Introduction
Glaucoma is a major cause of blindness,
characterized by progressive axonal pathology and death of retinal
ganglion cells, leading to structural changes in the optic nerve
head and irreversible vision loss. As a result, glaucoma is now
classified within a group of chronic neurodegenerative conditions
that collectively, are the leading cause of irreversible blindness
worldwide (1–6). As indicated by glaucoma epidemiological
surveys, primary open-angle and angle-closure glaucoma occur in the
majority of patients with glaucoma (7–9). A
number of hypotheses have been proposed regarding the pathogenesis
of glaucoma, however there is currently no hypothesis that fully
explains the pathological changes occurring. Although the
mechanisms underlying glaucoma are not well understood, it has been
established that glaucoma does not result from a single
pathological mechanism, but rather a combination of pathways
influenced by genes, age and environment (10–18).
Despite this, there remain many unsolved problems, such as the
effect of nuclear transcription factor regulation in glaucoma, that
warrant further study (19).
The role of trabecular meshwork cells, including
their mechanisms of apoptosis, in the pathogenesis of glaucoma is a
current focus of research. Previous studies have documented that
trabecular meshwork endothelial cells may directly alter the
composition of the extracellular matrix during oxidative stress,
resulting in elevated intraocular pressure (IOP) and glaucoma
(20–24). Nuclear factor (erythroid-derived
2)-like 2 (Nrf2) is a central regulator of cellular oxidation
reactions and serves a key role in cell defense mechanisms against
oxidative stress (25–29). However, the expression and functions
of Nrf-2 in trabecular meshwork cells are currently unknown.
Therefore, the present study evaluated the functions of Nrf-2 in
glaucoma.
Using reverse transcription-quantitative polymerase
chain reaction (RT-qPCR) and western blot analysis, the levels of
Nrf2 in glaucoma trabecular meshwork (GTM) cells were assessed. It
was observed that Nrf2 expression was downregulated in GTM cells,
relative to human trabecular meshwork (HTM) cells. Results from a
transfection assay demonstrated that Nrf2 overexpression markedly
increased the viability of GTM and HTM cells, while significantly
decreasing their rates of apoptosis. Furthermore, western blot
analysis indicated that Nrf2 regulated the expression of
apoptosis-related proteins. Collectively, these results suggest an
association between Nrf-2 expression and glaucoma, thus offering a
potential therapeutic target for the treatment of glaucoma.
Materials and methods
Cell culture
HTM and GTM cells were kindly provided by Yishui
Central Hospital (Shandong, China). HTM and GTM cells were grown in
an adherent cell culture with Dulbecco's modified Eagle medium
(DMEM) supplemented with 20% fetal bovine serum (both Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) at 37°C, 5%
CO2 and 100% humidity in a controlled incubator. Cells
were subcultured in a humidified 5% CO2 incubator at
37°C when they reached a confluence of 70–90%, then washed twice
with D-Hanks solution.
Plasmid and small interfering RNA
(siRNA) transfection
Plasmids expressing siRNA targeting Nrf2 (siNrf2),
overexpressing Nrf2 and control siRNA (non-silencing) were
synthesized by Shanghai GenePharma Co., Ltd. (Shanghai, China).
Cells were seeded in 6-well plates and cultured in DMEM overnight
at 37°C in a humidified atmosphere of 95% air and 5%
CO2. Subsequently, cell transfections were conducted
using Lipofectamine® 2000 reagent (Invitrogen; Thermo
Fisher Scientific, Inc.), according to the manufacturer's
protocol.
RT-qPCR
Total mRNA was isolated from the transfected cell
groups and untransfected cells, as previously described (30). Complementary DNA (cDNA) was produced
by reverse transcription using an iScript™ cDNA Synthesis kit
(Bio-Rad Laboratories, Inc., Hercules, CA, USA), according to the
manufacturer's instructions. Levels of mRNA expression were
measured by SYBR-Green-based qPCR using a SYBR® Green
Master mix (Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. The primer sequences used were as follows:
Nrf2, forward 5′-ATGGATTTGATTGACATACTTT-3′ and reverse
5′-ACTGAGCCTGATTAGTAGCAAT-3′; and GAPDH, forward
5′-TCCTGCACCACCAACTGCTTAG-3′ and reverse
5′-ATGGGCAGTGATGGCATGGACT-3′. qPCR conditions were as follows:
Initial denaturation was performed at 95°C for 15 sec, followed by
30 cycles of 95°C for 30 sec, 61°C for 5 sec, 72°C for 15 sec, and
a final extension at 72°C for 10 min. GAPDH gene expression was
used as a reference. mRNA expression levels were quantified using
the 2−ΔΔCq method (31).
Cell viability assay
The transfected HTM and GTM cells during the
logarithmic growth phase were cultured in 96-well plates with DMEM
(5×104 cells/ml) at 37°C. Four repeat cultures were
completed for each cell transfection group. Following culture for
24, 48, 72 and 96 h, 20 µl fresh DMEM supplemented with 0.5 mg/ml
MTT (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added to
each well and incubated for 4 h at 37°C. A total of 200 µl dimethyl
sulfoxide (DMSO; Sigma-Aldrich; Merck KGaA) was added to each well.
