Introduction
Pancreatic cancer (PC) is one of the most malignant
solid tumors in humans that is characterized by its late diagnosis,
rapid progression, early metastasis and chemoresistance (1,2).
Although an increasing number of therapies, including surgical
resection, chemotherapy and radiotherapy, have been used in recent
years, the overall 5-year survival rate is still <5% (2,3). Recent
studies have indicated that microRNAs (miRs) have a critical role
in the progression of PC (4,5). Therefore, a detailed understanding of
the miR-based molecular mechanisms of PC malignancy may provide
useful insights into the identification of biomarkers and
development of novel therapeutic strategies for PC.
miRs are small, non-coding RNA molecules, which
post-transcriptionally regulate gene expression by directly binding
to the 3′-untranslated region (UTR) of their target genes and
induce target mRNA degradation or suppress target mRNA translation
(6). An individual miR typically has
multiple target genes with partially complementary mRNA sequences,
whereas a single gene can be targeted by several miRs. Previous
studies have demonstrated that miR-634 acts as tumor suppressor in
glioma, gastric carcinoma, hepatocellular carcinoma, nasopharyngeal
carcinoma, ovarian cancer and cervical cancer (7–12).
However, to the best of our knowledge, the function and mechanism
of miR-634 in PC progression has not been fully elucidated.
Therefore, elucidating the function and mechanism of miR-634 in PC
is important.
The aim of the present study was to illustrate the
expression level of miR-634 in PC tissues and cell lines, its
association with PC progression and the underlying molecular
mechanisms in order to identify whether miR-634 serves as a tumor
suppressor in PC.
Materials and methods
Cell culture
Human pancreatic cancer lines (Capan-2, PANC-1,
SW1990 and COLO357) were purchased from the Cell Resource Center,
Chinese Academy of Science Committee (Shanghai, China). The human
immortal ductal cell line HPDE was obtained from American Type
Culture Collection (Manassas, VA, USA). Cancer cells were cultured
in Dulbecco's modified Eagle's medium (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.). HPDE
cells were maintained in keratinocyte serum-free medium (Gibco;
Thermo Fisher Scientific, Inc.) supplemented with bovine pituitary
extract and epidermal growth factor (Gibco; Thermo Fisher
Scientific, Inc.). All cells were maintained in a humidified
incubator at 37°C with 5% CO2.
Human tissue samples
A total of 30 paired PC and normal adjacent tissues
were collected from patients with PC (19 males and 11 females; age
range, 41–69 years old) who had undergone pancreaticoduodenectomy
at LanLing County Hospital (Linyi, China) between June 2015 and
December 2016. A total of 28 patients had PDAC and two patients had
adenosquamous carcinoma of the pancreas. According to the criteria
of the American Joint Commission on Cancer (13), these patients were divided into three
stages (stage I, n=6; stage II, n=16; stage III, n=8). Furthermore,
20 patients had lymph node metastasis and 10 patients had no lymph
node metastasis (no other organ metastasis was indicated). The
patients had not received chemotherapy or radiation therapy prior
to surgery. The samples were frozen and stored at −80°C until total
RNA extraction was performed. The Research Ethics Committee of
LanLing County Hospital (Linyi, China) approved the present study.
All patients provided their signed consent to the research.
Cell transfection
miR-634 mimic
(5-AACCAGCACCCCAACUUUGGACGGTATTCGCACTGGATACGACGAACTTT-3),
miR-negative control (NC; 5-ACUACUGAGUGACAGUAGA-3), miR-634
inhibitor (5-CACUACUUUUGUGUCCCACUU-3) and antiNC
(5-CAGUACUUUUGUGUAGUACAA-3) were purchased from Guangzhou RiboBio
Co., Ltd. (Guangzhou, China). The open reading frame of heat
shock-related 70 kDa protein 2 (HSPA2) was inserted into pcDNA3.1
vector (Invitrogen; Thermo Fisher Scientific, Inc.) to generate the
pcDNA3.1/HSPA2 overexpression vector. A total of 5×105
cells were transfected with 2.5 µg NC, miR-634 mimic, antiNC,
miR-634 inhibitor, empty vector or HSPA2 overexpression vector
using Lipofectamine 2000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol.
Overexpression efficiency was analyzed via western blot analysis.
After 48 h of transfection, the cells were harvested and used for
further experiments.
Colony formation assay
PANC-1 cells transfected with miR-634 mimic, miR-634
inhibitor or miR-634 mimic and HSPA2 plasmid were seeded at 400
cells/well in 6-well plates at 37°C. After 10 days, the colonies
were fixed with 4% paraformaldehyde at room temperature for 10 min
and stained with 0.5% crystal violet at room temperature for 10
min. The number of colonies (>50 cells) was counted under a
light microscope (magnification, ×100).
Cell Counting Kit-8 (CCK) assay
Cell viability was analyzed using CCK-8 (Beyotime
Institute of Biotechnology, Haimen, China). Briefly, PANC-1 cells
(3,000 cells/well) transfected with miR-634 mimic, miR-634
inhibitor or miR-634 mimic and HSPA2 plasmid in 200 µl DMEM were
plated into 96-well plates. After 0, 24, 48 and 72 h of seeding, 10
µl of CCK-8 solution was added into each well. Following 2 h of
incubation, the optical density values at 450 nm of each well were
measured using a microplate reader.
