Introduction
One-lung ventilation (OLV) is a commonly used
machine-controlled respiratory management method in clinical
thoracic surgery. OLV, as a non-physiological method, may cause
non-ventilation lung collapse in patients undergoing thoracotomy.
Pulmonary collapse can cause hypoxic and ischemic phenomena, and
hypoxia can easily cause lung injury. If ventilated lung tidal
volume is higher than double-lung ventilation, it can cause
high-grade lung injury and eventually acute lung injury (ALI)
(1,2). During OLV process, the ratio of
intrapulmonary ventilation/blood flow can be deregulated.
Hypoventilation may cause hypoxia and ischemic damage and
destruction of cells. In the process of restoring blood supply, it
can also increase the damage of cells, cause pathological and
physiological changes in the lungs, and induce ischemia-reperfusion
injury, and hypoxemia followed by acute inflammatory reactions and
oxidative stress reactions, causing acute injury to multiple organs
throughout the body (3,4). Studies have shown that prolonged OLV
and mechanical stimuli resulting from surgery can lead to the
activation of inflammatory cells in patients, and the release of
inflammatory factors may cause increased inflammatory reactions in
the lungs and increased lung injury in patients, which in turn
increase the risk of postoperative death (5). Therefore, the application of effective
measures for the protection of lung tissue in patients undergoing
OLV open surgery will improve the prognosis of patients.
Dexmedetomidine (DEX) is an α2 receptor agonist that
binds to α2 receptors and has anti-anxiety, sedative,
anti-inflammatory, antioxidative and inhibition of sympathetic
effects. DEX is commonly used in clinical anesthesia (6). Studies have shown that DEX can play a
protective role in organ hypoxia and ischemic injury (7). However, the anti-inflammatory and lung
protective effects of DEX-assisted anesthesia during surgery have
not been elucidated. In this study, we established a rat model of
lung injury induced by OLV and explored the role of DEX in OLV.
Materials and methods
Experimental animals
A total of 92 purebred inbred strain Sprague-Dawley
rats (5–11 weeks, 260–310 g) were purchased from Yison BIO
(Shanghai, China) [SCXK (Shanghai) 2009–0037]. Rats were raised in
a clean environment with good ventilation (21–26°C, humidity of
51–57%), they were allowed to access food and water freely, and
were fasted for 6 h before the experiments. Animal experiments were
approved by the Ethics Committee of the Affiliated Hospital of
Qingdao University (Qingdao, China) and the experimental procedures
were in compliance with the ‘Guidelines for the Protection and Use
of Laboratory Animals of the National Institutes of Health’
(8).
Main instruments, reagents, and
medications
Rat ventilator was purchased from Shanghai Yuyan
Instruments Co., Ltd. (Shanghai, China). Small animal physiological
monitor was purchased from the STARR Life Sciences Corp.™ (Oakmont,
PA, USA). DEX injection was purchased from Jiangsu Enhua
Pharmaceutical Co., Ltd. (Jiangsu, China) (batch no. H20110085).
Interleukin (IL)-6, IL-10 and tumor necrosis factor-α (TNF-α)
detection kits were purchased from Wuhan AmyJet Scientific, Inc.
(Wuhan, China). Malondialdehyde (MDA) and superoxide dismutase
(SOD) were purchased from Shanghai Xueman Biotechnology Co., Ltd.
(Shanghai, China).
Animal model preparation
Following the principle of similar body weight, the
rats were divided into group A, B, C and D, with 23 cases in each
group. According to the methods described by Pruszkowski et
al (9), an OLV rat model was
established. Modeling method: rats were intraperitoneally injected
with 3% pentobarbital at a dose of 40 mg/kg. After anesthesia, the
rats were fixed in supine position and limbs were fixed. Trachea
was incised and tracheal intubation was performed. Rats in group A
were subjected to bilateral ventilation for 2 h. Catheter (~3 cm)
was placed into the right main bronchus of the rats in groups B, C
and D, and the catheter was connected to a ventilator to perform
right lung OLV for 2 h. Mechanical ventilation parameters: RR=55
times/min, I:E=1:1, VT=10 ml/kg, FiO2 100%, PEEP=0. During
ventilation process, the left lobe was not opened. After
replantation, the endotracheal tube was retracted to 2 cm of the
main trachea. RR was adjusted to 65 times/min, and VT was set to 10
ml/kg for bilateral ventilation for 10 min. Transfemoral puncture
catheter was performed and connected to a physiological monitor to
monitor blood pressure, oxygen saturation, and heart rate. Blood
pressure was maintained at 70–130 mmHg, blood oxygen saturation was
maintained at ~99%, and heart rate was 250–380/min.
Experimental grouping
Before modeling (15 min), rats in group C were
injected with 5 ml of 0.9% sodium chloride. Rats in group D were
injected with DEX at a speed of 5 µg/kg/h for microinjection pump
for 50 min. Group A rats were ventilated in both lungs for 2 h.
Rats in groups B, C and D were subjected to OLV for 2 h and
bilateral ventilation for 10 min.