Cells incubated with just DMSO were used as controls. Optical
densities of each well were subsequently measured at an absorbance
of 492 nm.
Apoptosis assay
Cells transfected with siNrf2 for 28 h were seeded
in a 6-well plate at a density of 2×105 cells/well in
DMEM. Cell apoptosis was assayed using the FITC Annexin V Apoptosis
Detection kit (Beijing Biosynthesis Biotechnology Co., Ltd.,
Beijing, China), according to the manufacturer's instructions.
Stained cells were analyzed with a fluorescence-activated cell
sorting Calibur flow cytometer (BD Biosciences, San Jose, CA, USA).
The data were analyzed using FlowJo v. 9.0 software (Tree Star,
Inc., Ashland, OR, USA). The percentage of total apoptotic events
was defined as the sum of cells in the early (Annexin V positive/PI
negative) and late (Annexin V positive/PI positive) stages of
apoptosis, as described previously (32).
Western blot analysis
Protein was extracted from cells transfected with
Nrf2, siNrf2 or control using radioimmunoprecipitation assay buffer
(Beyotime Institute of Biotechnology, Shanghai, China). Protein
samples (30 µg/lane) were separated by 10–12% SDS-PAGE, blotted
onto polyvinylidene difluoride membranes, blocked in 5% fresh
non-fat milk in phosphate-buffered saline (PBS)-Triton X-100 (0.1%
Triton in PBS) for 1 h at room temperature. Subsequently, the
membranes were incubated with primary antibodies: Nrf2 (ab31163),
BCL2-Associated X (Bax; ab32503), B-cell lymphoma (BCL)-2
(ab32124), p53 (ab1101), phospho (p)-p53 (ab1431) and GAPDH
(ab8245; all Abcam, Cambridge, USA; all 1:1,000) overnight at 4°C.
Thereafter, membranes were incubated with corresponding horseradish
