Introduction
Free flap grafting has been widely applied in the
restructuring of soft tissue or skin defects caused by trauma or
burns. However, flap surgery results in a high rate of morbidity
correlated with regional flap necrosis, which is observed in 7–20%
of free flaps (1).
Ischemia/reperfusion injury (IRI) is caused by the reperfusion of
blood to previously ischemic tissue, which causes extreme cellular
injury (2,3). Reperfusion of blood into ischemic
tissue can cause a cascade of inflammatory processes, leading to
damage to vascular and endothelial cells, capillary narrowing and
tissue necrosis (4,5). In addition to serious necrotic injury,
ischemia also activates the caspase system, which rapidly leads to
apoptosis and cell death (6).
Botulinum toxin type A (BTXA) has been demonstrated
to increase flap survival through chemical denervation (7) and chemical delay (8). In fact, alterations in the vascular
endothelial cells following IRI are also critical, as IRI can also
affect microvessels. Endothelial cells, which are the basic
component of the vasculature, serve a significant role in
maintaining vessel function, for instance, in cytokine secretion
and regulation of vascular tone. For a free flap, endothelial cells
are essential to provide a blood supply, oxygen transportation and
adjustment of skin microcirculation (9,10). IRI
of a skin flap causes a change in the function of the endothelial
cells, such as increased production of reactive oxygen species,
induction of inflammatory response and even apoptosis (11). Endothelial cell injury resulting from
ischemia/reperfusion causes disruption of the vascular endothelium
and dysregulation of vascular tension, which ultimately leads to
worsening of the damage. Thus, protection of the endothelial cells
is an important way of alleviating skin flap necrosis.
BTXA, a polypeptide, is produced by the bacterium
Clostridium botulinum. BTXA has been widely used in clinical
practice since its authorization by the Food and Drug
Administration (12). It has been
demonstrated to exert protective effects on skin flaps in animals
by reducing inflammation, ameliorating blood flow and attenuating
necrosis (13). Despite significant
advances in the use of BTXA in IRI (7,8,13), the exact mechanism of BTXA needs to
be further established.
Autophagy is a lysosomal-dependent catabolic pathway
that recycles proteins and organelles in cells (14), and is considered to serve a
significant role in maintaining cellular homeostasis (15). It can be activated by various
stressors, such as ischemia, hypoxia or cell starvation, and
autophagy activity usually contributes to cell adaptation or
survival (16–18). By contrast, inordinate and
dysregulated autophagy may contribute to cellular dysfunction or
apoptosis (19–21). Protective autophagy can be induced in
various tissues, including cardiomyocytes and cerebral neurons
(22,23). However, the effect of autophagy in
reperfusion damage of human dermal microvascular endothelial cells
(HDMECs) remains unclear. Previous research revealed that BTXA
increases survival and attenuates apoptosis in skin flaps (24). Therefore, it can be hypothesized that
the protective effects of BTXA against IRI in HDMECs may be a
result of the induction of autophagy.
A previous experiment confirmed that BTXA attenuates
endothelial apoptosis during IRI (13); however, the underlying mechanism
remains unclear. Thus, the present study used an in vitro
model of hypoxia/reoxygenation (H/R) in HDMECs to determine the
role of BTXA and to confirm the effects of autophagy.
Materials and methods
Cell extraction and culture
HDMECs were harvested from the upper eyelid tissues
of 23 female patients with blepharoplasty (age range, 18–25 years)
from October 2016 to November 2017. The present study was approved
by the Ethics Committee of Anzhen Hospital (Beijing, China).
Subsequent to washing with cold phosphate-buffered saline (PBS),
the skin flaps were cut into sections of 0.5×0.5×0.1 cm. As
previously described (25), the skin
flaps were treated with the neutral protease, dispase II
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany), to separate and
remove the epidermal layer from the dermis. Next, the dermis was
digested using collagenase I (Sigma-Aldrich; Merck KGaA) to obtain
a cell suspension containing HDMECs, which were cultivated for 1
week. Following treatment with trypsin to create a single cell
suspension, HDMECs were purified using a CD31 microbead kit
(Miltenyi Biotec GmbH, Bergisch Gladbach, Germany). The cells were
directly transferred into culture or subjected to a second
purification. The present study was authorized by the Ethics
Committee of Anzhen Hospital (Beijing, China; approval no.
2015021X), and all patients provided signed informed consent.
The cells were cultured and maintained in
endothelial cell medium (ECM; cat. no. 1001; ScienCell Research
Laboratories, Inc., Carlsbad, CA, USA) containing 5% fetal bovine
serum, 1% endothelial cell growth supplement and 1%
penicillin/streptomycin at 37°C in a humidified incubator (5%
CO2). The experimental cells were treated with BTXA
(0.1, 0.2, 0.4, 0.8, 1.6 or 3.2 U/ml) for 12 h before induction of
hypoxia. Control cells were treated with PBS for the same period of
time.
H/R treatment
The culture medium was replaced with fresh
serum-free ECM and then the cell cultures were placed in a hypoxic
incubator, containing a gas mixture comprising 90% N2,
5% O2 and 5% CO2, for 8 h. To imitate
ischemia/reperfusion in vitro, cells were then incubated for
a further 24 h under normal conditions.
BTXA preparation
BTXA is available as a freeze-dried power, which
must be kept in cold storage. The powder was dissolved in ECM and
then stored at 4°C and used within 4 h. The solution was adjusted
to a final concentration of 10 U/ml by the addition of ECM.
Chloroquine diphosphate salt (CQ)
As an autophagy inhibitor, CQ (cat. no. C6628,
Sigma-Aldrich; Merck KGaA) primarily inhibits autophagy by
suppressing the formation of autolysosomes. The powder was
dissolved in DMSO and then stored at −20°C. Cells were treated with
25 µM CQ for 2 h prior to BTXA treatment.