Transwell assay
Cell metastasis was determined using transwell
chambers (Corning, Inc., Corning, NY, USA). For the invasion assay,
the upper sides of the filters were coated with 50 µl Matrigel (BD
Biosciences, San Jose, CA, USA). For the invasion and migration
assays, 5×104 cells in 200 µl of serum-free DMEM were
seeded in the upper chamber. The lower chamber was filled with DMEM
supplemented with 5% FBS. Following incubation at 37°C in an
atmosphere containing 5% CO2 for 24 h, cells on the
lower filter were fixed with 10% methanol at room temperature for
20 min, stained with 0.5% crystal violet at room temperature for 10
min and counted under a light microscope (magnification, ×100).
Flow cytometry
Cell apoptosis was assessed using flow cytometry
with staining of the cells using the Annexin V/propidium iodide
(PI) kit (KeyGEN BioTHCH, Nanjing, Jiangsu, China) according to the
manufacturer's protocols. The samples were analyzed using a
fluorescence-activated cell sorter system (Cytomics FC 500 MPL;
Beckman Coulter, Inc., Brea, CA, USA) to evaluate the apoptotic
levels according to the manufacturer's protocol. Data were analyzed
using ModFit LT 3.0 (Verity Software House, Inc., Topsham, ME,
USA).
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA from cells was extracted using TRIzol
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) and then
reverse-transcribed using a reverse transcription kit (Takara
Biotechnology Co., Ltd., Dalian, China) following the
manufacturer's protocol. qPCR was conducted with All-in-One miRNA
qPCR Detection kit (AOMD-Q020, GeneCopoeia, Inc., Rockville, MD,
USA) on a CFX96 Real-Time PCR Detection System supplied with
analytical software (Bio-Rad Laboratories, Inc., Hercules, CA,
USA). PCR conditions were as follows: 40 cycles of 95°C for 15 sec,
60°C for 15 sec and 72°C for 45 sec. To determine HSPA2 mRNA
expression levels, GAPDH served as an internal control. To examine
the expression levels of miR-634, U6 acted as the internal control.
The relative expression of miR-634 and HSPA2 was calculated
utilizing the comparative 2ΔΔCq method (14). The primers used for amplification
were as follows: U6, forward 5′-CTCGCTTCGGCAGCACA-3′ and reverse
5′-AACGCTTCACGAATTTGCGT-3′; GAPDH, forward
5′-AGAAGGCTGGGGCTCATTTG-3′ and reverse 5′-AGGGGCCATCCACAGTCTTC-3′;
miR-634, forward 5′-CAGTCTCAAACCAGCACC-3′ and reverse
5′-TATGGTTGTTCACGACTCCTTCAC-3′; and HSPA2, forward
5′-AAACTTTACCAAGGTGGTCCTG-3′ and reverse
5′-GCTTAGTCCACTTCTTCGATGG-3′.
Western blot analysis
Proteins were extracted using
radioimmunoprecipitation lysis buffer (Thermo Fisher Scientific,
Inc.) and the protein content was determined using the
Bicinchoninic Acid Protein assay kit (Beyotime Institute of
Biotechnology). Equal amounts of the protein (50 µg/lane) from
lysates of PANC-1 cells were subjected to SDS-PAGE (10% gels) and
then transferred to polyvinylidene difluoride membranes. The
membranes were blocked with 5% non-fat milk for 60 min at room
temperature, followed by incubation with primary antibodies against
HSPA2 (cat. no. ab108416; 1:500; Abcam, Cambridge, MA, USA) and
GAPDH (cat. no. AF0006; 1:1,000; Beyotime Institute of
Biotechnology) overnight at 4°C. Membranes were then washed with
0.1% Tween-20 in PBS (PBST) three times at room temperature. Then,
they were incubated with horseradish peroxidase-conjugated goat
anti-rabbit secondary antibodies (cat. no. sc-2004; 1:3,000; Santa
Cruz Biotechnology, Inc. Santa Cruz, CA, USA) at room temperature
for 1 h. The proteins of interest were detected by the enhanced
chemiluminescence detection system (Sea Biotech, Shanghai, China).
Finally, the intensity of protein bands was detected using
Image-Pro Plus 6.0 software (Media Cybernetics, Inc., Rockville,
MD, USA). GAPDH served as the loading control.
Target prediction
The TargetScanHuman database and TargetScanHuman
Release 7.1 software (http://www.targetScan.org) were used to predict the
potential target gene of miR-634.
RNA immunoprecipitation (RIP)
RIP assays were performed using the Imprint RNA
Immunoprecipitation kit (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany) with the protein argonaute-2 (AGO2) antibodies (cat. no.
2897; 1:500; Cell Signaling Technology, Inc., Danvers, MA, USA) or
IgG antibodies (cat. no. A6066; 1:500; Sigma-Aldrich; Merck KGaA)
for 2 h at room temperature. The mRNA expression levels of miR-634
and HSPA2 in the immunoprecipitates were detected using qPCR
analysis.
Dual-luciferase reporter assay
The wild-type (WT) or mutant (MUT) HSPA2-3′UTR which
contained the miR-634 binding sites was inserted into the
luciferase genes in the pGL3 vectors (Promega Corporation, Madison,
WI, USA). PANC-1 cells were co-transfected with 0.1 mg pGL3-WT
HSPA2-3′-UTR or 0.1 mg pGL3-MUT HSPA2-3′-UTR and 10 nM miR-634
mimic or 10 nM miR-634 inhibitor using Lipofectamine 2000 reagent.