peroxidase-conjugated secondary antibodies (ab6721; ab6788;
1:5,000; Abcam) for 1 h at room temperature. Immunoreactive protein
bands were developed by enhanced chemiluminescence western blotting
substrate (Pierce; Thermo Fisher Scientific, Inc.) and analyzed
using Image Gauge v. 4.0 software (FujiFilm Science Lab, Tokyo,
Japan).
Statistical analysis
All experiments were repeated three times. Results
are presented as the mean ± standard deviation. Statistical
analyses were performed using SPSS 19.0 software (IBM SPSS, Armonk,
NY, USA). P-values were calculated using one-way analysis of
variance and P<0.05 was considered to indicate a statistically
significant result.
Results
Nrf2 expression is upregulated in HTM
cells
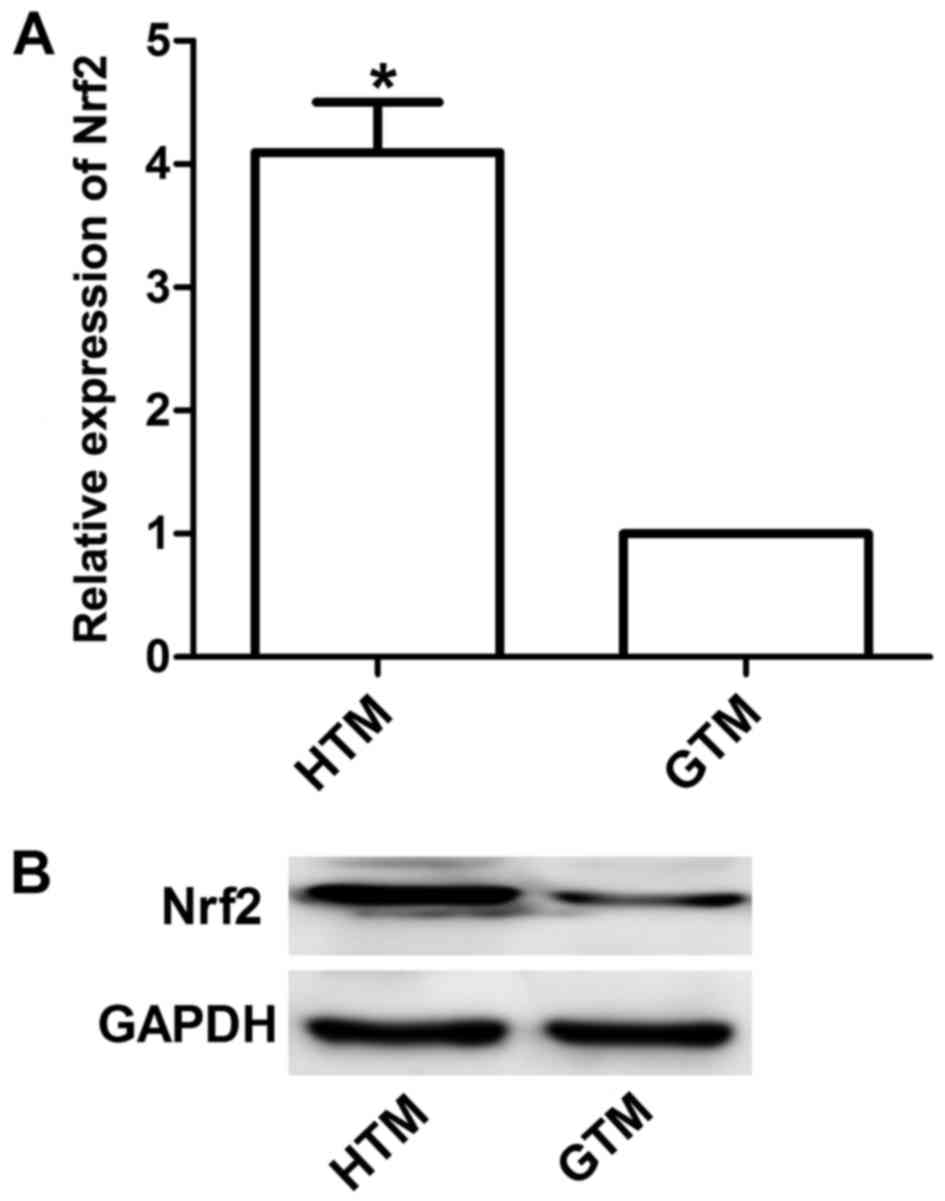
Western blotting and RT-qPCR were performed to
detect Nrf2 expression. Results demonstrated that the expression of
Nrf2 was significantly upregulated in HTM cells compared with GTM
cells (P<0.05; Fig. 1). This was
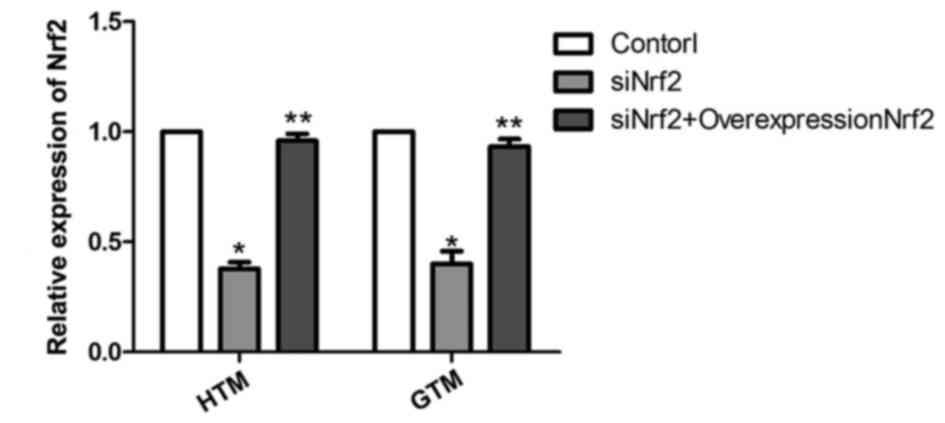
demonstrated to be significant at the mRNA level by subsequent
analysis of HTM and GTM cells transfected with siNrf2 to
downregulate Nrf2 expression. As depicted in Fig. 2, the mRNA expression levels of Nrf2
were effectively regulated by siNrf2, with significant decreases in
Nrf2 observed in siNrf2 transfectants compared with the control
(P<0.05). In addition, the mRNA expression levels of Nrf2
overexpression plasmid together with siNrf2 were significantly
upregulated in HTM and GTM cells compared with cells transfected
with siNrf2 alone (P<0.01).
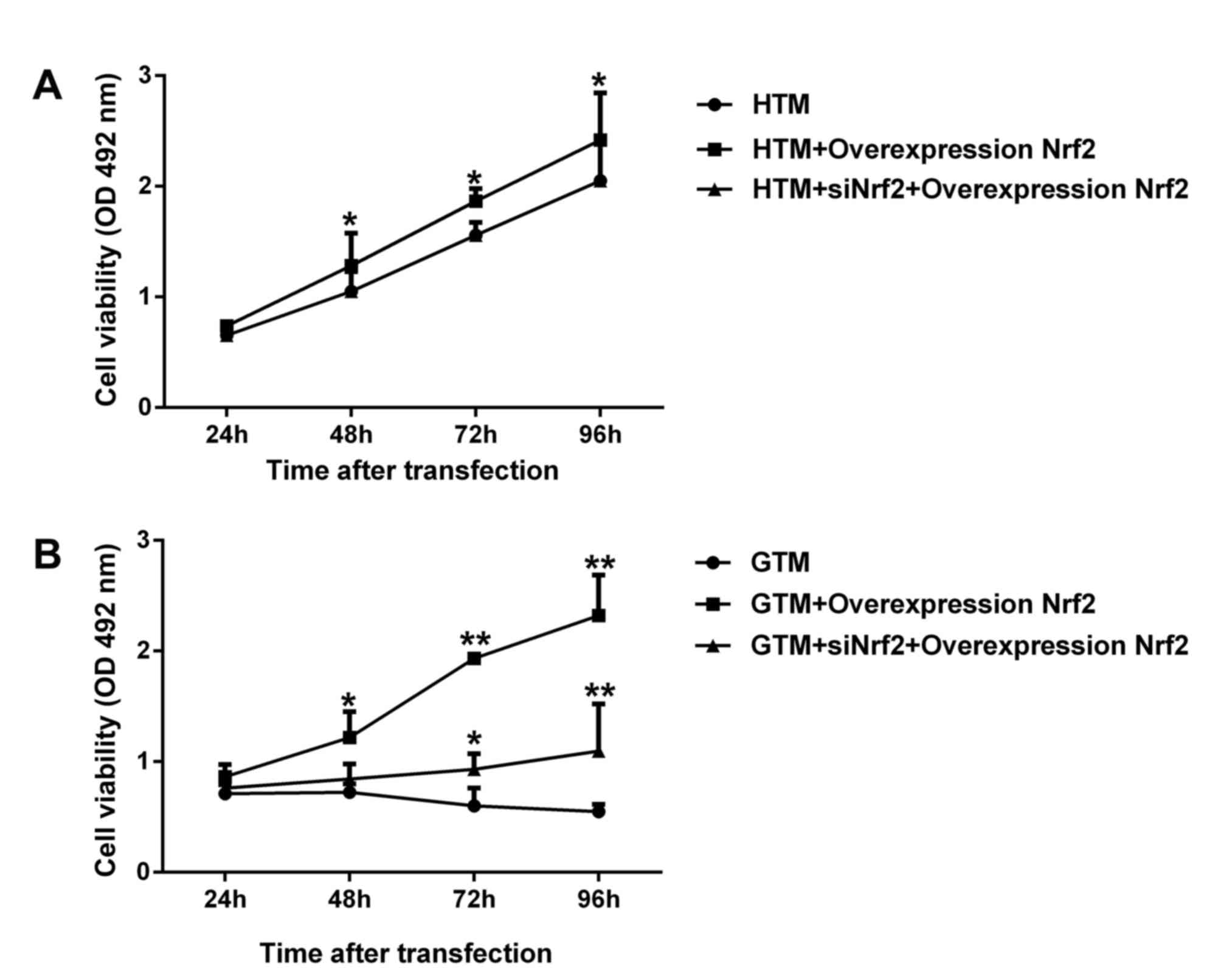
Nrf2 increases the viability of HTM
and GTM cells
To determine the effect of Nrf2 expression on GTM