Determination of apoptosis using flow
cytometry
The apoptosis rate was assessed by flow cytometry
using a fluorescein isothiocyanate (FITC) Annexin V Apoptosis
Detection kit I (cat. no. 556547; BD Biosciences, Franklin Lakes,
NJ, USA). According to the manufacturer's protocol, the cells were
harvested and resuspended in 1X binding buffer at a concentration
of 1×106 cells/ml. Next, 100 µl of the solution
(1×105 cells) was transferred to a 5 ml culture tube,
and 5 µl FITC Annexin V and 5 µl propidium iodide (PI) were added.
The mixture was incubated for 15 min at room temperature in the
dark. Prior to analysis by flow cytometry, 300 µl of 1X binding
buffer was added to each tube. The following controls were used in
the flow cytometry experiment: Unstained cells, cells stained with
FITC Annexin V and cells stained with PI.
Western blot analysis
HDMECs were washed twice with cold PBS and exposed
to lysis buffer containing a protease inhibitor for 20 min,
followed by centrifugation at 15,000 × g for 15 min at 4°C. The
protein concentration was determined using a bicinchoninic acid
assay kit (cat. no. 23227, Thermo Fisher Scientific, Inc.), and
protein content was adjusted to achieve equal concentration and
volumes. Next, protein samples were analyzed by 12.5%
SDS-polyacrylamide gel electrophoresis and transferred onto a
nitrocellulose membrane. Samples were then incubated with
monoclonal rabbit primary antibodies at 4°C for 12 h, including
anti-light chain 3 (LC3; 1:1,000; cat. no. 12741), anti-Beclin-1
(1:2,000; cat. no. 3495) and anti-GAPDH (1:2,000; cat. no. 5174;
all from Cell Signaling Technology, Inc., Danvers, MA, USA).
Secondary antibody incubation was then performed using alkaline
phosphatase goat anti-rabbit immunoglobulin G (cat. no. 7074; Cell
Signaling Technology, Inc.). Protein bands were visualized using
Enhanced Chemiluminescence (Thermo Fisher Scientific, Inc.),
according to the manufacturer's protocol, Protein expression was
quantified using an Odyssey Infrared Imaging system (Gene Company,
Ltd., Beijing, China).
Immunofluorescence staining
Immunofluorescence staining was performed to
determine the expression of LC3. Briefly, the cells were washed
with PBS then fixed with 4% neutral-buffered formaldehyde for 15
min at room temperature. Cells were permeabilized with 0.1% Triton
X-100 in Tris-buffered saline for 15 min and then incubated with 5%
bovine serum albumin in PBS for 30 min at room temperature. Next,
the cells were incubated with anti-LC3 antibody (cat. no. 12741;
Cell Signaling Technology, Inc.) in 3% bovine serum albumin-PBS at
a dilution of 1:100 overnight at 4°C in a humidified chamber.
Subsequent to washing with PBS, the cells were incubated with a
FITC-conjugated goat anti-rabbit antibody (cat. no. 4413; Cell
Signaling Technology, Inc.) at a dilution of 1:200 in PBS at 37°C
for 1 h in the dark. Finally, nuclei were counterstained with DAPI.
Slides were observed and images were captured under a fluorescence