Observation indicators
After the rat model was established, rats were
anesthetized by intraperitoneal injection of pentobarbital sodium
(45 mg/kg), and the rats in each group were sacrificed by
decapitation. Left lung tissue was collected, homogenized and
centrifuged at 3,000 × g for 10 min at 4°C to collect the
supernatant. Concentrations of IL-6, IL-10 and TNF-α in the lung
tissue of rats were detected by ELISA double-antibody sandwich
method. MDA concentration and SOD activity in the lung tissue of
rats were detected by radioimmunoassay (Beyotime Institute of
Biotechnology, Shanghai, China), and the detection scheme was
carried out according to the manufacturer's instructions. The right
lung tissue was taken and washed with physiological saline at 4°C,
and liquid was absorbed using filter paper. Lung tissue wet weight
(W) was weighed, and the lung tissue dry weight (D) was measured
after being placed in a 70% electric oven for 24 h to calculate the
W/D of the lung tissue.
Statistical methods
SPSS 19.0 [Yiyun (Shanghai) Information Technology
Co., Ltd., Shanghai, China] was used for statistical analysis.
Measured data were expressed as mean ± standard deviation (SD).
Chi-square test was used for comparisons of count data. Comparisons
among multiple groups were performed by one-way analysis of
variance, followed by LSD-t post hoc test. P<0.05 was considered
to indicate a statistically significant difference.
Results
General information of the four groups
of rats
A rat in each group C and D died due to anesthesia
and the success rate was 95.65% (22/23). Rats in both groups A and
B were successfully modeled. There was no significant difference in
sex, age, body weight, indoor temperature and indoor humidity among
the four groups (p>0.05) (Table
I).
 | Table I.General information of the four groups
of rats [n (%)] (mean ± SD). |
Table I.
General information of the four groups
of rats [n (%)] (mean ± SD).
| Items | A (n=23) | B (n=23) | C (n=22) | D (n=22) | χ2 | P-value |
|---|
| Sex |
|
|
|
| 2.391 | 0.492 |
| Male | 15 (65.22) | 11 (47.83) | 13 (59.09) | 10 (45.45) |
|
|
|
Female | 8
(34.78) | 12 (52.17) | 9
(40.91) | 12 (54.55) |
|
|
| Indoor temperature
(°C) | 23.98±1.39 | 24.03±1.12 | 23.83±1.27 | 24.18±1.22 | 0.291 | 0.831 |
| Indoor humidity
(%) | 53.87±1.33 | 54.42±1.26 | 54.31±1.02 | 54.51±0.98 | 1.360 | 0.260 |
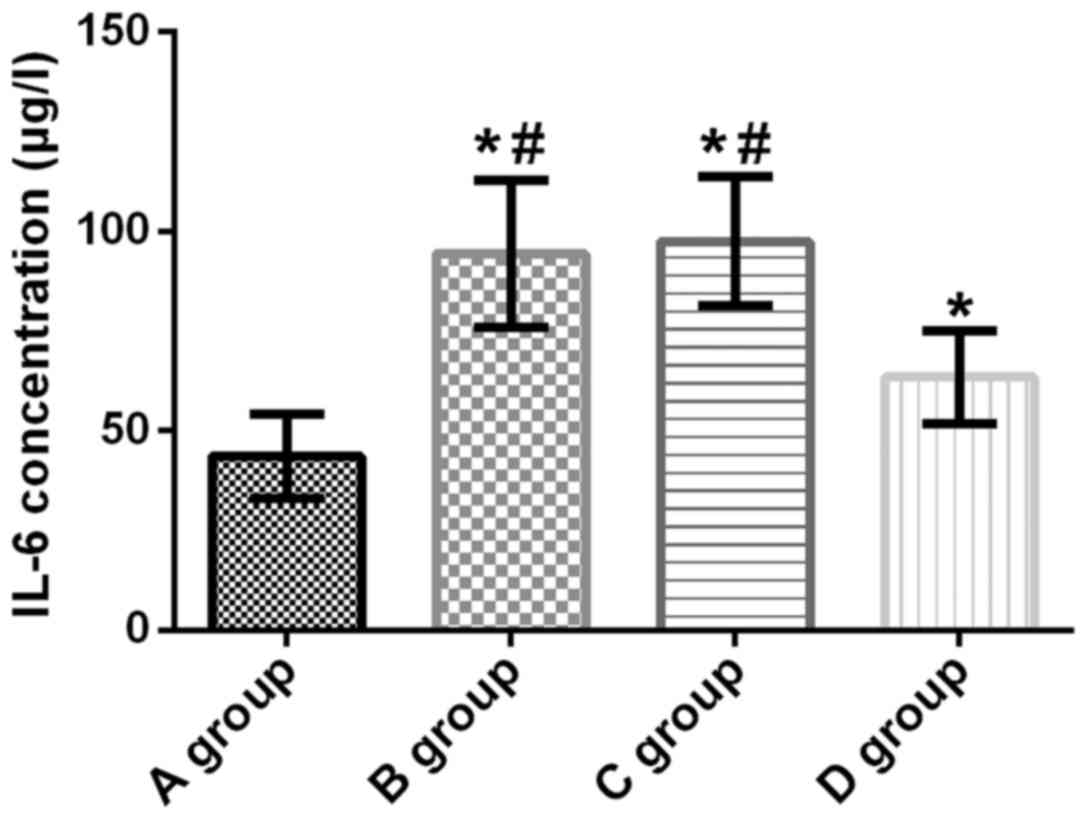
IL-6 concentration in lung tissue of
rats
The concentration of IL-6 in lung tissue of rats in
groups A, B, C and D was 43.63±10.62, 94.34±18.45, 97.58±16.17 and
63.41±11.54 µg/l, respectively. Compared with group A, IL-6
concentration in the lung tissue of rats in groups B, C and D was
significantly increased (t=11.420, p<0.001; t=13.290,
p<0.001; t=5.987, p<0.001). Compared with groups B and C, the
L-6 concentration in group D was significantly decreased (t=6.706,
p<0.001; t=8.068, p<0.001). There was no significant
difference in the concentration of IL-6 between groups B and C
(t=0.625, p=0.535) (Fig. 1).