At 48 h after transfection, the cells were gathered and analyzed
via Dual-Luciferase Reporter Assay system (GeneCopoeia, Inc.)
according to the manufacturer's protocol. The activity of firefly
luciferase was normalized to the corresponding Renilla
luciferase activity.
Statistical analysis
All the experiments were performed at least three
times. Quantitative values were expressed as the mean ± standard
error of the mean. Statistical analyses were performed using SPSS
13.0 software (SPSS, Inc., Chicago, IL, USA). Data of more than two
groups were analyzed using one-way analysis of variance with
Tukey's post hoc test. Statistical analysis of miR-634 and HSPA2
expression between PC tissues and control tissues was evaluated
using the paired Student's t-test. The statistical analysis of
unpaired two groups was evaluated using an unpaired Student's
t-test. The correlations between miR-634 expression levels and the
HSPA2 mRNA expression levels in PC tissues were analyzed using
Spearman's rank test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Expression levels of miR-634 in human
PC tissues and cell lines
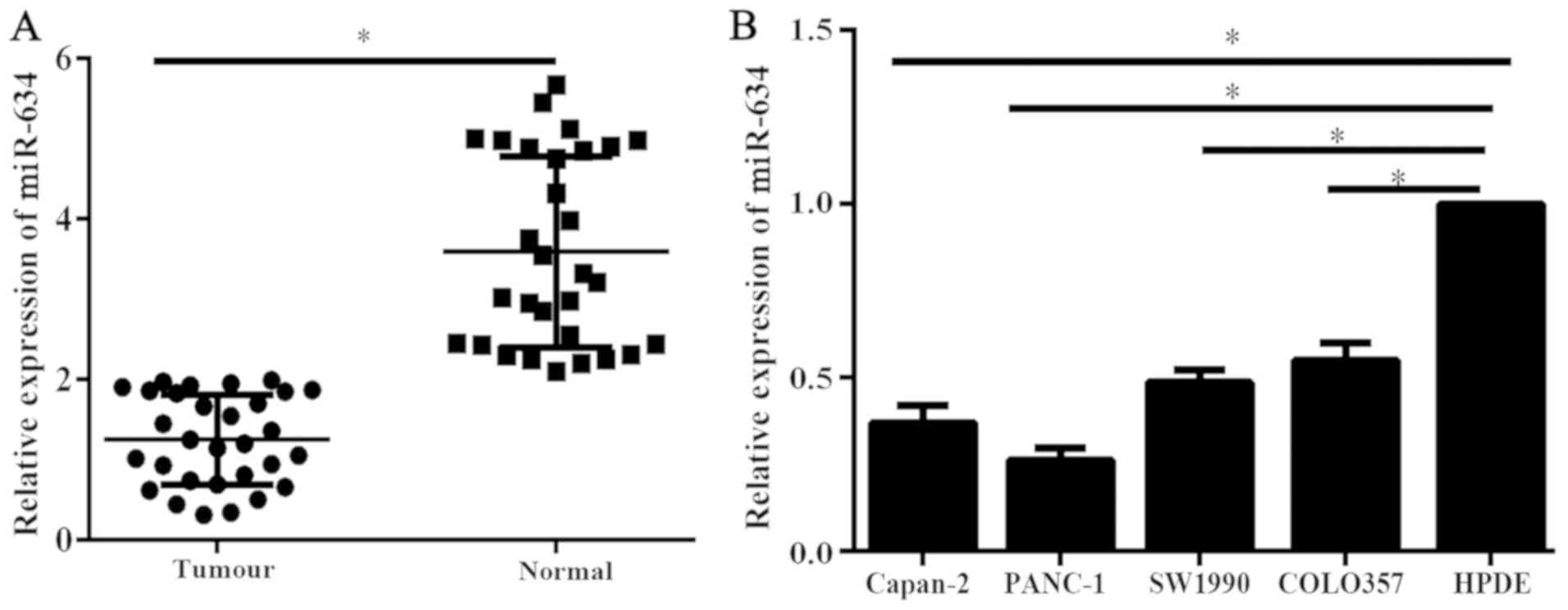
Expression levels of miR-634 in 30 PC tissues and
corresponding adjacent normal tissues were analyzed using qPCR. The
expression levels of miR-634 in PC tissues was significantly
reduced compared with that in adjacent normal tissues (Fig. 1A). Furthermore, the miR-634
expression levels were assessed by RT-qPCR in PC cell lines and the
immortal human pancreatic ductal cell line HPDE. Results of RT-qPCR
indicated that miR-634 expression levels were significantly reduced
in the PC cells compared with HPDE cells (Fig. 1B). These data suggested that the low
expression of miR-634 may be associated with the malignant process
of PC.
miR-634 inhibits the viability and
proliferation of PANC-1 cells
PANC-1 cells were identified to have the lowest
expression level of miR-634 (Fig.
1B). Therefore, PANC-1 cells were used for functional testing.