microscope (Olympus Corporation, Tokyo, Japan).
Transmission electron microscopy
Following treatment, cells were washed twice with
cold PBS, harvested with trypsin and then centrifuged at 1,200 × g
for 5 min at room temperature. The supernatant was discarded, and
cells were fixed with 2.5% glutaraldehyde at 4°C for 3 h.
Subsequently, the samples were dehydrated by an acetone gradient
and embedded in Epon 812 resin (cat. no. 14120; Electron Microscopy
Sciences, Hatfield, PA, USA), followed by semi-thin section optical
positioning and ultra-thin sectioning. The sections were then
double-stained with uranyl acetate and lead citrate. A transmission
electron microscope was used to record images.
Statistical analysis
Statistical analysis was conducted using IBM SPSS
version 19.0 software (IBM Corp., Armonk, NY, USA). Differences
among the groups were analyzed by one-way analysis of variance,
while pairwise comparisons within groups were conducted using the
Student-Newman-Keuls q test. P<0.05 was considered to denote a
statistically significant difference. All the data are presented as
the mean ± standard deviation.
Results
BTXA attenuated
ischemia/reperfusion-induced apoptosis of HDMECs
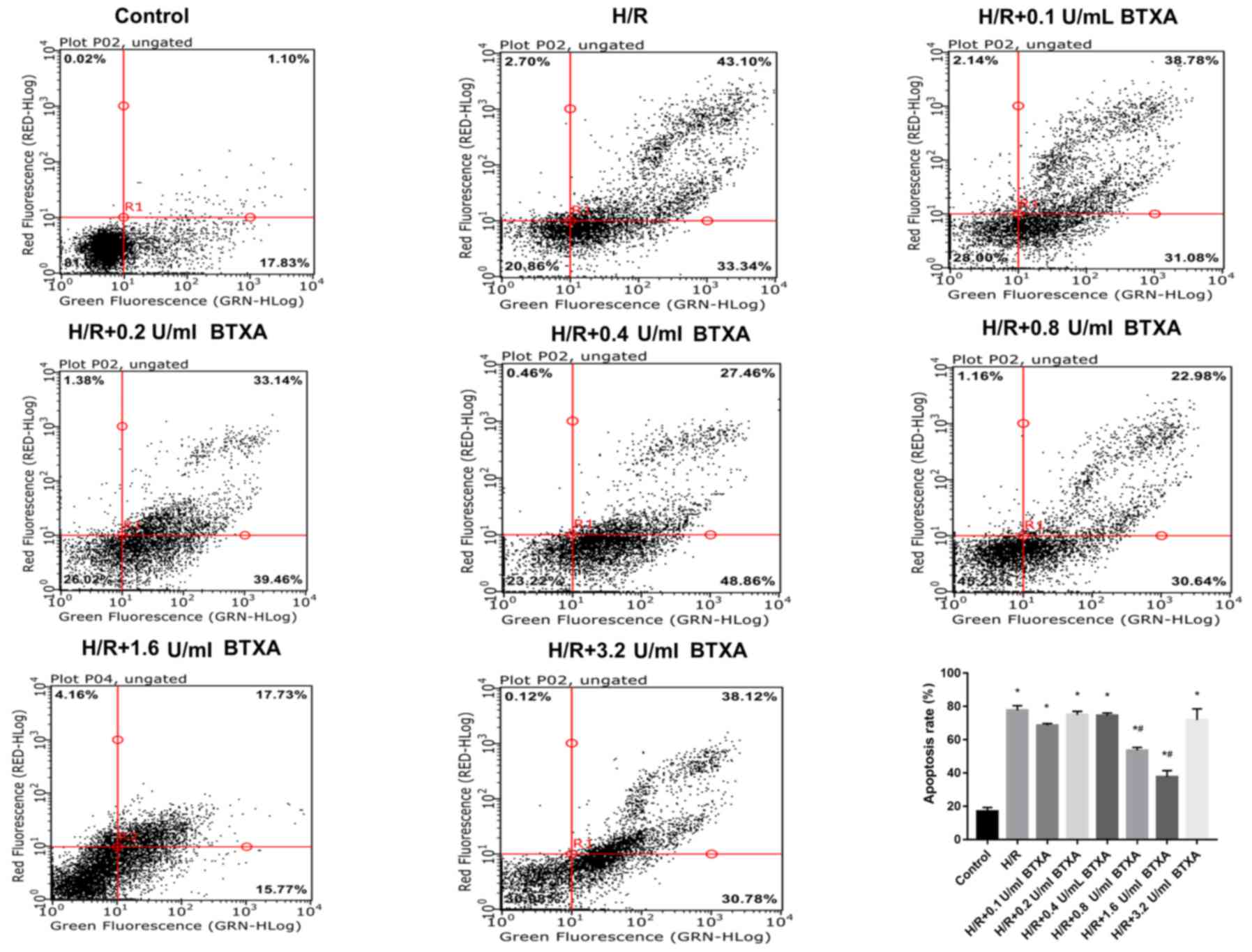
HDMECs were hypersensitive to H/R. To identify
whether BTXA protects HDMECs from apoptosis, HDMECs were treated
with different concentrations of BTXA (0.1, 0.2, 0.4, 0.8, 1.6 or
3.2 U/ml) for 12 h prior to exposure to hypoxia. Flow cytometric
analysis was initially conducted to assess the apoptosis rate of
cells in all the treatment groups. The results demonstrated that,
compared with the control group, the rate of apoptosis was
significantly increased following H/R exposure. Flow cytometric
analysis indicated that BTXA treatment significantly decreased the
rate of apoptosis following H/R induction in a dose-dependent
manner (Fig. 1). While BTXA did not
have a protective role at low concentrations (0.1, 0.2 and 0.4
U/ml), the protective effect gradually increased with increasing
concentrations of BTXA (0.8 and 1.6 U/ml) and was strongest at a
concentration of 1.6 U/ml. However, the protection disappeared at a
concentration of 3.2 U/ml BTXA. Thus, BTXA decreased the rate of
apoptosis following H/R induction in a dose-dependent manner, and
treatment with 1.6 U/ml BTXA produced the peak beneficial effect
(Fig. 1). These results suggested
that an appropriate concentration of BTXA may attenuate H/R-induced
damage.
BTXA protects against H/R-induced
injury in HDMECs through the activation of autophagy
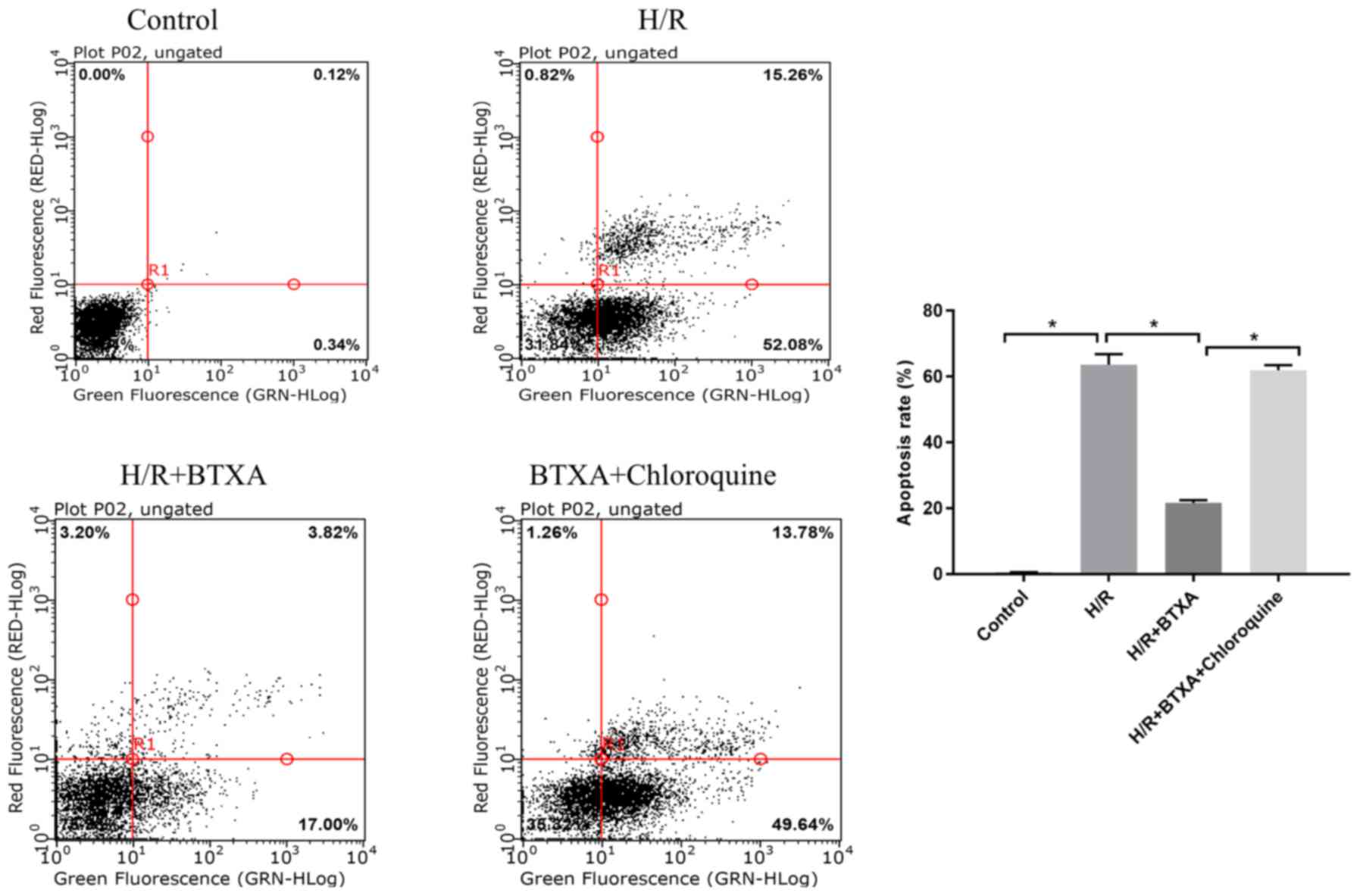
Using CQ as an autophagy inhibitor, the present
study aimed to confirm the effect of BTXA in activating autophagy
in HDMECs exposed to H/R. CQ effectively inhibited autophagy by
inactivating the lysosomal enzymes and blocking the formation of
the autolysosomes. Compared with the H/R group, BTXA significantly
attenuated the apoptotic rate, while the protective effect of BTXA
was abolished by CQ (Fig. 2).