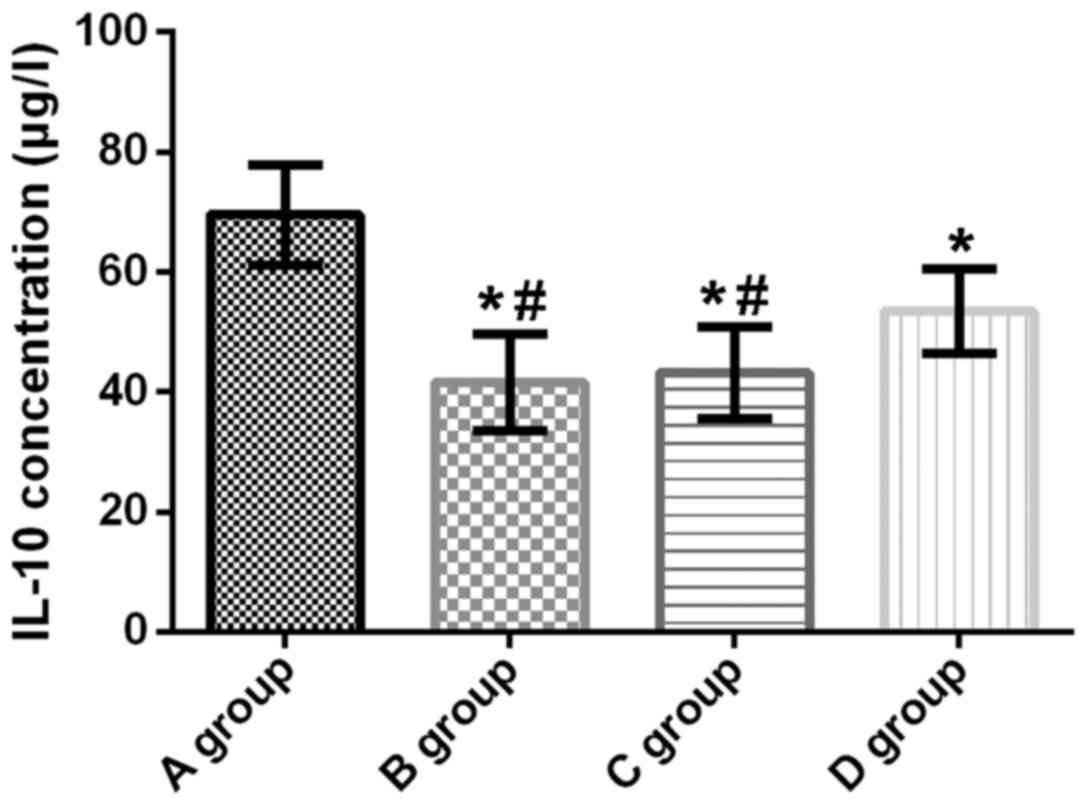
Concentration of IL-10 in lung tissue
of rats
The concentration of IL-10 in the lungs of rats in
groups A, B, C and D was 69.52±8.41, 41.63±8.02, 43.18±7.64 and
53.45±7.01 µg/l, respectively. Compared with group A, IL-10
concentration in the lung tissue of rats in groups B, C and D was
significantly decreased (t=11.510, p<0.001; t=10.980,
p<0.001; t=6.946, p<0.001). Compared with groups B and C, the
IL-10 concentration in group D was significantly increased
(t=5.254, p<0.001; t=4.646, p<0.001). There was no
significant difference in IL-10 concentration between groups B and
C (t=0.663, p=0.510) (Fig. 2).
