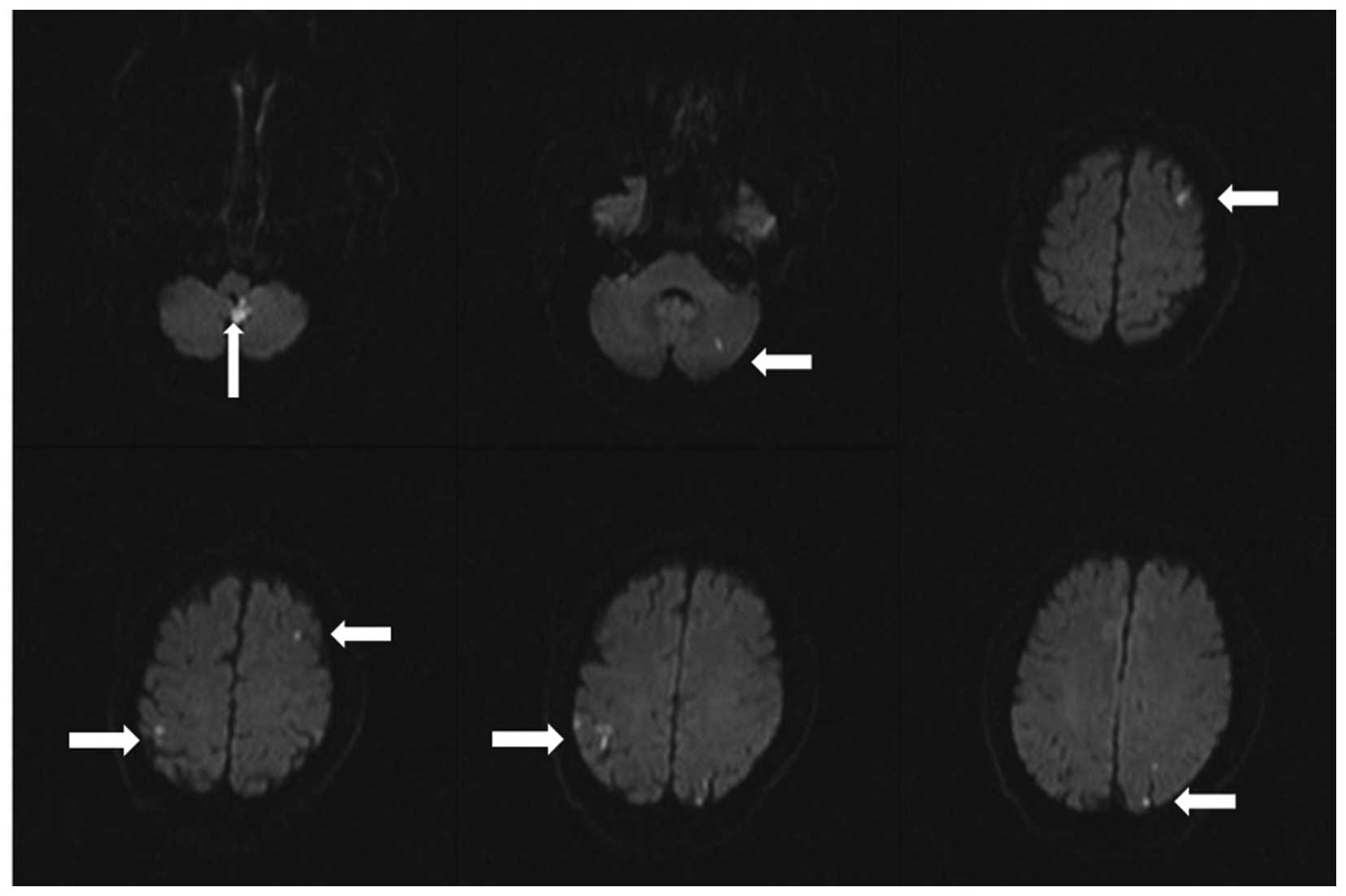
eye (Fig. 3). Magnetic resonance
imaging of the brain revealed multiple new-onset acute cerebral
infarctions (Fig. 4). The patient
was then administered therapies for the purpose of dilating the
blood vessels including regular aspirin and anticoagulants. Visual
acuity improved to 0.5 at the final visit 6 months after the
procedure, and the fundus photography revealed that the symptoms of
retinal whitening and edema had improved. However, visible
intravascular emboli and vascular occlusion was still present and
are observed as a white line in Fig.