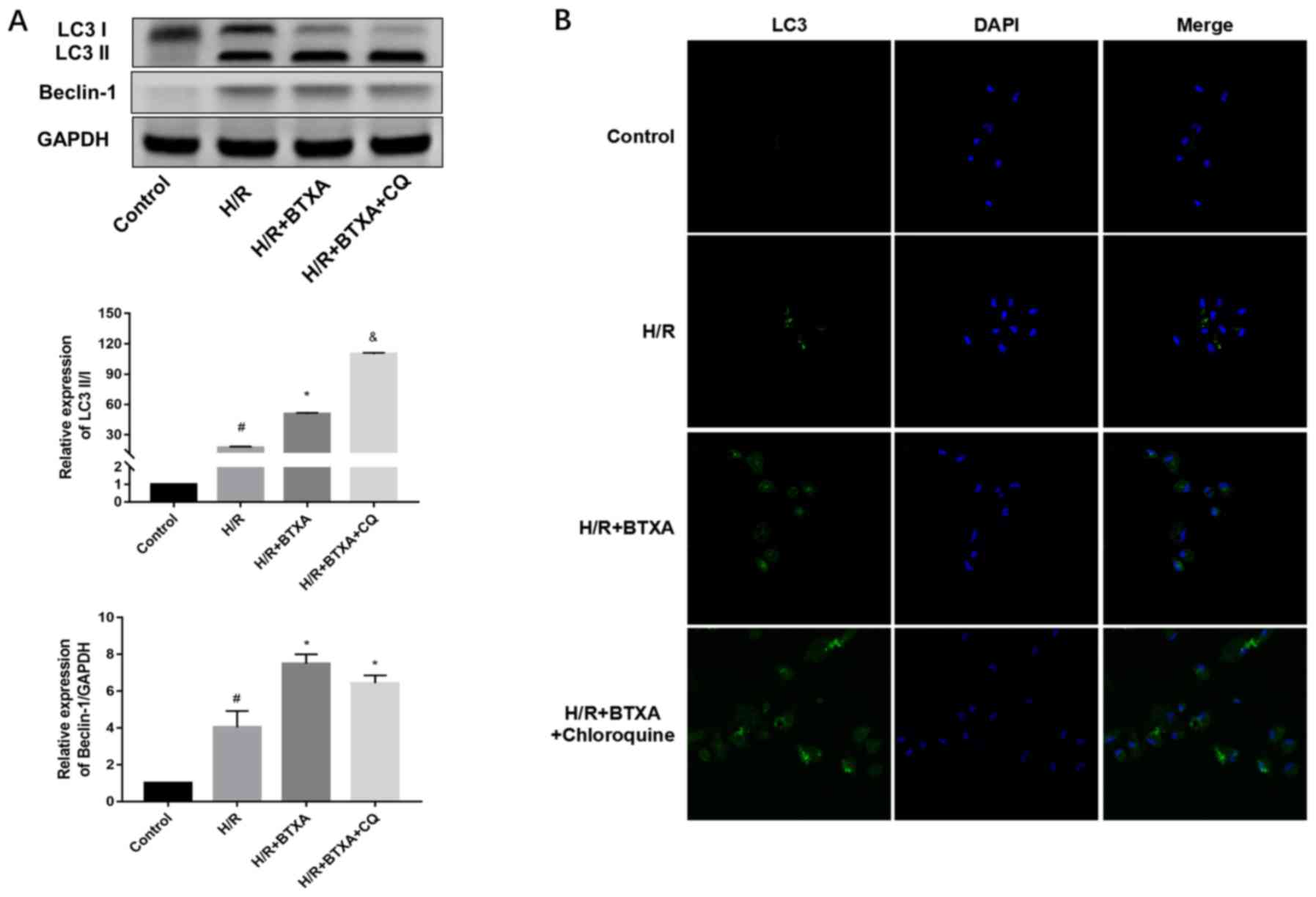
Next, the levels of LC3 and Beclin-1 in each group
were measured by western blot analysis. Compared with the control
group, H/R exposure caused significant conversion of LC3-I to
LC3-II and increased the expression of Beclin-1 (Fig. 3A). The results also demonstrated
that, when the cells were treated with 1.6 U/ml BTXA, the ratio of
LC3-II/LC3-I and Beclin-1 expression were significantly increased
compared with the H/R group (Fig.