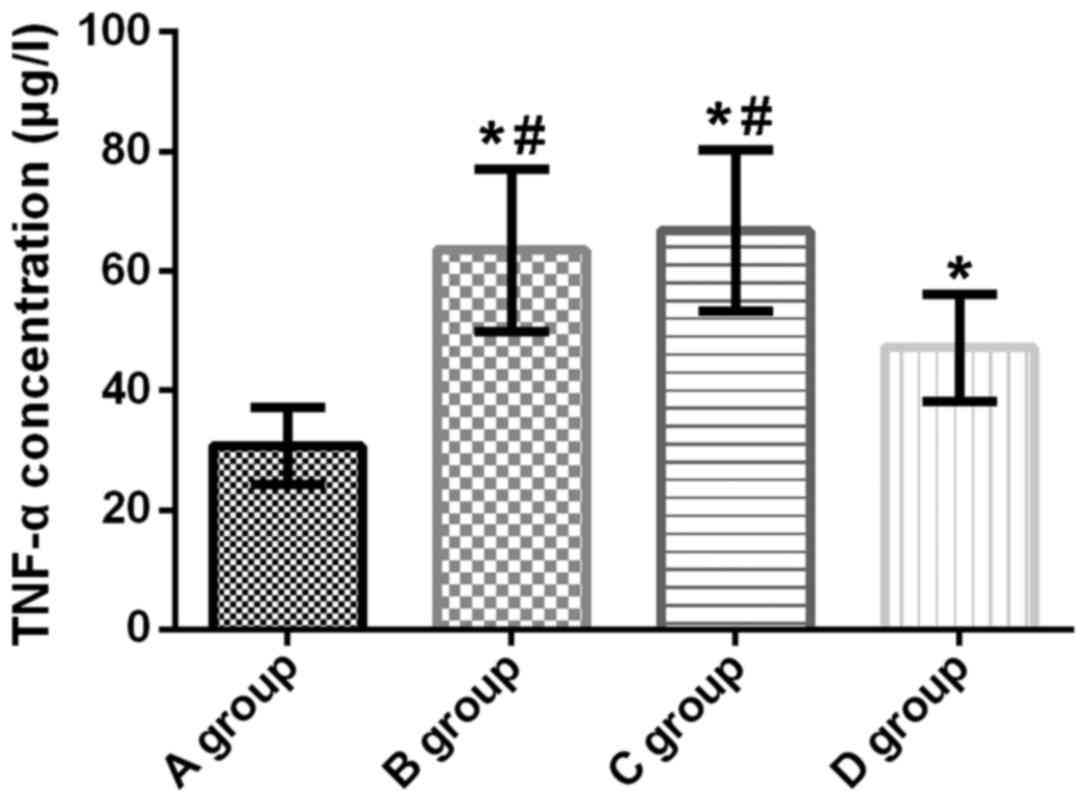
TNF-α concentration in lung tissue of
rats
The concentration of TNF-α in the lung tissue of
rats in groups A, B, C and D was 30.74±6.41, 63.54±13.58,
66.79±13.86, and 47.21±8.97 µg/l, respectively. Compared with group
A, the concentration of TNF-α in the lung tissue of rats in groups
B, C and D was significantly increased (t=10.460, p<0.001;
t=11.280, p<0.001; t=7.111, p<0.001). Compared with groups B
and C, the concentration of TNF-α in group D was significantly
decreased (t=5.563, p<0.001; t=4.725, p<0.001). There was no
significant difference in the concentration of TNF-α between groups
B and C (t=0.794, p=0.431) (Fig.
3).
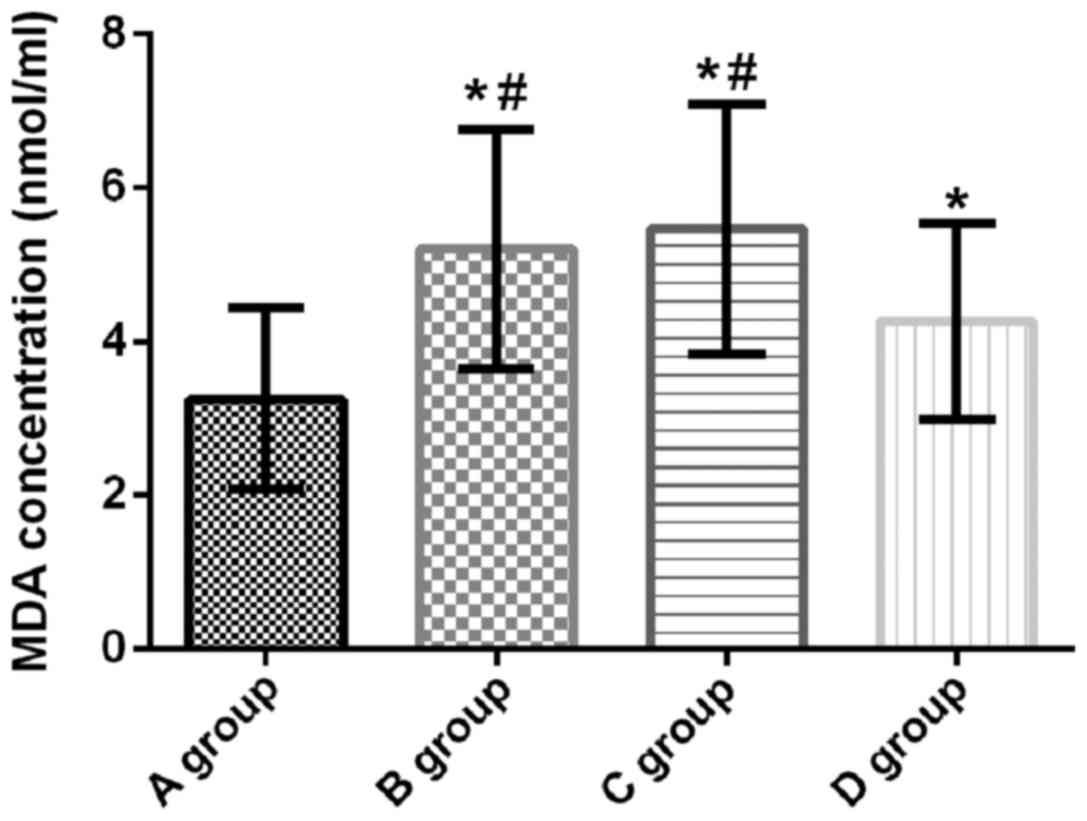
MDA concentration in lung tissue of
rats
MDA concentration in the lung tissue of rats in
groups A, B, C and D was 3.25±1.18, 5.21±1.56, 5.46±1.63, and
4.26±1.28 nmol/ml, respectively. Compared with group A, MDA
concentration in the lung tissue of rats in groups B, C and D was
significantly increased (t=4.767, p<0.001; t=5.227, p<0.001;
t=2.754, p=0.008). Compared with groups B and C, the MDA
concentration in group D was significantly decreased (t=2.228,
p=0.031; t=2.716, p=0.009). There was no significant difference in
the concentration of MDA between groups B and C (t=0.525, p=0.601)
(Fig. 4).
