Introduction
Cervical cancer is one of the most important
reproductive health problems for adult women worldwide (1) and the second most common cancer among
women worldwide (2). Approximately
500,000 newly diagnosed cases of cervical cancer are identified
each year and at least 200,000 cases succumb to the disease
(3). Cervical cancer incidence rates
are high in developing countries, where more than 80% of the
cervical cancer cases occur in the world (4). It is widely recognized that the leading
cause of cervical cancer is persistent infection with specific
types of the human papillomavirus (5). The complex process from normal tissue
to cervical cancer involves going through mild dysplasia, moderate
dysplasia, severe dysplasia, carcinoma in situ, and
infiltrating carcinoma (6). This
complex progression involves the abnormal expression of numerous
oncogenes and tumor suppressor genes (7). The current primary treatment for
cervical cancer is surgery, radiotherapy and chemotherapy (8). Although improvements have been made,
there remain limitations associated with the current treatment
options (9). Recurrent cervical
cancer and metastasis occur frequently in patients with advanced
cervical cancer (10). It is
therefore important to investigate new and effective therapeutic
targets for the treatment of cervical cancer.
MicroRNAs (miRNAs) are a class of small non-coding,
single-stranded RNAs 19–25 nucleotides in length, found in
eukaryotic cells. miRNAs can regulate post-transcriptional gene
expression through mRNA degradation or translation inhibition of
target mRNAs through the complete or partial binding in the 3′
untranslated regions (3′UTR) of target mRNAs (11,12).
Previous studies have demonstrated that miRNAs are involved in the
development of various types of cancer (13–16).
Abnormal expression of miRNAs is associated with the occurrence and
development of cervical cancer through the regulation of target
gene expression, which include oncogenes and tumor suppressor genes
(17–21). MicroRNA-425-5p (miR-425-5p) was
previously revealed to be upregulated in cervical cancer (22), however the cellular function of
miR-425-5p in cervical cancer remains unknown. The aim of the
current study was to investigate the cellular function of
miR-425-5p and its underlying mechanism in cervical cancer.
Materials and methods
Cell culture
Human cervical cancer cell lines HeLa, SiHa, C-33A
and ME-180, as well as the human cervical epithelium cell line
End1/E6E7 were purchased from American Type Culture Collection
(Manassas, VA, USA). HeLa, SiHa and C-33A cells were grown in high
glucose Dulbeco's modified Eagle medium and ME-180 cells were grown
in RPMI 1640 medium (both Invitrogen; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum
(Invitrogen; Thermo Fisher Scientific, Inc.), 1%
streptomycin-penicillin solution and maintained at 37°C in a 5%
CO2-humidified incubator. Cells were passaged every 2–3
days. End1/E6E7 cells were grown in Keratinocyte serum-free medium
(Gibco; Thermo Fisher Scientific, Inc.) supplemented with 0.1 ng/ml
human recombinant epithelial growth factor, 0.05 mg/ml bovine
pituitary extract (both Santa Cruz Biotechnology, Inc., Dallas, TX,
USA), 1% streptomycin-penicillin solution and maintained at 37°C in
a 5% CO2-humidified incubator.
Dual-luciferase reporter assay
TargetScan bioinformatics software (www.targetscan.org/vert_71) was used to predict
the putative target genes of miR-425–5. Apoptosis-inducing factor
mitochondria-associated 1 (AIFM1) was identified as a potential
target of miR-425-5p. The QuikChange Site-Directed Mutagenesis kit
(Agilent Technologies, Inc., Santa Clara, CA, USA) was used,
according to the manufacturer's protocol, to make a point mutation
in the miR-425-5p binding domain on the 3′UTR of AIFM1. To confirm
direct target binding, the wild-type (WT) 3′UTR AIFM1 or the mutant
(MUT) 3′UTR AIFM1 were cloned into the dual-luciferase reporter
vector pmiR-RB-REPORT™ (Guangzhou RiboBio Co., Ltd., Guangzhou,
China). HeLa cells were co-transfected with 100 ng WT-AIFM1 or 100
ng MUT-AIFM1 and 50 nM miR-425-5p mimic (forward,
5′-AAUGACACGAUCACUCCCGUUGA-3′ and reverse,
5′-AACGGGAGUGAUCGUGUCAUUUU-3′) or 50 nM mimic control (forward,
5′-UUCUCCGAACGUGUCACGUTT-3′ and reverse,
5′-ACGUGACACGUUCGGAGAATT-3′) using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. MiR-425-5p mimic and mimic control were
purchased from GenePharma Co., Ltd. (Shanghai, China). Following
incubation for 48 h, luciferase activity was detected using a
Dual-Luciferase® Reporter assay system (Promega
Corporation, Madison, WI, USA), according to the manufacturer's
protocol. Firefly luciferase activity was normalized to Renilla
luciferase activity.
Cell transfection
HeLa cells were seeded into a 6-well plate at a
density of 1×106 cells/well and cultured at 37°C for 24
h. MiR-425-5p inhibitor and inhibitor control were obtained from
GenePharma Co., Ltd. Cells were transfected with 100 nM miR-425-5p
inhibitor (5′-AGGCGAAGGAUGACAAAGGGAA-3′), 100 nM inhibitor control
(5′-CAGUACUUUUGUGUAGUACAA-3′), 10 µM control-siRNA (cat. no. 36869;
Santa Cruz Biotechnology, Inc.), 10 µM AIFM1-siRNA (cat. no. 26926;
OriGene Technologies, Inc., Rockville, MD, USA), miR-425-5p
inhibitor + control-siRNA, or miR-425-5p inhibitor + AIFM1-siRNA
using Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Following incubation for 48 h, transfection efficiency was
measured.
Cell proliferation assay
Cell viability was measured by MTT assay. Following
a 48-h cell transfection, HeLa cells were seeded into 96-well
plates at a density of 1×104 cells/per well and cultured
for 24 h. Following incubation, 20 ml MTT solution (0.5 mg/ml;
Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added to each
well and further incubated at 37°C for 4 h. Cell viability was
determined by measuring the absorbance at a wavelength of 570 nm
using a FLUOstar® Omega Microplate Reader (BMG Labtech
GmbH, Ortenberg, Germany).