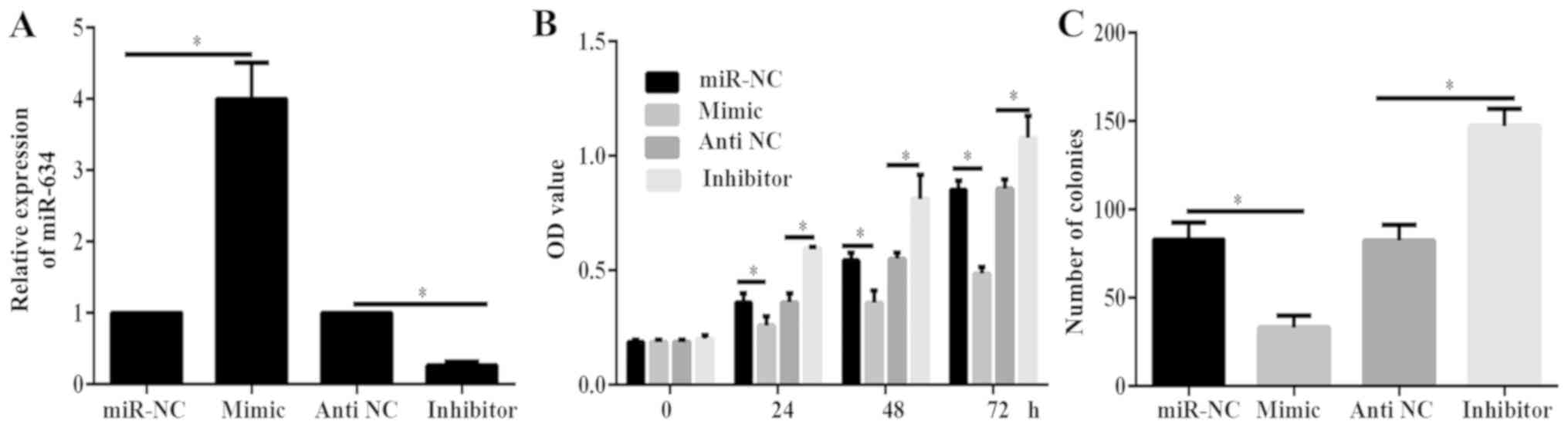
PANC-1 cells were transfected with miR-634 mimics, miR-NC, miR-634
inhibitor or antiNC. Results of RT-qPCR indicated that miR-634
mimic could significantly increase miR-634 expression, while
miR-634 inhibitor significantly decreased miR-634 expression in
PANC-1 cells (Fig. 2A). Results of
CCK-8 and colony formation assays demonstrated that the viability
and proliferation of cells in the miR-634 mimics group was
significantly decreased compared with that in the miR-NC group
(Fig. 2B and C). By contrast, the
viability and proliferation of cells in the miR-634 inhibitor group
was significantly increased than that of cells in the antiNC group
(Fig. 2B and C). These data
demonstrated that miR-634 inhibited PC growth.
miR-634 inhibits the migration and
invasion of PANC-1 cells and enhances the apoptosis rate
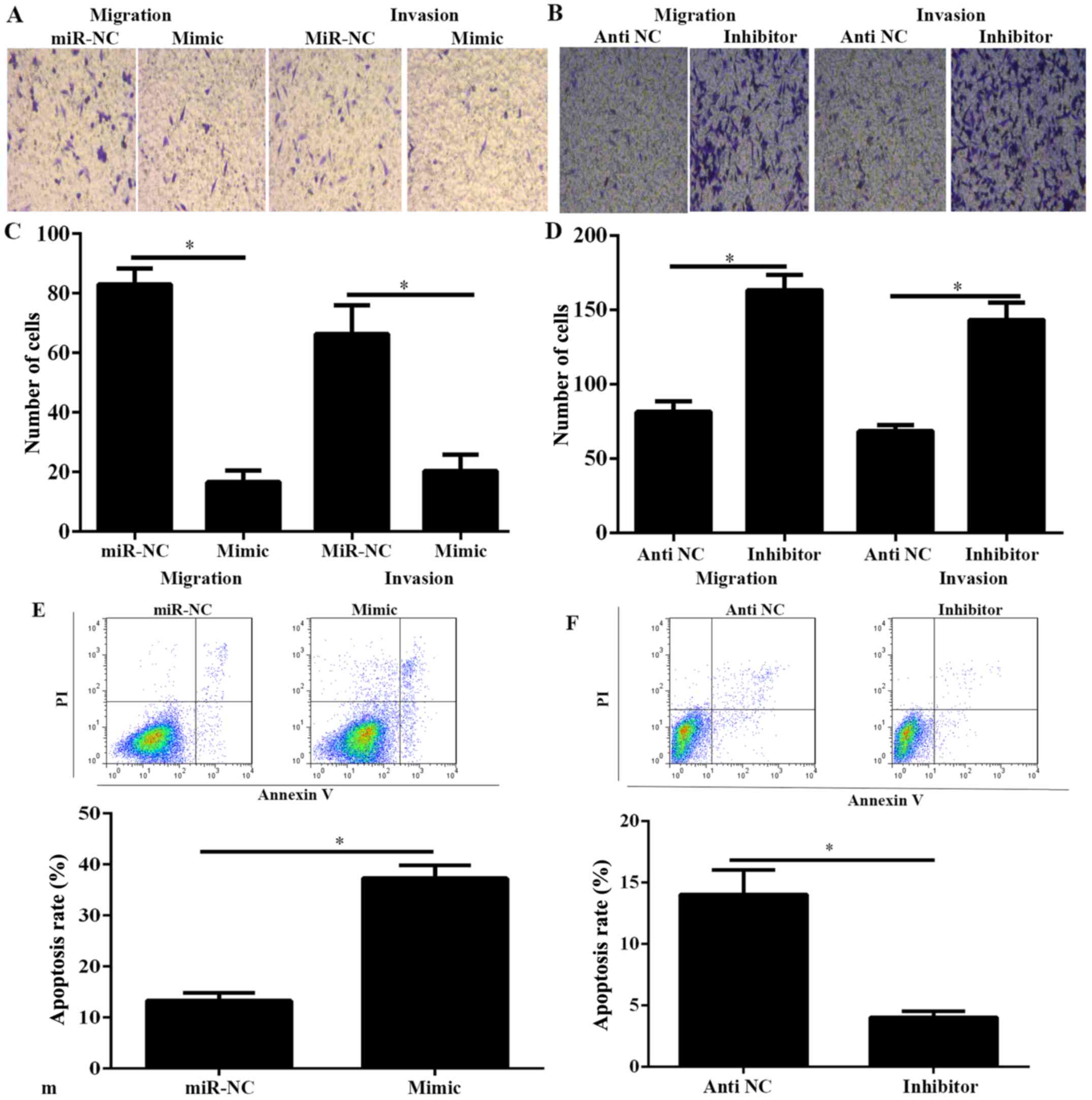
PC is characterized by early metastasis (2). Therefore, to demonstrate the function