and HTM cell viability, cells were transfected with Nrf2
overexpression plasmid alone and Nrf2 overexpression together with
siNrf2. As demonstrated in Fig. 3A,
regulation of Nrf2 expression significantly enhanced the viability
of GTM and HTM cells. In HTM cells, overexpression Nrf2
significantly improved cell viability (P<0.05), while Nrf2
overexpression together with siNrf2 demonstrated no significant
change compared with the respective non-transfected control. In GTM
cells, overexpression of Nrf2 and Nrf2 overexpression together with
siNrf2 significantly promoted cell viability (P<0.05; Fig. 3B). These results indicated that the
greatest increases in GTM and HTM cell viability were observed in
Nrf2 overexpression plasmid alone. Interestingly, the viability of
untransfected GTM cells decreased after 48 h, while all other cell
groups remained in a proliferative phase throughout the 96-h assay
period.
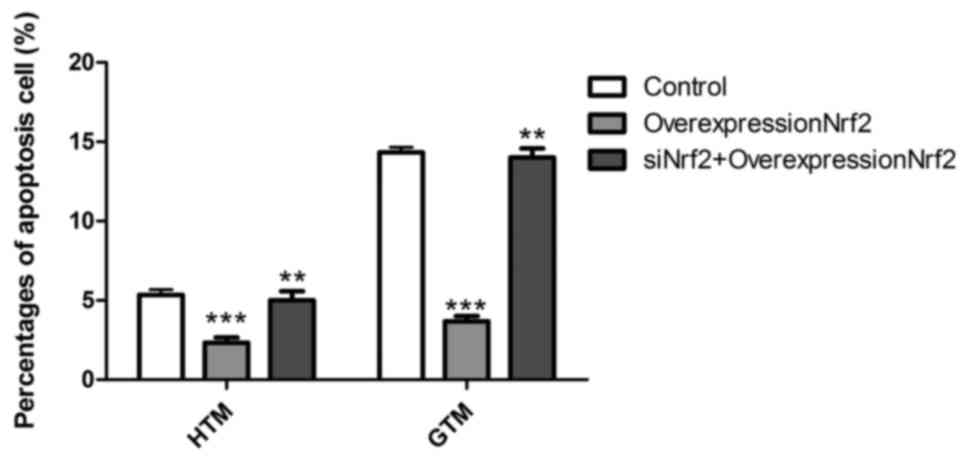
Nrf2 decreases the apoptotic rate of
HTM and GTM cells
To determine the effect of Nrf2 expression on GTM
and HTM cell apoptosis, cells were transfected with Nrf2
overexpression plasmid alone and in combination with siNrf2. As
depicted in Fig. 4, overexpression
of Nrf2 significantly decreased the rate of apoptosis in GTM and
HTM cells (P<0.001). This effect was most prominent in GTM
cells, due to their higher starting rate of apoptosis, when
compared to HTM cells. In turn, transfection overexpression Nrf2
together with siNrf2 significantly reversed the lowered rate of
apoptosis than Nrf2 overexpressing cells (P<0.01). These results
suggest that Nrf2 may regulate GTM and HTM cells through induction
of apoptosis.