Flow cytometric analysis of
apoptosis
Cell apoptosis was analyzed using the Annexin
V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis
detection kit (cat. no. 70-AP101-100; MultiSciences, Hangzhou,
Zhejiang, China). Following a 48-h cell transfection, HeLa cells
were harvested with 0.25% trypsin, washed with PBS and subsequently
stained with 5 µl Annexin V-FITC and 5 µl PI for 30 min at room
temperature without light. Early and late apoptotic cells were
subsequently analyzed using a flow cytometer (BD Biosciences,
Franklin Lakes, NJ, USA), and data were analyzed using WinMDI
software (version 2.5; Purdue University Cytometry Laboratories;
www.cyto.purdue.edu/flowcyt/software/Catalog.htm).
Western blot analysis
Total protein was extracted from cells using
radioimmunoprecipitation assay buffer (Beyotime Institute of
Biotechnology, Haimen, China), according to the manufacturer's
protocol. Total protein was quantified using a bicinchoninic acid
assay kit (Pierce; Thermo Fisher Scientific, Inc.) and 30 mg
protein/lane was separated via SDS-PAGE on a 12% gel. The separated
proteins were transferred onto polyvinylidene fluoride membranes
(EMD Millipore, Billerica, MA, USA) and blocked for 1 h at room
temperature with 5% skimmed milk. The membranes were incubated with
primary antibodies against AIFM1 (1:1,000; cat. no. BA3715-1;
Boster Biological Technology, Pleasanton, CA, USA), DNA damage
regulated autophagy modulator 1 (DRAM; 1:1,000; cat. no. 208160;
Abcam, Cambridge, MA, USA), cytochrome c (1:1,000; cat. no. 11940),
caspase-3 (1:1,000; cat. no. 9665), caspase-9 (1:1,000; cat. no.
9502) and β-actin (1:1,000; cat. no. 4970; all Cell Signaling
Technology Inc., Danvers, MA, USA) overnight at 4°C. Following
primary incubation, membranes were incubated with horseradish
peroxidase-conjugated secondary antibody, anti-rabbit IgG (1:2,000;
cat. no. 7074; Cell Signaling Technology, Inc.) for 2 h at room
temperature. Protein bands were visualized using the enhanced
chemiluminescence Western Blotting Detection kit (EMD
Millipore).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cells using
TRIzol® reagent (Invitrogen, Thermo Fisher Scientific,
Inc.), according to the manufacturer's protocol. Total RNA was
reverse transcribed into cDNA using the TaqMan™ MicroRNA
Reverse Transcription kit (Applied Biosystems; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. qPCR
was subsequently performed using the SYBR® Premix Ex
Taq™ II kit (Takara Bio, Inc., Otsu, Japan). The
following primer pairs were used for the qPCR: GAPDH forward,
5′CTTTGGTATCGTGGAAGGACTC3′ and reverse, 5′GTAGAGGCAGGGATGATGTTCT3′;
and U6 forward, 5′GCTTCGGCAGCACATATACTAAAAT3′ and reverse,
5′CGCTTCACGAATTTGCGTGTCAT3′; miR-425-5p forward,
5′TGCGGAATGACACGATCACTCCCG3′ and reverse, 5′CCAGTGCAGGGTCCGAGGT3′;
AIFM1 forward, 5′TTGAGAATGGTGGTGTGGCT3′ and reverse,
5′AGACTTCTTGGAGTACCTCCTGT3′; caspase-3 forward,
5′AGAACTGGACTGTGGCATTG3′ and reverse, 5′CACAAAGCGACTGGATGAAC3′;
caspase-9 forward, 5′TGTTTCCGAGCGAGGGATTT3′ and reverse,
5′CGCAGGAAGGTTTTGGGGTA3′; DRAM forward, 5′AGACTCCATCTTTTCACCCAAA3′