3A). The addition of CQ suppressed the formation of the
autolysosomes, which was characterized by a further increase of the
LC3-II/LC3-I ratio. However, the expression the other autophagic
marker, Beclin-1, was not markedly influenced by CQ. These findings
were supported by the results of the immunofluorescence study, with
immunofluorescence staining with the LC3 antibody revealing that
BTXA and CQ treatment increased LC3-II punctate dots (Fig. 3B). Taken together, the results
demonstrated that induction of autophagy explained the protective
mechanism of BTXA against H/R-caused injury. At the same time, the
study results confirmed that autophagy is a lysosome-dependent
protein degradation pathway.
Transmission electron microscopy
observations
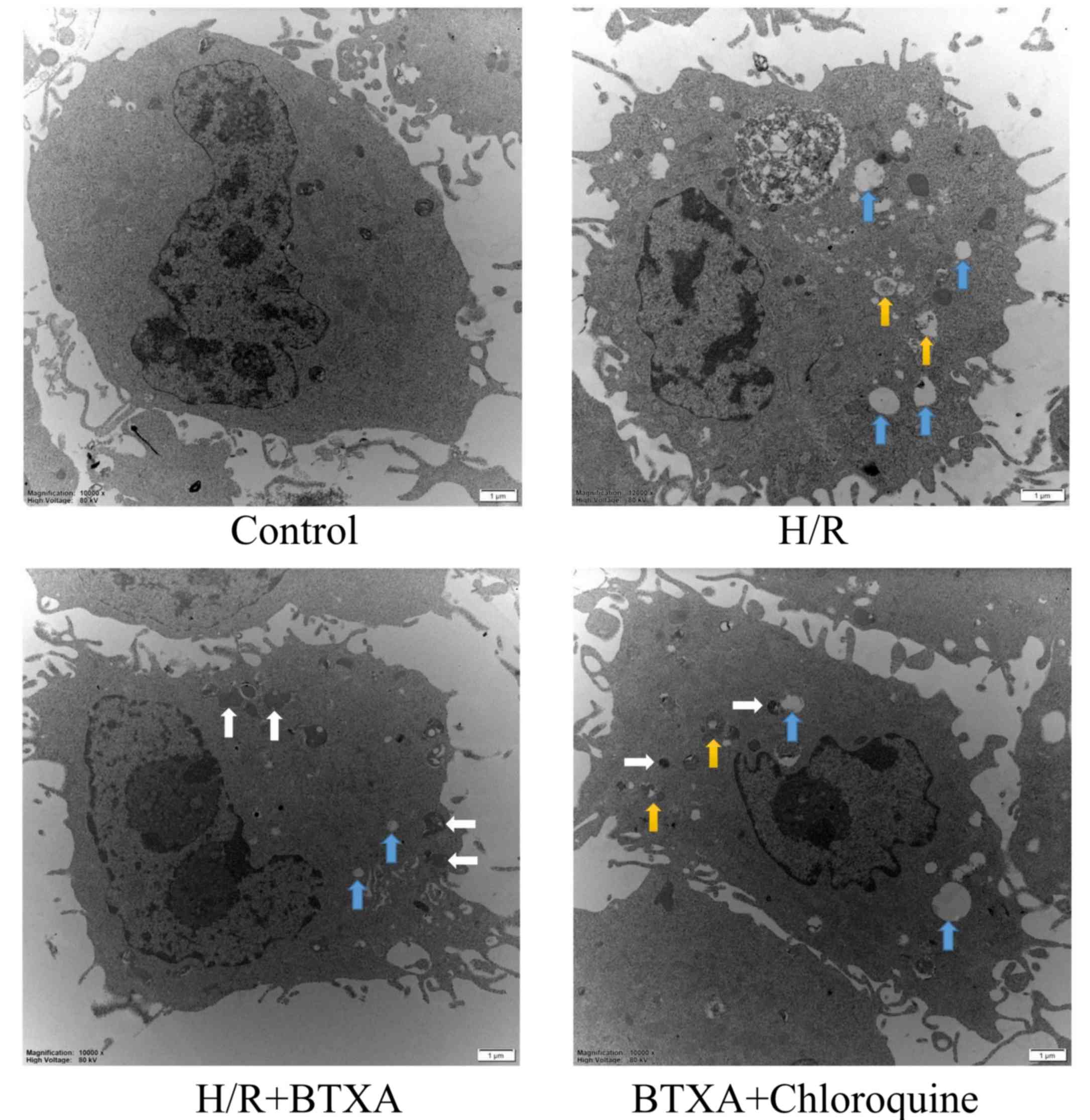
Transmission electron microscopy analyses revealed
that HDMECs in the control group exhibited typical endothelial
features, and were visible as oval cells with a large central
nucleus and intact mitochondria and endoplasmic reticulum (ER;
Fig. 4). HDMECs in the H/R group
exhibited clear ultrastructural lesions, including cell swelling,
appearance of numerous vesicles, ER dilation, mitochondrial
swelling and vacuolization. Compared with the H/R group,
pretreatment with BTXA evidently alleviated ultrastructural lesions
and increased autophagosome formation. However, CQ aggravated
ultrastructural lesions and reduced autolysosome formation,
indicating that autophagy alleviated H/R-induced injury (Fig. 4). The results indicated that BTXA
alleviated H/R-induced injury by inducing protective autophagy.
Discussion
Over the past few decades, BTXA has been widely used
in clinical practice, particularly in plastic surgery, and
satisfactory effects on facial rejuvenation have been obtained
(26–29). As the research continues, there has
been a broad spectrum of indications for the use of BTXA in
neuromuscular disorders, such as spasmodic torticollis, spasms of
the extremities and anal fissures (30–34).
Furthermore, researchers have observed that BTXA has beneficial
effects on ischemic skin flaps and Raynaud's disease (35–37).
Results have demonstrated that BTXA is able to regulate the
vascular tone and improve blood flow, while a number of studies
have demonstrated that BTXA improved the survival of critical
ischemic skin flaps in animal models (38–40).
BTXA has beneficial effects on skin flap IRI, accounted for by its
anti-inflammatory effect and chemical delay.