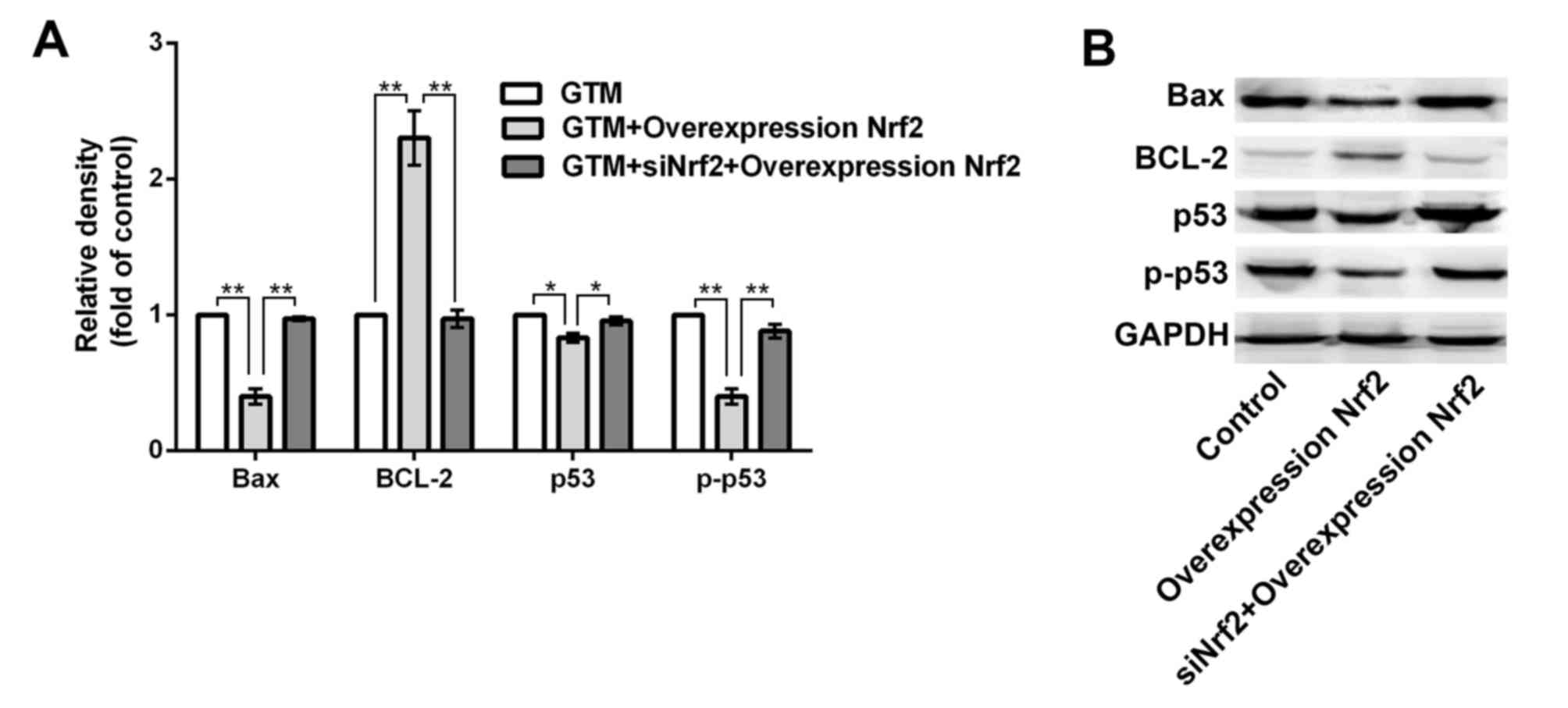
Nrf2 regulates the expression of
apoptosis-related proteins
BCL-2, bcl-2-like protein 4 (Bax), tumor suppressor
protein p53 and phosphorylated (p)-p53 are all apoptosis-related
proteins, with BCL-2 considered to be a key anti-apoptotic factor
(33,34). Therefore, to determine whether Nrf2
regulates the expression of apoptosis-related proteins, levels of
BCL-2, Bax, p53 and p-p53 were evaluated by western blotting and
densitometric analysis following Nrf2 overexpression in GTM cells.
As depicted in Fig. 5,
overexpression of Nrf2 significantly upregulated BCL-2 (P<0.01),
while significantly downregulating Bax, p53 and p-p53 expression
(P<0.01), relative to control cells. In turn, transfection with
siNrf2 significantly reversed the altered expression of BCL-2
(P<0.01), Bax (P<0.01), p53 (P<0.05) and p-p53 (P<0.01)
compared with Nrf2 overexpressing cells.
Discussion
Glaucoma is a retinal neuropathy that can lead to
permanent blindness, and is associated with elevated intraocular
pressure (IOP), due to fibrosis and degeneration of the trabecular
meshwork (35). It is the second
leading cause of progressive vision loss and is expected to affect
80 million people worldwide by 2020 (36). The etiology of glaucoma is complex
and is generally considered to arise due to a combination of
factors, including genes, age and environmental factors (37).