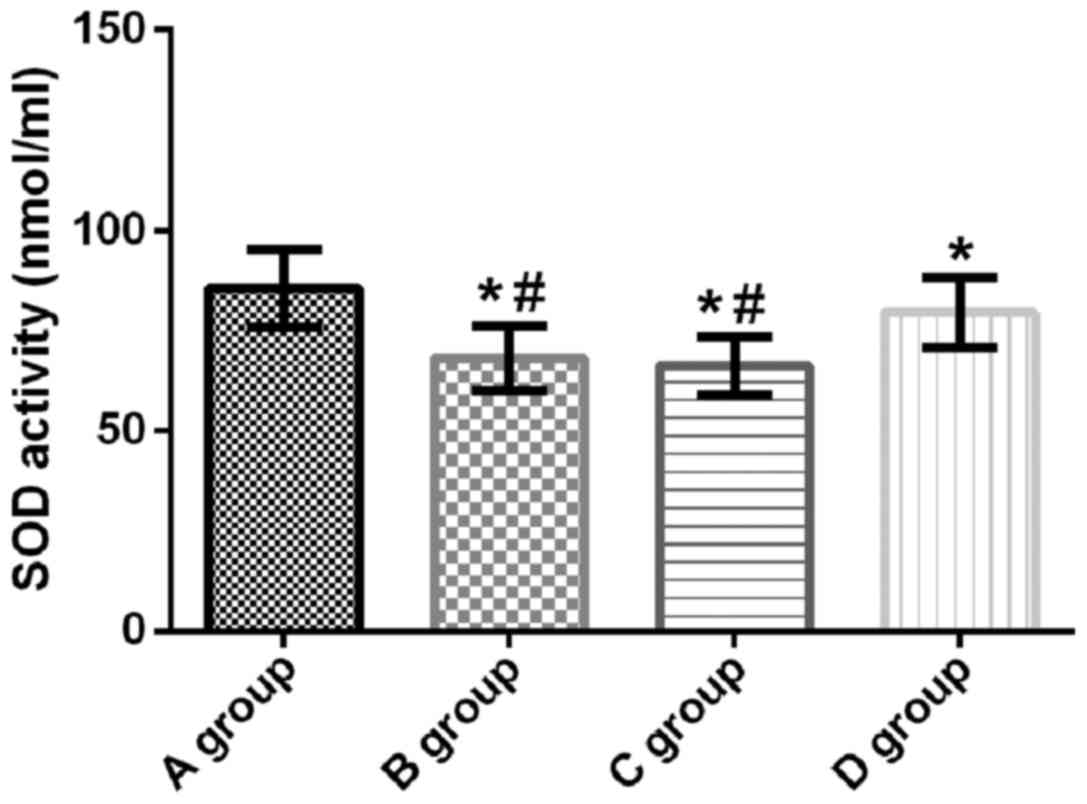
SOD activity in lung tissue of
rats
SOD activity in lung tissue of rats in groups A, B,
C and D was 85.63±9.64, 68.14±8.06, 66.46±7.24 and 79.72±8.77 U/ml,
respectively. Compared with group A, SOD activity in groups B, C
and D was significantly decreased (t=6.675, p<0.001; t=7.516,
p<0.001; t=2.148, p=0.037). Compared with groups B and C, the
SOD activity in group D was significantly increased (t=4.615,
p<0.001; t=5.469, p<0.001). There was no significant
difference in SOD activity between groups B and C (t=0.734,
p=0.466) (Fig. 5).
