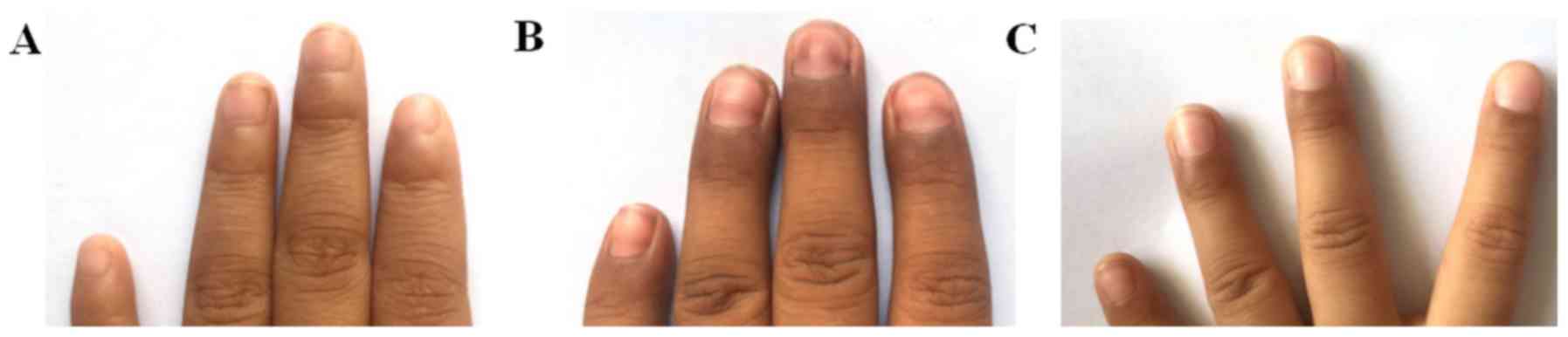
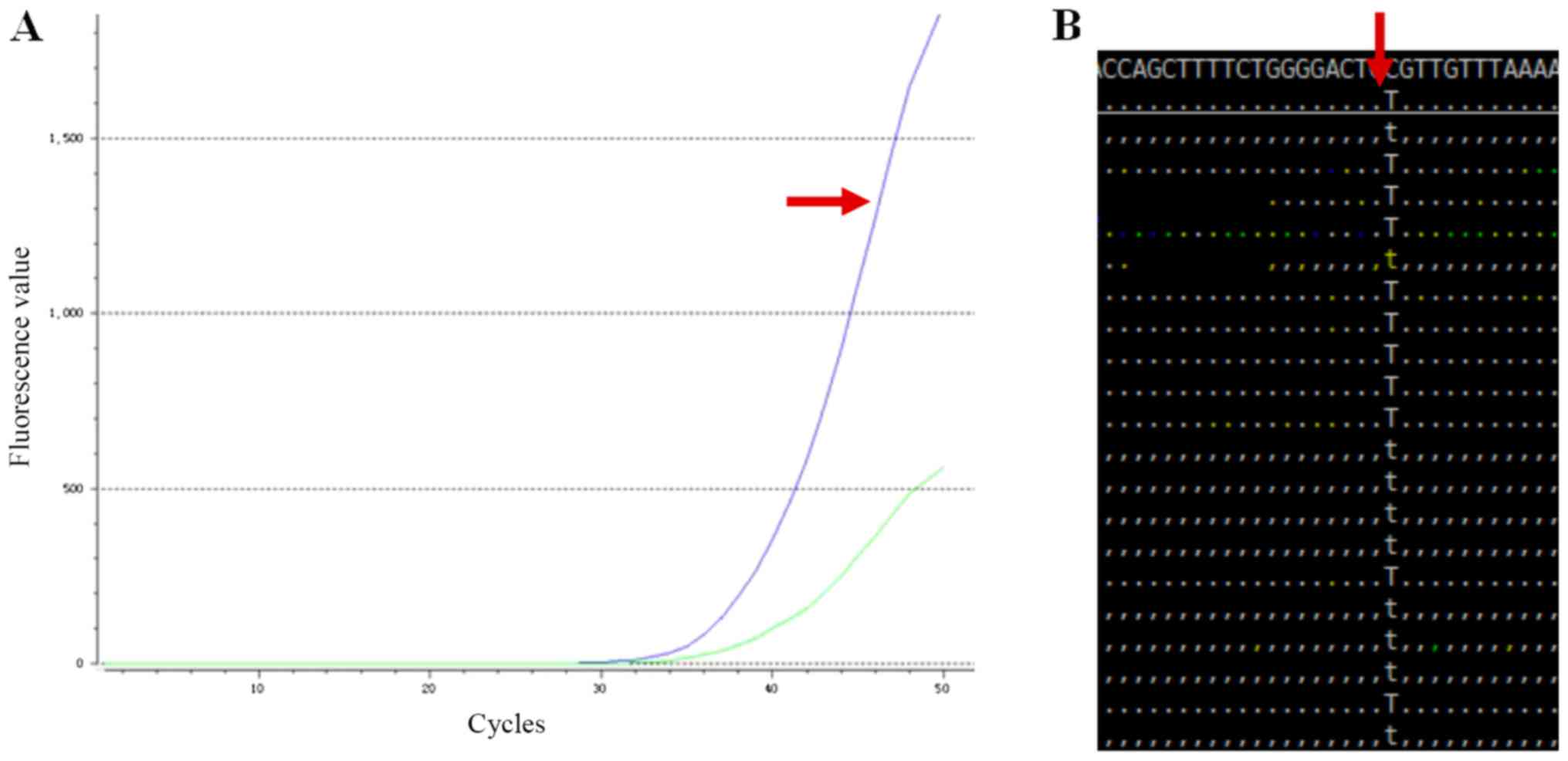
|
1
|
Pui CH, Carroll WL, Meshinchi S and Arceci
RJ: Biology, risk stratification, and therapy of pediatric acute
leukemias: An update. J Clin Oncol. 29:551–565. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lopez-Lopez E, Gutierrez-Camino A,
Bilbao-Aldaiturriaga N, Pombar-Gomez M, Martin-Guerrero I and
Garcia-Orad A: Pharmacogenetics of childhood acute lymphoblastic
leukemia. Pharmacogenomics. 15:1383–1398. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Silverman LB, Declerck L, Gelber RD,
Dalton VK, Asselin BL, Barr RD, Clavell LA, Hurwitz CA, Moghrabi A,
Samson Y, et al: Results of Dana-farber cancer institute consortium
protocols for children with newly diagnosed acute lymphoblastic
leukemia (1981–1995). Leukemia. 14:2247–2256. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rudin S, Marable M and Huang RS: The
promise of pharmacogenomics in reducing toxicity during acute
lymphoblastic leukemia maintenance treatment. Genomics Proteomics
Bioinformatics. 15:82–93. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cooper SL and Brown PA: Treatment of
pediatric acute lymphoblastic leukemia. Pediatr Clin North Am.
62:61–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hunger SP and Mullighan CG: Acute
lymphoblastic leukemia in children. N Engl J Med. 373:1541–1552.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Arya LS, Kotikanyadanam SP, Bhargava M,
Saxena R, Sazawal S, Bakhshi S, Khattar A, Kulkarni KP, Adde M,
Vats TS and Magrath I: Pattern of relapse in childhood all:
Challenges and lessons from a uniform treatment protocol. J Pediatr
Hemato Oncol. 32:370–375. 2010. View Article : Google Scholar
|
|
8
|
Mei L, Ontiveros EP, Griffiths EA,
Thompson JE, Wang ES and Wetzler M: Pharmacogenetics predictive of
response and toxicity in acute lymphoblastic leukemia therapy.