and reverse, 5′GCTCTTCACCTTTCAAGCCTAA3′; cytochrome c forward,
5′TGCCACACTGTTGAAGCCGGT3′ and reverse, 5′GATCTGCACGCTCGTTTGCCT3′.
The following thermocycling conditions were used for the qPCR:
Initial denaturation at 95°C for 10 min; 35 cycles of 95°C for 15
sec and 55°C for 40 sec. The relative mRNA expression levels were
quantified using the 2−ΔΔCq method and normalized to the
internal reference gene, U6 or GAPDH (23).
Statistical analysis
Data are presented as the mean ± standard deviation
of at least three independent experiments. All statistical analyses
were performed using SPSS software (version 17.0; SPSS, Inc.,
Chicago. IL, USA). Student's t-test was performed for comparison
analysis between two groups. One-way analysis of variance followed
by Tukey's post hoc test was performed for analyze differences
among multiple groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
miR-425-5p expression is upregulated
in cervical cancer
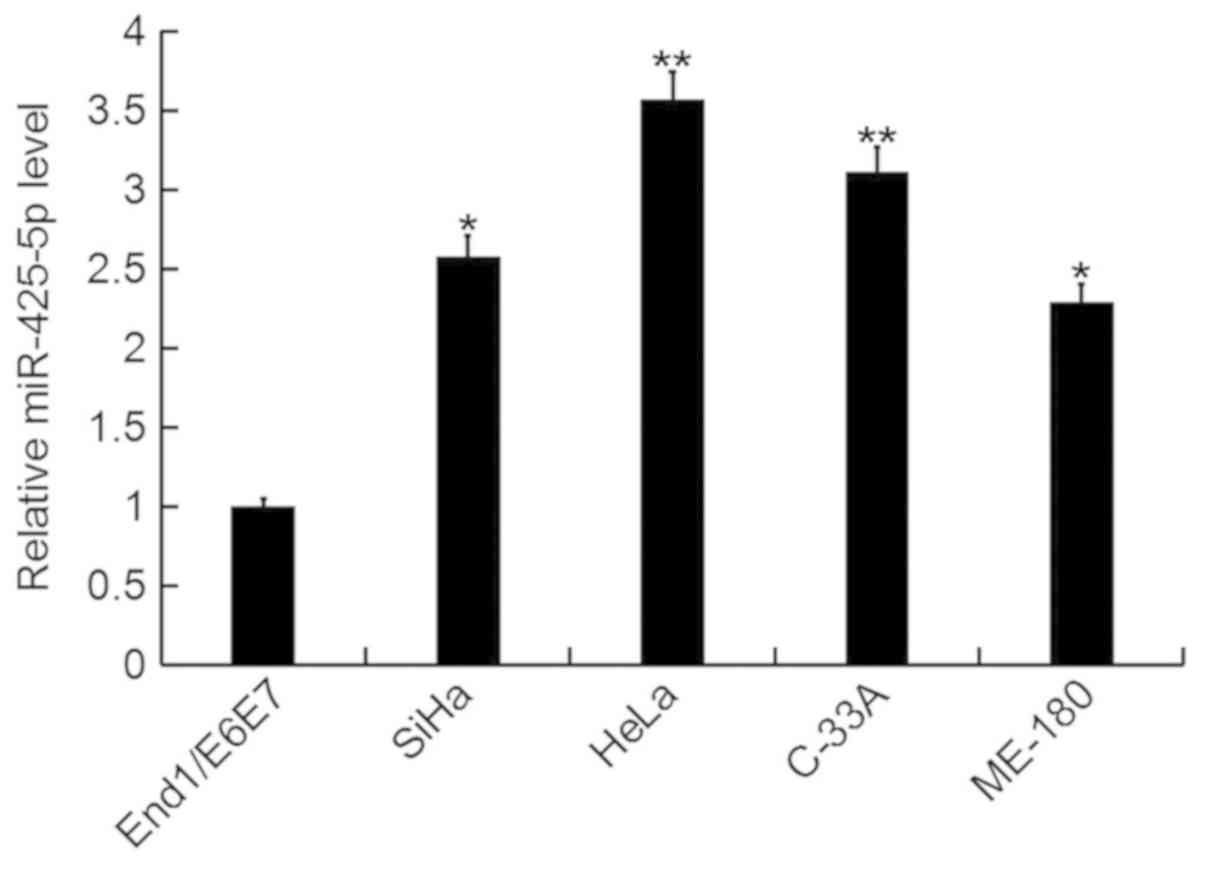
In the current study, the expression level of
miR-425-5p was determined by RT-qPCR in several human cervical
cancer cell lines (HeLa, SiHa, C-33A and ME-180), as well as the
human normal cervical epithelium cell line End1/E6E7. The
miR-425-5p expression level was significantly increased in human
cervical cancer cell lines compared with the normal cervical
epithelium cell line (Fig. 1). As
the highest level of miR-425-5p expression was detected in HeLa
cells, these were selected for all subsequent experiments.
AIFM1 is a target gene of
miR-425-5p
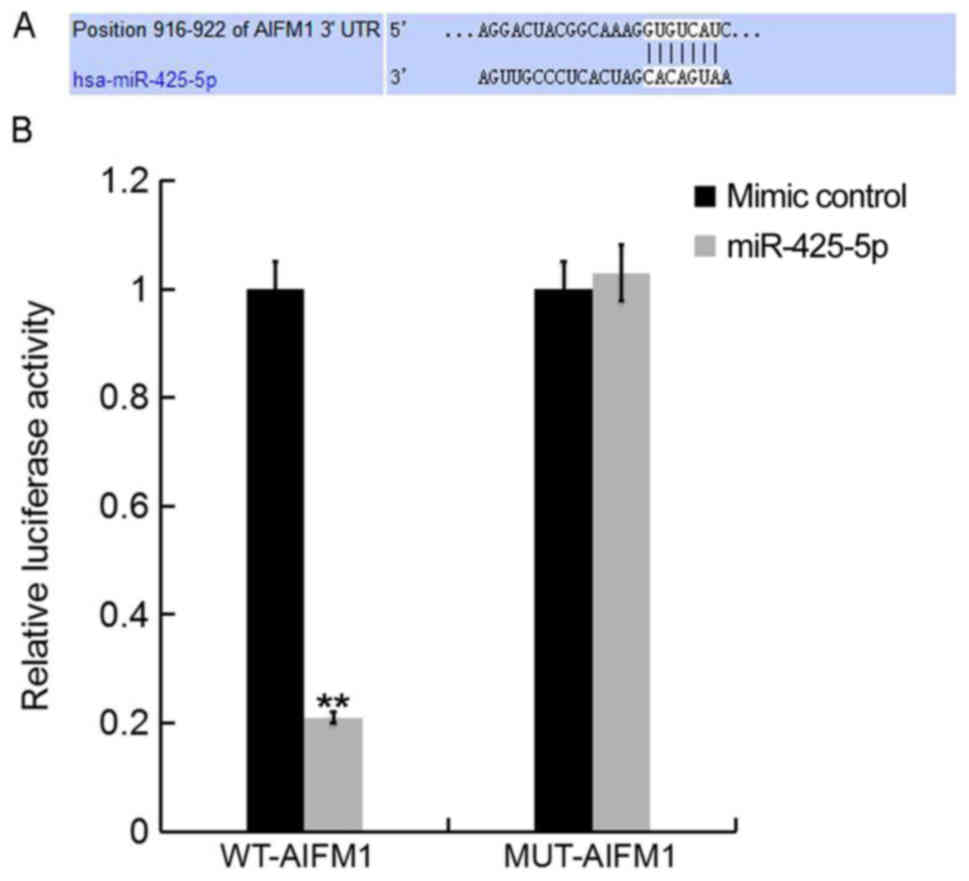
To further investigate the role of miR-425-5p in
human cervical cancer, potential targets of miR-425-5p were
examined. TargetScan bioinformatics software was used to identify
AIFM1 as a putative target gene of miR-425-5p (Fig. 2A). To confirm whether miR-425-5p
directly regulates AIFM1 expression via interaction with the
predicted binding sites, luciferase reporter assays were performed.
Following co-transfection with miR-425-5p mimic, the luciferase
reporter activity of WT-AIFM1 was significantly decreased compared
with the luciferase reporter activity of MUT-AIFM1 (Fig. 2B). The results suggest that AIFM1 is
a direct target of miR-425-5p.
Downregulation of miR-425-5p enhances
AIFM1 expression in HeLa cells
To investigate the effect of miR-425-5p on cervical
cancer, HeLa cells were transfected with miR-425-5p inhibitor,
inhibitor control, AIFM1-siRNA, control-siRNA, miR-425-5p inhibitor
+ AIFM1-siRNA or miR-425-5p inhibitor + control-siRNA,
respectively. Following incubation for 48 h, transfection
efficiency was measured. The current study demonstrated that the
miR-425-5p inhibitor significantly decreased miR-425-5p expression
in HeLa cells (Fig. 3A). In
addition, the mRNA and protein expression levels of AIFM1 were
decreased in HeLa cells following transfection with AIFM1-siRNA
(Fig. 3B and C). Furthermore, the
mRNA and protein expression levels of AIFM1 were increased
following transfection with miR-425-5p inhibitor compared with the
control group. However, AIFM1-siRNA reversed the effects observed
with miR-425-5p inhibitor (Fig. 3D and
E).
