Introduction
Sepsis is a systemic inflammatory response syndrome
caused by infection with bacteria, fungi and/or viruses (1,2).
Although sepsis is caused by infection, the development of sepsis
is based on a series of inflammatory responses and the pathological
processes associated with them (3,4).
Biomarkers in serum may be used to diagnose for patients with
sepsis (5). Although numerous
strategies have been explored for the treatment of sepsis, affected
patients still have an increased risk of mortality (6,7). A
previous study also demonstrated that a reduction of inflammatory
factors significantly improved metabolic parameters that are
beneficial for survival in septic rats (8). In addition, distinct shock phenotypes
were apparent, and the effect of early resuscitation according to
protocols has been examined in selected cohorts of patients with
septic shock (9).
Sepsis is associated with a complex systemic
inflammation, which may lead to immune dysfunction, abnormal
coagulant function and tissue damage caused by infection with
various pathogenic microorganisms, as well as by toxins (10,11).
Various studies suggested that sepsis is closely associated with
the pathological processes of inflammation (12,13),
which may require further elucidation. Sepsis is thought to result
from acute and postponed inflammatory responses via the increasing
of plasma inflammatory cytokines levels and leukocytes in affected
patients (14,15). A previous study also has indicated
the role of inflammatory responses in the progression of sepsis,
suggesting inhibition of inflammatory responses may contribute to
the alleviation of jejunum injury in rats with sepsis (16). In addition, a randomized controlled
trial has suggested that reducing lipopolysaccharide-triggered
inflammation is beneficial for the treatment of human sepsis by
downregulation of the plasma levels of interleukin (IL)-6, IL-8 and
tumor necrosis factor (TNF)-α (17).
Furthermore, Kaplan et al (18) have indicated that reducing
inflammation through inhibition of nuclear factor (NF)-κB is an
efficient strategy for the treatment of sepsis patients. These
studies suggest that inflammation is associated with the
progression of pathology of sepsis and may be potential target for
its treatment.
Systemic inflammatory response syndrome is thought
to be associated with the progression of sepsis patients (19). Therefore, detection of markers of the
inflammatory response may be considered as a way to evaluate the
prognosis of sepsis. The purpose of the present study was to
investigate the expression levels of inflammatory cytokines between
patients with sepsis and healthy individuals. The importance of
inflammatory responses in the evaluation of patients with sepsis
was also investigated.
Materials and methods
Ethical approval and participant
consent
A total of 122 patients with sepsis who were
admitted to the intensive care unit (ICU) at Daqing Oil Field
General Hospital (Daqing, China) without any medication prior to
the determination of the clinical parameters and 106 healthy
individuals (who did not exhibit symptoms of sepsis or
inflammation) were recruited for clinical analysis between January
2014 and August 2015. All patients and healthy volunteers provided
written informed consent. The study was approved by the ethics
committee of Daqing Oil Field General hospital (Daqing, China).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) assay
A total of 15 ml peripheral venous blood was
obtained from each patient with sepsis. Human peripheral blood
monouclear cells (hPBMCs) were separated by density gradient
centrifugation using 10 ml of 0.01% cesium chloride at 4,000 × g
for 30 min at 4°C. Total RNA was extracted from hPBMCs cells using
RNAzol (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany), and
RNase-free DNase was applied to digest total DNA at 37°C for 15
min. The RNeasy kit was then used to purify RNA to adjust its
concentration to 1 µg/µl. A total of 2 µg RNA was used as the
template to synthetize complementary (c)DNA by reacting with
reverse transcriptase at 37°C for 120 min, at 99°C for ٤ min and at
4°C for 3 min using the High Capacity cDNA Reverse Transcription
kit (Thermo Fisher Scientific, Inc., Waltham, MA, USA) according to
the manufacturer's protocol. Subsequently, PCR was employed to
amplify the cDNA of neutrophil gelatinase-associated lipocalin
(NGAL), peptidoglycan recognition protein (PRP), cluster of
differentiation (CD)64, procalcitonin (PCT), NF-κB-p65, inhibitor
of NF-κB (IκBα), IL-1, IL-17, TNF-α and IL-6 using the primers
listed in Table I to determine the
transcription level of the respective mRNA. PCR amplification had
preliminary denaturation at 95°C for 1 min, followed by 40 cycles
of 95°C for 30 sec, 58°C for 30 sec and 72°C for 10 min. The
reaction volume was a total of 20 µl containing 50 ng genomic cDNA,
200 µM dNTPs, 200 µM primers, and Taq DNA polymerase and SYBR-Green
(both 2.5 U; Thermo Fisher Scientific, Inc.). β-Actin was used as
the housekeeping gene for the internal control group. Eventually,
agarose electrophoresis with 1% ethidium bromide was adopted to
check PCR-amplified products. Changes in relative mRNA expression
were calculated by the 2−ΔΔCq method (20). The results are expressed as the fold
change compared with the control.
 | Table I.Sequences of primers used for
polymerase chain reaction. |
Table I.
