Introduction
Cervical cancer, the third most common malignant
tumor type endangering women's health, has a high incidence rate in
developing countries due to limitation of medical treatment for
health conditions and lack of cervical cancer screening (1,2). An
annual estimate of 10,000 cases are newly diagnosed and 30,000
succumb to cervical cancer in China (3). With the development of detection
methods, early-stage diagnosis, which has an important role in the
prevention and treatment of cancer, has been successfully performed
and been of benefit to the patients.
The implementation of the Pap smear test, a single
method to screen for cervical cancer, has decreased the incidence
rate of this malignancy. However, due to the low coverage, low
dependence, high specificity subjective judgment and the discomfort
of the patients, the method is not widely used in the clinic
(4–7). The Thinprep liquid-based cytological
test (TCT) provides the screening efficiency of cervical cytology
with a better medium for cervical cells and avoidance of the
disadvantages of the Pap smear test. However, cytological screening
cannot be widely used for the prevention and treatment of cervical
cancer, due to a series of weaknesses, such as limitation of
morphological examination, tiredness of vision as well as low
sensitivity and specificity (8,9).
In the past decade, cervical cancer was evidenced to
be closely correlated with HR-HPV infection (10). HPV testing has been approved to be a
reasonable choice and to be widely used in the screening of
cervical pre-cancerous diseases (11,12). The
risk of disease also depends on HPV types, along with the patient's
genotype and environmental factors such as smoking (10). To date, >200 types of HPV have
been recognized and it has been suggested that HR-HPV including
types 16, 18, are able to induce cervical cancer, particularly in
patients with chronic HR-HPV infection (13).
More than 50 years of experience in the US and
European countries have revealed that cytological screening on its
own or combined with HPV testing for feedback obviously decreased
the incidence rate of cervical cancer (14). However, HPV testing during the first
line of cervical cancer screening has remained controversial. HPV
testing was reported to be more effective and sensitive than
cytological methods and may replace cytology as the first-line
screening of cervical cancer (15).
However, HPV testing may also have high probability of ignoring
cervical diseases that may be diagnosed by other methods of
detection. The American Cancer Society, the American Society for
Colposcopy and Cervical Pathology and the American Society of
Clinical Pathology have proposed that the combination of cytology
and HPV testing significantly increase the sensitivity in cervical
cancer screens (16). In addition
HR-HPV testing in subjects with abnormal TCT results may increase
the sensitivity of cervical cancer detection and decrease the
missed diagnosis rate (10).
Further studies suggested that the controversial
role of HPV testing in the screening of cervical cancer may be due
to differences in study populations. A meta-analysis revealed that
subjects display significant inter-regional differences (17).
As a screening guide must take several factors and
the real conditions of the district into consideration, the present
study was performed. In 2008, Beijing commenced the screening of
women for cervical and breast cancer free of charge and implemented
this screening in the entire city in 2009 with good social
feedback. Through this cervical cancer screening, the detection
rate of cervical pre-cancerous lesions and early cervical cancer
was significantly increased (18).
Liaocheng People's Hospital (Lioacheng, China) in Shandong province
began to screen women for cervical and breast cancer in January
2013; this service was provided free of charge in the first
instance and once every two years thereafter. Until January 2014,
nearly 20,000 married women residing in Liaocheng underwent
cervical cancer screening. The present study focused on subjects
with abnormal cervical cytology findings. HR-HPV testing was
performed and an epidemiological analysis was conducted in order to
effectively identify individuals with a high risk of cervical
cancer. The present study may increase the screening efficacy, save
resources and provide data for screening individuals with abnormal
cytology results.
Materials and methods
Ethics statement
All of the testing procedures were in accordance
with the ethical committee of Liaocheng People's Hospital and the
international ethical guidelines for biomedical research involving
humans (19). Patients provided
informed written consent and agreed with their data being included
in the present study.
Study design
According to the plan of Liaocheng (China) to screen
women who were permanent residents for cervical and breast cancer,
Liaocheng People's Hospital (Liaocheng, China) subjected 20,017
women to cervical cytology testing from January 2013 to January
2014, among which 937 cases had abnormal results. Abnormalities on
liquid-based cytology are defined according to the Bethesda 2001
directive and include atypical squamous cell of unknown
significance (ASCUS), atypical glandular cells of unknown
significance (AGCUS), low-grade squamous intraepithelial lesions
(LSIL), high-grade squamous intraepithelial lesion (HSIL), squamous
cell carcinoma (SCC) and adenocarcinoma (ACA). In the present
study, liquid-based cytology results of ASCUS or worse (ASCUS+)
were considered abnormal (20).
Patients with abnormal results were informed by trained staff via
telephone communication, and were advised to undergo further
treatment at the hospital. A file was generated for each of these
patients and an information management platform was built in order
to screen high-risk patients. A total of 785 patients were recalled
for repeated CTC and HR-HPV-DNA testing. Their age ranged from 21
to 65 years with an average age of 36±9 years and none of them were
pregnant.
