Introduction
Ovarian cancer is one of the most common
malignancies in women, and may be classified as epithelial ovarian
cancer (EOC) and ovarian cancer of germ cell origin (1,2).
Statistics indicate that the mortality rate of EOC places it at
fourth position among female cancers, and the mortality rates as a
result of EOC morbidity have been high in recent years (3,4).
Previous studies suggest that it is difficult to diagnose EOC in
the early stages in the ovary exists in the specific shady position
of pelvis, which also results in a 5-year survival rate of 15–30%
(5,6). Therefore, to explore several key
factors for EOC diagnosis in the early stage may be of some use to
clinical practice.
MicroRNAs (miRNAs) are a class of endogenous ~22 nt
RNAs that serve crucial roles in the human body through targeting
mRNAs at the post-transcriptional level (7). Several miRNAs represent a novel class
of genes that have important roles as negative regulators of gene
expression in a number of diseases in previous studies. For
example, Yang et al (8)
showed that miR-214 was able to induce cell survival and cisplatin
resistance via targeting phosphatase and tensin homolog in ovarian
cancer, and Vecchione et al (9) revealed that miR-484 functions
independently in ovarian cancer by modulating vascular endothelial
growth factor (VEGF) through VEGFB signaling. In addition, several
papers have indicated that miR-9 may function as a tumor suppressor
in certain types of cancer, such as ovarian cancer and colon cancer
(10,11). One such paper by Tang et al
(12) determined that miR-9 inhibits
cell proliferation, migration and invasion, and is a tumor
suppressor in ovarian serous carcinoma through targeting the
Talin-1 gene. In addition, cytokine stromal cell-derived factor
(SDF-1, also known as CXCL12), a small proinflammatory
chemoattractant cytokine that is able to bind to a specific
G-protein coupled seven-span transmembrane receptor of CXCR4, is a
major regulator of cell trafficking and adhesion (13). Increasing evidence has indicated that
the SDF-1/CXCR4 pathway is crucial in promoting tumor cell
proliferation and enhancing cell invasion and tumor angiogenesis by
activating the downstream signal proteins in variety of tumors,
such as ovarian carcinoma, oral cancer and colorectal carcinoma
(14,15). Although several papers in the
literature have reported the importance of the role of miR-9 in
ovarian cancer development, the association between miR-9 and the
SDF-1/CXCR4 pathway in ovarian cancer proliferation, in addition to
its underlying mechanism, have yet to be elucidated.
In the present study, miR-9 was transfected into
human ovarian cancer OVCAR-3 cells for the purpose of analyzing the
effects of miR-9 in ovarian cancer progression. Comprehensive
experimental methods were used to detect the expression levels of
CXCR4 mRNA, and to measure the effects of miR-9 on OVCAR-3 cell
proliferation, invasion and cell apoptosis. The current study aimed
to investigate the potential role of miR-9 in ovarian cancer
progression and its potential mechanism, and may provide a
theoretical basis for future research concerning ovarian cancer
diagnosis and treatment.
Materials and methods
Cell culture and cell
transfection
The human EOC OVCAR-3 cell line (American Type
Culture Collecction, Manassas, VA, USA) was cultivated in RPMI 1640
medium (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) supplemented with 20% fetal bovine serum (FBS; Sigma-Aldrich,
St. Louis, MO, USA) and incubated at 37°C in 5% CO2. The
miR-9 mimics were purchased from Sangon Biotech Co., Ltd.
(Shanghai, China), and synthetic small duplex sequences of
miR-9-RNA were bioprocessed into mature miR-9 in cells. Total cells
were separated into three groups as follows: i) Blank (cells
transfected without miR-9 RNA); ii) negative control (NC) sequence
was used to eliminate any potential non-sequence specific effects;
and iii) experimental group (cells transfected with miR-9 RNA).
Briefly, OVCAR-3 cells in the logarithmic phase were transferred
onto 6-well plates, followed by the transfection of miR-9 mimics
into cells according to the protocol supplied with the
Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.).
Subsequently, cells were cultivated using RPMI-1640 medium without
antibiotics, and Lipofectamine 2000 and miR-9 mimics were diluted
with serum-free medium. Primers were as follows: Sense,
5′-GGGTCTTTGGTTATCTAGC-3′ and antisense, 5′-TGCGTGTCGTGGAGTC-3′ for
miR-9 amplification; and sense, 5′-UUCUCCGAACGUGUCACGUTT-3′ and
antisense, 5′-ACGUGACACGUUCGGAGAATT-3′ for the silenced miR-9
vector construction.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
The OVCAR-3 cells in three groups collected at 48 h
underwent grinding in liquid nitrogen and were then washed with PBS
buffer (pH 7.4). The total RNA from OVCAR-3 cells was isolated
using TRIzol extraction reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol
(16), and the isolated RNA was
treated with RNase-free Dnase I (Promega Corporation, Madison, WI,
USA) to remove genomic DNA. The concentration and purity of
extracted RNA was detected using SMA4000 UV–VIS (Merinton
Instrument, Inc., Ann Arbor, MI, USA). The purified RNA (0.5 µg/µl)
was used for cDNA synthesis with the PrimerScript 1st Strand cDNA
Synthesis kit (Takara Biotechnology Co., Ltd., Dalian, China).