Sequences of primers used for
polymerase chain reaction.
| Gene name | Reverse (5′-3′) | Forward (5′-3′) |
|---|
| TNF-α |
TCCAGACTTCCTTGAGACA |
GGCGATTACAGACACAACT |
| IL-1 |
GGCTGCTTCCAAACCTTTGA |
GAAGACACGGATTCCATGGT |
| IL-6 |
GTGAGGAACAAGCCAGAG |
TGACCAGAAGAAGGAATGC |
| IL-17 |
ATGCACAGCCACCGCGACTT |
CTTCATGACTGCCTCCAAGTAG |
| PRP |
GCTTGGCACACCTTTTCACATACC |
GTCCTCATTCGGGGCACATTCTG |
| IκBα |
CTTCCTCCTCTTCCTCCTC |
GCCATCTTCACGCTAAGG |
| NF-κB-p65 |
GAGTCAGAGTTCACGGAGTTC |
CATGTTCTTTCAGCCCCTTTG |
| PCT |
GGAGAGCACGCCATGAAG |
AAGATTCGCATGCGGTAGAG |
| β-actin |
CGGAGTCAACGGATTTGGTC |
AGCCTTCTCCATGGTCGTGA |
ELISA
Plasma samples were immediately prepared from
peripheral venous blood by centrifugation (2,000 × g at 4°C for 10
min). Serum levels of TNF-α (cat. no. MBS6080), IL-1 (cat. no.
MBS700340), IL-6 (cat. no. MBS3205), high mobility group box 1
(HMGB1; cat. no. MBS4108) and C-reactive protein (CRP; cat. no.
MBS910284; all from Thermo Fisher Scientific, Inc.) were analyzed
in patients with sepsis using ELISA kits according to the
manufacturer's protocols. The balance of T-helper cell type 1
(Th1)/Th2 cytokines was analysed using Th1/Th2 Human Th1/Th2 (7
plex) Multiplex Immunoassay kit (cat. no. ab213389; Abcam,
Cambridge, UK) according to the manufacturer's protocol. The serum
levels of these cytokines were measured with a micro-plate reader
at 570 nm.
Flow cytometry
From the peripheral blood drawn from the patients
with sepsis, total leukocytes were extracted using a Human
Leukocyte Extract Kit (Invitrogen; Thermo Fisher Scientific, Inc.).
The proportion of lymphocytes, granulocytes and mononuclear cells
among total leukocytes in the plasma was analyzed by flow cytometry
as previously described (21). The
hPBMCs was also incubated with fluorescein isothiocyanate-labelled
CD25 (1:1,000; cat. no. ab210335; Abcam) for 12 h at 4°C. The
proportion of hPBMCs presenting the activation marker CD25 was
analysed by flow cytometry using FCS Express™ 4 IVD (De
Novo Software, Glendale, CA, USA).
Statistical analysis
Statistical analysis was performed using SPSS 19.0
software (IBM Corp., Armonk, NY, USA) and Excel (version 2010;
Microsoft Corp., Redmond, WA, USA). All values are expressed as the
mean ± standard error of the mean of triplicate experiments.
Differences between groups were analyzed using Student's two-tailed
t-test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Characteristics of patients with
sepsis
A total of 122 patients with sepsis and 106 healthy
individuals were recruited for the present clinical analysis. The
ratios of males and females among the groups of sepsis patients and
healthy individuals were approximately equal. The mean age was
45.4±18.3 and 47.2±١8.6 years in sepsis patients and healthy
individuals, respectively. The characteristics of patients with
sepsis are summarized in Table II.
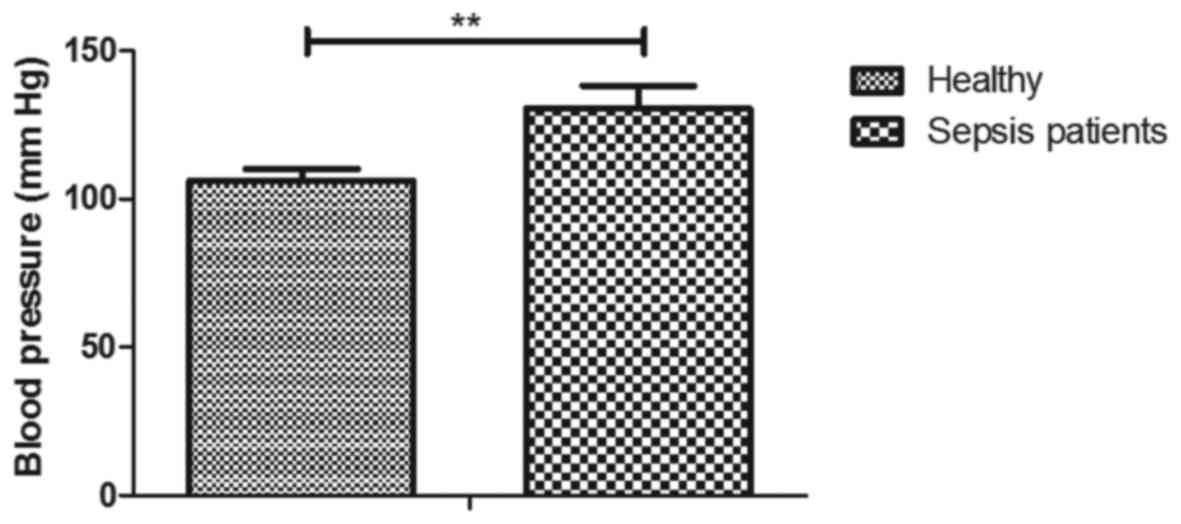
The blood pressure was higher in sepsis patients than that in
healthy individuals (130 vs. 106 mm Hg; P<0.01; Fig. 1).
 | Table II.Characteristics of patients with
sepsis at an intensive care unit. |
Table II.