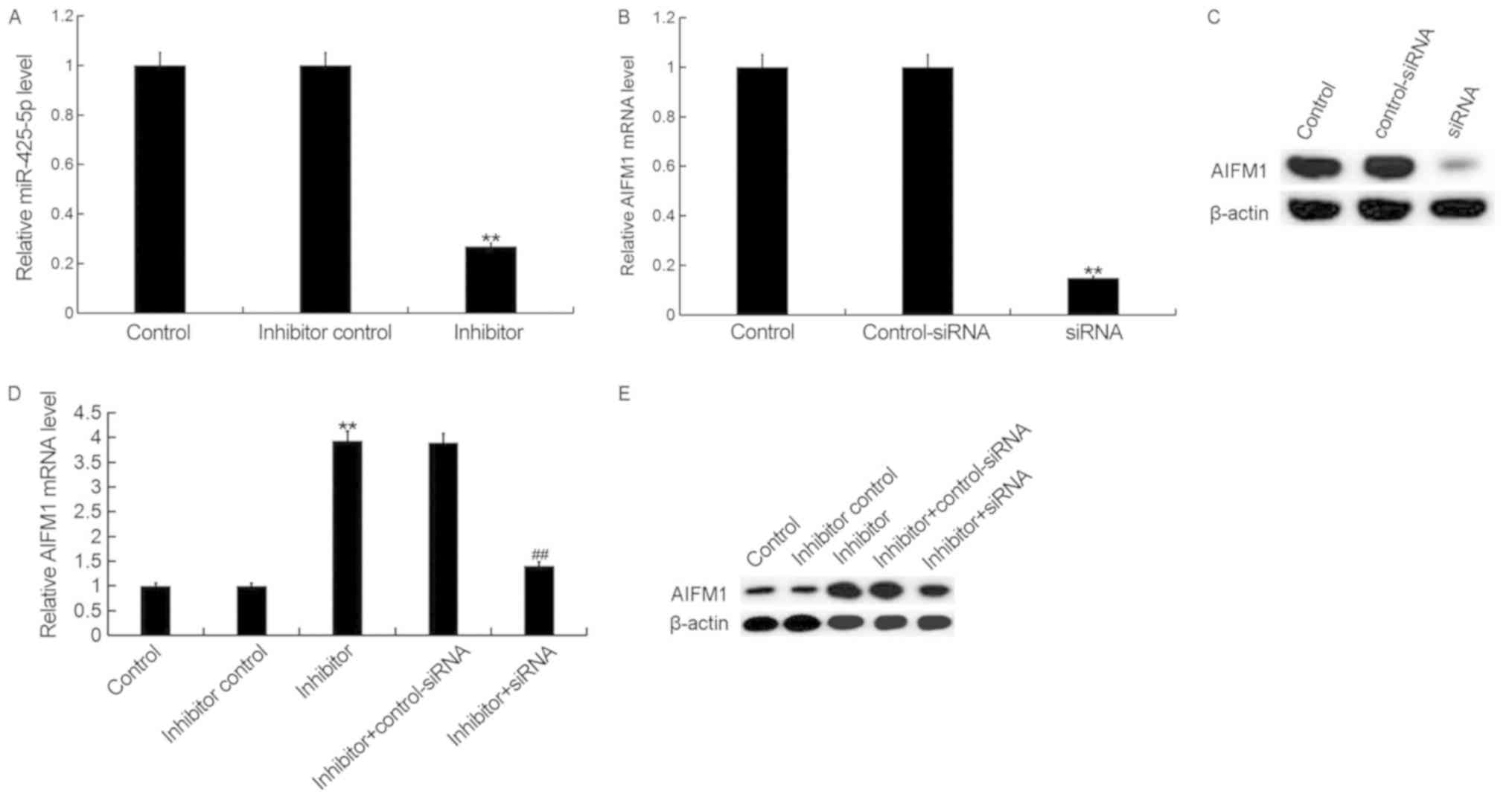 | Figure 3.miR-425-5p inhibitor enhances AIFM1
expression in HeLa cells. (A) The relative expression level of
miR-425-5p was determined by reverse transcription-quantitative
polymerase chain reaction in HeLa cells following transfection with
miR-425-5p inhibitor and inhibitor control. The (B) mRNA and (C)
protein expression level of AIFM1 was analyzed following
transfection with AIFM1-siRNA or control-siRNA. The (D) mRNA and
(E) protein expression level of AIFM1 was analyzed following
transfection with miR-425-5p inhibitor, inhibitor control,
miR-425-5p inhibitor + AIFM1-siRNA or miR-425-5p inhibitor +
control-siRNA. Data are presented as the mean ± standard deviation.