Primers used for target amplification were as follows: Sense,
5′-CTTCTTAACTGGCATTGTGG-3′ and antisense,
5′-ACTGAACCTGACCGTACAGTGATGACAAAG-3′. RT-qPCR was performed using
an Eppendorf Mastercycler (Brinkmann Instruments, Westbury, NY,
USA) using the SYBR ExScript RT-qPCR Kit (Takara Biotechnology Co.,
Ltd.). A total reaction system of 20 µl volume contained 1 µl cDNA
from the aforementioned PCR, 10 µl SYBR Premix EX Taq, 1 µl of each
of the primers (10 µM), and 7 µl ddH2O. The PCR program
was as follows: Denaturation at 95°C for 2 min; followed by 45
cycles at 95°C for 10 sec, 59°C for 20 sec and 72°C for 30 sec. The
2−ΔΔCT method was used to determine the relative gene
expression levels. Melting curve analysis of amplification products
was performed at the climax of each PCR to confirm that only one
product was amplified and detected. GAPDH was used as the internal
control.
3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl tetrazolium bromide (MTT)
assay
Cell survival rate and sensitivity of OVCAR-3 tumor
cells at different time points following transfection with miR-9 or
NC vectors were detected using MTT assay as previously described
(17). Briefly, 200 µl OVCAR-3 tumor
cells in the logarithmic phase were transfected into 24-well plates
with RPMI 1640 medium supplemented with 10% FBS for 24 h. Each
assay was performed six times and repeated three times
independently. Subsequently, MTT (5 mg/ml; Sigma-Aldrich) was added
into the 24-well plates to combine with tumor cells, and incubated
at 37°C for 4 h. Mixtures were centrifuged at 1,000 × g for 10 min,
followed by removal of the supernatant. Next, 150 µl dimethyl
sulfoxide was added to the harvested cells to dissolve the
formazan. The absorbance of cells in each plate was measured at 570
nm using a microplate reader (THERMOMultiskan; M3, Fort Washington,
PA, USA).
Cell invasion assay
The invasive ability of OVCAR-3 cells was detected
on Matrigel-coated (0.8 mg/ml) Transwell inserts with 8 µm pore
size (BD Biosciences, Franklin Lakes, NJ, USA). The miR-9 RNA and
scrambled OVCAR-3 cells were starved for 24 h, then harvested.
Subsequent to this, cells (300 µl; cell density, 1×105
cells/ml) were seeded into the upper well of the chamber containing
serum free medium. The lower chamber contained RPMI 1640 with 10%
BSA. Following an incubation period of 12 h, cells were invaded
onto the membrane and fixed with 70% ethanol and stained with 0.1%
crystal violet and sealed on slides. The number of cells across the
membrane was calculated using a light microscope.
Cell apoptosis assay
Effect of miR-9 on OVCAR-3 tumor cell apoptosis was
quantified by flow cytometry using Annexin V-fluorescein
isothiocyanate (FITC) cell apoptosis kit (Invitrogen; Thermo Fisher
Scientific, Inc.) according to manufacturer's protocol (18). Briefly, the OVCAR-3 cells were
transfected with miR-9 or NC vectors for 36 h, followed by the
replacement of cell culture medium with serum-free RPMI 1640
medium. Total cells were harvested and washed using PBS buffer (pH
7.4) three times, and resuspended in the staining buffer provided
in the kit. Following this, 5 µl Annexin V-FITC and 5 µl propidium
iodide (PI) were mixed with the cells. After being cultivated at
room temperature for 10 min, mixtures were analyzed using the
FACScan flow cytometry (BD Biosciences). Annexin V-positive and
PI-negative cells were considered to be apoptotic cells.
Western blot analysis
OVCAR-3 tumor cells at 48 h were lysed in
radioimmunoprecipitation assay (Sangon Biotech Co., Ltd.) lysate
containing phenylmethanesufonyl fluoride and centrifuged at 8,000 ×
g for 10 min at 4°C. Supernatant was collected to determine
the concentration of proteins using a bicinchoninic acid protein
assay kit (Pierce Biotechnology, Inc., Rockford, IL, USA). A total
of 25 µg protein per cell lysates was then subjected to 10%
SDS-PAGE and transferred onto a polyvinylidene fluoride membrane
(Merck KGaA, Darmstadt, Germany). The membrane was blocked in
Tris-buffered saline/Tween 20 (TBST) with 5% non-fat milk for 1 h,
and subsequently incubated with rabbit anti-human extracellular
signal-regulated kinase 1 (ERK1; ab17942), ERK2 (ab32081) or matrix
metalloproteinase-9 (MMP-9; ab38898; all Abcam, Cambridge, UK)
monoclonal antibodies (1:100) overnight at 4°C, then incubated with
horseradish peroxidase labeled goat anti-rat secondary antibody
(1:1,000; ab7010; Abcam) at room temperature for 1 h. Subsequently,
the membrane was washed using the X1 TBST buffer for 10 min three
times. Detection was performed using the development of X-ray after
chromogenic substrate with an enhanced chemiluminescence method. In
addition, β-actin (Sigma-Aldrich) served as the internal
control.