Cytology testing
The sampling of the secretion specimens was strictly
performed during the non-menstrual period. No operation was
performed within 3 days prior to sampling. For sampling, a cotton
swab was used to obtain a secretion specimen from the cervix by
wiping, and a cervical brush (ThinPrep 2000; Hologic, Inc.,
Beijing, China) was then applied to the cervical mouth and rotated
clockwise for 4–5 circles. The cervical brush head was then put
into a tube containing cell preservation liquid
(PreservCyt® Solution; Hologic, Inc.). Tubes were kept
in an upright position. Cervical liquid-based cytology tests were
then performed by experienced cytology experts at the Department of
Pathology of Liaocheng People's Hospital.
HPV DNA testing
Methods clinically used for HPV testing include
cytology, immunohistochemistry, in situ hybridization, dot
blot hybridization, nucleic acid blotting and polymerase chain
reaction (PCR). In 2014, the US Food and Drug Administration (FDA)
approved Cobas® HPV testing applied on its own in the
first-line screening of women aged ≥25 years (20). This method will decrease the status
of cervical cytology in screening and become a method for
diagnostically stratifying patients. At present, two major HR-HPV
testing methods are used in China: The first one is the Hybrid
Capture 2 HPV test for quantitative testing of HR-HPV DNA, which is
an established standard worldwide, and served as a comparator in
the guidelines for accurate HPV testing methods (21–23). The
second is a gene chip technology-based test, which combines PCR and
reverse dot blot hybridization (24). The two methods are used to collect
information about the probands' HPV infection status of the genital
tract at different time-points, and they are widely used in initial
screens for cervical diseases and follow-up visits.
In the present study, cervical cell samples from
patients were kept for 2 weeks, within which one equal part of the
samples of patients with abnormal cytology findings was transported
to the central laboratory and stored at a temperature of 4°C, where
HPV-DNA testing was performed. Regular HPV testing included HPV-DNA
sampling, DNA amplification, in situ hybridization detection
and data processing with Luminex Data Collector version 1.7
(Luminex Corp., Austin, TX, USA). The procedure was performed
according to the protocol of the Nucleic Acid Genotyping kit for
Human Papillomavirus (cat. no. HPV27) provided by Shanghai Tellgen
Life Science Co., Ltd. (Shanghai, China).
HPV genotype testing
Cervical cell samples were subjected to PCR for
in vitro amplification of HPV DNA using the Nucleic Acid
Genotyping kit for Human Papillomavirus. The Applied
Biosystems® 7500 Real-Time PCR System (Thermo Fisher
Scientific, Waltham, MA, USA) was used as a platform, and according
to the hybridization principle, DNA probes were hybridized on a
low-density gene chip with attached nucleic acid probes. This
experiment can test 27 HPV sub-types, which are divided into 17
high-risk and 10 low-risk HPV types (23). Through analysis of high-risk sub-type
cases, HR-HPV includes HPV16, −18, −31, −33, −35, −39, −45, −51-53,
−56, −58, −59, −66, −26, −68 and −73. Positivity is defined as ≥103
copies/ml HR-HPV DNA. Infection with ≥2 of the 17 types was defined
as a multi-type infection. In the current study, differences in
multi-type HR-HPV infection between groups with different TCT
results were statistically significant.
Statistical analysis
SPSS 18.0 (SPSS, Inc., Chicago, IL, USA) was used
for comparative and descriptive analysis. χ2 testing was
used when comparing more than two groups, and data were compared in
a line × column Table. P<0.05 was considered to indicate a
statistically significant difference.
Results
Cases with abnormal TCT results
Among the 785 cases with abnormal findings on TCT,
repeated examination identified ASCUS/AGC in 478, LSIL in 175, HSIL
in 127, and SCC/ACA in 5. In these groups, the infective rate of
HR-HPV was 50.2, 77.1, 89.0 and 100%, respectively. The HPV
positivity rate was significantly associated with the degree of
cervical cell abnormality according to the TCT (χ2=88.1;
P<0.05; Table I).
 | Table I.Comparison between TCT results and
positivity rate of high-risk human papillomavirus infection. |
Table I.