Characteristics of patients with
sepsis at an intensive care unit.
| Characteristic | Patients with
sepsis | Healthy
individuals |
|---|
| Number | 122 | 106 |
| Age (years) |
45.4±18.3 | 47.2±18.6 |
| Male/female | 60/62 | 50/56 |
| APACHE II
score | 15.2±7.8 | 0 |
| Type of
infection |
|
|
| Acute
pyelonephritis | 28 | 0 |
|
Intra-abdominal infection | 32 | 0 |
| Lung
infection | 29 | 0 |
|
Bloodstream infection | 33 | 0 |
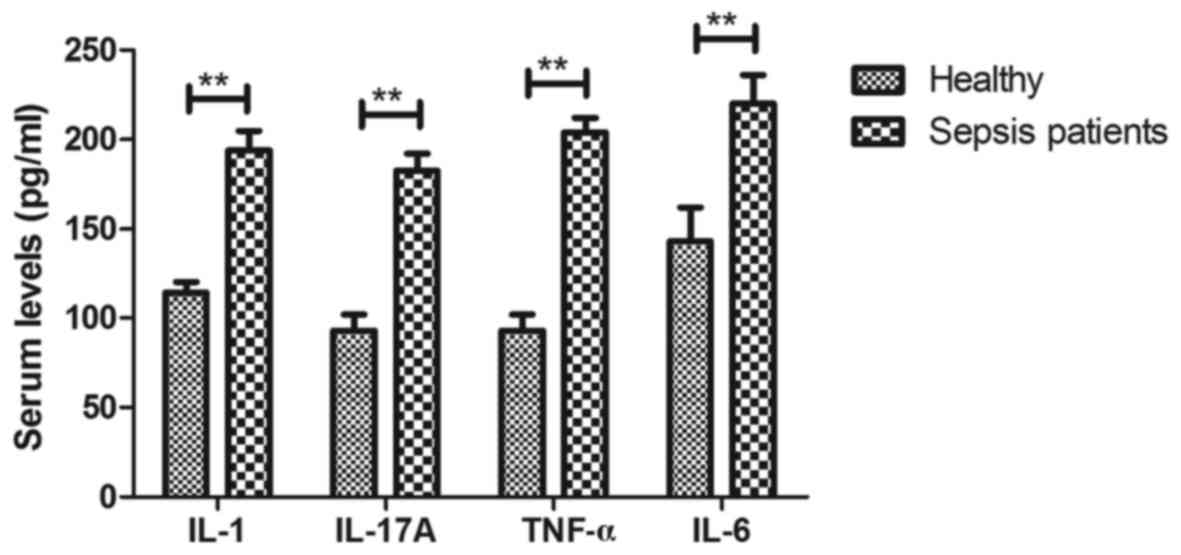
Inflammatory cytokines are increased
in serum of patients with sepsis at the ICU
Changes of inflammatory cytokines in the serum of
patients with sepsis were analyzed. The serum levels of IL-1,
IL-17, TNF-α and IL-6 were identified to be upregulated in sepsis
patients compared with those in healthy individuals (P<0.01;
Fig. 2). These results suggest that
inflammatory cytokines were obviously upregulated in the serum of
patients with sepsis at the ICU.
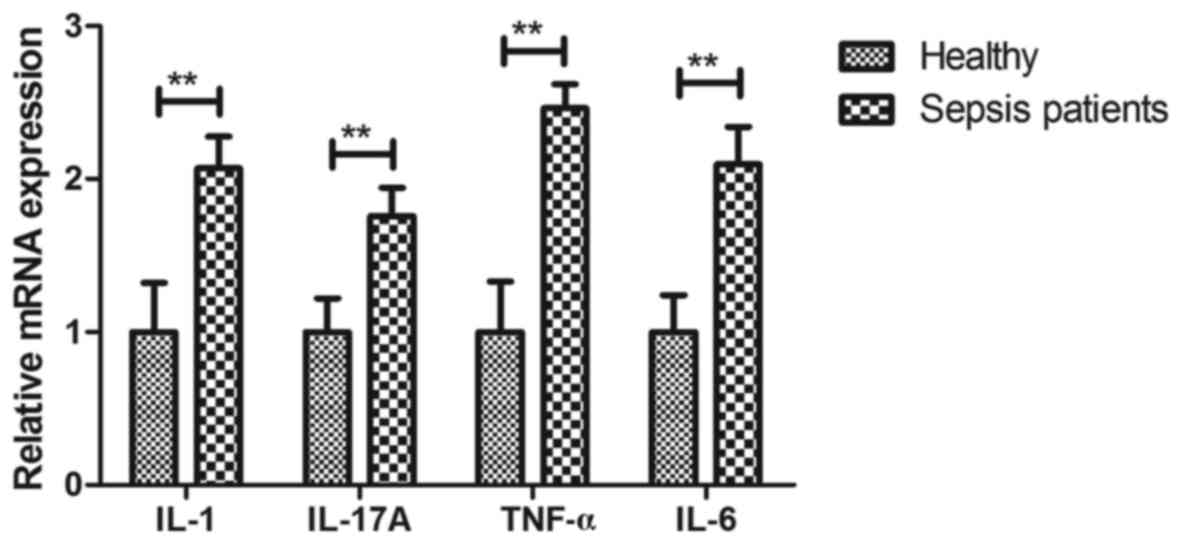
Analysis of inflammatory cytokine mRNA
expression in hPBMCs of patients with sepsis at the ICU
The gene expression levels of inflammatory cytokines
in hPBMCs of patients with sepsis were assessed in the present
study. The mRNA expression levels of IL-1 (2.07-fold), IL-17
(1.76-fold), TNF-α (2.46-fold) and IL-6 (1.96-fold) were markedly
up-regulated in hPBMCs of patients with sepsis compared with that
in healthy individuals (P<0.01; Fig.