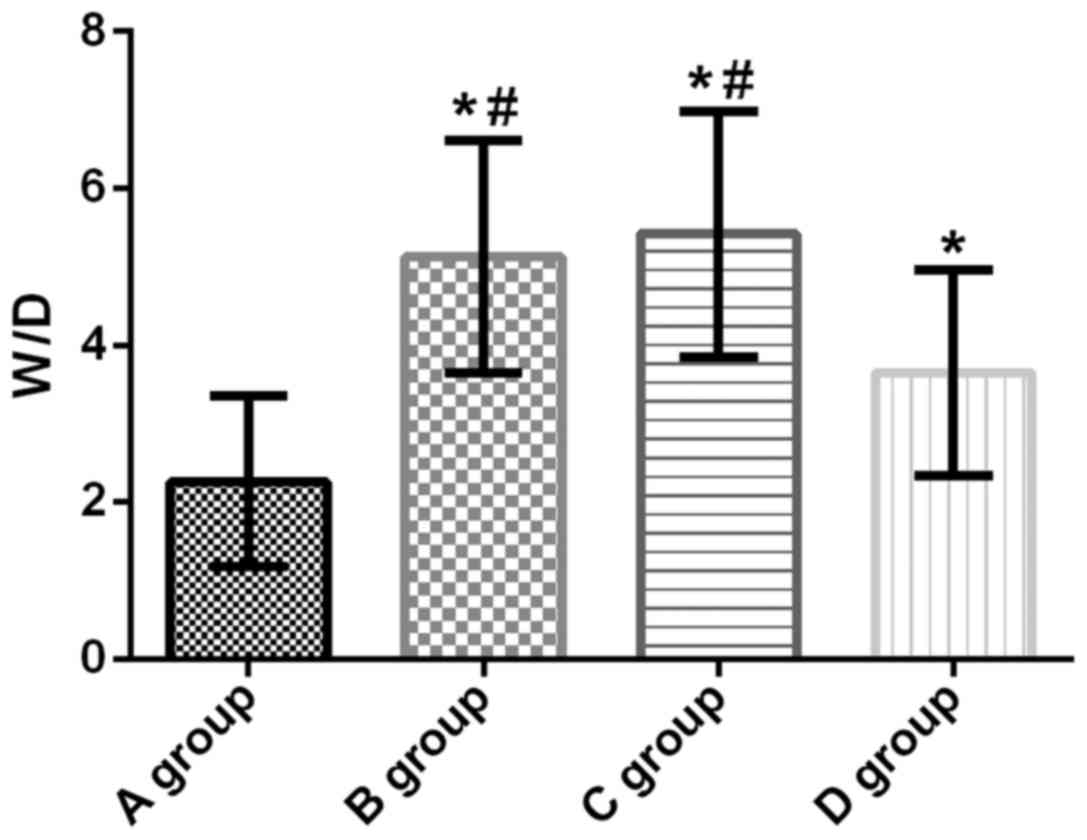
W/D of lung tissue of rats in all four
groups
W/D of lung tissues of rats in groups A, B, C and D
was 2.26±1.09, 5.13±1.48, 5.42±1.57 and 3.65±1.31, respectively.
Compared with group A, W/D in lung tissue of groups B, C and D was
significantly increased (t=7.488, p<0.001; t=7.873, p<0.001;
t=3.876, p=0.037). Compared with groups B and C, the W/D in group D
was significantly decreased (t=3.546, p=0.001; t=4.060,
p<0.001). There was no significant difference in lung tissue W/D
between groups B and C (t=0.637, p=0.527) (Fig. 6).
Discussion
OLV is the use of single- or double-lumen
endotracheal intubation to open the lungs of patients with open
chest. The purpose of OLV is to obtain enough surgical vision, in
order to reduce the ipsilateral lung traction, improve the clarity
of the lungs, and separate the lungs to avoid effects of cancer
cells and secretions from the affected lung on the healthy one
(10). However, although OLV brings
convenience to the operation, it may also cause lung injury of
different severity. OLV may not only cause ALI, but also affect
other organs, resulting in their functional disorder and affecting
patients' prognosis and survival rate (11). OLV can also cause severe inflammatory
reactions in the body. Excessive inflammatory response to the lungs
is a progressive process of injury that can progress from ALI to
systemic inflammatory response syndrome and multiple organ
dysfunction syndrome (12). Studies
have shown that the mortality rate from intra- and postoperative
cardiac surgery caused by ALI is ~3% (13). Therefore, reducing the OLV
inflammatory response and lung injury has important clinical
significance.
DEX is a highly selective α2 adrenoceptor agonist.
Sedative effect of DEX is achieved by its high affinity for α2
distributed in the central nervous system. α2 is mainly distributed
in locus coeruleus in the brain and plays an important role
in regulating awakening and sleeping, and it can also inhibit the
central sympathetic activity and reduce the release of inflammatory
mediators (14). Abnormal
upregulation of the proinflammatory mediators of the lungs and
neutrophil infiltration can all lead to inflammatory reactions in
the body, resulting in inflammatory factors that cause tissue
damage and lung injury. Inflammatory response is the main cause of
lung injury (15). Concentrations of
IL-6, IL-10, and TNF-α reflect the severity of inflammation in the
body and are important inflammatory factors (16,17).
IL-6 plays an important role in the body's immune and stress
response, and has both pro- and anti-inflammatory effects. When the
body is stimulated by external factors, the concentration of
circulating IL-6 can be gradually increased (18). IL-10 has a strong inhibitory effect
on the production of pro-inflammatory cytokines, and is a potent
inhibitory cytokine to protect the body from inflammation (19). TNF-α is a start factor in ALI, it
plays an initial role in the development and progression of
inflammatory reactions, and has immunomodulatory and anti-infective
effects (20). Akdis et al
(21) have shown that IL-10 can
inhibit the release of IL-6 and TNF-α from monocytes and increase
the level of TNF-α receptor, thus reducing the adhesion of white
blood cells to vascular endothelial cells, inhibiting the
expression of endothelial cell adhesion factors and reducing lung
injury. The results of this study revealed that compared with group
A, the concentrations of IL-6 and TNF-α in the lungs of rats in
groups B, C and D were significantly increased, and the
concentration of IL-10 significantly decreased; compared with
groups B and C, the concentrations of IL-6 and TNF-α in the lungs
of rats in group D were significantly decreased, but the
concentration of IL-10 was significantly increased. It is suggested
that the application of DEX can reduce the inflammatory reaction in
the lung and play a protective role during OLV ventilation. We
speculate that DEX can control the inflammatory response by
inhibiting macrophage hyper-responsiveness and protecting
macrophage activity (22). Tan et
al (23) have also reported that
DEX can reduce the severity of inflammation and the incidence of
lung injury in OLV in sepsis rats, and it protects the lungs by
inhibiting the release of inflammatory factors.