**P<0.01 vs. control group; ##P<0.01 vs. inhibitor
group. AIFM1, apoptosis-inducing factor mitochondria-associated 1;
miR, microRNA; siRNA, small interfering RNA; control, untransfected
HeLa cells; inhibitor control, HeLa cells transfected with
inhibitor control; inhibitor, HeLa cells transfected with
miR-425-5p inhibitor; control-siRNA, HeLa cells transfected with
control siRNA; siRNA, HeLa cells transfected with AIFM1-siRNA;
inhibitor + control-siRNA, HeLa cells co-transfected with
miR-425-5p inhibitor and control siRNA; inhibitor + siRNA, HeLa
cells co-transfected with miR-425-5p inhibitor and AIFM1-siRNA. |
Downregulation of miR-425-5p inhibits
HeLa cell viability
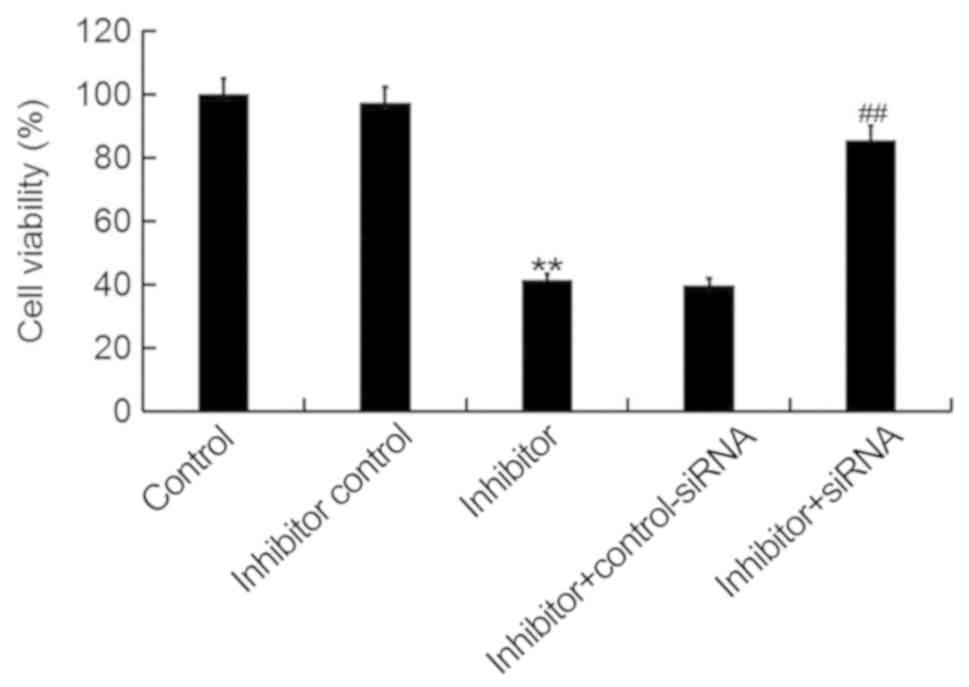
To investigate the cellular function of miR-425-5p
in cervical cancer, the MTT assay was used to examine the viability
of HeLa cells following transfection with miR-425-5p inhibitor,
inhibitor control, miR-425-5p inhibitor + AIFM1-siRNA or miR-425-5p
inhibitor + control-siRNA. The current study demonstrated that the
miR-425-5p inhibitor significantly decreased HeLa cell viability
compared with the control group. However, AIFM1-siRNA significantly
reversed the effect of miR-425-5p inhibitor on HeLa cell viability
(Fig. 4).
Downregulation of miR-425-5p induces
HeLa cell apoptosis
To further investigate the cellular function of
miR-425-5p in cervical cancer, flow cytometry was used to examine
cell apoptosis in HeLa cells following transfection with miR-425-5p
inhibitor, inhibitor control, miR-425-5p inhibitor + AIFM1-siRNA or
miR-425-5p inhibitor + control-siRNA. The current study
demonstrated that the miR-425-5p inhibitor significantly increased
HeLa cell apoptosis compared with the control group. However,
AIFM1-siRNA significantly reversed the effect of miR-425-5p
inhibitor on HeLa cell apoptosis (Fig.
5A and B).
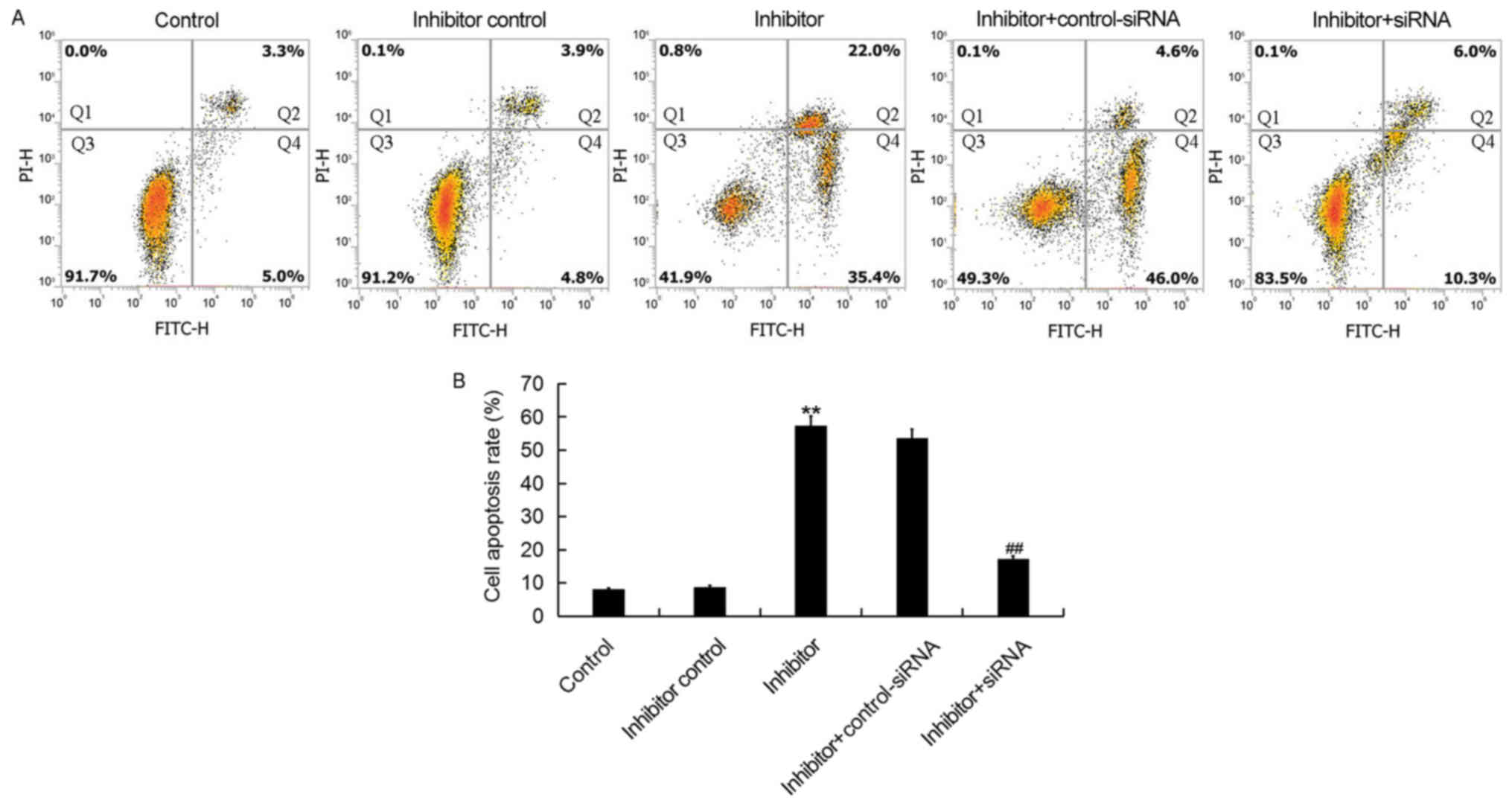 | Figure 5.miR-425-5p inhibitor induces HeLa cell
apoptosis. (A) Early apoptosis (Q2) and late apoptosis (Q4) was
detected by flow cytometry in HeLa cells following transfection
with miR-425-5p inhibitor, inhibitor control, miR-425-5p inhibitor
+ AIFM1-siRNA or miR-425-5p inhibitor + control-siRNA. (B) The
effect of miR-425-5p knockdown on HeLa cell apoptosis was examined.