Statistical analysis
All data are expressed as the mean ± standard error
of the mean. Independent sample t-test was used to calculate the
difference among groups (Blank compared with control; miR-9
compared with Blank, and miR-9 compared with control) using
GraphPad Prism software (version 5.0; GraphPad Software, Inc., San
Diego, CA, USA). P<0.05 was defined as statistically
significant.
Results
miR-9 inhibited CXCR4 expression in
ovarian tumor cells
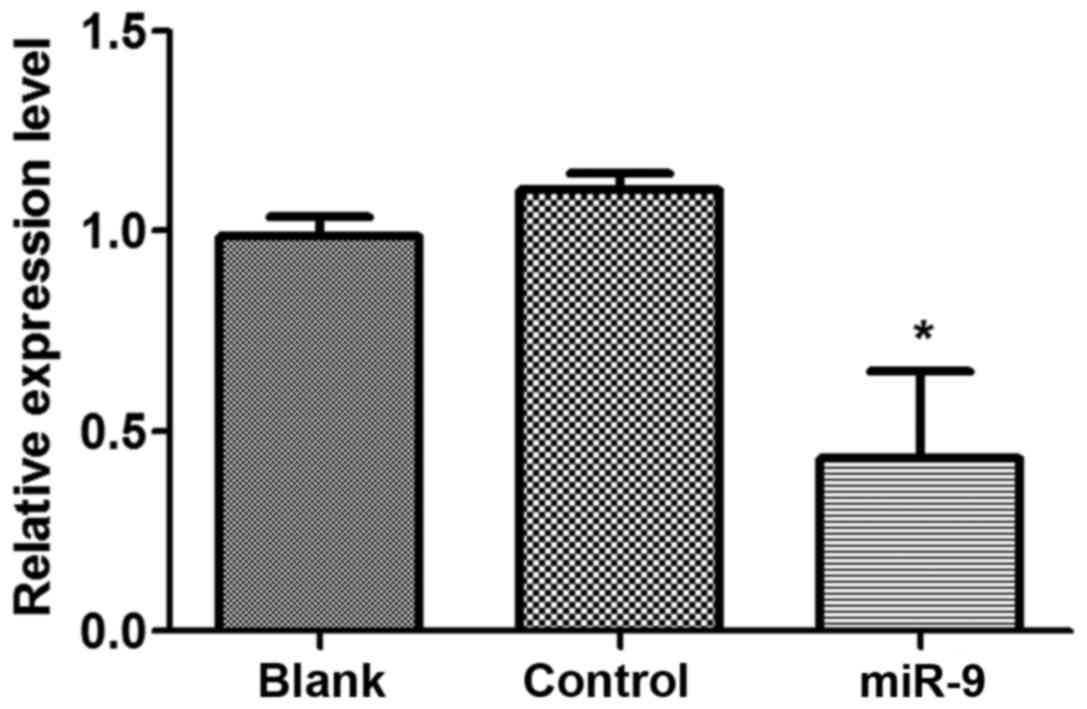
In order to analyze the effect of miR-9 on CXCR4
expression levels in OVCAR-3 tumor cells, shRNA vectors of miR-9 or
NC were transfected into OVCAR-3 cells (Fig. 1). The results indicated that CXCR4
expression levels in OVCAR-3 cells that were transfected with miR-9
declined significantly compared with those in the control group
(P<0.05), other than this, there was no significant difference
in CXCR4 expression levels in the Blank group compared with the NC
group (P>0.05).