IRI is common in clinical practice and can lead to
severe complications (41). Plastic
surgeons have already recognized the problem and have searched for
a better way to fight IRI. Our previous study demonstrated that
ischemia/reperfusion caused serious necrosis of skin flaps, while
BTXA pretreatment increased the survival of flaps in an animal
experiment (42). The H/R HDMEC
in vitro model was also established, and the results
illustrated that the apoptosis rate of HDMECs following H/R was
markedly increased.
Autophagy has been reported to be protective against
hypoxia or chemically-induced oxidative stress in several
endothelial cell lines (43–46). LC3, as the main autophagy marker
(47), is the main component of
autophagosomes and exists in two molecular forms, including LC3-I
(18 KDa) and LC3-II (16 KDa), and the LC-3II is eventually degraded
through lysosomes. LC3-II is formed from cytosolic LC3-I (23) during autophagy activation and the
ratio of LC3-II/I significantly increases. LC3-II is mainly
degraded by the autolysosome pathway. As an autophagy inhibitor, CQ
mainly inhibits autophagy by suppressing the formation of
autolysosomes. As a result, LC3-II cannot be degraded by
autolysosomes, and a large amount of LC3-II accumulates in cells.
Beclin-1 is also an important autophagy marker and serves a
critical role in autophagy.
Multiple studies (23,48) have
already indicated that activation of autophagy exerts a protective
effect in human umbilical vein endothelial cells. While apoptosis
is considered to be a process of programmed cell death, autophagy
is programmed cell survival. These processes share the same
stimulating factors and regulatory proteins, however, they have
different thresholds. Autophagy is a type of intracellular defense
mechanism that degrades destroyed organelles and recycles proteins,
allowing cells to store more nutrition and protect themselves from
death (49). However, dysregulated
autophagy may induce apoptosis and may also cause certain diseases.
Several studies demonstrated that the upregulation of autophagy has
beneficial effects in some diseases, including AIDS, autoimmune
diseases and neurodegenerative diseases (50–54);
however, its role in cancer is controversial (55–58). The
present study revealed that BTXA promoted protective autophagy in
HDMECs in an in vitro model of IRI. To the best of our
knowledge, this is the first study to prove that BTXA protects
H/R-treated HDMECs in vitro by inducing autophagy.
Based on the model of H/R injury, HDMECs were
treated with BTXA for 12 h in an attempt to understand the effect
of autophagy and its regulation by BTXA. The results demonstrated
that autophagy had an antiapoptotic rather than a proapoptotic
effect on HDMECs during the H/R period, and that BTXA protected
HDMECs from H/R-induced injury in a dose-dependent manner. However,
the BTXA treatment had a protective effect only at suitable
concentrations, whereas other concentrations of BTXA may exacerbate
apoptosis.
To further verify the role of autophagy in the
protective effect of BTXA against H/R-induced injury, the autophagy
inhibitor CQ was added into the culture medium and the protective
effect of BTXA was blocked. CQ, a weakly-alkaline drug, inhibited
the autophagic activity by disturbing the degradation of
autophagosomes by autophagolysosomes. As a result, excessive
LC3-II, an autophagy marker, was stored within cells. Furthermore,
it was observed that simultaneous treatment with CQ also increased
the cell apoptosis rate. Compared with the BTXA group, CQ
significantly inhibited the formation of autolysosomes. These
observations further suggested that protective autophagy caused by
BTXA may be a potential mechanism underlying the beneficial effects
of BTXA on IRI.
Although the present study demonstrated that
activation of autophagy by BTXA may ameliorate IRI, certain
limitations remain. Firstly, in the experimental design, only one
autophagy inhibitor was used. Although this confirmed the results,
the underlying reason is not clear. In subsequent experiments, the
experimental design will be improved. In addition, the signaling
pathways involved in autophagy remain unclear, and the study only
presented the results of in vitro experiments. Consequently,
further research is required to address these issues.
In conclusion, the present study revealed for the
first time that autophagy serves as a protective mechanism of BTXA
in H/R-treated endothelial cells in vitro. Thus, the study
confirmed that autophagy activation may be a crucial strategy for
ischemia/reperfusion-induced skin flap injury.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation Grants of China (grant nos. 81571922 and
81272130), and the Beijing Natural Science Foundation Grants of
China (grant no. 7172065).
Availability of data and materials
The datasets used and analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HL conceived and designed experiments; YS and JC
performed the experiments; YS and CC analyzed the data; and YS
wrote the manuscript. All authors agreed and approved the final
version of the manuscript.
Ethics approval and consent to
participate
The study was authorized by the Ethics Committee of
Anzhen Hospital (Beijing, China; approval no. 2015021X), and all
patients provided signed informed consent.
Patient consent for publication
All patients agree to the publication of this
study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Clemens MW, Higgins JP and Wilgis EF:
Prevention of anastomotic thrombosis by botulinum toxin a in an
animal model. Plast Reconstr Surg. 123:64–70. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pretto EA Jr: Reperfusion injury of the
liver. Transplant Proc. 23:1912–1914. 1991.PubMed/NCBI
|
|
3
|
Woolfson RG, Millar CG and Neild GH:
Ischaemia and reperfusion injury in the kidney: Current status and
future direction. Nephrol Dial Transplant. 9:1529–1531.
1994.PubMed/NCBI
|
|
4
|
Carroll WR and Esclamado RM:
Ischemia/reperfusion injury in microvascular surgery. Head Neck.
22:700–713. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kasuya A, Sakabe J and Tokura Y: Potential
application of in vivo imaging of impaired lymphatic duct to
evaluate the severity of pressure ulcer in mouse model. Sci Rep.
4:41732014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang WZ, Fang XH, Stephenson LL, Khiabani
KT and Zamboni WA: Ischemia/reperfusion-induced necrosis and
apoptosis in the cells isolated from rat skeletal muscle. J Orthop
Res. 26:351–356. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Küçüker I, Tuncer S, Sencan A, Bircan F,
Cağlar E, Elmas C and Ayhan S: The effect of surgical and chemical
denervation on ischaemia/reperfusion injury of skeletal muscle. J
Plast Reconstr Aesthet Surg. 65:240–248. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Akcal A, Sevim KZ, Yesilada A, Kiyak V,
Sucu DO, Tatlidede HS, Sakiz D and Kaya H: Comparison of
perivascular and intramuscular applied botulinum toxin a
pretreatment on muscle flap ischemia-reperfusion injury and
chemical delay. J Craniofac Surg. 24:278–283. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Curin Y, Ritz MF and Andriantsitohaina R:
Cellular mechanisms of the protective effect of polyphenols on the
neurovascular unit in strokes. Cardiovasc Hematol Agents Med Chem.