3). These results suggest that the gene expression levels of
inflammatory cytokines are upregulated in hPBMCs of patients with
sepsis in an ICU setting.
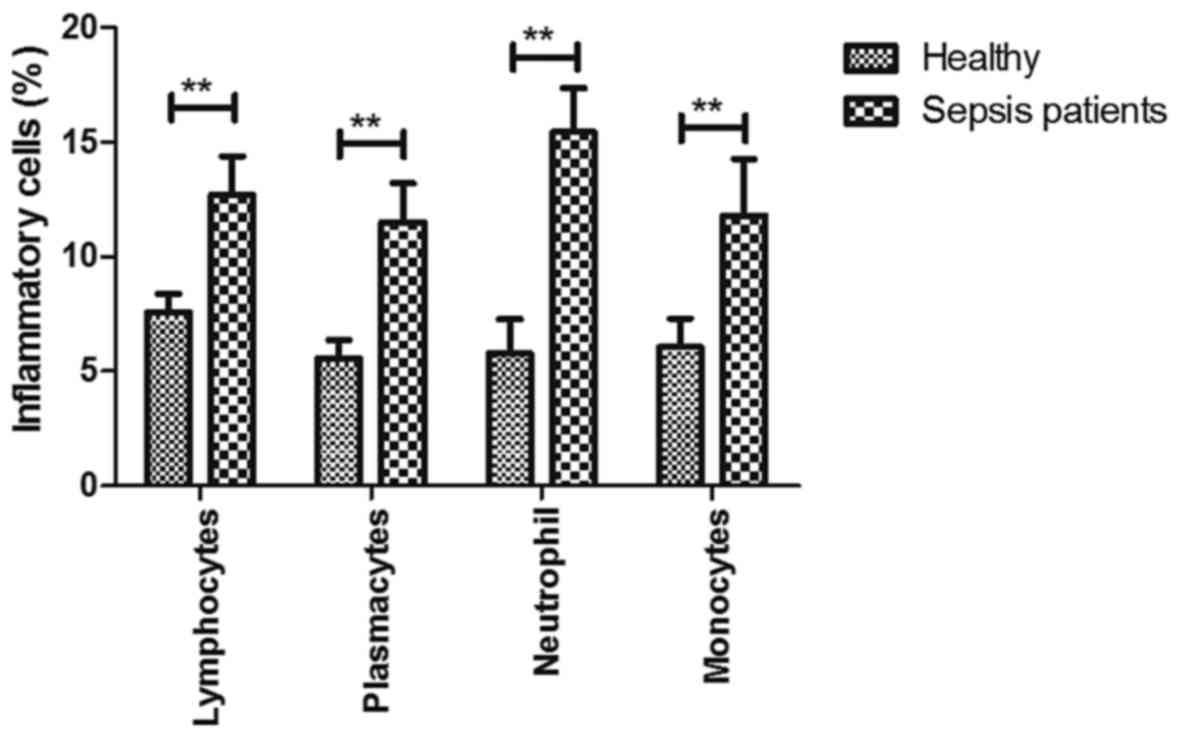
Analysis of inflammatory cells in
serum of patients with sepsis at the ICU
To explore the role of inflammation in the
progression of sepsis, changes in the ratio of inflammatory cells
were investigated in serum in patients with sepsis. As presented in
Fig. 4, the percentages of
lymphocytes, plasmacytes, neutrophils and monocytes among the total
leukocytes were increased in patients with sepsis at the ICU
compared with those in healthy individuals (P<0.01). These
results suggest that inflammatory cells are upregulated in serum of
patients with sepsis at the ICU.