Comparison between TCT results and
positivity rate of high-risk human papillomavirus infection.
| TCT typing | Total cases (n) | Positive (n) | Negative (n) | Positive rate
(%) | χ2 | P-value |
|---|
| ASCUS/AGC | 478 | 240 | 238 | 50.2 | 88.1 | 0.001 |
| LSIL | 175 | 135 | 40 | 77.1 |
|
|
| HSIL | 127 | 113 | 14 | 89.0 |
|
|
| SCC/ACA |
5 |
5 |
0 | 100.0 |
|
|
| Total | 785 | 493 | 292 | 62.8 |
|
|
Epidemiological distribution of
HR-HPV
Of the 17 genotypes of HR-HPV covered by the testing
kit, all were found in the screened subjects except HPV26. The
infection rate of HR-HPV was 62.8% (493/785). Fig. 1 presents the 5 most frequently
identified HR-HPV infection types, including HPV16 (21.5%,
169/785), HPV52 (12.2%, 96/785), HPV58 (9.8%, 77/785), HPV33 (9.7%,
76/785) and HPV18 (7.5%, 59/785). The HPV infection rate was high
among cases of HSIL and cases of SCC and ACA. Single-type HR-HPV
infection was encountered in 45.0% (353/785), while multi-type
HR-HPV infection (including ≥2 types) was found in 17.8% (140/785).
There were 98 cases of two-type infection, 37 cases of three-type
infection, 2 of four-type infection, 2 of five-type infection and 1
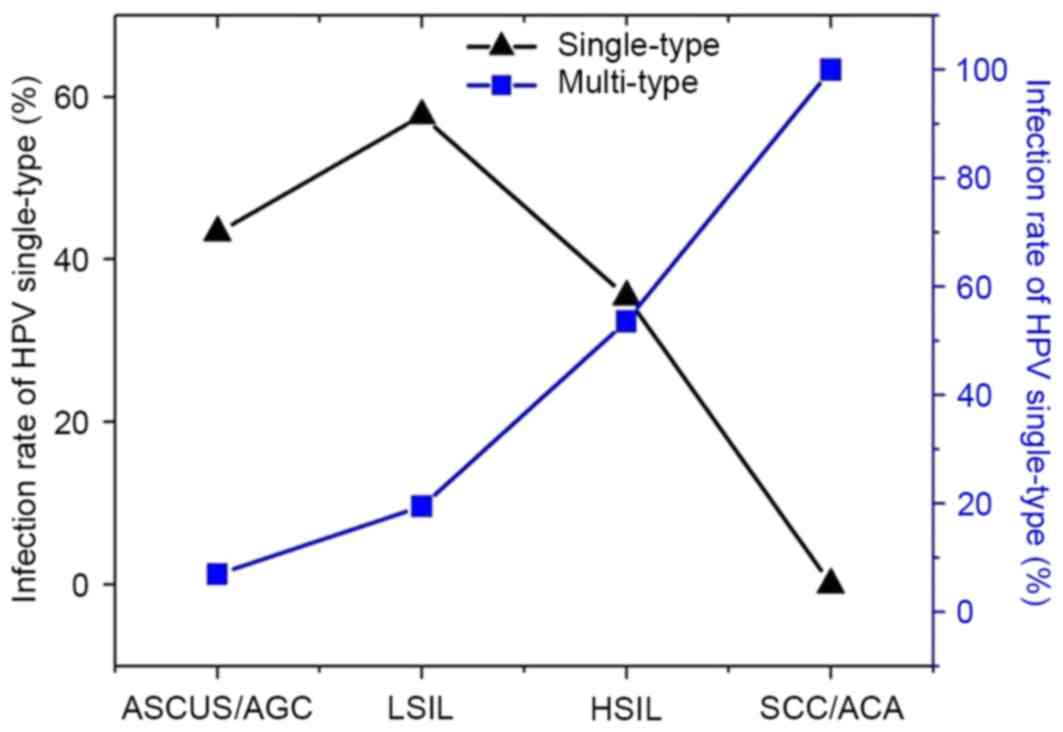
case of six-type infection. As presented in Fig. 1 and Table
II, there were 478 cases of ASCUS and AGC, 207 of which
possessed single-type infection (43.3%) and 33 cases possessed
two-type infection (6.9%). A total of 175 cases of LSIL were
identified, among which 101 cases had single-type infection (57.7%)
and 34 had multi-type infection (19.4%; two- and three-type). There
were 127 cases of HSIL, 45 of which had single-type HR-HPV
infection and 68 had multi-type infection (53.5%; two- and
four-type infection). A total of 5 cases of SCC and ACA were
identified, all of which had multi-type infection; among them, 2
cases were of three-type infection, 1 of four-type, 1 of five-type
and 1 of six-type infection (Tables
II and III, and Fig. 2).
 | Table II.Comparison of infection rate [n, (%)]
of single- and multi-type high-risk human papillomavirus in
subjects with different TCT results in Liaocheng. |
Table II.