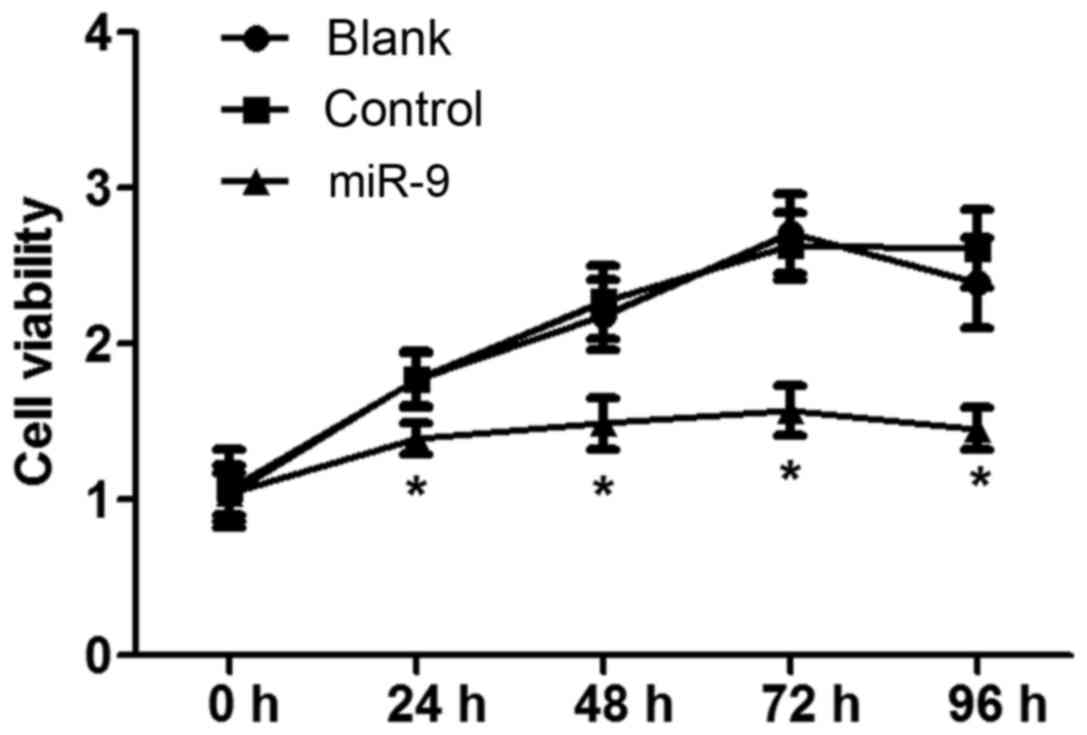
MTT assay
The effect of miR-9 on ovarian tumor OVCAR-3 cell
vitality was detected using an MTT assay (Fig. 2). The results indicated that there
was no significant difference in cell vitality between the Blank
and NC groups with increasing time (P>0.05). However, cell
vitality in the miR-9 group was significantly decreased compared
with that in the Blank and NC groups with increasing time
(P<0.05), indicating that miR-9 may inhibit OVCAR-3 cell
vitality.
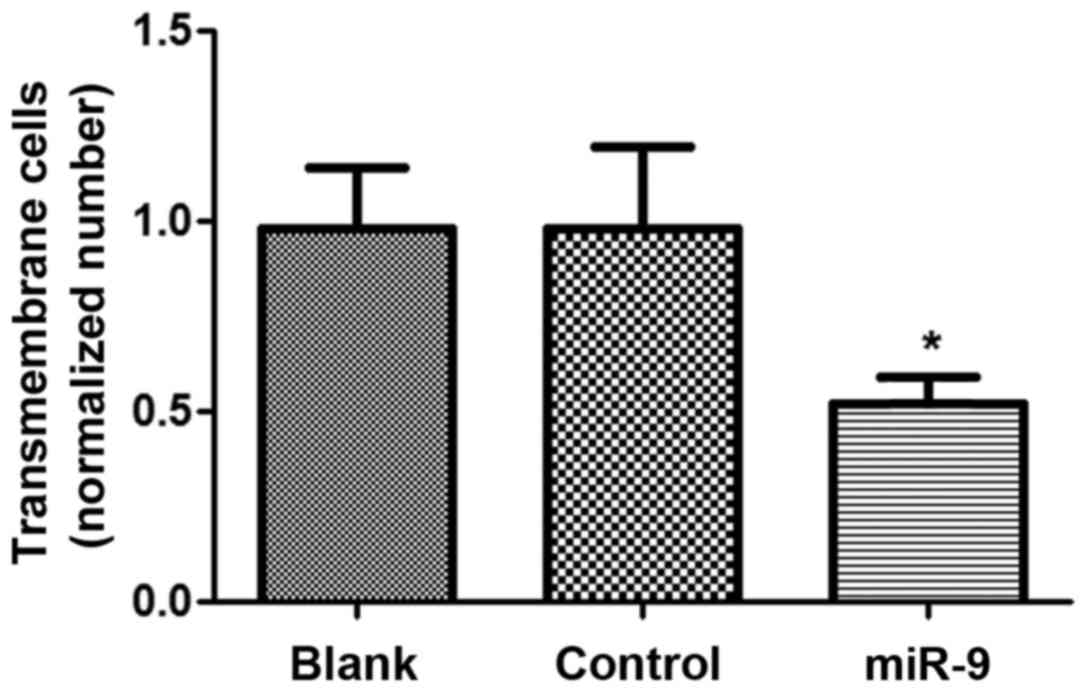
Cell invasion assay
The cell invasive ability of OVCAR-3 cells
transfected with miR-9 was assessed using the Matrigel method
(Fig. 3). There was no significant
difference in cell invasive ability between the Blank and control
groups (P>0.05), however, the cell number of OVCAR-3 cells in
the miR-9 group was significantly declined compared with the Blank
and NC groups (P<0.05), suggesting that miR-9 may suppress the
invasive ability of ovarian tumor OVCAR-3 cells.