of miR-634 on the migration and invasion of PANC-1 cells, the
migration and invasion of PANC-1 cells was investigated using
Transwell migration and invasion assays. Results indicated that
miR-634 mimic significantly decreased the capability of migration
and invasion, whereas miR-634 inhibitor significantly increased the
capability of migration and invasion (Fig. 3A-D). Furthermore, cell apoptosis was
investigated in PANC-1 cells transfected with miR-634 mimic or
inhibitor. As demonstrated in Fig. 3E
and F, overexpression of miR-634 could significantly increase
the cell apoptosis rate in PANC-1 cells, whereas miR-634 silencing
promoted the opposite results.
HSPA2 is a direct target of miR-634 in
PANC-1 cells
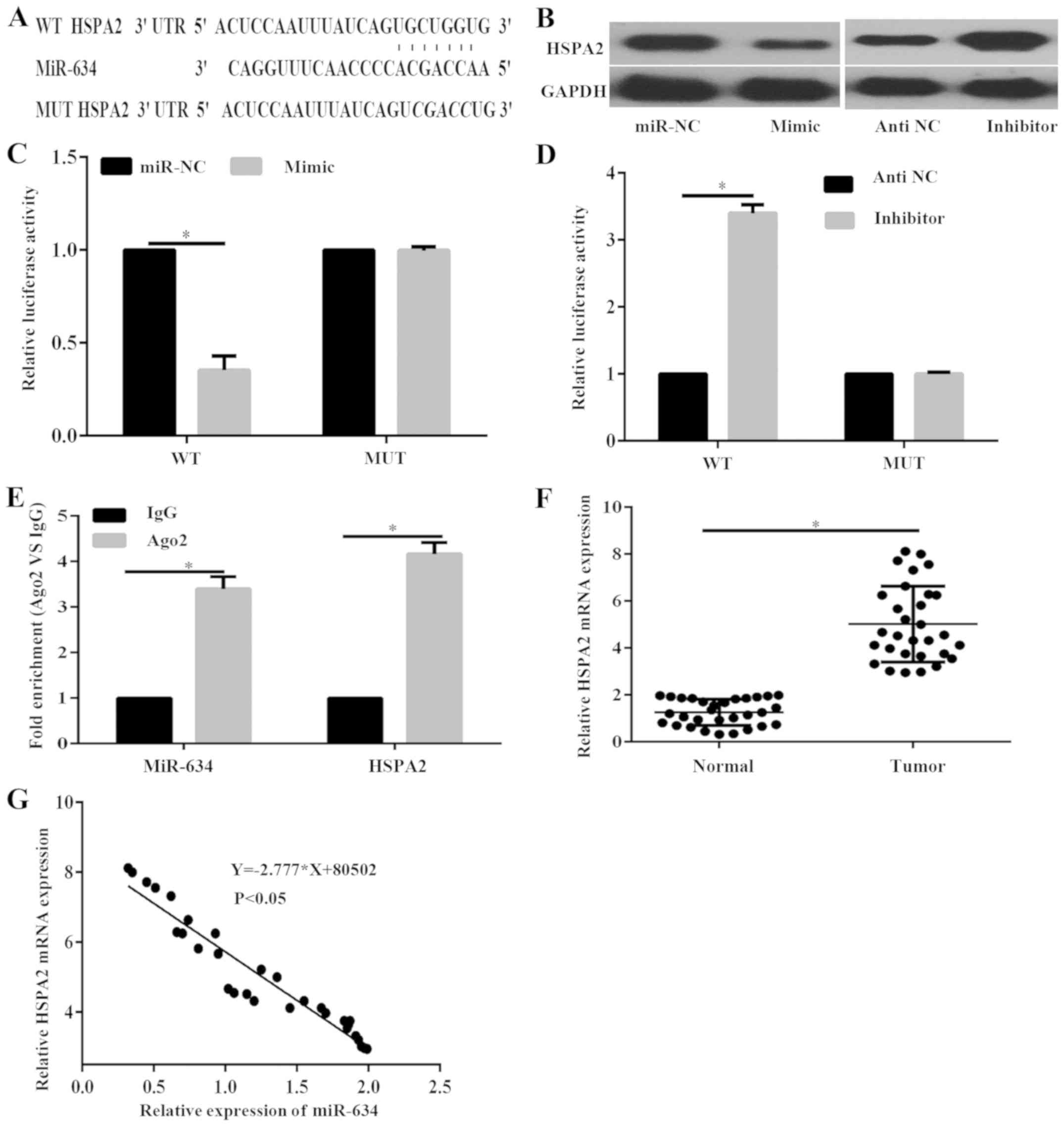
To identify the molecular mechanism underlying the
suppressive role of miR-634 in PC progression, Targetscan was used
to predict the potential target genes of miR-634. Analysis
indicated that the 3′-UTR of HSPA2 contained predicted binding
sites for miR-634 (Fig. 4A).