Analysis of biomarkers of inflammatory
cytokines in patients with sepsis at the ICU
The presentation of biomarkers of inflammatory
cytokines on hPBMCs of patients with sepsis at the ICU was then
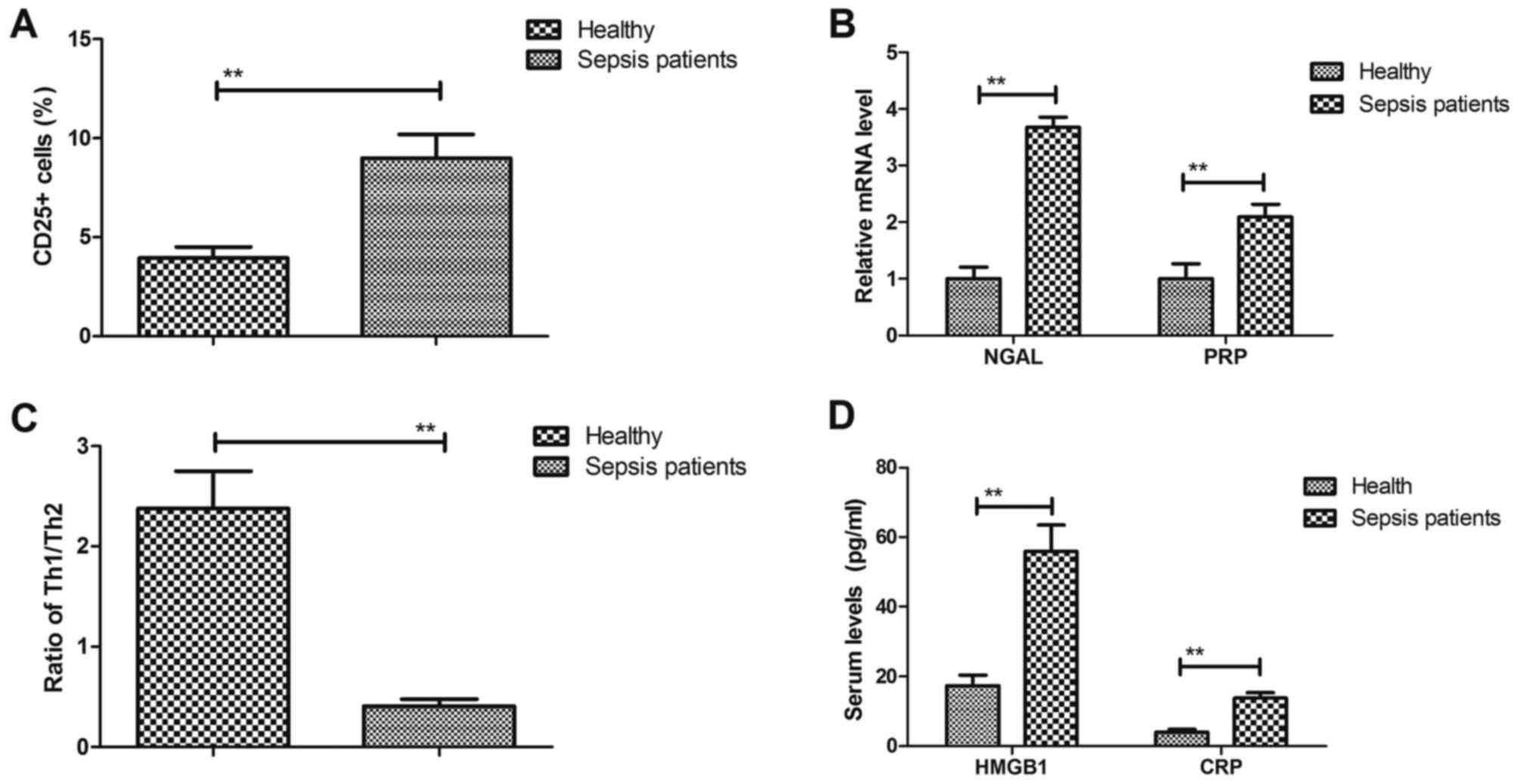
analyzed. The results indicated that the proportion of hPBMCs
presenting the activation marker CD25 was significantly increased
in sepsis patients compared with that in healthy individuals (9.00
vs. 3.95%; P<0.01; Fig. 5A). As
presented in Fig. 5B, NGAL and PRP
were significantly upregulated in hPBMCs of sepsis patients
compared with those from healthy individuals (3.67- and 2.09-fold
for NGAL and PRP, respectively). The balance of T-helper cell type
1 (Th1)/Th2 cytokines was decreased in patients with sepsis (2.38
vs. 0.41%; P<0.01; Fig. 5C). In
addition, the serum levels of HMGB1 and C-reactive protein (CRP)
were increased in sepsis patients compared with those in healthy
individuals (HMGB1, 55.95 vs. 16.88; CRP, 13.80 vs. 4.10;
P<0.01; Fig. 5D). These results
suggest that biomarkers of inflammatory cytokines are upregulated
in serum of patients with sepsis in an ICU setting.