OLV can cause inflammation in the lungs. Prolonged
infiltration of inflammatory factors can lead to thickening of the
lung parenchyma and significant congestion, causing pulmonary
edema. Lung tissue W/D is an important indicator of lung water
content. Changes in W/D ratio reflect lung edema and degree of lung
injury (24). During OLV, occurrence
of unilateral pulmonary atrophy will lead to ischemia, hypoxia, and
increased oxygen-free radical production and strengthened
scavenging ability in lung tissue. Reperfusion may occur in the
atrophic lung tissue, and during this process, excessive
oxygen-free radicals will be generated, which can induce lipid
peroxidation and the formation of lipid peroxides (25). Abnormal increase in the concentration
of MDA indicates strong lipid peroxidation and severe damage to the
mitochondrial and cell membrane (26). SOD is the scavenger of oxygen-free
radicals whose activity reflects the antioxidant capacity. Thus,
changes in MDA concentration and SOD activity reflect the degree of
lung injury (27). The results of
this study showed that compared with group A, MDA concentration and
W/D in lung tissue of rats in groups B, C and D were significantly
increased, but SOD activity was significantly decreased. Compared
with groups B and C, MDA concentration and W/D in group D were
significantly decreased, but SOD activity was significantly
increased, suggesting that DEX could interfere with oxygen-free
radicals and inhibit lipid peroxidation, and can protect the lung
by inhibiting the release of inflammatory factors and relieving
pulmonary edema. Shen et al (28) have also shown that the protective
effect of DEX on myocardium is mainly achieved by free radical
scavenging and antioxidative activities.
In this study, the rats were all screened strictly.
No difference in sex, age, body weight, indoor temperature and
indoor humidity was found among all four groups, indicating the
high reliability of our data. The establishment of OLV model in
rats is simple and of low cost with favorable use value. However,
there are still some limitations. Pathogenesis of lung injury in
OLV rat model was not investigated and clinical application of DEX
was not elucidated in this study. Therefore, we hope that in the
next study, we will be able to select the research objects in
clinic and study in depth the pathogenesis of lung injury caused by
OLV, in order to further confirm the results of the present
study.
In summary, DEX can inhibit the production of
inflammatory factors in the development and progress of pulmonary
inflammation and lipid peroxidation, relieve pulmonary edema and
reduce lung injury after OLV to protect the lung.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JW and XY conceived and designed the study and
assisted with the animal model preparation. LJ and HD were
responsible for ELISA. WF and CC performed the radioimmunoassay.
JW, XY and SW contributed to the statistical analysis. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
the Affiliated Hospital of Qingdao University (Qingdao, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lu Y, Dai W, Zong Z, Xiao Y, Wu D, Liu X
and Wong Chun GT: Bronchial blocker versus left double-lumen
endotracheal tube for one-lung ventilation in right video-assisted
thoracoscopic surgery. J Cardiothorac Vasc Anesth. 32:297–301.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
El Tahan MR, Pasin L, Marczin N and
Landoni G: Impact of low tidal volumes during one-lung ventilation.
A meta-analysis of randomized controlled trials. J Cardiothorac
Vasc Anesth. 31:1767–1773. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Murphy E and Shelley B: Response to:
‘Postoperative pulmonary complications, pulmonary and systemic
inflammatory responses after lung resection surgery with prolonged
one-lung ventilation. Randomised controlled trial comparing
intravenous and inhalational anaesthesia’. Br J Anaesth.
120:411–412. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Xu Z, Gu L, Bian Q, Li P, Wang L, Zhang J
and Qian Y: Oxygenation, inflammatory response and lung injury
during one lung ventilation in rabbits using inspired oxygen
fraction of 0.6 vs. 1.0. J Biomed Res. 31:56–64. 2016.PubMed/NCBI
|
|
5
|
de la Gala F, Piñeiro P, Reyes A, Vara E,
Olmedilla L, Cruz P and Garutti I: Postoperative pulmonary
complications, pulmonary and systemic inflammatory responses after
lung resection surgery with prolonged one-lung ventilation.