2B.
Discussion
The retinal artery, as a terminal artery, is the
only source of nutrition for the inner retina and there are no
anastomoses which exit among its branches. Once occlusion has
occurred, the subsequent loss of retinal blood supply may result in
a decrease in vision and visual field. Causes of CRAO and BRAO
include cardiac disease, retinal artery thrombosis, vascular spasm
and vasculitis (3). Embolisms from
the carotid bifurcation are considered to be the most common cause
of CRAO and BRAO (4). CAS is
currently the predominant method employed for the treatment of
carotid stenosis; however, the procedure may result in the
occlusion of the retinal artery. Retinal artery embolization during
CAS is a rare but serious complication that can lead to partial or
total blindness (5).
The internal carotid artery (ICA) gives rise to the
ophthalmic arteries, therefore ICA stenosis may result in an
insufficient blood supply to the inner retina. Thus, retinal artery
occlusion may occur as a result of embolisms induced by carotid
stenosis, as the flow of blood may flush the debris of
atherosclerotic plaque toward the retinal artery. In CAS, debris
may be flushed to the retinal artery during the placement of
carotid stents and embolic protection devices (EPDs). In addition,
the anastomosis of external carotid (ECA) circulation to the
internal carotid artery (i.e., from the orbital branch of the
middle meningeal artery to ophthalmic artery) may form with carotid
stenosis (6), and 8–35% of
preoperative carotid stenosis patients have reverse flow in the
ophthalmic artery prior to surgery (7,8).
Transient blindness, amaurosis fugax, has previously
been reported in patients with a fully occluded ipsilateral
internal carotid, unequivocally indicating the collateral pathway
to be a mechanism for retinal embolization (2). The passage of debris through the
aforementioned collaterals poses a significant risk as flushing
debris through the orbital branch of the middle meningeal artery
towards the ECA, in particular, may lead to retinal embolization
(9). The absence of EPDs to filter
the debris may also lead to retinal embolization following CAS.
Additionally, the opening of ECA prior to opening ICA during CAS
surgery may result in retinal embolization (10,11).
Embolization resulting in retinal arterial occlusion
following CAS surgery, despite the use of EPDs, has been reported
in the literature. Vos et al (12) discovered that retinal embolization
occurred in 6/118 patients (4%) undergoing CAS with EPDs, two of
whom (1.7%) were symptomatic. Wilentz et al (2) reported retinal emboli in 5/33 stenting
procedure patients (15%) performed using distal EPDs, two of whom
presented with additional small retinal infarcts. The mean rate of
embolization per CAS procedure has been reported as 74 particles
per stenotic lesion (13).
Fortunately, of the embolic material produced following CAS, few
are large enough to cause retinal damage; typically, emboli >20
mm cause symptomatic retinal artery occlusions in CAS (2).
In the case detailed in the present study, a large
number of emboli were observed within the retinal vessels, and a
large quantity of emboli were observed which led to intracranial
vascular embolism. This conclusion indicated that an increased
quantity of embolus is flushed to intracranial vessels in CAS.
Investigation into the correlation between retinal artery embolism
and intracranial vascular embolism has therefore gained increasing
interest. With regard to the aforementioned discoveries, monitoring
of patients following CAS is necessary to avoid retinal artery
embolism.
The current therapies for the treatment of BRAO and
CRAO include the use of acute and chronic measures (14,15).
Acute treatment involves modalities which lower the intraocular
pressure, including enhanced retinal oxygenation and ocular
massage, whilst chronic treatments aim to decrease platelet
adhesiveness with therapies such as aspirin, dipyridamole or
sulfinpyrazone to prevent the dislodging of further emboli and
thrombus formation. However, no therapy has been conclusively
demonstrated to be efficacious (16).