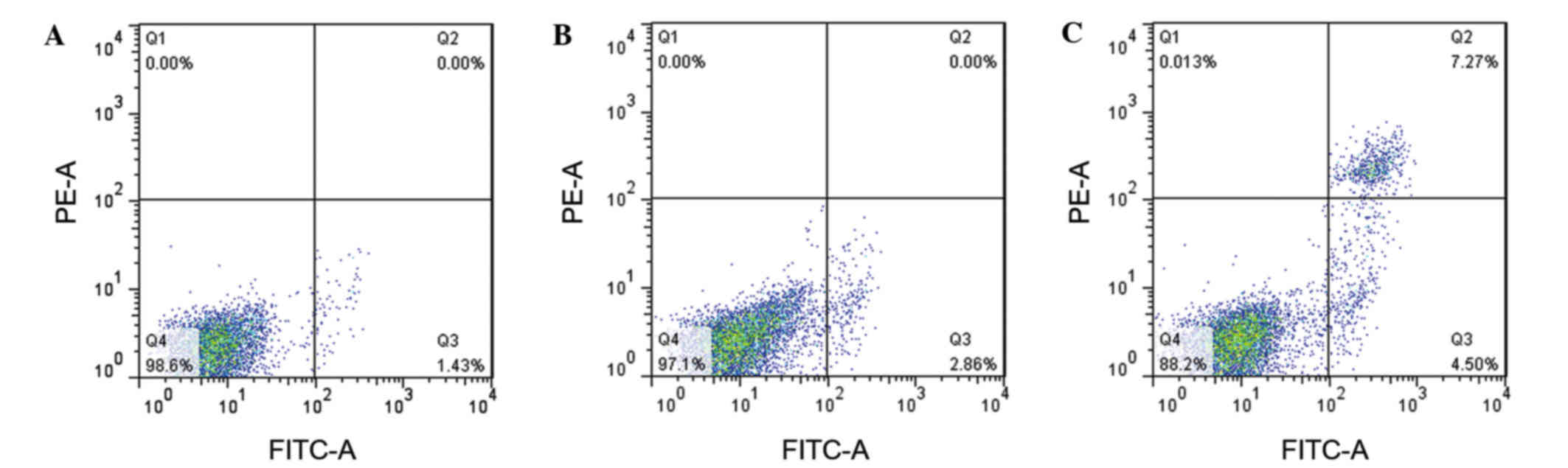
Cell apoptosis
In order to assess the effect of miR-9 on OVCAR-3
cell apoptosis, Annexin V was used to measure the apoptotic cells
(Fig. 4). The results revealed that
the apoptotic cells in the Blank and NC groups were 1.43 and 2.86%,
respectively (Fig. 4A and B). The
amount of apoptotic cells in the miR-9 group was 4.5% (Fig. 4C), implying that miR-9 may accelerate
OVCAR-3 cell apoptosis.
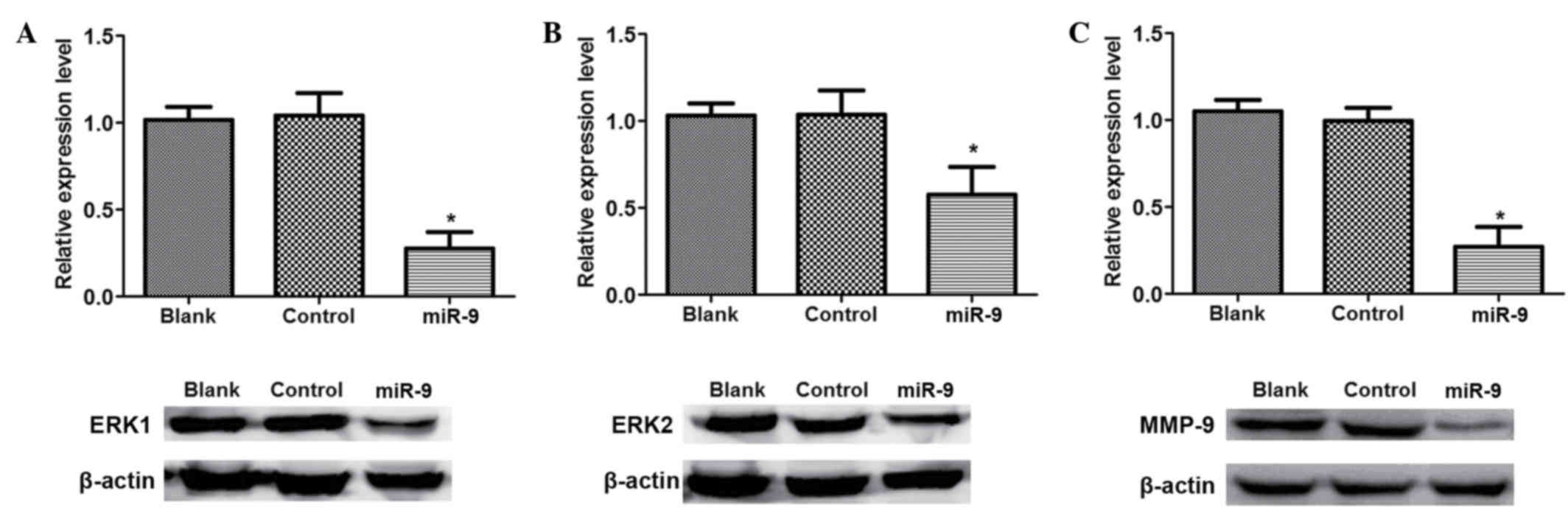
Western blot analysis
To identify the association between miR-9 with the
SDF-1/CXCR4 signal pathway in ovarian cancer, western blot analysis
was conducted to detect the signal pathway associated proteins
(Fig. 5). There was no significant
difference in ERK1, ERK2 and MMP-9 protein expression levels
between Blank and control group (P>0.05; Fig. 5). However, the expression levels of
ERK1, ERK2 and MMP-9 in miR-9 groups were significantly reduced
compared with those in the Blank or NC groups (P<0.05).
Discussion
Ovarian cancer is one of the most prominent
malignancies detected in females worldwide (1,2), and the
mortality rates associated with EOC has led it to be ranked fourth
among female cancers, and mortality resulting from EOC morbidity
has been particularly high in recent years (3,4). Despite
the importance of the role of miR-9 in ovarian cancer development
being widely reported in the literature (10,12), the
association between miR-9 and the SDF-1/CXCR4 pathway in ovarian
cancer proliferation and its potential mechanism have yet to be
fully elucidated. In the present study, the effect of miR-9 on
ovarian cancer cell proliferation was analyzed by transfecting
miR-9 RNA into OVCAR-3 cells. The current data indicated that miR-9
was able to downregulate the mRNA expression levels of CXCR4,
inhibit cell proliferation, suppress cell invasion and induce cell
apoptosis of OVCAR-3 cells. Furthermore, miR-9 was able to
downregulate the protein expression levels of ERK1, ERK2 and MMP-9
in OVCAR-3 cells.
The current data displayed that miR-9 was able to
inhibit cell proliferation, suppress invasive ability and induce
cell apoptosis of OVCAR-3 cells. Prior studies have demonstrated
that inhibiting cell proliferation, suppressing cell invasion and
inducing cell apoptosis could block tumor metastasis (19,20).
Laios et al (11)
demonstrated that miR-9 blocked cell proliferation and