Comparison of infection rate [n, (%)]
of single- and multi-type high-risk human papillomavirus in
subjects with different TCT results in Liaocheng.
| TCT type | Cases tested (n) | Single-type | Multi-type | χ2 | P-value |
|---|
| ASCUS/AGC | 478 | 207 (43.3) | 33 (6.9) | 94.7 | <0.01 |
| LSIL | 175 | 101 (57.7) | 34
(19.4)a |
|
|
| HSIL | 127 | 45
(35.4) | 68
(53.5)a,b |
|
|
| SCC/ACA |
5 |
0 (0.0) | 5
(100.0)a,b |
|
|
 | Table III.Epidemiological distribution of
HR-HPV [positive rate in n (%)] for subjects with different TCT
results in Liaocheng. |
Table III.
Epidemiological distribution of
HR-HPV [positive rate in n (%)] for subjects with different TCT
results in Liaocheng.
| HR-HPV type | Screened subjects
(n=785) | ASCUS/AGC
(n=478) | LSIL (n=175) | HSIL (n=127) | SCC/ACA (n=5) |
|---|
| HPV16 | 169 (21.5) | 76 (15.9) | 35 (20) | 54 (42.5) | 4 (80) |
| HPV18 | 59 (7.5) | 21 (4.4) | 19 (10.9) | 15 (11.8) | 4 (80) |
| HPV31 | 36 (4.6) | 15 (3.1) | 6 (3.4) | 14 (11) | 1 (20) |
| HPV33 | 76 (9.7) | 31 (6.5) | 20 (11.4) | 23 (18.1) | 2 (40) |
| HPV35 | 2 (0.3) | 2 (0.4) | 0 (0) | 0 (0) | 0 (0) |
| HPV39 | 9 (1.1) | 9 (1.9) | 0 (0) | 0 (0) | 0 (0) |
| HPV45 | 2 (0.3) | 1 (0.2) | 1 (0.6) | 0 (0) | 0 (0) |
| HPV51 | 24 (3.1) | 9 (1.9) | 3 (1.7) | 12 (9.4) | 0 (0) |
| HPV52 | 96 (12.2) | 28 (5.9) | 25 (14.3) | 40 (31.5) | 3 (60) |
| HPV53 | 23 (2.9) | 14 (2.9) | 6 (3.4) | 2 (1.6) | 1 (20) |
| HPV56 | 51 (6.5) | 20 (4.2) | 15 (8.6) | 15 (11.8) | 1 (20) |
| HPV58 | 77 (9.8) | 24 (5) | 22 (12.6) | 29 (22.8) | 2 (40) |
| HPV59 | 9 (1.1) | 5 (1) | 4 (2.3) | 0 (0) | 0 (0) |
| HPV66 | 18 (2.3) | 6 (1.3) | 9 (5.1) | 2 (1.6) | 1 (20) |
| HPV68 | 24 (3.1) | 10 (2.1) | 11 (6.3) | 2 (1.6) | 1 (20) |
| HPV73 | 2 (0.3) | 2 (0.4) | 0 (0) | 0 (0) | 0 (0) |
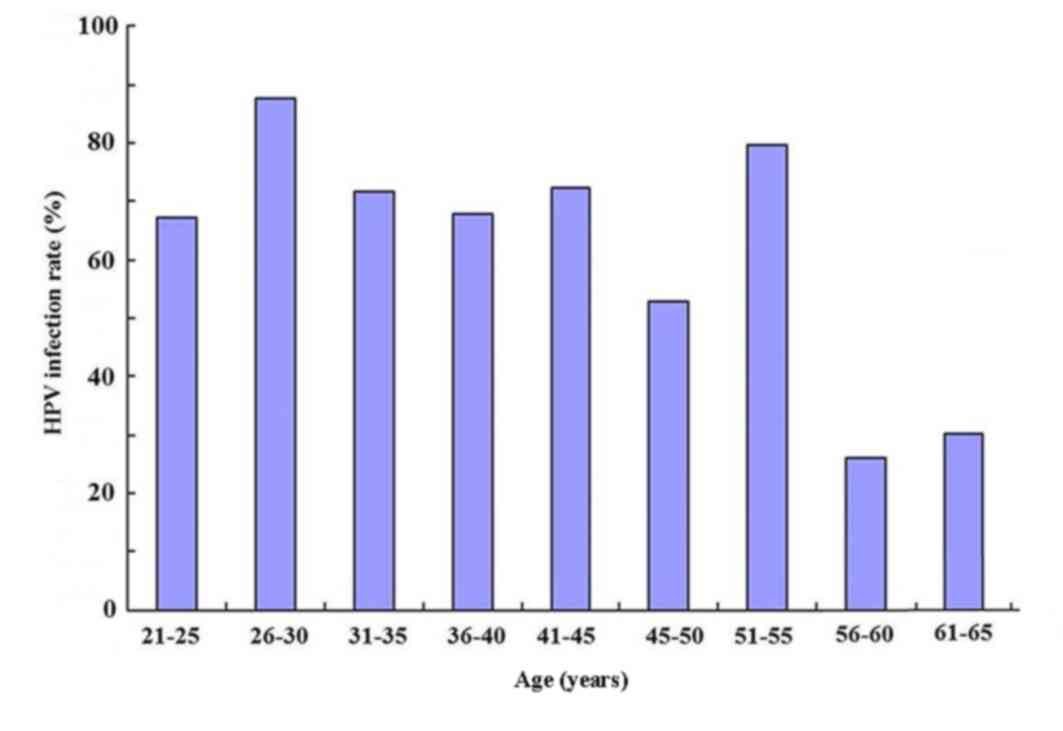
Age distribution of HR-HPV-infected
patients
As presented in Fig.