4:277–288. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xie R, Li X, Ling Y, Shen C, Wu X, Xu W
and Gao X: Alpha-lipoic acid pre- and post-treatments provide
protection against in vitro ischemia-reperfusion injury in cerebral
endothelial cells via Akt/mTOR signaling. Brain Res. 1482:81–90.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Olmez I and Ozyurt H: Reactive oxygen
species and ischemic cerebrovascular disease. Neurochem Int.
60:208–212. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Arnold PB, Fang T, Songcharoen SJ, Ziakas
G and Zhang F: Inflammatory response and survival of pedicled
abdominal flaps in a rat model after perivascular application of
botulinum toxin type a. Plast Reconstr Surg. 133:491e–498e. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schweizer DF, Schweizer R, Zhang S, Kamat
P, Contaldo C, Rieben R, Eberli D, Giovanoli P, Erni D and Plock
JA: Botulinum toxin A and B raise blood flow and increase survival
of critically ischemic skin flaps. J Surg Res. 184:1205–1213. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mizushima N, Levine B, Cuervo AM and
Klionsky DJ: Autophagy fights disease through cellular
self-digestion. Nature. 451:1069–1075. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Klionsky DJ and Emr SD: Autophagy as a
regulated pathway of cellular degradation. Science. 290:1717–1721.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nakai A, Yamaguchi O, Takeda T, Higuchi Y,
Hikoso S, Taniike M, Omiya S, Mizote I, Matsumura Y, Asahi M, et
al: The role of autophagy in cardiomyocytes in the basal state and
in response to hemodynamic stress. Nat Med. 13:619–624. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kroemer G, Marino G and Levine B:
Autophagy and the integrated stress response. Mol Cell. 40:280–293.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Moreau K, Luo S and Rubinsztein DC:
Cytoprotective roles for autophagy. Curr Opin Cell Biol.
22:206–211. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Levine B and Yuan J: Autophagy in cell
death: An innocent convict? J Clin Invest. 115:2679–2688. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fulda S and Kogel D: Cell death by
autophagy: Emerging molecular mechanisms and implications for
cancer therapy. Oncogene. 34:5105–5113. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Green DR and Levine B: To be or not to be?
How selective autophagy and cell death govern cell fate. Cell.
157:65–75. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cui H, Li X, Li N, Qi K, Li Q, Jin C,
Zhang Q, Jiang L and Yang Y: Induction of autophagy by Tongxinluo
through the MEK/ERK pathway protects human cardiac microvascular
endothelial cells from hypoxia/reoxygenation injury. J Cardiovasc
Pharmacol. 64:180–190. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dong W, Xiao S, Cheng M, Ye X and Zheng G:
Minocycline induces protective autophagy in vascular endothelial
cells exposed to an in vitro model of ischemia/reperfusion-induced
injury. Biomed Rep. 4:173–177. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ghanbarzadeh K, Tabatabaie OR, Salehifar
E, Amanlou M and Khorasani G: Effect of botulinum toxin A and
nitroglycerin on random skin flap survival in rats. Plast Surg
(Oakv). 24:99–102. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang HC, Zhang HF, Guo WY, Su H, Zhang KR,
Li QX, Yan W, Ma XL, Lopez BL, Christopher TA and Gao F: Hypoxic
postconditioning enhances the survival and inhibits apoptosis of
cardiomyocytes following reoxygenation: Role of peroxynitrite
formation. Apoptosis. 11:1453–1460. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cordivari C, Misra VP, Catania S and Lees
AJ: New therapeutic indications for botulinum toxins. Mov Disord. 8
Suppl 19:S157–S161. 2004. View Article : Google Scholar
|
|
27
|
Bhidayasiri R and Truong DD: Expanding use
of botulinum toxin. J Neurol Sci. 235:1–9. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rohrich RJ, Janis JE, Fagien S and Stuzin
JM: The cosmetic use of botulinum toxin. Plast Reconstr Surg. 112
Suppl 5:(177S): 188S, 188S, 192S; discussion. 189S–191S. 2003.
|
|
29
|
Klein AW: The therapeutic potential of
botulinum toxin. Dermatol Surg. 30:452–455. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Maria G, Brisinda G, Bentivoglio AR,
Cassetta E, Gui D and Albanese A: Botulinum toxin injections in the
internal anal sphincter for the treatment of chronic anal fissure:
Long-term results after two different dosage regimens. Ann Surg.
228:664–669. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Patel S and Martino D: Cervical dystonia:
From pathophysiology to pharmacotherapy. Behav Neurol. 26:275–282.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Winner PK, Sadowsky CH, Martinez WC,
Zuniga JA and Poulette A: Concurrent onabotulinumtoxinA treatment
of cervical dystonia and concomitant migraine. Headache.
52:1219–1225. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Olver J, Esquenazi A, Fung VS, Singer BJ
and Ward AB; Cerebral Palsy Institute, : Botulinum toxin
assessment, intervention and aftercare for lower limb disorders of
movement and muscle tone in adults: International consensus
statement. Eur J Neurol. 2 Suppl 17:S57–S73. 2010. View Article : Google Scholar
|
|
34
|
Schulte-Baukloh H: Botulinum toxin for
neurogenic bladder dysfunction. Urologe A. 51:198–203. 2012.(In
German). View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Arnold PB, Merritt W, Rodeheaver GT,
Campbell CA, Morgan RF and Drake DB: Effects of perivascular
botulinum toxin-A application on vascular smooth muscle and flap
viability in the rat. Ann Plast Surg. 62:463–467. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sycha T, Graninger M, Auff E and Schnider
P: Botulinum toxin in the treatment of raynaud's phenomenon: A
pilot study. Eur J Clin Invest. 34:312–313. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Neumeister MW: Botulinum toxin type A in
the treatment of raynaud's phenomenon. J Hand Surg Am.