angiogenesis, induced cell apoptosis and repressed invasion in
ovarian cancer. Additionally, Guo et al (21) revealed that cell growth was inhibited
by miR-9 through NF-kB1 signaling in ovarian cancer. In addition,
Tan et al (22) established
that miR-9 reduced cell invasion in SK-Hep-1 cells, consequently
suppressing tumor metastasis. In a study by Lujambio et al
(23), the methylation status of
miR-9 was certified to be associated with cancer metastasis by
influencing biological processes such as cell apoptosis,
differentiation and proliferation. Based on the current results, we
hypothesize that miR-9 may function to suppress ovarian cancer
metastasis by influencing cell invasion, proliferation and
apoptosis.
Furthermore, the current results revealed that CXCR4
was downregulated by miR-9 in OVCAR-3 cells. CXCR4 is the receptor
for SDF-1 (CXCL12 cytokine) and is involved in cancer progression
through certain signals (24). It
has been demonstrated that endocrine disrupting chemicals are able
to promote cell growth in ovarian cancer through the CXCL-12-CXCR4
signaling pathway (25). In
addition, Popple et al (26)
revealed that SDF-1 was able to induce the proliferation of tumor
cells and thus may be an independent predictor of poor survival in
ovarian cancer. Furthermore, Lu et al (27) identified that miR-9 acted as a tumor
suppressor in nasopharyngeal carcinoma by repressing CXCR4
expression, and miR-9 has been determined to be an inhibitor on
cell proliferation in oral squamous cell carcinoma by suppressing
CXCR4 expression (28). Conversely,
the expression levels of ERK1, ERK2 and MMP-9 were also
downregulated by miR-9 in this study. The activated ERK1/2 was able
to induce CXCR4 expression in cancers (29) and Yu et al (30) found that CXCR4 enhanced squamous cell
carcinoma migration and invasion by inducing MMP-9 expression
through the ERK signaling pathway. MMP-9 serves a crucial role in
tumor invasion and metastasis (31),
and it has been reported that SDF-1 enhances cell invasion of
ovarian cancer by upregulating MMP-9 expression (32). In accordance with previous results,
CXCR4 and MMP-9, ERK1/2 expressions were lower in the miR-9 group
compared with the controls, indicating that miR-9 may be a
suppressor of ovarian cancer metastasis by downregulating the
SDF-1/CXCR4 pathway by suppressing ERK1/2 and MMP-9 expression
levels.