2, different age groups had a different infection rate of HPV.
χ2 (line × column) testing revealed χ2=377
and the difference was statistically significant (P<0.05).
Further comparison at α=0.0014 demonstrated that the age groups
with the highest infection rates were those of 26–30 and 51–55
years, while the infection rate was lowest in patients aged 56–60
and 61–65 years.
Discussion
Epidemiological and biological data have confirmed
that continuous HR-HPV infection is the main cause of cervical
cancer and its pre-cancerous lesions. There is a dispute with
regard to whether HPV testing should be used as a primary screening
method for cervical cancer. It is worth mentioning that cytology is
not effective in the screening of cervical intraepithelial
neoplasia grade 2+ (CIN2+) lesions. Analysis of European and North
American cases indicated that the sensitivity rate is only 53%,
giving rise to a new era of molecular testing for HPV DNA (24). In May 2003, the US FDA approved the
joint application of TCT and HPV-DNA HC2 testing for the primary
screening of women aged ≥30 years (17). In 2005, the World Health
Organization's International Agency for Research on Cancer (IARC)
recommended HR-HPV testing for use in the primary screening of
cervical cancer (22). Stratified
management of HR-HPV-positive patients is performed according to
cytological testing results. Once women were tested as double
negative, they are not tested for ≥5 years, but a test is mandatory
after 10 years (25); these
guidelines are applied in Liaocheng People's Hospital. Cervical
cancer screening guidelines by the American College of Obstetrics
and Gynecology was published in 2014 proposed that HR-HPV testing
is not recommended for women under the age of 30 years. The reason
is that transient infections are common and even if they test
positive, the infection will be cleared over a period of time. HPV
infection may be transient and only a small number of patients
infected will develop cancer after a long incubation period. As
there are >20 sub-types (13),
testing for single HPV types is not sufficient and a typing test is
necessary. In recent years, the Chinese government has listed
cervical cancer screening for women at appropriate ages as a major
public health service, which is funded by the government.
Therefore, the screening effect was evaluated in different regions,
institutions and job positions, which may promote continuous
improvement of cervical cancer screening work and ensure that the
government's policy is implemented. The present study screened
patients with abnormal TCT findings and subjected them to HR-HPV
testing. It provided an evaluation of the epidemiological
distribution of HPV in the local area and allows for comparison
with previous studies. The age for testing was 21–65 years, with
the lower limit of the age range being earlier than that reported
in recent studies (10,26). The purpose of the present study was
to identify HR-HPV infection rates in patients with different
abnormal TCT results as well as age distribution of infected
patients and common genotypes of HPV in the local area through
joint testing. The results may lead to the improvement of the
effective screening rate of cervical lesions and ensure the future
prevention and control of cervical cancer. In the follow-up study,
patients whose cytology result was ASCUS were not further assessed
in order to avoid unnecessary and intrusive inspection and decrease
vaginal examination for HPV-negative patients. However, this did
not reduce the discovery of CIN2+ and CIN3+ in patients with ASCUS
(27). The diagnostic reliability is
improved by eliminating suspicious ASCUS or low-grade lesions.
Patients who had an increased risk of developing cervical cancer
were differentiated from those with a low risk, which promoted the
appropriate use of vaginal examination and pathological biopsy. In
the follow-up study, screening time was relaxed for women negative
on HPV testing.
Recent random comparisons among follow-up visits for
HPV screening indicated that HPV DNA testing detects more
high-level lesions than cytology (22). HPV-negative women have a low risk of
developing CIN, and screening for them is performed at intervals of
≥5 years (27). This largely reduces
the screening cost as well as the psychological and economic burden
of women screened. The present study found that HPV genotypes in
different stages of cervical pre-cancerous lesions were
significantly different. The infection rate of HR-HPV increased
with the degree of cytological abnormality. This verified that
HR-HPV infection is associated with the development of cervical
lesions and it is the cause of cervical cancer. This result
indicated that HR-HPV infection may predict high-level cervical
lesions and high-degree abnormalities on cytology may suggest a
high probability of HPV infection. Therefore, it is important for
patients with abnormal cytological findings to be subjected to a
HPV typing test. Individual evaluation may be performed in order to
predict the risk of cervical lesions and determine a personal
treatment plan. This is in consistent with the results of previous
studies (28,29).