Several genes have been identified as contributing
factors (38). Although numerous
studies have been conducted, the mechanisms underlying glaucoma
remain unknown. Due to its location, the trabecular meshwork serves
a key role in aqueous fluid circulation, thus dysfunction of the
trabecular meshwork may be a key contributing factor in the onset
of primary open-angle glaucoma (39).
In addition, the trabecular meshwork is considered
to be involved in the regulation of IOP, due to observations that
glaucoma trabecular meshwork cells undergo increased rates of
apoptosis in a high IOP state (40).
Nrf2 is a key transcription factor in the regulation of antioxidant
and Phase II detoxification gene expression, and is activated by
oxidative stress and the presence of electrophiles. It has been
observed that Nrf2 exerts protective effects in both normal and
cancer cells during cell stress, thereby serving key roles in the
development of cancer, including gastric, and skin cancer (41–45). It
is considered that Nrf2 protects cells from oxidative stress
through overproduction of antioxidants and detoxification proteins
(46,47). Ran et al (48) demonstrated that the regulatory
effects of Nrf2 on microRNA-29b expression influenced the
proliferation of Tenon's capsule fibroblasts obtained from patients
with glaucoma. Sun et al (49) also observed that NRF2 may determine
the therapeutic response of hepatocellular carcinoma cells to
ferroptosis-targeted therapies.
To determine the underlying mechanisms regarding the
effects of Nrf2 on trabecular meshwork cells, particularly during
glaucoma, the present study evaluated cell behaviors associated
with Nrf2 expression. Levels of Nrf2 in HTM and GTM cells were
evaluated by western blotting and RT-qPCR, with observations that
Nrf2 was downregulated in GTM cells relative to HTM cells. To
elucidate the role of Nrf2 in GTM cell apoptosis, levels of Nrf2
were subsequently regulated using siNrf2 and overexpression
plasmid, and it was observed that overexpression of Nrf2 stimulated
proliferation and inhibited apoptosis in GTM and HTM cells.
The current study demonstrated that overexpression
of Nrf2 had regulatory effects on the expression of
apoptosis-related proteins. Upregulation of the anti-apoptotic
factor BCL-2, and downregulation of Bax, p53 and p-p53 were all
observed following Nrf2 overexpression. Collectively, these
findings suggest that Nrf2 serves a key role in the regulation of
trabecular meshwork cells in glaucoma. Specifically, overexpression
of Nrf2 may attenuate apoptosis of GTM cells by regulating
apoptosis-related proteins.
In conclusion, the present results indicate a novel
role of Nrf2 within trabecular meshwork cells during glaucoma and
may offer insight into the underlying mechanisms of glaucoma. It
was principally demonstrated that Nrf2 may have regulatory effects
on trabecular meshwork cell apoptosis, suggesting that Nrf2 is a
potential therapeutic target in the prevention and treatment of
glaucoma.
References
|
1
|
Akram MU, Tariq A, Khalid S, Javed MY,
Abbas S and Yasin UU: Glaucoma detection using novel opticdisc
localization, hybrid feature set and classification techniques.
Australas Phys Eng Sci Med. 38:643–655. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hua Z, Fang Q, Sha X, Yang R and Hong Z:
Role of retinal nerve fiber layer thickness and optic disk
measurement by OCT on early diagnosis of glaucoma. Eye Sci.
30:7–12. 2015.PubMed/NCBI
|
|
3
|
Pinchuk L, Riss I, Batlle JF, Kato YP,
Martin JB, Arrieta E, Palmberg P, Parrish RK II, Weber BA, Kwon Y
and Parel JM: The development of a micro-shunt made from poly
(styrene-block-isobutylene-block-styrene) to treat glaucoma. J
Biomed Mater Res B Appl Biomater. 105:211–221. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wilson GN, Inman DM, Denger-Crish CM,
Smith MA and Crish SD: Early pro-inflammatory cytokine elevations
in the DBA/2J mouse model of glaucoma. J Neuroinflammation.
12:1762015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nuschke AC, Farrell SR, Levesque JM and
Chauhan BC: Assessment of retinal ganglion cell damage in
glaucomatous optic neuropathy: Axon transport, injury and soma los.
Exp Eye Res. 141:111–124. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vidal-Sanz M, Salinas-Navarro M,
Nadal-Nicolás FM, Alarcón-Martínez L, Valiente-Soriano FJ, De
Imperial JM, Avilés-Trigueros M, Agudo-Barriuso M and
Villegas-Pérez MP: Understanding glaucomatous damage: Anatomical
and functional data from ocular hypertensive rodent retinas. Prog
Retin Eye Res. 31:1–27. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baskaran M, Foo RC, Cheng CY,
Narayanaswamy AK, Zheng YF, Wu R, Saw SM, Foster PJ, Wong TY and
Aung T: The prevalence and types of glaucoma in an urban Chinese
population: The Singapore Chinese eye study. JAMA Ophthalmol.