Data are presented as the mean ± standard deviation. **P<0.01
vs. control group; ##P<0.01 vs. inhibitor group.
siRNA, small interfering RNA; FITC, fluorescein isothiocyanate;
control, untransfected HeLa cells; inhibitor control, HeLa cells
transfected with inhibitor control; inhibitor, HeLa cells
transfected with miR-425-5p inhibitor; inhibitor + control-siRNA,
HeLa cells co-transfected with miR-425-5p inhibitor and control
siRNA; inhibitor + siRNA, HeLa cells co-transfected with miR-425-5p
inhibitor and AIFM1-siRNA; PI, propidium iodide. |
Downregulation of miR-425-5p
upregulates the expression of cytochrome c, caspase-3, caspase-9
and DRAM in HeLa cells
To investigate the regulatory effect of miR-425-5p
in cervical cancer, the expression of four pro-apoptotic genes,
cytochrome c, caspase-3, caspase-9 and DRAM were examined in HeLa
cells following transfection with miR-425-5p inhibitor, inhibitor
control, miR-425-5p inhibitor + AIFM1-siRNA or miR-425-5p inhibitor
+ control-siRNA (Fig. 6). The
current study demonstrated that the miR-425-5p inhibitor
significantly upregulated both the protein and mRNA expression
levels of all four pro-apoptotic genes analyzed, compared with the
control group. However, the enhanced expression of these
pro-apoptotic genes was significantly reversed by AIFM1
knockdown.
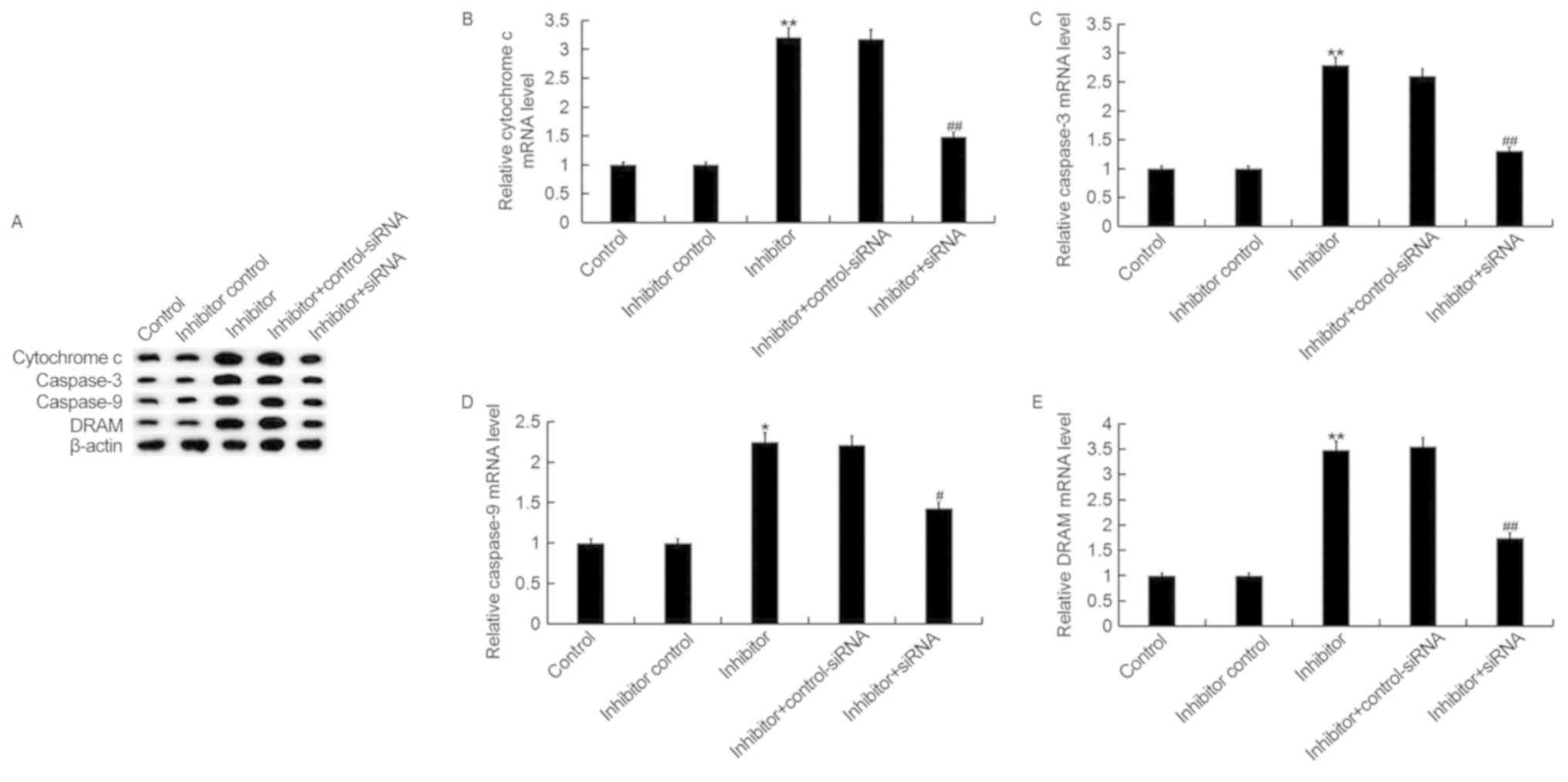 | Figure 6.miR-425-5p inhibitor upregulates
pro-apoptotic gene expression in HeLa cells. (A) The protein
expression levels of cytochrome c, caspase-3, caspase-9 and DRAM
were determined by western blot analysis in HeLa cells following
transfection with miR-425-5p inhibitor, inhibitor control,
miR-425-5p inhibitor + AIFM1-siRNA or miR-425-5p inhibitor +
control-siRNA. The mRNA expression levels of (B) cytochrome c, (C)
caspase-3, (D) caspase-9 and (E) DRAM were determined by reverse
transcription-quantitative polymerase chain reaction in HeLa cells
following transfection with miR-425-5p inhibitor, inhibitor
control, miR-425-5p inhibitor + AIFM1-siRNA or miR-425-5p inhibitor
+ control-siRNA. Data are presented as the mean ± standard
deviation. *P<0.05 and **P<0.01 vs. control group;
#P<0.05 and ##P<0.01 vs. inhibitor
group. DRAM, DNA damage regulated autophagy modulator 1; siRNA,
small interfering RNA; control, untransfected HeLa cells; inhibitor
control, HeLa cells transfected with inhibitor control; inhibitor,
HeLa cells transfected with miR-425-5p inhibitor; inhibitor +
control-siRNA, HeLa cells co-transfected with miR-425-5p inhibitor
and control siRNA; inhibitor + siRNA, HeLa cells co-transfected
with miR-425-5p inhibitor and AIFM1-siRNA. |
Discussion
Increasing evidence suggests that miRNAs are
involved in the development of various types of cancer (13–16).