In the present study, multi-type infection mostly
comprised two-type infection, but hybrid infection of three, five
or six types also occurred. Multi-type HR-HPV infection is more
dangerous than single-type infection with regard to the generation
of cervical cancer and HSIL, which has been demonstrated by
previous studies (30,31). In the present study, differences in
multi-type HR-HPV infection between groups with different TCT
results were statistically significant. Although the impact of
multi-type infection on the molecular mechanism and epidemiology of
cervical cancer remains to be fully elucidated, follow-up visits
for multi-type infection patients should be increased and further
examination should be performed when necessary. The HPV genotype
distribution has inter-regional differences and the 15 common
sub-types reported by the IARC are HPV16, −18, −31, −33, −35, −39,
−45, −51, −52, −56, −58, −59, −66, −68 and −73 (32).
The present study found that HPV16 was common in
Liaocheng (China), followed by HPV52 and −58, while HPV18 was
uncommon. However, HPV52 and −58 were associated with a lower
incidence rate of cervical cancer than HPV18, indicating that HPV18
may have a greater carcinogenic effect or accelerate the
development of cancer to a greater extent than the former
types.
A recent study demonstrated that in a susceptible
population, the 9vHPV vaccine prevented diseases associated with
HPV31, −33, −45, −52 and −58, led to the production of antibodies
against HPV6, −11, −16 and −18 and did not perform worse than the
qHPV vaccine (33). HPV typing
revealed the genotype distribution in the region, which may provide
scientific evidence for future screening and vaccine
development.
A large-scale epidemiological survey identified age
differences among HPV-infected individuals in different regions and
populations (34). In line with
this, the present study also found that the HR-HPV infection rate
differs among different age groups. Among the 785 patients with
abnormal TCT findings, the age groups of 26–30 and 51–55 years had
the highest infection rates of 87.7 and 79.7%, respectively. The
infection rate was low women aged >55 at 28.6%. The rate of HPV
infection is high in women aged >30 years due to frequent sexual
intercourse. However, their immune function is relatively strong,
and most of the HPV infections are only transient and may be
eliminated by the immune system. With increasing age, the function
of the immune system decreases and the HPV infection rate
increases. Changes of estrogen and progesterone levels in the
peri-menopausal period are relatively large and is affect the
metaplasia of the cervical epithelium. HPV replication may be
supported by squamous epithelial metaplasia through its natural
proliferation and differentiation process, thus increasing the HPV
infection rate. Therefore, follow-up visits are important for women
in the peri-menopausal period. A previous study demonstrated that
with the polarization of cervical pre-cancerous lesions regarding
the incidence age of cervical cancer, attention should be paid to
women around the age of 30 years and those after the age of 50
years (35). Follow-up visits should
be increased for HPV-positive patients. This has an important role
in the prevention and treatment of cervical cancer.
In conclusion, the present study focused on the
epidemiology of HPV infection in a population of patients with
abnormal findings on cervical cytology in Liaocheng (China).
According to the screening results, as compared with cytological
screening on its own, HR-HPV typing is an effective screening
method for cervical cancer. Identification of high-risk patients
may strengthen the management of cervical cancer screening in the
region in order to perform individualized treatment. Patients with
abnormalities on cervical cytology in the region had a high
infection rate of HPV16, −52, −58, −33 and −18. Multi-type
infection was found to increase the risk of disease. In the region,
high-risk individuals are mostly aged 26–30 and 51–55 years, and
should therefore be subjected to stringent screening and follow-up
visits. The genotype distribution in the region may provide
scientific evidence for future screenings and vaccine
development.
References
|
1
|
Zheng X, Liang P, Zheng Y, Yi P, Liu Q,
Han J, Huang Y, Zhou Y, Guo J and Li L: Clinical significance of
hTERC gene detection in exfoliated cervical epithelial cells for
cervical lesions. Int J Gynecol Cancer. 23:758–790. 2013.
View Article : Google Scholar
|
|
2
|
Schmeink CE, Bekkers RL, Massuger LF and
Melchers WJ: The potential role of self-sampling for high-risk
human papillomavirus detection in cervical cancer screening. Rev
Med Virol. 21:139–153. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhao R, Zhang WY, Wu MH, Zhang SW, Pan J,
Zhu L, Zhang YP, Li H, Gu YS and Liu XZ: Human papillomavirus
infection in Beijing, People's Republic of China: A
population-based study. Br J cancer. 101:1635–1640. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
McPhail S, Elliss-Brookes L, Shelton J,
Ives A, Greenslade M, Vernon S, Morris EJ and Richards M: Emergency
presentation of cancer and short-term mortality. Br J Cancer.