Notably, previous studies have revealed that HSPA2 correlates with
PC development and progression (15,16). The
protein expression levels of HSPA2 in PANC-1 cells under the
regulation of miR-634 were investigated. Results demonstrated that
miR-634 mimic markedly decreased the protein expression of HSPA2,
whereas miR-634 inhibitor markedly enhanced the protein expression
of HSPA2 in PANC-1 cells (Fig. 4B).
Furthermore, the dual-luciferase reporter assay was performed to
confirm that miR-634 could direct target HSPA2. Results indicated
that the luciferase activity in cells transfected with miR-634
mimic was significantly decreased compared with that of cells
transfected miR-NC in the HSPA2-3′-UTR-wild type (WT) group;
however, there was no significant difference in the
HSPA2-3′-UTR-mutant (MUT) group (Fig.
4C). By contrast, luciferase activity was significantly
increased in cells treated with miR-634 inhibitor compared with
those treated with antiNC in the HSPA2-3′-UTR-WT group (Fig. 4D). However, luciferase activity was
not significantly different in the HSPA2-the 3′-UTR-MUT group
(Fig. 4D).
To further confirm the interaction between miR-634
and the HSPA2 3′-UTR, RIP assay was performed. The RIP assay was
performed with the AGO2 antibody. In the RNA extracted from the
precipitated AGO2 protein, it was possible to detect significantly
increased enrichment of the miR-634 and HSPA2 3′-UTR in the Ago2
group compared with IgG in PANC-1 cells (Fig. 4E), indicating that miR-634 and the
HSPA2 3′-UTR existed in an RNA-induced silencing complex. These
data suggested that miR-634 directly targets HSPA2 by binding to
its 3′-UTR region in PANC-1 cells. In addition, RT-qPCR results
indicated that the expression level of HSPA2 in PC tissues was
significantly increased compared with adjacent normal tissues
(Fig. 4F). Furthermore, Pearson
correlation analysis demonstrated that miR-634 was negatively
correlated with HSPA2 mRNA expression (Fig. 4G). These data demonstrated that HSPA2
is a direct target of miR-634 in PANC-1 cells.
HSPA2 is associated with
miR-634-induced suppression in PANC-1 cells
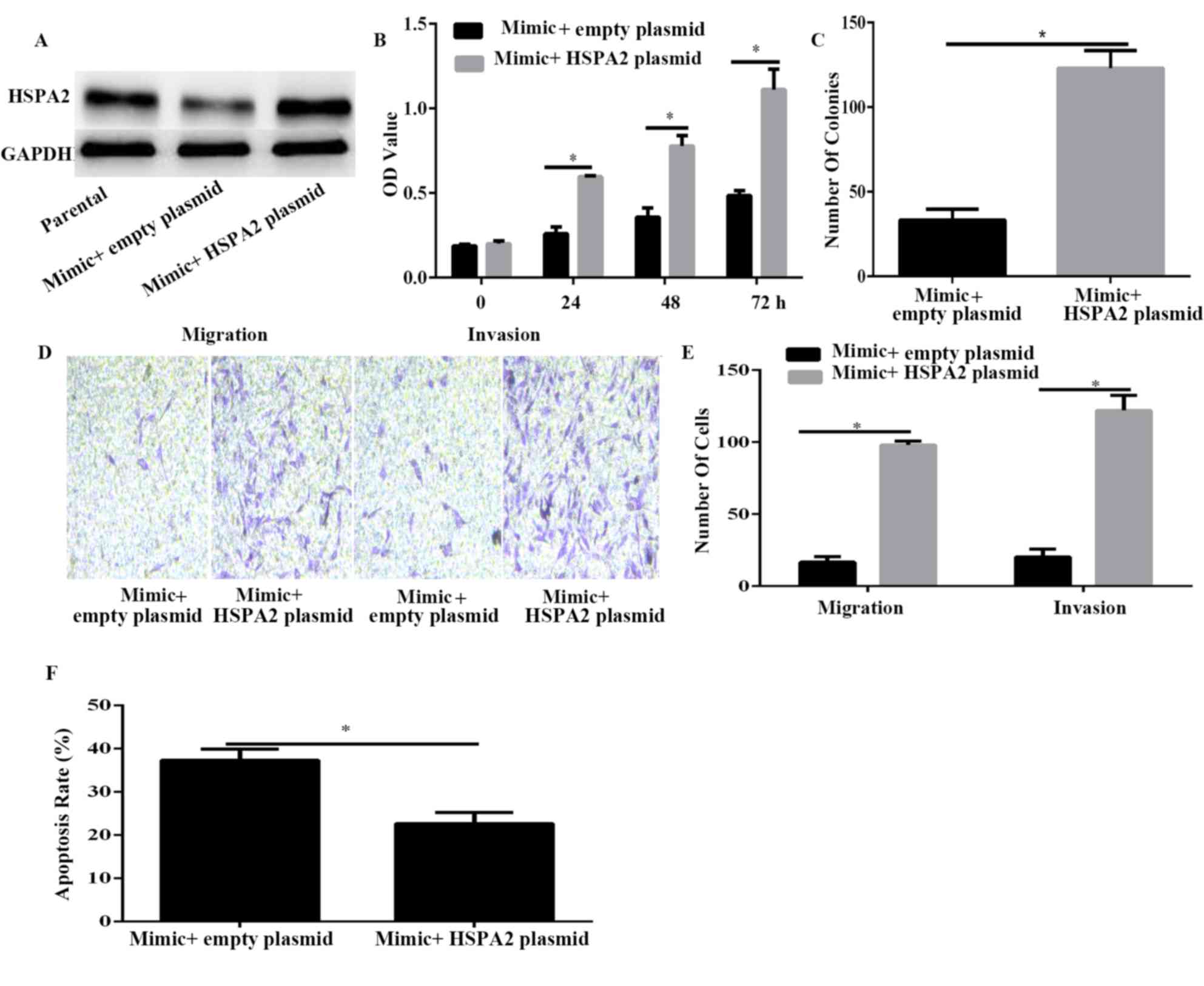
To confirm that HSPA2 was a functional target of
miR-634, HSPA2 was overexpressed using HSPA2 plasmid in
miR-634-overexpressed PANC-1 cells (Fig.
5A). Results of CCK-8 and colony formation assays demonstrated
that HSPA2 overexpression significantly reversed the inhibitory
effect induced by miR-634 mimic on cell viability and proliferation
(Fig. 5B and C). Furthermore, the
Transwell assay results demonstrated that HSPA2 overexpression
significantly reversed the inhibitory effect induced by miR-634
mimic on cell migration and invasion (Fig. 5D and E). Furthermore, the results of
flow cytometry indicated that HSPA2 overexpression could decrease
PANC-1 cell apoptosis induced by miR-634 overexpression. These data
indicated that HSPA2 may be a functional mediator of miR-634 in
PANC-1 cells.
Discussion
miRs act as post-transcriptional gene regulators in
the etiology of various pathological events, including apoptosis,