Previous reports have indicated that miRNAs serve a regulatory role
in cell proliferation, differentiation, apoptosis, migration and
metabolism (12,24,25).
Studies have revealed that enhanced expression levels of miR-425-5p
exist in different types of cancer, which suggest that miR-425-5p
may have multiple functions in the development of cancer (22,26).
Studies previously demonstrated that miR-425-5p can promote
invasion and metastasis in hepatocellular carcinoma, colorectal
cancer and gastric cancer (27–29).
The aim of the current study was to investigate the
cellular function of miR-425-5p and its underlying mechanism in
cervical cancer. In the current study, the expression level of
miR-425-5p was determined by RT-qPCR in several human cervical
cancer cell lines including HeLa, SiHa, C-33A and ME-180, as well
as the human normal cervical epithelium cell line End1/E6E7.
miR-425-5p expression was significantly increased in human cervical
cancer cell lines compared with the normal cervical epithelium cell
line. In addition, HeLa cells expressed the highest level of
miR-425-5p and were selected for all subsequent experiments.
To further investigate the role of miR-425-5p in
human cervical cancer, potential targets of miR-425-5p were
examined in HeLa cells using the TargetScan software. Bioinformatic
analysis identified AIFM1 as a putative target gene of miR-425-5p
and this was verified by dual-luciferase reporter assay. AIFM1,
which is located in the mitochondrion intermembrane space, serves a
role in the regulation of cell apoptosis (30–33).
The effect of miR-425-5p on cervical cancer cell
viability and apoptosis was examined in HeLa cells. Downregulation
of miR-425-5p significantly decreased cell viability and induced
cell apoptosis, however, the inhibitory effect exerted by the
miR-425-5p inhibitor was significantly reversed by AIFM1 knockdown.
These results suggest that miR-425-5p inhibitor may function as a
tumor suppressor in cervical cancer by enhancing the expression of
AIFM1. AIFM1 is a phylogenetically conserved mitochondrial
flavoprotein with NADH oxidation and potent apoptosis-inducing
activity (30–32). AIFM1 induces mitochondria to release
the apoptogenic proteins cytochrome c and caspase-9, thereby
initiating apoptosis (31,33). A previous study demonstrated that
overexpression of AIFM1 induced apoptosis by promoting the
transcription of caspase-3 and DRAM in hepatoma cells (33).
To investigate the underlying molecular mechanism of
miR-425-5p in cervical cancer, the expression of pro-apoptotic
genes including cytochrome c, caspase-3, caspase-9 and DRAM was
examined in HeLa cells. The current study demonstrated that both
the protein and mRNA expression levels of cytochrome c, caspase-3,
caspase-9 and DRAM were significantly upregulated following
transfection with miR-425-5p inhibitor. However, the enhanced
expression of all four pro-apoptotic genes was significantly
reversed by AIFM1 knockdown.
The present study investigated the cellular function
of miR-425-5p and its underlying mechanism in cervical cancer,
however in vivo studies and clinical trial data are required
to validate the preliminary in vitro results obtained in the
current study. Furthermore, the cellular function of AIFM1 alone in
cervical cancer needs to be further investigated.
In conclusion, the current study demonstrated that
miR-425-5p was upregulated in cervical cancer, and this may
contribute to cervical cancer development by inhibiting AIFM1
expression. Furthermore, inhibition of miR-425-5p decreased
cervical cancer cell viability and induced cell apoptosis.
Therefore, the miR-425-5p/AIFM1 axis may serve a role in cervical
cancer progression and this may be a promising therapeutic target
for the treatment of patients.
Acknowledgements
Not applicable.
Funding
The present study was supported by a grant from the
Shijiazhuang Science and Technology Bureau of China 2013 Science
and Technology Project (grant no. 131462543).
Availability of data and materials
All datasets used and/or generated during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
YZ designed the study. YZ, YY, and RL analyzed the
data. YM, GT and QC analyzed the data and prepared the manuscript.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Forouzanfar MH, Foreman KJ, Delossantos
AM, Lozano R, Lopez AD, Murray CJ and Naghavi M: Breast and
cervical cancer in 187 countries between 1980 and 2010: A
systematic analysis. Lancet. 378:1461–1484. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
De Sanjose S, Quint WG, Alemany L, Geraets
DT, Klaustermeier JE, Lloveras B, Tous S, Felix A, Bravo LE, Shin
HR, et al: Human papillomavirus genotype attribution in invasive
cervical cancer: A retrospective cross-sectional worldwide study.
Lancet Oncol. 11:1048–1056. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li Z, Wang H, Wang Z and Cai H: MiR-195
inhibits the proliferation of human cervical cancer cells by
directly targeting cyclin D1. Tumor Biol. 37:6457–6463. 2016.
View Article : Google Scholar
|
|
4
|
zur Hausen H: Papillomaviruses and cancer:
From basic studies to clinical application. Nat Rev Cancer.