109:2027–2034. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jemal A, Ward E and Thun M: Declining
death rates reflect progress against cancer. PLoS One. 5:e95842010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Aggarwal P, Batra S, Gandhi G and Zutshi
V: Comparison of Papanicolaou test with visual detection tests in
screening for cervical cancer and developing the optimal strategy
for low resource settings. Int J Gynecol Cancer. 20:862–868. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wentzensen N and Silver MI: Biomarkers for
cervical cancer prevention programs: The long and winding road from
discovery to clinical use. J Low Genit Tract Dis. 20:191–194. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Belinson JL, Hu S, Niyazi M, Pretorius RG,
Wang H, Wen C, Smith JS, Li J, Taddeo FJ, Burchette RJ and Qiao YL:
Prevalence of type-specific human papillomavirus in endocervical,
upper and lower vaginal, perineal and vaginal self-collected
specimens: Implications for vaginal self-collection. Int J cancer.
127:1151–1157. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Belinson JL, Pretorius RG, Enerson C,
Garcia F, Cruz EP, Belinson SE, García E Yeverino and Brainard J:
The Mexican Cervical Cancer Screening Trial (MECCS): Self-sampling
for human papillomavirus with unaided visual inspection as a
secondary screen. Int J Gynecol Cancer. 19:27–32. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Agorastos T, Chatzistamatiou K,
Katsamagkas T, Koliopoulos G, Daponte A, Constantinidis T and
Constantinidis TC; HERMES study group, : Primary screening for
cervical cancer based on high-risk human papillomavirus (HPV)
detection and HPV 16 and HPV 18 genotyping, in comparison to
cytology. PLoS One. 10:e01197552015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ronco G, Dillner J, Elfström KM, Tunesi S,
Snijders PJ, Arbyn M, Kitchener H, Segnan N, Gilham C, Giorgi-Rossi
P, et al: Efficacy of HPV-based screening for prevention of
invasive cervical cancer: Follow-up of four European randomised
controlled trials. Lancet. 383:524–532. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cuzick J, Cadman L, Mesher D, Austin J,
Ashdown-Barr L, Ho L, Terry G, Liddle S, Wright C, Lyons D and
Szarewski A: Comparing the performance of six human papillomavirus
tests in a screening population. Br J Cancer. 108:908–913. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bouvard V, Baan R, Straif K, Grosse Y,
Secretan B, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Freeman C,
Galichet L, et al: A review of human carcinogens-Part B: Biological
agents. Lancet Oncol. 10:321–322. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhao C, Li Z, Nayar R, Levi AW, Winkler
BA, Moriarty AT, Barkan GA, Rao J, Miller F, Fan F, et al: Prior
high-risk human papillomavirus testing and Papanicolaou test
results of 70 invasive cervical carcinomas dianosed in 2012:
Results of a retrospective multicenter study. Arch Pathol Lab Med.
139:184–188. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wright TC, Stoler MH, Behrens CM, Sharma
A, Zhang G and Wright TL: Primary cervical cancer screening with
human papillomavirus: End of study results from the ATHENA study
using HPV as the first-line screening test. Gynecol Oncol.
136:189–197. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Saslow D, Solomon D, Lawson HW, Killackey
M, Kulasingam SL, Cain J, Garcia FA, Moriarty AT, Waxman AG, Wilbur
DC, et al: American Cancer Society, American Society for Colposcopy
and Cervical Pathology, and American Society for Clinical Pathology
screening guidelines for the prevention and early detection of
cervical cancer. Am J Clin Pathol. 137:516–542. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schiffman M, Boyle S, Raine-Bennett T,
Katki HA, Gage JC, Wentzensen N, Kornegay JR, Apple R, Aldrich C,
Erlich HA, et al: The role of human papillomavirus genotyping in
cervical cancer screening: A large-scale evaluation of the cobas
HPV test. Cancer Epidemiol Biomarkers Prev. 24:1304–1310. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang YZ, Ma JF, Zhao FH, Xiang XE, Ma ZH,
Shi YT, Hu SY and Qiao YL: Three-year follow-up results of visual
inspection with acetic acid/Lugol's iodine (VIA/VILI) used as an
alternative screening method for cervical cancer in rural areas.
Chin J Cancer. 29:4–8. 2010.(In Chinese). View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Council for International Organizations of
Medical Sciences, . International ethical guidelines for biomedical
research involving human subjects. Bull Med Ethics. 1–23. 2002.
|
|
20
|
Solomon D, Davey D, Kurman R, Moriarty A,
O'Connor D, Prey M, Raab S, Sherman M, Wilbur D, Wright T Jr, et
al: The 2001 bethesda system: Terminology for reporting results of
cervical cytology. JAMA. 287:2114–2119. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Stoler MH, Wright TC Jr, Sharma A, Apple
R, Gutekunst K and Wright TL; ATHENA (Addressing THE Need for
Advanced HPV Diagnostics) HPV Study Group, : High-risk human
papillomavirus testing in women with ASC-US cytology: Results from
the ATHENA HPV study. Am J Clin Pathol. 135:468–475. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cox JT, Castle PE, Behrens CM, Sharma A,
Wright TC Jr and Cuzick J; Athena HPV Study Group, : Comparison of
cervical cancer screening strategies incorporating different
combinations of cytology, HPV testing, and genotyping for HPV
16/18: Results from the ATHENA HPV study. Am J Obstet Gynecol.