In conclusion, the current study investigated the
effect of miR-9 in ovarian cancer metastasis and investigated
potential underlying mechanisms. Overall, the current study
ascertained that miR-9 downregulates CXCR4 expression in ovarian
cancer OVCAR-3 cells and inhibits OVCAR-3 cell proliferation and
suppresses cell invasiveness. In addition, miR-9 induces OVCAR-3
cell apoptosis and is a tumor inhibitor for ovarian cancer via the
SDF-1/CXCR4 pathway. Thus, miR-9 may function as a promising tumor
therapy by inhibiting cell proliferation, suppressing invasion and
inducing cell apoptosis via the downregulation of the SDF-1/CXCR4
pathway in ovarian cancer. The present study may provide a basis
for future research pertaining to the diagnosis of ovarian cancer.
However, further experimental studies are required to explore the
complex mechanisms underlying the involvement of miR-9 in ovarian
cancer.
References
|
1
|
Miow QH, Tan TZ, Ye J, Lau JA, Yokomizo T,
Thiery JP and Mori S: Epithelial-mesenchymal status renders
differential responses to cisplatin in ovarian cancer. Oncogene.
34:1899–1907. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nik NN, Vang R, Shih IeM and Kurman RJ:
Origin and pathogenesis of pelvic (ovarian, tubal and primary
peritoneal) serous carcinoma. Ann Rev Pathol. 9:27–45. 2014.
View Article : Google Scholar
|
|
3
|
Marcus CS, Maxwell GL, Darcy KM, Hamilton
CA and McGuire WP: Current approaches and challenges in managing
and monitoring treatment response in ovarian cancer. J Cancer.
5:25–30. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tone AA, McConechy MK, Yang W, Ding J, Yip
S, Kong E, Wong KK, Gershenson DM, Mackay H, Shah S, et al:
Intratumoral heterogeneity in a minority of ovarian low-grade
serous carcinomas. BMC Cancer. 14:9822014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dillman RO, DePriest C, Ellis R and de
Leon C: 5-year survival for patients with metastatic melanoma who
had no evidence of disease at time of treatment with patient
specific tumor stem cell vaccines. Cancer Res. 74 Suppl 19:1972014.
View Article : Google Scholar
|
|
7
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang H, Kong W, He L, Zhao JJ, O'Donnell
JD, Wang J, Wenham RM, Coppola D, Kruk PA, Nicosia SV and Cheng JQ:
MicroRNA expression profiling in human ovarian cancer: miR-214
induces cell survival and cisplatin resistance by targeting PTEN.
Cancer Res. 68:425–433. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vecchione A, Belletti B, Lovat F, Volinia
S, Chiappetta G, Giglio S, Sonego M, Cirombella R, Onesti EC,
Pellegrini P, et al: A microRNA signature defines chemoresistance
in ovarian cancer through modulation of angiogenesis. Proc Natl
Acad Sci USA. 110:9845–9850. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cekaite L, Rantala JK, Bruun J, Guriby M,
Agesen TH, Danielsen SA, Lind GE, Nesbakken A, Kallioniemi O, Lothe
RA and Skotheim RI: MiR-9, −31, and −182 deregulation promote
proliferation and tumor cell survival in colon cancer. Neoplasia.
14:868–879. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Laios A, O'Toole S, Flavin R, Martin C,
Kelly L, Ring M, Finn SP, Barrett C, Loda M, Gleeson N, et al:
Potential role of miR-9 and miR-223 in recurrent ovarian cancer.
Mol Cancer. 7:352008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tang H, Yao L, Tao X, Yu Y, Chen M, Zhang
R and Xu C: miR-9 functions as a tumor suppressor in ovarian serous
carcinoma by targeting TLN1. Int J Mol Med. 32:381–388.
2013.PubMed/NCBI
|
|
13
|
Teicher BA and Fricker SP: CXCL12
(SDF-1)/CXCR4 pathway in cancer. Clin Cancer Res. 16:2927–2931.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yu Y, Shi X, Shu Z, Xie T, Huang K, Wei L,
Song H, Zhang W and Xue X: Stromal cell-derived factor-1
(SDF-1)/CXCR4 axis enhances cellular invasion in ovarian carcinoma
cells via integrin β1 and β3 expressions. Oncol Res. 21:217–225.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kinouchi M, Uchida D, Kuribayashi N,
Tamatani T, Nagai H and Miyamoto Y: Isolation of a novel
metastasis-related microRNA, miR-518c-5p, induced by the stromal
cell-derived factor (SDF)-1/CXCR4 system in oral cancer. Cancer
Res. 74 Suppl 19:14462014. View Article : Google Scholar
|
|
16
|
Hummon AB, Lim SR, Difilippantonio MJ and
Ried T: Isolation and solubilization of proteins after TRIzol
extraction of RNA and DNA from patient material following prolonged
storage. Biotechniques. 42:467–470. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Szotek PP, Pieretti-Vanmarcke R, Masiakos
PT, Dinulescu DM, Connolly D, Foster R, Dombkowski D, Preffer F,
Maclaughlin DT and Donahoe PK: Ovarian cancer side population
defines cells with stem cell-like characteristics and mullerian
inhibiting substance responsiveness. Proc Natl Acad Sci USA.