35:2085–2092. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim YS, Roh TS, Lee WJ, Yoo WM and Tark
KC: The effect of botulinum toxin A on skin flap survival in rats.
Wound Repair Regen. 17:411–417. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Uchiyama A, Yamada K, Perera B, Ogino S,
Yokoyama Y, Takeuchi Y, Ishikawa O and Motegi S: Protective effect
of botulinum toxin A after cutaneous ischemia-reperfusion injury.
Sci Rep. 5:90722015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Park TH, Lee SH, Park YJ, Lee YS, Rah DK
and Kim SY: Presurgical botulinum toxin a treatment increases
angiogenesis by hypoxia-inducible factor-1α/vascular endothelial
growth factor and subsequent superiorly based transverse rectus
abdominis myocutaneous flap survival in a rat model. Ann Plast
Surg. 76:723–728. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Tang YH, Pennington LA, Scordino JW,
Alexander JS and Lian T: Dynamics of early stem cell recruitment in
skin flaps subjected to ischemia reperfusion injury.
Pathophysiology. 23:221–228. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Huang L: Beneficial effect of botulinum
toxin A on secondary ischaemic injury of skin flaps in rats. Br J
Oral Maxillofac Surg. 56:144–147. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Karalliedde LD and Kappagoda CT: The
challenge of traditional chinese medicines for allopathic
practitioners. Am J Physiol Heart Circ Physiol. 297:H1967–H1969.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang L, Liu Y, Lu XT, Wu YL, Zhang C, Ji
XP, Wang R, Liu CX, Feng JB, Jiang H, et al: Traditional Chinese
medication Tongxinluo dose-dependently enhances stability of
vulnerable plaques: A comparison with a high-dose simvastatin
therapy. Am J Physiol Heart Circ Physiol. 297:H2004–H2014. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wu T, Harrison RA, Chen X, Ni J, Zhou L,
Qiao J, Wang Q, Wei J, Xin D and Zheng J: Tongxinluo (Tong xin luo
or Tong-xin-luo) capsule for unstable angina pectoris. Cochrane
Database Syst Rev. 18:D44742006.
|
|
46
|
Chen WQ, Zhong L, Zhang L, Ji XP, Zhao YX,
Zhang C, Jiang H, Wu YL and Zhang Y: Chinese medicine tongxinluo
significantly lowers serum lipid levels and stabilizes vulnerable
plaques in a rabbit model. J Ethnopharmacol. 124:103–110. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Klionsky DJ, Abdalla FC, Abeliovich H,
Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M,
Agostinis P, Aguirre-Ghiso JA, et al: Guidelines for the use and
interpretation of assays for monitoring autophagy. Autophagy.
8:445–544. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang JS, Tseng CY and Chao MW: Diesel
exhaust particles contribute to endothelia apoptosis via autophagy
pathway. Toxicol Sci. 156:72–83. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chandrika BB, Yang C, Ou Y, Feng X, Muhoza
D, Holmes AF, Theus S, Deshmukh S, Haun RS and Kaushal GP:
Endoplasmic reticulum stress-induced autophagy provides
cytoprotection from chemical hypoxia and oxidant injury and
ameliorates renal ischemia-reperfusion injury. PLoS One.
10:e1400252015. View Article : Google Scholar
|
|
50
|
Nicoletti F, Fagone P, Meroni P, McCubrey
J and Bendtzen K: mTOR as a multifunctional therapeutic target in
HIV infection. Drug Discov Today. 16:715–721. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Donia M, McCubrey JA, Bendtzen K and
Nicoletti F: Potential use of rapamycin in HIV infection. Br J Clin
Pharmacol. 70:784–793. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Nicoletti F, Lapenta C, Donati S, Spada M,
Ranazzi A, Cacopardo B, Mangano K, Belardelli F, Perno C and Aquaro
S: Inhibition of human immunodeficiency virus (HIV-1) infection in
human peripheral blood leucocytes-SCID reconstituted mice by
rapamycin. Clin Exp Immunol. 155:28–34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Donia M, Mangano K, Amoroso A, Mazzarino
MC, Imbesi R, Castrogiovanni P, Coco M, Meroni P and Nicoletti F:
Treatment with rapamycin ameliorates clinical and histological
signs of protracted relapsing experimental allergic
encephalomyelitis in Dark Agouti rats and induces expansion of
peripheral CD4+CD25+Foxp3+ regulatory T cells. J Autoimmun.
33:135–140. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bao XH, Naomoto Y, Hao HF, Watanabe N,
Sakurama K, Noma K, Motoki T, Tomono Y, Fukazawa T, Shirakawa Y, et
al: Autophagy: Can it become a potential therapeutic target? Int J
Mol Med. 25:493–503. 2010.PubMed/NCBI
|
|
55
|
Maksimovic-Ivanic D, Fagone P, McCubrey J,
Bendtzen K, Mijatovic S and Nicoletti F: HIV-protease inhibitors
for the treatment of cancer: Repositioning HIV protease inhibitors
while developing more potent NO-hybridized derivatives? Int J
Cancer. 140:1713–1726. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Han Y, Fan S, Qin T, Yang J, Sun Y, Lu Y,
Mao J and Li L: Role of autophagy in breast cancer and breast
cancer stem cells (Review). Int J Oncol. 52:1057–1070.
2018.PubMed/NCBI
|
|
57
|
Zhang X, Cheng Q, Yin H and Yang G:
Regulation of autophagy and EMT by the interplay between p53 and
RAS during cancer progression (Review). Int J Oncol. 51:18–24.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Horie R, Nakamura O, Yamagami Y, Mori M,
Nishimura H, Fukuoka N and Yamamoto T: Apoptosis and antitumor
effects induced by the combination of an mTOR inhibitor and an
autophagy inhibitor in human osteosarcoma MG63 cells. Int J Oncol.
48:37–44. 2016. View Article : Google Scholar : PubMed/NCBI
|