208:184.e1–e184.e11. 2013. View Article : Google Scholar
|
|
23
|
Meijer CJ, Berkhof J, Castle PE, Hesselink
AT, Franco EL, Ronco G, Arbyn M, Bosch FX, Cuzick J, Dillner J, et
al: Guidelines for human papillomavirus DNA test requirements for
primary cervical cancer screening in women 30 years and older. Int
J Cancer. 124:516–520. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Söderlund-Strand A, Rymark P, Andersson P,
Dillner J and Dillner L: Comparison between the Hybrid Capture II
test and a PCR-based human papillomavirus detection method for
diagnosis and posttreatment follow-up of cervical intraepithelial
neoplasia. J Clin Microbiol. 43:3260–3266. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Saslow D, Solomon D, Lawson HW, Killackey
M, Kulasingam SL, Cain J, Garcia FA, Moriarty AT, Waxman AG, Wilbur
DC, et al: American Cancer Society, American Society for Colposcopy
and Cervical Pathology and American Society for Clinical Pathology
screening guidelines for the prevention and early detection of
cervical cancer. Am J Clin Pathol. 137:516–542. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Siddiqa A, Zainab M, Qadri I, Bhatti MF
and Parish JL: Prevalence and genotyping of high risk human
papillomavirus in cervical cancer samples from Punjab, Pakistan.
Viruses. 6:2762–2777. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cuzick J, Arbyn M, Sankaranarayanan R, Tsu
V, Ronco G, Mayrand MH, Dillner J and Meijer CJ: Overview of human
papillomavirus-based and other novel options for cervical cancer
screening in developed and developing countries. Vaccine. 26 Suppl
10:K29–K41. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gage JC, Schiffman M, Katki HA, Castle PE,
Fetterman B, Wentzensen N, Poitras NE, Lorey T, Cheung LC and
Kinney WK: Reassurance against future risk of precancer and cancer
conferred by a negative human papillomavirus test. J Natl Cancer
Inst. 106:pii: dju1532014. View Article : Google Scholar
|
|
29
|
Srodon M, Dilworth H Parry and Ronnett BM:
Atypical squamous cells, cannot exclude high-grade squamous
intraepithelial lesion: Diagnostic performance, human
papillomavirus testing, and follow-up results. Cancer. 108:32–38.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Crowe E, Pandeya N, Brotherton JM, Dobson
AJ, Kisely S, Lambert SB and Whiteman DC: Effectiveness of
quadrivalent human papillomavirus vaccine for the prevention of
cervical abnormalities: Case-control study nested within a
population based screening programme in Australia. BMJ.
348:g14582014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Massad LS, Einstein MH, Huh WK, Katki HA,
Kinney WK, Schiffman M, Solomon D, Wentzensen N and Lawson HW:
2012; ASCCP Consensus Guidelines Conference: 2012 updated consensus
guidelines for the management of abnormal cervical cancer screening
tests and cancer precursors. 121:pp. 1–846, 2013.
|
|
32
|
Kitchener HC, Almonte M, Thomson C,
Wheeler P, Sargent A, Stoykova B, Gilham C, Baysson H, Roberts C,
Dowie R, et al: HPV testing in combination with liquid-based
cytology in primary cervical screening (ARTISTIC): A randomised
controlled trial. Lancet Oncol. 10:672–682. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Strander B, Hällgren J and Sparén P:
Effect of ageing on cervical or vaginal cancer in Swedish women
previously treated for cervical intraepithelial neoplasia grade 3:
Population based cohort study of long term incidence and mortality.
BMJ. 348:f73612014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hu Y, Qian HZ, Sun J, Gao L, Yin L, Li X,
Xiao D, Li D, Sun X, Ruan Y, et al: Anal human papillomavirus
infection among HIV-infected and uninfected men who have sex with
men in Beijing, China. J Acquir Immune Defic Syndr. 64:103–114.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ogilvie GS, van Niekerk DJ, Krajden M,
Martin RE, Ehlen TG, Ceballos K, Peacock SJ, Smith LW, Kan L, Cook
DA, et al: A randomized controlled trial of Human Papillomavirus
(HPV) testing for cervical cancer screening: Trial design and
preliminary results (HPV FOCAL Trial). BMC Cancer. 10:1112010.
View Article : Google Scholar : PubMed/NCBI
|