Randomized controlled trial comparing intravenous and inhalational
anaesthesia. Br J Anaesth. 119:655–663. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mahmoud M, Ishman SL, McConnell K, Fleck
R, Shott S, Mylavarapu G, Gutmark E, Zou Y, Szczesniak R and Amin
RS: Upper airway reflexes are preserved during dexmedetomidine
sedation in children with Down syndrome and obstructive sleep
apnea. J Clin Sleep Med. 13:721–727. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang SL, Duan L, Xia B, Liu Z, Wang Y and
Wang GM: Dexmedetomidine preconditioning plays a neuroprotective
role and suppresses TLR4/NF-κB pathways model of cerebral ischemia
reperfusion. Biomed Pharmacother. 93:1337–1342. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Harriss DJ, Macsween A and Atkinson G:
Standards for ethics in sport and exercise science research: 2018
Update. Int J Sports Med. 38:1126–1131. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pruszkowski O, Dalibon N, Moutafis M,
Jugan E, Law-Koune JD, Laloë PA and Fischler M: Effects of propofol
vs. sevoflurane on arterial oxygenation during one-lung
ventilation. Br J Anaesth. 98:539–544. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Clayton-Smith A, Bennett K, Alston RP,
Adams G, Brown G, Hawthorne T, Hu M, Sinclair A and Tan J: A
Comparison of the efficacy and adverse effects of double-lumen
endobronchial tubes and bronchial blockers in thoracic surgery: A
systematic review and meta-analysis of randomized controlled
trials. J Cardiothorac Vasc Anesth. 29:955–966. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lohser J and Slinger P: Lung injury after
one-lung ventilation: A review of the pathophysiologic mechanisms
affecting the ventilated and the collapsed lung. Anesth Analg.
121:302–318. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Blank RS, Colquhoun DA, Durieux ME,
Kozower BD, McMurry TL, Bender SP and Naik BI: Management of
one-lung ventilation: Impact of tidal volume on complications after
thoracic surgery. Anesthesiology. 124:1286–1295. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Primieri P, Ancona P and Gualtieri E:
Unusual airways management during one-lung ventilation in thoracic
surgery. Saudi J Anaesth. 11:225–227. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mantz J, Josserand J and Hamada S:
Dexmedetomidine: New insights. Eur J Anaesthesiol. 28:3–6. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Iwata M, Inoue S, Kawaguchi M, Takahama M,
Tojo T, Taniguchi S and Furuya H: Jugular bulb venous oxygen
saturation during one-lung ventilation under sevoflurane- or
propofol-based anesthesia for lung surgery. J Cardiothorac Vasc
Anesth. 22:71–76. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Aisiku IP, Yamal JM, Doshi P, Benoit JS,
Gopinath S, Goodman JC and Robertson CS: Plasma cytokines IL-6,
IL-8, and IL-10 are associated with the development of acute
respiratory distress syndrome in patients with severe traumatic
brain injury. Crit Care. 20:2882016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Huang SR, Ma AY, Liu Y and Qu Y: Effects
of inflammatory factors including plasma tumor necrosis factor-α in
the clinical treatment of acute respiratory distress syndrome.
Oncol Lett. 13:5016–5020. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Heink S, Yogev N, Garbers C, Herwerth M,
Aly L, Gasperi C, Husterer V, Croxford AL, Möller-Hackbarth K,
Bartsch HS, et al: Trans-presentation of IL-6 by dendritic cells is
required for the priming of pathogenic TH17 cells. Nat Immunol.
18:74–85. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ip WKE, Hoshi N, Shouval DS, Snapper S and
Medzhitov R: Anti-inflammatory effect of IL-10 mediated by
metabolic reprogramming of macrophages. Science. 356:513–519. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Choi ST, Kang EJ, Ha YJ and Song JS:
Levels of plasma-soluble triggering receptor expressed on myeloid
cells-1 (sTREM-1) are correlated with disease activity in
rheumatoid arthritis. J Rheumatol. 39:933–938. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Akdis M, Aab A, Altunbulakli C, Azkur K,
Costa RA, Crameri R, Duan S, Eiwegger T, Eljaszewicz A, Ferstl R,
et al: Interleukins (from IL-1 to IL-38), interferons, transforming
growth factor β, and TNF-α: Receptors, functions, and roles in
diseases. J Allergy Clin Immunol. 138:984–1010. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kuru S, Bozkirli OB, Barlas AM, Duymus ME,
Senes M, Yumusak N, Yilmaz C and Kismet K: The preventive effect of
dexmedetomidine against postoperative intra-abdominal adhesions in
rats. Int Surg. 100:87–95. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tan F, Chen Y, Yuan D, Gong C, Li X and
Zhou S: Dexmedetomidine protects against acute kidney injury
through downregulating inflammatory reactions in endotoxemia rats.
Biomed Rep. 3:365–370. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jiang W, Luo F, Lu Q, Liu J, Li P, Wang X,
Fu Y, Hao K, Yan T and Ding X: The protective effect of Trillin
LPS-induced acute lung injury by the regulations of inflammation
and oxidative state. Chem Biol Interact. 243:127–134. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Heerdt PM and Stowe DF: Single-lung
ventilation and oxidative stress: A different perspective on a
common practice. Curr Opin Anaesthesiol. 30:42–49. 2017.PubMed/NCBI
|
|
26
|
Tsikas D: Assessment of lipid peroxidation
by measuring malondialdehyde (MDA) and relatives in biological
samples: Analytical and biological challenges. Anal Biochem.
524:13–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fan Z, Yao J, Li Y, Hu X, Shao H and Tian
X: Anti-inflammatory and antioxidant effects of curcumin on acute
lung injury in a rodent model of intestinal ischemia reperfusion by
inhibiting the pathway of NF-κB. Int J Clin Exp Pathol.
8:3451–3459. 2015.PubMed/NCBI
|
|
28
|
Shen J and Fu G, Jiang L, Xu J, Li L and
Fu G: Effect of dexmedetomidine pretreatment on lung injury
following intestinal ischemia-reperfusion. Exp Ther Med.
6:1359–1364. 2013. View Article : Google Scholar : PubMed/NCBI
|