In conclusion, ophthalmic evaluation is important
following CAS due to the possibility of embolic occlusion of the
retinal artery. Patients must be advised of the risk of permanent
vision loss as a result of CAS, and neurologists performing the
procedure are require to be vigilant of this potential
postoperative complication of CAS.
References
|
1
|
Faries PL, Chaer RA, Patel S, Lin SC,
DeRubertis B and Kent KC: Current management of extracranial
carotid artery disease. Vasc Endovascular Surg. 40:165–175. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wilentz JR, Chati Z, Krafft V and Amor M:
Retinal embolization during carotid angioplasty and stenting:
Mechanisms and role of cerebral protection systems. Catheter
Cardiovasc Interv. 56:320–327. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kimura K, Hashimoto Y, Ohno H, Uchino M
and Ando M: Carotid artery disease in patients with retinal artery
occlusion. Intern Med. 35:937–940. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hayreh SS, Podhajsky PA and Zimmerman MB:
Retinal artery occlusion: Associated systemic and ophthalmic
abnormalities. Ophthalmology. 116:1928–1936. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Karjalainen K: Occlusion of the central
retinal artery and retinal branch arterioles. A clinical,
tonographic and fluorescein angiographic study of 175 patients.
Acta Ophthalmol Suppl. 109:1–95. 1971.PubMed/NCBI
|
|
6
|
Cohn EJ Jr, Sandager GP, Benjamin ME,
Lilly MP, Hanna DJ and Flinn WR: Assessment of ocular perfusion
after carotid endarterectomy with color-flow duplex scanning. J
Vasc Surg. 29:665–671. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Costa VP, Kuzniec S, Molnar LJ, Cerri GG,
Puech-Leão P and Carvalho CA: The effects of carotid endarterectomy
on the retrobulbar circulation of patients with severe occlusive
carotid artery disease. An investigation by color Doppler imaging.
Ophthalmology. 106:306–310. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rosenkranz K, Langer R and Felix R:
Transcranial Doppler sonography: Collateral pathways in internal
carotid artery obstructions. Angiology. 42:819–826. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sfyroeras GS, Bessias N, Moulakakis KG,
Lyra S, Kotsikoris I, Andrikopoulos V and Liapis CD: New cerebral
ischemic lesions after carotid endarterectomy. Ann Vasc Surg.
27:883–887. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Madycki G, Staszkiewicz W and Gabrusiewicz
A: Carotid plaque texture analysis can predict the incidence of
silent brain infarcts among patients undergoing carotid
endarterectomy. Eur J Vasc Endovasc Surg. 31:373–380. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shillito J Jr and Rockett FX: Retinal
artery embolism: A complication of carotid endarterectomy. J
Neurosurg. 20:718–720. 1963. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vos JA, van Werkum MH, Bistervels JH,
Ackerstaff RG, Tromp SC and van den Berg JC: Retinal embolization
during carotid angioplasty and stenting: Periprocedural data and
follow-up. Cardiovasc Intervent Radiol. 33:714–719. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jordan WD Jr, Voellinger DC, Doblar DD,
Plyushcheva NP, Fisher WS and McDowell HA: Microemboli detected by
transcranial Doppler monitoring in patients during carotid
angioplasty versus carotid endarterectomy. Cardiovasc Surg.
7:33–38. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Beiran I, Goldenberg I, Adir Y, Tamir A,
Shupak A and Miller B: Early hyperbaric oxygen therapy for retinal
artery occlusion. Eur J Ophthalmol. 11:345–350. 2001.PubMed/NCBI
|
|
15
|
Rumelt S, Dorenboim Y and Rehany U:
Aggressive systematic treatment for central retinal artery
occlusion. Am J Ophthalmol. 128:733–738. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fraser SG and Adams W: Interventions for
acute non-arteritic central retinal artery occlusion. Cochrane
Database Syst Rev. 1:CD0019892009.PubMed/NCBI
|