103:11154–11159. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shirali S, Aghaei M, Shabani M, Fathi M,
Sohrabi M and Moeinifard M: Adenosine induces cell cycle arrest and
apoptosis via cyclinD1/Cdk4 and Bcl-2/Bax pathways in human ovarian
cancer cell line OVCAR-3. Tumor Biol. 34:1085–1095. 2013.
View Article : Google Scholar
|
|
19
|
Zetter BR: Angiogenesis and tumor
metastasis. Ann Rev Med. 49:407–424. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Steeg PS: Tumor metastasis: Mechanistic
insights and clinical challenges. Nat Med. 12:895–904. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guo LM, Pu Y, Han Z, Liu T, Li YX, Liu M,
Li X and Tang H: MicroRNA-9 inhibits ovarian cancer cell growth
through regulation of NF-kappaB1. FEBS J. 276:5537–5546. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tan HX, Wang Q, Chen LZ, Huang XH, Chen
JS, Fu XH, Cao LQ, Chen XL, Li W and Zhang LJ: MicroRNA-9 reduces
cell invasion and E-cadherin secretion in SK-Hep-1 cell. Med Oncol.
27:654–660. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lujambio A, Calin GA, Villanueva A, Ropero
S, Sánchez-Céspedes M, Blanco D, Montuenga LM, Rossi S, Nicoloso
MS, Faller WJ, et al: A microRNA DNA methylation signature for
human cancer metastasis. Proc Natl Acad Sci USA. 105:13556–13561.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Obermajer N, Muthuswamy R, Odunsi K,
Edwards RP and Kalinski P: PGE2-induced CXCL12 production and CXCR4
expression controls the accumulation of human MDSCs in ovarian
cancer environment. Cancer Res. 71:7463–7470. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hall JM and Korach KS: Endocrine
disrupting chemicals promote the growth of ovarian cancer cells via
the ER-CXCL12-CXCR4 signaling axis. Mol Carcinog. 52:715–725. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Popple A, Durrant L, Spendlove I, Rolland
P, Scott IV, Deen S and Ramage JM: The chemokine, CXCL12, is an
independent predictor of poor survival in ovarian cancer. Br J
Cancer. 106:1306–1313. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lu J, Luo H, Liu X, Peng Y, Zhang B, Wang
L, Xu X, Peng X, Li G, Tian W, et al: miR-9 targets CXCR4 and
functions as a potential tumor suppressor in nasopharyngeal
carcinoma. Carcinogenesis. 35:554–563. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yu T, Liu K, Wu Y, Fan J, Chen J, Li C,
Yang Q and Wang Z: MicroRNA-9 inhibits the proliferation of oral
squamous cell carcinoma cells by suppressing expression of CXCR4
via the Wnt/β-catenin signaling pathway. Oncogene. 33:5017–5027.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hatano K, Yamaguchi S, Nimura K, Murakami
K, Nagahara A, Fujita K, Uemura M, Nakai Y, Tsuchiya M, Nakayama M,
et al: Residual prostate cancer cells after docetaxel therapy
increase the tumorigenic potential via constitutive signaling of
CXCR4, ERK1/2 and c-Myc. Mol Cancer Res. 11:1088–1100. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yu T, Wu Y, Helman JI, Wen Y, Wang C and
Li L: CXCR4 promotes oral squamous cell carcinoma migration and
invasion through inducing expression of MMP-9 and MMP-13 via the
ERK signaling pathway. Mol Cancer Res. 9:161–172. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Roomi M, Monterrey J, Kalinovsky T, Rath M
and Niedzwiecki A: In vitro modulation of MMP-2 and MMP-9 in human
cervical and ovarian cancer cell lines by cytokines, inducers and
inhibitors. Oncol Rep. 23:605–614. 2010.PubMed/NCBI
|
|
32
|
Yu Y, Li H, Xue B, Jiang X, Huang K, Ge J,
Zhang H and Chen B: SDF-1/CXCR7 axis enhances ovarian cancer cell
invasion by MMP-9 expression through p38 MAPK pathway. DNA Cell
Biol. 33:543–549. 2014. View Article : Google Scholar : PubMed/NCBI
|