133:874–880. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Song W, Shan L, Cheng F, Fan P, Zhang L,
Qu W, Zhang Q and Yuan H: Prevalence of glaucoma in a rural
northern China adult population: A population-based survey in kailu
county, inner mongolia. Ophthalmology. 118:1982–1988. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tham YC, Li X, Wong TY, Quigley HA, Aung T
and Cheng CY: Global prevalence of glaucoma and projections of
glaucoma burden through 2040: A systematic review and
meta-analysis. Ophthalmology. 121:2081–2090. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wostyn P, van Dam D, Audenaert K, Killer
HE, De Deyn PP and De Groot V: A new glaucoma hypothesis: A role of
glymphatic system dysfunction. Fluids Barriers CNS. 12:162015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cuchra M, Markiewicz L, Mucha B, Pytel D,
Szymanek K, Szemraj J, Szaflik J, Szaflik JP and Majsterek I: The
role of base excision repair in the development of primary open
angle glaucoma in the Polish population. Mutat Res. 778:26–40.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mousa A, Kondkar AA, Al-Obeidan SA, Azad
TA, Sultan T, Osman E and Abu-Amero KK: Association of total
antioxidants level with glaucoma type and severity. Saudi Med J.
36:671–677. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Almasieh M, Wilson AM, Morquette B, Vargas
JL Cueva and Di Polo A: The molecular basis of retinal ganglion
cell death in glaucoma. Prog Retin Eye Res. 31:152–181. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lebrun-Julien F and Di Polo A: Molecular
and cell-based approaches for neuroprotection in glaucoma. Optom
Vis Sci. 85:417–424. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Doucette LP, Rasnitsyn A, Seifi M and
Walter MA: The interactions of genes, age, and environment in
glaucoma pathogenesis. Surv Ophthalmol. 60:310–326. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu P, Zhang M, Shoeb M, Hogan D, Tang L,
Syed MF, Wang CZ, Campbell GA and Ansari NH: Metal chelator
combined with permeability enhancer ameliorates oxidative
stress-associated neurodegeneration in rat eyes with elevated
intraocular pressure. Free Radic Biol Med. 69:289–299. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Vohra R, Tsai JC and Kolko M: The role of
inflammation in the pathogenesis of glaucoma. Surv Ophthalmol.
58:311–320. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nickells RW, Howell GR, Soto I and John
SW: Under pressure: Cellular and molecular responses during
glaucoma, a common neurodegeneration with axonopathy. Annu Rev
Neurosci. 35:153–179. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Motallebipour M, Rada-Iglesias A, Jansson
M and Wadelius C: The promoter of inducible nitric oxide synthase
implicated in glaucoma based on genetic analysis and nuclear factor
binding. Mol Vis. 11:950–957. 2005.PubMed/NCBI
|
|
20
|
Itakura T, Peters DM and Fini ME:
Glaucomatous MYOC mutations activate the IL-1/NF-κB inflammatory
stress response and the glaucoma marker SELE in trabecular meshwork
cells. Mol Vis. 21:1071–1084. 2015.PubMed/NCBI
|
|
21
|
Chen WS, Cao Z, Krishnan C and Panjwani N:
Verteporfin without light stimulation inhibits YAP activation in
trabecular meshwork cells: Implications for glaucoma treatment.
Biochem Biophys Res Commun. 466:221–225. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pattabiraman PP and Rao PV: Hic-5
regulates actin cytoskeletal reorganization and expression of
fibrogenic markers and myocilin intrabecular meshwork cells. Invest
Ophthalmol Vis Sci. 56:5656–5669. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stothert AR, Fontaine SN, Sabbagh JJ and
Dickey CA: Targeting the ER-autophagy system in the trabecular
meshwork to treat glaucoma. Exp Eye Res. 144:38–45. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Morgan JT, Raghunathan VK, Chang YR,
Murphy CJ and Russell P: The intrinsic stiffness of human
trabecular meshwork cells increases with senescence. Oncotarget.
6:15362–15374. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sachdeva MM, Cano M and Handa JT: Nrf2
signaling is impaired in the aging RPE given an oxidative insult.
Exp Eye Res. 119:111–114. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ruiz S, Pergola PE, Zager RA and Vaziri
ND: Targeting the transcription factor Nrf2 to ameliorate oxidative
stress and inflammation in chronic kidney disease. Kidney Int.
83:1029–1041. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu Z, Wei Y, Gong J, Cho H, Park JK, Sung
ER, Huang H, Wu L, Eberhart C, Handa JT, et al: Nrf2 plays a
protective role in diabetic retinopathy in mice. Diabetologia.
57:204–213. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ko SY, Chang SS, Lin IH and Chen HI:
Suppression of antioxidant Nrf-2 and downstream pathway in H9c2
cells by advanced glycation end products (AGEs) via ERK
phosphorylation. Biochimie. 118:8–14. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Njayou FN, Amougou AM, Tsayem R Fouemene,
Manjia J Njikam, Rudraiah S, Bradley B, Manautou JE and Fewou
Moundipa P: Antioxidant fractions of Khaya grandifoliola C.DC. and
Entada africana Guill. et Perr. induce nuclear translocation of
Nrf2 in HC-04 cells. Cell Stress Chaperones. 20:991–1000. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fransson L, Rosengren V, Saha TK,
Grankvist N, Islam T, Honkanen RE, Sjöholm Å and Ortsäter H:
Mitogen-activated protein kinases and protein phosphatase 5 mediate
glucocorticoid-induced cytotoxicity in pancreatic islets and
β-cells. Mol Cell Endocrinol. 383:126–136. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jung HS, Rajasekaran N, Song SY, Kim YD,
Hong S, Choi HJ, Kim YS, Choi JS, Choi YL and Shin YK: Human
papillomavirus E6/E7-specific siRNA potentiates the effect of
radiotherapy for cervical cancer in vitro and in vivo. Int J Mol
Sci. 16:12243–12260. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Qin YU, Wang H, Liu ZY, Liu J and Wu JZ:
Realgar quantum dots induce apoptosis and necrosis in HepG2 cells
through endoplasmic reticulum stress. Biomed Rep. 3:657–662.