Analysis of biomarkers of inflammation
in hPBMCs of patients with sepsis at the ICU
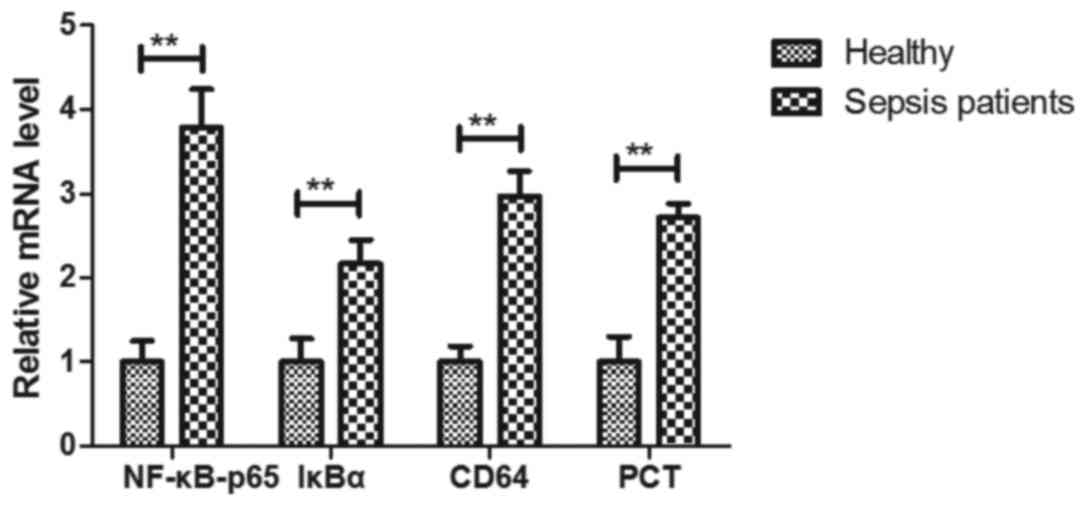
The gene expression levels of biomarkers of
inflammation in hBPMCs of patients with sepsis at the ICU were then
explored. The results indicated that the gene expression levels of
NF-κB-p65, IκBα, CD64 and PCT were higher in hPBMCs than those in
healthy individuals (NF-κB-p65, 3.79-fold; IκBα, 2.17-fold; CD64,
2.97-fold; PCT, 2.72-fold; P<0.01; Fig. 6). These results suggest that the mRNA
expression of inflammatory biomarkers in hPBMCs was upregulated in
patients with sepsis in an ICU setting.
Discussion
Sepsis can leads to accumulation of bacterial
endotoxins, release of inflammatory mediators and immune
dysfunction (22,23). Inflammatory responses are the major
reason leading to mortality of patients with sepsis (24,25). The
present study analyzed the changes of serum inflammatory cytokines
and markers in hPBMCs of patients with sepsis in an ICU setting.
The present results indicated that the serum levels of inflammatory
cytokines, biomarkers and inflammatory cells were upregulated in
patients with sepsis compared with those in healthy
individuals.
Circulating inflammatory cytokines have been
identified as mediators of damage in patients with sepsis (26). The present study reported the
upregulation of the inflammatory cytokines IL-1, IL-17, TNF-α and
IL-6 in sepsis patients compared with those in healthy individuals.
Cross (27) suggested that targeting
IL-1/IL-17A may improve therapeutic outcomes for patients with
sepsis. Studies have also demonstrated that the serum levels of the
cytokines TNF-α and IL-6 were increased and that the TNF-α-308 G/A
polymorphism is associated with post-operative sepsis (28–31). The
present study indicated that the serum levels of inflammatory
cytokines and the gene expression levels of IL-1, IL-17, TNF-α and
IL-6 in hPBMCs were upregulated in patients with sepsis at the ICU
compared with those in healthy individuals. Of note, the serum
concentrations of inflammatory cells, including lymphocytes,
plasmacytes, neutrophil and monocytes among the leukocytes, were
markedly increased in patients with sepsis compared with those in
healthy individuals.
HMGB1 is regarded as a predicator of organ
dysfunction in patients with severe sepsis (32,33). The
role of CRP in sepsis has been reported in multidisciplinary
studies on patients in the ICU setting (34,35). The
present study reported that the serum levels of HMGB1 and CRP were
significantly increased in sepsis patients compared with those in
healthy individuals. Wang et al (36) reported that attenuating
CD4+ CD25+ T-regulatory cells ameliorated
burn sepsis. In addition, plasma NGAL has been used to diagnose
acute kidney injury in patients with systemic inflammatory disease
and sepsis (37). Furthermore,
peptidoglycan recognition protein is associated with inflammation
in patients with bacterial infection (38,39). The
present results suggest that biomarkers of inflammatory cytokines
are upregulated in serum of patients with sepsis at the ICU
compared with that in healthy individuals.
Studies have indicated that inhibition of NF-κB
activation is beneficial for the treatment of experimental sepsis
(40,41). Song et al (42) reported that benzenediamine derivate
FC-98 exhibited anti-inflammatory efficacy against sepsis injury in
mice via suppression of c-Jun N-terminal kinase, NF-κB and
interferon regulatory transcription factor 3 signaling pathways.
The present study indicated that NF-κB gene expression levels are
higher in hPBMCs of patients with sepsis than those in healthy
individuals, which may provide a potential target for the treatment
of sepsis.
In conclusion, the present study indicated an
upregulation of inflammatory cytokines in the serum of patients
with sepsis. These results suggest that inflammatory cytokines are
potential targets for the treatment of sepsis patients. It was also
indicated that the balance of Th1/Th2 cytokines was decreased in
patients with sepsis in an ICU setting compared with that in
healthy individuals, which may provide a potential diagnostic tool
for patients with sepsis.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LW performed the experiments. HZ designed and
analysed the experiments. DW analysed the data from the
experiments.
Ethical approval and consent to
participate
All patients and healthy volunteers provided written
informed consent. The study was approved by the ethics committee of
Daqing Oil Field General hospital (Daqing, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Guild GN III, Wu B and Scuderi GR:
Articulating vs. static antibiotic impregnated spacers in revision
total knee arthroplasty for sepsis. A systematic review. J
Arthroplasty. 29:558–563. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Neto Serpa A, Veelo DP, Peireira VG, de
Assunção MS, Manetta JA, Espósito DC and Schultz MJ: Fluid
resuscitation with hydroxyethyl starches in patients with sepsis is
associated with an increased incidence of acute kidney injury and
use of renal replacement therapy: A systematic review and
meta-analysis of the literature. J Crit Care. 29:1852014.
|
|
3
|
Neto AS, Pereira VG, Manetta JA, Espósito
DC and Schultz MJ: Association between static and dynamic thenar
near-infrared spectroscopy and mortality in patients with sepsis: A
systematic review and meta-analysis. J Trauma Acute Care Surg.