proliferation, migration and invasion (6). Abnormal expression of miRs has been
considered to impact cancer progression. Several studies have
revealed that miR-634 functions as a biomarker and has significant
roles in various types of cancer, including glioma, gastric
carcinoma, hepatocellular carcinoma, nasopharyngeal carcinoma,
ovarian cancer and cervical cancer (7–12).
However, to the best of our knowledge, no study has investigated
the role of miR-634 in PC thus far. In the present study, it was
revealed that miR-634 was significantly downregulated in PC.
Furthermore, miR-634 overexpression significantly inhibited the
viability, proliferation, migration and invasion of PC cells,
whereas its inhibition led to opposite effects. The findings
demonstrated that miR-634 acts as a tumor suppressor by suppressing
its target gene HSPA2.
To date, few miR-634 targets have been
experimentally validated. In the present study, luciferase reporter
assays and western blot analysis demonstrated that HSPA2 is a
direct miR-634 target. HSPA2, as a testis-specific protein, serves
a critical role in spermatogenesis (17). A previous study demonstrated that
human tumor tissues can express HSPA2 at high levels (18). The polymorphism of HSPA2 at position
1,267 has been suggested to be associated with carcinogenesis in
some types of malignant cancer tissues, including lung cancer,
cervical cancer, oesophageal squamous cell carcinoma and
hepatocellular carcinoma (19–22). A
recent study has indicated that overexpressed HSPA2 is correlated
with tumor angiogenesis and poor prognosis in PC, and may have an
important role in PC progression and serve as a potential biomarker
for the prediction of adverse prognosis in pancreatic carcinoma
(15). However, the specific
molecular mechanisms underlying the observed increase of HSPA2 in
PC remain unclear. In the present study, it was demonstrated that
miR-634 specifically targets HSPA2 in human PC cells. HSPA2
expression was upregulated in PC tissues compared with adjacent
normal tissues, and the levels were inversely correlated with
miR-634 expression in PC tissues. Furthermore, HSPA2 expression was
significantly decreased in miR-634-overexpressing cells, and forced
HSPA2 expression reversed the phenotypes associated with miR-634
overexpression. Thus, to the best of our knowledge, the present
study provided evidence for the first time that miR-634 can
suppress PC cell growth by inhibiting HSPA2. Notably, miR-634 can
regulate other genes, including CYR61, JAG1, Rab1A and DHX33
(7–9); therefore, further assessment of the
molecular mechanism of miR-634 in PC is required. A recent study
has revealed that miR-634 could directly regulate Jagged 1
expression in gastric cancer (8);
however, the present study failed to identify any significant
association between miR-634 and JAG1 in PC (data not shown). This
may be due to tissue specificity. Future studies should be
performed to further identify the mechanism of miR-634 inhibition
on the progression of PC and the biological function of miR-634 in
other types of cancer.