2015.PubMed/NCBI
|
|
34
|
Lee JY, Jee SB, Park WY, Choi YJ, Kim B
and Kim YH, Jun do Y and Kim YH: Tumor suppressor protein p53
promotes 2-methoxyestradiol-induced activation of Bak and Bax,
leading to mitochondria-dependent apoptosis in human colon cancer
HCT116 cells. J Microbiol Biotechnol. 24:1654–1663. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wordinger RJ, Sharma T and Clark AF: The
role of TGF-β2 and bone morphogenetic proteins in the trabecular
meshwork and glaucoma. J Ocul Pharmacol Ther. 30:154–162. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen X, Xu Y, Duan L, Zhang Z, Wong DWK
and Liu J: Multiple ocular diseases detection by graph regularized
multi-label learning. 2014.
|
|
37
|
Casson RJ, Chidlow G, Ebneter A, Wood JP,
Crowston J and Goldberg I: Translational neuroprotection research
in glaucoma: A review of definitions and principles. Clin Exp
Ophthalmol. 40:350–357. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Janulevičiene I, Ehrlich R, Siesky B,
Nedzelskienė I and Harris A: Evaluation of hemodynamic parameters
as predictors of glaucoma progression. J Ophthalmol.
2011:1643202011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Crabb JW, Yuan X, Crabb JS, Putliwala TM,
Clark AF and Bollinger KE: Quantitative proteomic studies implicate
mitochondrial dysfunction in the trabecular meshwork in glaucoma
pathology. 2011.
|
|
40
|
Tamm ER, Braunger BM and Fuchshofer R:
Intraocular pressure and the mechanisms involved in resistance of
the aqueous humor flow in the trabecular meshwork outflow pathways.
Prog Mol Biol Transl Sci. 134:301–314. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zheng H, Nong Z and Lu G: Correlation
between nuclear factor E2-related factor 2 expression and gastric
cancer progression. Med Sci Monit. 21:2893–2899. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Leone A, Roca MS, Ciardiello C,
Terranova-Barberio M, Vitagliano C, Ciliberto G, Mancini R, Di
Gennaro E, Bruzzese F and Budillon A: Vorinostat synergizes with
EGFR inhibitors in NSCLC cells by increasing ROS via up-regulation
of the major mitochondrial porin VDAC1 and modulation of the
c-Myc-NRF2-KEAP1 pathway. Free Radic Biol Med. 89:287–299. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang D, Ma Y, Yang X, Xu X, Zhao Y, Zhu Z,
Wang X, Deng H, Li C, Gao F, et al: Hypermethylation of the Keap1
gene inactivates its function, promotes Nrf2 nuclear accumulation,
and is involved in arsenite-induced human keratinocyte
transformation. Free Radic Biol Med. 89:209–219. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Rushworth SA, Bowles KM and MacEwan DJ:
High basal nuclear levels of Nrf2 in acute myeloid leukemia reduces
sensitivity to proteasome inhibitors. Cancer Res. 71:1999–2009.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ohnuma T, Anzai E, Suzuki Y, Shimoda M,
Saito S, Nishiyama T, Ogura K and Hiratsuka A: Selective
antagonization of activated Nrf2 and inhibition of cancer cell
proliferation by procyanidins from Cinnamomi Cortex extract. Arch
Biochem Biophys. 585:17–24. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Pastorek M, Müller P and Vojtěšek B: Nrf2-
two faces of antioxidant system regulation. Klin Onkol. 28 Suppl
2:2S26–2S31. 2015.(In Czech). View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Tertil M, Golda S, Skrzypek K, Florczyk U,
Weglarczyk K, Kotlinowski J, Maleszewska M, Czauderna S, Pichon C,
Kieda C, et al: Nrf2-heme oxygenase-1 axis in mucoepidermoid
carcinoma of the lung: Antitumoral effects associated with
down-regulation of matrix metalloproteinases. Free Radic Biol Med.
89:147–157. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ran W, Zhu D and Feng Q: TGF-β2 stimulates
Tenon's capsule fibroblast proliferation in patients with glaucoma
via suppression of miR-29b expression regulated by Nrf2. Int J Clin
Exp Pathol. 8:4799–4806. 2015.PubMed/NCBI
|
|
49
|
Sun X, Ou Z, Chen R, Niu X, Chen D, Kang R
and Tang D: Activation of the p62-Keap1-NRF2 pathway protects
against ferroptosis in hepatocellular carcinoma cells. Hepatology.
63:173–184. 2016. View Article : Google Scholar : PubMed/NCBI
|