76:226–233. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Patel A, Laffan MA, Waheed U and Brett SJ:
Randomised trials of human albumin for adults with sepsis:
Systematic review and meta-analysis with trial sequential analysis
of all-cause mortality. BMJ. 349:g45612014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nelson GE, Mave V and Gupta A: Biomarkers
for sepsis: A review with special attention to India. Biomed Res
Int. 2014:2643512014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Trivedi V, Bavishi C and Jean R: Impact of
obesity on sepsis mortality: A systematic review. J Crit Care.
30:518–524. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang C, Chi C, Guo L, Wang X, Guo L, Sun
J, Sun B, Liu S, Chang X and Li E: Heparin therapy reduces 28-day
mortality in adult severe sepsis patients: A systematic review and
meta-analysis. Crit Care. 18:5632014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Silva LS, Catalão CH, Felippotti TT,
Oliveira-Pelegrin GR, Petenusci S, de Freitas LA and Rocha MJ:
Curcumin suppresses inflammatory cytokines and heat shock protein
70 release and improves metabolic parameters during experimental
sepsis. Pharm Biol. 55:269–276. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Williams JM, Greenslade JH, Dymond CA, Chu
K, Brown AF and Lipman J: Characteristics, treatment and outcomes
for all emergency department patients fulfilling criteria for
septic shock: A prospective observational study. Eur J Emerg Med.
25:97–104. 2016.
|
|
10
|
Gille-Johnson P, Hansson KE and Gårdlund
B: Severe sepsis and systemic inflammatory response syndrome in
emergency department patients with suspected severe infection.
Scand J Infect Dis. 45:186–193. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fan PC, Chang CH, Tsai MH, Lin SM, Jenq
CC, Hsu HH, Chang MY, Tian YC, Hung CC, Fang JT, et al: Predictive
value of acute kidney injury in medical intensive care patients
with sepsis originating from different infection sites. Am J Med
Sci. 344:83–89. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen Z, Ding X, Jin S, Pitt B, Zhang L,
Billiar T and Li Q: WISP1-αvβ3 integrin signaling positively
regulates TLR-triggered inflammation response in sepsis induced
lung injury. Sci Rep. 6:288412016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chung HY, Hupe DC, Otto GP, Sprenger M,
Bunck AC, Dorer MJ, Bockmeyer CL, Deigner HP, Gräler MH and Claus
RA: Acid sphingomyelinase promotes endothelial stress response in
systemic inflammation and sepsis. Mol Med. 22:2016. View Article : Google Scholar
|
|
14
|
Park JH, Jang JH, Choi EJ, Kim YS, Lee EJ,
Jung ID, Han HD, Wu TC, Hung CF, Kang TH and Park YM: Annexin A5
increases survival in murine sepsis model by inhibiting
HMGB1-mediated pro-inflammation and coagulation. Mol Med. 22:2016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dwivedi DJ, Grin PM, Khan M, Prat A, Zhou
J, Fox-Robichaud AE, Seidah NG and Liaw PC: Differential expression
of PCSK9 modulates infection, inflammation, and coagulation in a
murine model of sepsis. Shock. 46:672–680. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen YK, Xu YK, Zhang H, Yin JT, Fan X,
Liu DD, Fu HY and Wan B: Emodin alleviates jejunum injury in rats
with sepsis by inhibiting inflammation response. Biomed
Pharmacother. 84:1001–1007. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Matzneller P, Strommer S, Drucker C,
Petroczi K, Schörgenhofer C, Lackner E, Jilma B and Zeitlinger M:
Colistin reduces LPS-triggered inflammation in a human sepsis model
In vivo: A randomized controlled trial. Clin Pharmacol Ther.
101:773–781. 2016. View
Article : Google Scholar
|
|
18
|
Kaplan D, Casper TC, Elliott CG, Men S,
Pendleton RC, Kraiss LW, Weyrich AS, Grissom CK, Zimmerman GA and
Rondina MT: VTE incidence and risk factors in patients with severe
sepsis and septic shock. Chest. 148:1224–1230. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chou HL, Han ST, Yeh CF, Tzeng IS, Hsieh
TH, Wu CC, Kuan JT and Chen KF: Systemic inflammatory response
syndrome is more associated with bacteremia in elderly patients
with suspected sepsis in emergency departments. Medicine
(Baltimore). 95:e56342016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-delta delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Filipova J, Rihova L, Vsianska P, Kufova
Z, Kryukova E, Kryukov F and Hajek R: Flow cytometry in
immunoglobulin light chain amyloidosis: Short review. Leuk Res.
July 13–2015.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Markwart R, Condotta SA, Requardt RP,
Borken F, Schubert K, Weigel C, Bauer M, Griffith TS, Förster M,
Brunkhorst FM, et al: Immunosuppression after sepsis: Systemic
inflammation and sepsis induce a loss of naïve T-cells but no
enduring cell-autonomous defects in T-cell function. PloS One.