In conclusion, the present findings suggested that
miR-634 is downregulated in PC and serves as a tumor suppressor by
directly targeting HSPA2. Additionally, the results further
clarified the importance of the miR-634/HSPA2 molecular network in
PC development and may provide a novel therapeutic approach for
treating PC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
DRC and XWZ conceived and designed the experiments.
DRC, XLW, JWZ and XWZ conducted all of the experiments. JWZ and XWZ
wrote and revised the manuscript. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Medical Ethics
Committee of LanLing County Hospital (Shandong, China). All
patients included in this research were required to provide written
informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Li D, Xie K, Wolff R and Abbruzzese JL:
Pancreatic cancer. Lancet. 363:1049–1057. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Oettle H, Post S, Neuhaus P, Gellert K,
Langrehr J, Ridwelski K, Schramm H, Fahlke J, Zuelke C, Burkart C,
et al: Adjuvant chemotherapy with gemcitabine vs. observation in
patients undergoing curative-intent resection of pancreatic cancer:
A randomized controlled trial. JAMA. 297:267–277. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Diab M, Muqbil I, Mohammad RM, Azmi AS and
Philip PA: The role of microRNAs in the diagnosis and treatment of
pancreatic adenocarcinoma. J Clin Med. 5:E592016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yonemori K, Kurahara H, Maemura K and
Natsugoe S: MicroRNA in pancreatic cancer. J Hum Genet. 62:33–40.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tan Z, Zhao J and Jiang Y: MiR-634
sensitizes glioma cells to temozolomide by targeting CYR61 through
Raf-ERK signaling pathway. Cancer Med. 7:913–921. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Guo J, Zhang CD, An JX, Xiao YY, Shao S,
Zhou NM and Dai DQ: Expression of miR-634 in gastric carcinoma and
its effects on proliferation, migration, and invasion of gastric
cancer cells. Cancer Med. 7:776–787. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang CZ, Cao Y, Fu J, Yun JP and Zhang
MF: miR-634 exhibits anti-tumor activities toward hepatocellular
carcinoma via Rab1A and DHX33. Mol Oncol. 10:1532–1541. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
van Jaarsveld MT, van Kuijk PF, Boersma
AW, Helleman J, van IJcken WF, Mathijssen RH, Pothof J, Berns EM,
Verweij J and Wiemer EA: miR-634 restores drug sensitivity in
resistant ovarian cancer cells by targeting the Ras-MAPK pathway.
Mol Cancer. 14:1962015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cong J, Liu R, Wang X, Jiang H and Zhang
Y: MiR-634 decreases cell proliferation and induces apoptosis by
targeting mTOR signaling pathway in cervical cancer cells. Artif
Cells Nanomed Biotechnol. 44:1694–1701. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Peng X, Cao P, He D, Han S, Zhou J, Tan G,
Li W, Yu F, Yu J, Li Z and Cao K: miR-634 sensitizes nasopharyngeal
carcinoma cells to paclitaxel and inhibits cell growth both in
vitro and in vivo. Int J Clin Exp Pathol. 7:6784–6791.
2014.PubMed/NCBI
|
|
13
|
Chun YS, Pawlik TM and Vauthey JN: 8th
edition of the AJCC cancer staging manual: Pancreas and
hepatobiliary cancers. Ann Surg Oncol. 25:845–847. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhai LL, Xie Q, Zhou CH, Huang DW, Tang ZG
and Ju TF: Overexpressed HSPA2 correlates with tumor angiogenesis
and unfavorable prognosis in pancreatic carcinoma. Pancreatology.
17:457–463. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang H, Gao H, Liu C, Kong Y, Wang C and
Zhang H: Expression and clinical significance of HSPA2 in
pancreatic ductal adenocarcinoma. Diagn Pathol. 10:132015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dix DJ, Allen JW, Collins BW,
Poorman-Allen P, Mori C, Blizard DR, Brown PR, Goulding EH, Strong
BD and Eddy EM: HSP70-2 is required for desynapsis of synaptonemal
complexes during meiotic prophase in juvenile and adult mouse
spermatocytes. Development. 124:4595–4603. 1997.PubMed/NCBI
|
|
18
|
Scieglinska D and Krawczyk Z: Expression,
function, and regulation of the testis-enriched heat shock HSPA2
gene in rodents and humans. Cell Stress Chaperones. 20:221–235.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fu Y, Zhao H, Li XS, Kang HR, Ma JX, Yao
FF and Du N: Expression of HSPA2 in human hepatocellular carcinoma
and its clinical significance. Tumour Biol. 35:11283–11287. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang H, Chen W, Duan CJ and Zhang CF:
Overexpression of HSPA2 is correlated with poor prognosis in
esophageal squamous cell carcinoma. World J Surg Oncol. 11:1412013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Scieglinska D, Gogler-Piglowska A,
Butkiewicz D, Chekan M, Malusecka E, Harasim J, Habryka A and
Krawczyk Z: HSPA2 is expressed in human tumors and correlates with
clinical features in non-small cell lung carcinoma patients.
Anticancer Res. 34:2833–2840. 2014.PubMed/NCBI
|
|
22
|
Garg M, Kanojia D, Saini S, Suri S, Gupta
A, Surolia A and Suri A: Germ cell-specific heat shock protein 70-2
is expressed in cervical carcinoma and is involved in the growth,
migration, and invasion of cervical cells. Cancer. 116:3785–3796.
2010. View Article : Google Scholar : PubMed/NCBI
|