2:342–350. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Khan MJ, Castle PE, Lorincz AT, Wacholder
S, Sherman M, Scott DR, Rush BB, Glass AG and Schiffman M: The
elevated 10-year risk of cervical precancer and cancer in women
with human papillomavirus (HPV) type 16 or 18 and the possible
utility of type-specific HPV testing in clinical practice. J Natl
Cancer Inst. 97:1072–1079. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mariuzzi G, Santinelli A, Valli M, Sisti
S, Montironi R, Mariuzzi L, Alberti R and Pisani E: Cytometric
evidence that cervical intraepithelial neoplasia I and II are
dysplasias rather than true neoplasias. An image analysis study of
factors involved in the progression of cervical lesions. Anal Quant
Cytol Histol. 14:137–147. 1992.PubMed/NCBI
|
|
7
|
Zou DL, Zhou Q, Wang D, Guan L, Yuan L and
Li S: The downregulation of microRNA-10b and its role in cervical
cancer. Oncol Res. 24:99–108. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dueñas-Gonzalez A, Cetina L, Mariscal I
and de la Garza J: Modern management of locally advanced cervical
carcinoma. Cancer Treat Rev. 29:389–399. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ebina Y, Mikami M, Nagase S, Tabata T,
Kaneuchi M, Tashiro H, Mandai M, Enomoto T, Kobayashi Y, Katabuchi
H, et al: Japan Society of Gynecologic Oncology guidelines 2017 for
the treatment of uterinecervical cancer. Int J Clin Oncol. Oct
5–2018.(Epub ahead of print). PubMed/NCBI
|
|
10
|
Glick SB, Clarke AR, Blanchard A and
Whitaker AK: Cervical cancer screening, diagnosis and treatment
interventions for racial and ethnic minorities: A systematic
review. J Gen Intern Med. 27:1016–1032. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee RC, Feinbaum RL and Ambros V: The
C. elegans heterochronic gene lin-4 encodes small RNAs with
antisense complementarity to lin-14. Cell. 75:843–854. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu L, Ai J, Long H, Liu W, Wang X, Zuo Y,
Li Y, Wu Q and Deng Y: Intergrative microRNA and gene profiling
data analysis reveals novel biomarkers and mechanisms for lung
cancer. Oncotarget. 7:8441–8454. 2016.PubMed/NCBI
|
|
14
|
Tsai MM, Wang CS, Tsai CY, Huang HW, Chi
HC, Lin YH, Lu PH and Lin KH: Potential diagnostic, prognostic and
therapeutic targets of microRNAs in human gastric cancer. Int J Mol
Sci. 17(pii): E9452016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhao H, Li M, Li L, Yang X, Lan G and
Zhang Y: MiR-133b is down-regulated in human osteosarcoma and
inhibits osteosarcoma cells proliferation, migration and invasion,
and promotes apoptosis. PLoS One. 8:e835712013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kaukoniemi KM, Rauhala HE, Scaravilli M,
Latonen L, Annala M, Vessella RL, Nykter M, Tammela TL and
Visakorpi T: Epigenetically altered miR-193b targets cyclin D1 in
prostate cancer. Cancer Med. 4:1417–1425. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang X, Tang S, Le SY, Lu R, Rader JS,
Meyers C and Zheng ZM: Aberrant expression of oncogenic and
tumor-suppressive microRNAs in cervical cancer is required for
cancer cell growth. PLoS One. 3:e25572008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wilting SM, van Boerdonk RA, Henken FE,
Meijer CJ, Diosdado B, Meijer GA, le Sage C, Agami R, Snijders PJ
and Steenbergen RD: Methylation-mediated silencing and tumor
suppressive function of hsa-miR-24 in cervical cancer. Mol Cancer.
9:1672010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Long MJ, Wu FX, Li P, Liu M, Li X and Tang
H: MicroRNA-10a targets CHL1 and promotes cell growth, migration
and invasion in human cervical cancer cells. Cancer Lett.
324:186–196. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kogo R, How C, Chaudary N, Bruce J, Shi W,
Hill RP, Zahedi P, Yip KW and Liu FF: The microRNA-218~Survivin
axis regulates migration, invation, and lymph node metastasis in
cervical cancer. Oncotarget. 6:1090–1100. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Deng B, Zhang Y, Zhang S, Wen F, Miao Y
and Guo K: microRNA-142-3p inhibits cell proliferation and invasion
of cervical cancer cells by targeting FZD7. Tumor Biol.
36:8065–8073. 2015. View Article : Google Scholar
|
|
22
|
Sun L, Jiang R, Li J, Wang B, Ma C, Lv Y
and Mu N: MicoRNA-425-5p is a potential prognostic biomarker for
cervical cancer. Ann Clin Biochem. 54:127–133. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ebert MS and Sharp PA: Roles for microRNAs
in conferring robustness to biological processes. Cell.
149:515–524. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rogers K and Chen X: Biogenesis, turnover,
and mode of action of plant microRNAs. Plant Cell. 25:2383–2399.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Quan J, Li Y, Pan X, Lai Y, He T, Lin C,
Zhou L, Zhao L, Sun S, Ding Y, et al: Oncogenic miR-425-5p is
associated with cellular migration, proliferation and apoptosis in
renal cell carcinoma. Oncol Lett. 16:2175–2184. 2018.PubMed/NCBI
|
|
27
|
Fang F, Song T, Zhang T, Cui Y, Zhang G
and Xiong Q: MiR-425-5p promotes invasion and metastasis of
hepatocellular carcinoma cells through SCAI-mediated dysregulation
of multiple signaling pathways. Oncotarget. 8:31745–31757.
2017.PubMed/NCBI
|
|
28
|
Cristóbal I, Madoz-Gúrpide J, Rojo F and
García-Foncillas J: Potential therapeutic value of miR-425-5p in
metastatic colorectal cancer. J Cell Mol Med. 20:2213–2214. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang Z, Wen M, Guo J, Shi J, Wang Z, Tan
B, Zhang G, Zheng X and Zhang A: Clinical value of miR-425-5p
detection and its association with cell proliferation and apoptosis
of gastric cancer. Pathol Res Pract. 213:929–937. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Susin SA, Lorenzo HK, Zamzami N, Marzo I,
Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler
M, et al: Molecular characterization of mitochondrial
apoptosis-inducing factor. Nature. 397:441–446. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Joza N, Susin SA, Daugas E, Stanford WL,
Cho SK, Li CY, Sasaki T, Elia AJ, Cheng HY, Ravagnan L, et al:
Essential role of the mitochondrial apoptosis-inducing factor in
programmed cell death. Nature. 410:549–554. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cregan SP, Dawson VL and Slack RS: Role of
AIF in caspase-dependent and caspase-independent cell death.
Oncogene. 23:2785–2796. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu D, Liu M, Wang W, Pang L, Wang Z, Yuan
C and Liu K: Overexpression of apoptosis-inducing factor
mitochondrion-associated 1 (AIFM1) induces apoptosis by promoting
the transcription of caspase-3 and DRAM in hepatoma cells. Biochem
Biophys Res Commun. 498:453–457. 2018. View Article : Google Scholar : PubMed/NCBI
|