9:e1150942014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sharma D, Packiriswamy N, Malik A, Lucas
PC and Parameswaran N: Nonhematopoietic β-Arrestin-1 inhibits
inflammation in a murine model of polymicrobial sepsis. Am J
Pathol. 184:2297–2309. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang Y, Braun OO, Zhang S, Norström E and
Thorlacius H: Monocytes regulate systemic coagulation and
inflammation in abdominal sepsis. Am J Physiol Heart Circ Physiol.
308:H540–H547. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang S, Luo L, Wang Y, Gomez MF and
Thorlacius H: Nuclear factor of activated T cells regulates
neutrophil recruitment, systemic inflammation, and T-cell
dysfunction in abdominal sepsis. Infect Immun. 82:3275–3288. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
de Andrade JAA, Gayer CRM, Nogueira NPA,
Paes MC, Bastos VLFC, Neto JDCB, Alves SC Jr, Coelho RM, da Cunha
MGAT, Gomes RN, et al: The effect of thiamine deficiency on
inflammation, oxidative stress and cellular migration in an
experimental model of sepsis. J Inflamm (Lond). 11:112014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cross AS: IL-18/IL-1/IL-17A axis: A novel
therapeutic target for neonatal sepsis? Cytokine. 86:1–3. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gil M, Kim YK, Hong SB and Lee KJ:
Naringin decreases TNF-α and HMGB1 release from LPS-stimulated
macrophages and improves survival in a CLP-induced sepsis mice.
PloS One. 11:e01641862016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Montoya-Ruiz C, Jaimes FA, Rugeles MT,
López JÁ, Bedoya G and Velilla PA: Variants in LTA, TNF, IL1B and
IL10 genes associated with the clinical course of sepsis. Immunol
Res. 64:1168–1178. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Roderburg C, Benz F, Schüller F, Pombeiro
I, Hippe HJ, Frey N, Trautwein C, Luedde T, Koch A, Tacke F and
Luedde M: Serum levels of TNF receptor ligands are dysregulated in
sepsis and predict mortality in critically Ill patients. PloS One.
11:e01537652016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Baghel K, Srivastava RN, Chandra A, Goel
SK, Agrawal J, Kazmi HR and Raj S: TNF-α, IL-6, and IL-8 cytokines
and their association with TNF-α-308 G/A polymorphism and
postoperative sepsis. J Gastrointest Surg. 18:1486–1494. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Karlsson S, Pettila V, Tenhunen J,
Laru-Sompa R, Hynninen M and Ruokonen E: HMGB1 as a predictor of
organ dysfunction and outcome in patients with severe sepsis.
Intensive Care Med. 34:1046–1053. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Waterer GW: High-mobility group box 1
(HMGB1) as a potential therapeutic target in sepsis-more questions
than answers. Crit Care Med. 35:1205–1206. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sarathy VCY: Comparison of eosinophil
count and neutrophillymphocyte count ratio with C-reactive protein
levels in patients with sepsis. J Assoc Physicians India.
64:962016.PubMed/NCBI
|
|
35
|
Pradhan S, Ghimire A, Bhattarai B, Khanal
B, Pokharel K, Lamsal M and Koirala S: The role of C-reactive
protein as a diagnostic predictor of sepsis in a multidisciplinary
intensive care unit of a tertiary care center in Nepal. Indian J
Crit Care Med. 20:417–420. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang SX, Liu QY and Li Y: Lentinan
ameliorates burn sepsis by attenuating CD4+ CD25+ tregs. Burns.
42:1513–1521. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ralib Md A, Nor Mat MB and Pickering JW:
Plasma neutrophil gelatinase-associated lipocalin diagnosed acute
kidney injury in patients with systemic inflammatory disease and
sepsis. Nephrology (Carlton). 22:412–419. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dziarski R, Platt KA, Gelius E, Steiner H
and Gupta D: Defect in neutrophil killing and increased
susceptibility to infection with nonpathogenic gram-positive
bacteria in peptidoglycan recognition protein-S (PGRP-S)-deficient
mice. Blood. 102:689–697. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Michel T, Reichhart JM, Hoffmann JA and
Royet J: Drosophila toll is activated by gram-positive bacteria
through a circulating peptidoglycan recognition protein. Nature.
414:756–759. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li H, Qiu D, Gao Q, Wang H and Sun M:
Selectively activating melanocortin 4 receptor acts against rat
sepsis-induced acute liver injury via HMGB1/TLR4/NF-κβ signaling
pathway. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 32:1055–1059.
2016.(In Chinese). PubMed/NCBI
|
|
41
|
Zhao H, Li S and Zhang H, Wang G, Xu G and
Zhang H: Saikosaponin a protects against experimental sepsis via
inhibition of NOD2-mediated NF-κβ activation. Exp Ther Med.
10:823–827. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Song Y, Liu X, Yue H, Ji J, Dou H and Hou
Y: Anti-inflammatory effects of benzenediamine derivate FC-98 on
sepsis injury in mice via suppression of JNK, NF-κβ and IRF3
signaling pathways. Mol Immunol. 67:183–192. 2015. View Article : Google Scholar : PubMed/NCBI
|