Blood Rev. 29:243–249. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang DS, Yu CH, Chou WC, Chang YH, Lin CY,
Lin KH, Jou ST, Lu MY, Chang HH, Lin SW, et al: Adjusting dosage of
mercaptopurine By NUDT15 polymorphisms in childhood acute
lymphoblastic leukemia. Blood. 130:25662017.
|
|
10
|
Farfan MJ, Salas C, Canales C, Silva F,
Villarroel M, Kopp K, Torres JP, Santolaya ME and Morales J:
Prevalence of TPMT and ITPA gene polymorphisms and effect on
mercaptopurine dosage in Chilean children with acute lymphoblastic
leukemia. BMC Cancer. 14:2992014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nishii R, Moriyama T, Janke LJ, Yang W,
Suiter CC, Lin TN, Li L, Kihira K, Toyoda H, Hofmann U, et al:
Preclinical evaluation of NUDT15-guided thiopurine therapy and its
effects on toxicity and anti-leukemic efficacy. Blood.
131:2466–2474. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chiengthong K, Ittiwut C, Muensri S,
Sophonphan J, Sosothikul D, Seksan P, Suppipat K, Suphapeetiporn K
and Shotelersuk V: NUDT15 c.415C>T increases risk of
6-mercaptopurine induced myelosuppression during maintenance
therapy in children with acute lymphoblastic leukemia.
Haematologica. 101:e24–e26. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang JJ, Landier W, Yang W, Liu C, Hageman
L, Cheng C, Pei D, Chen Y, Crews KR, Kornegay N, et al: Inherited
NUDT15 variant is a genetic determinant of mercaptopurine
intolerance in children with acute lymphoblastic leukemia. J Clin
Oncol. 33:1235–1242. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang SK, Hong M, Baek J, Choi H, Zhao W,
Jung Y, Haritunians T, Ye BD, Kim KJ, Park SH, et al: A common
missensevariant in NUDT15 confers susceptibility to
thiopurine-induced leukopenia. Nat Genet. 46:1017–1020. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Singh M, Bhatia P, Khera S and Trehan A:
Emerging role of NUDT15 polymorphisms in 6-mercaptopurine
metabolism and dose related toxicity in acute lymphoblastic
leukaemia. Leuk Res. 62:17–22. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cui L, Li ZG, Chai YH, Yu J, Gao J, Zhu
XF, Jin RM, Shi XD, Zhang LP, Gao YJ, et al: Outcome of children
with newly diagnosed acute lymphoblastic leukemia treated with
CCLG-ALL 2008: The first nation-wide prospective multicenter study
in China. Am J Hematol. 93:913–920. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schmiegelow K, Nielsen SN, Frandsen TL and
Nersting J: Mercaptopurine/Methotrexate maintenance therapy of
childhood acute lymphoblastic leukemia: Clinical facts and fiction.
J Pediatr Hematol Oncol. 36:503–517. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ebbesen MS, Nersting J, Jacobsen JH,
Frandsen TL, Vettenranta K, Abramsson J, Wesenberg F and
Schmiegelow K: Incorporation of 6-thioguanine nucleotides into DNA
during maintenance therapy of childhood acute lymphoblastic
leukemia-the influence of thiopurine methyltransferase genotypes. J
Clin Pharmacol. 53:670–674. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goldberg R and Irving PM: Toxicity and
response to thiopurines in patients with inflammatory bowel
disease. Expert Rev Gastroenterol Hepatol. 9:891–900. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lennard L, Cartwright CS, Wade R and Vora
A: Thiopurine dose intensity and treatment outcome in childhood
lymphoblastic leukaemia: The influence of thiopurine
methyltransferase pharmacogenetics. Br J Haematol. 169:228–240.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wong FC, Leung AW, Kwok JS, Chan MH, Li CK
and Yuen YP: NUDT15 variant and thiopurine-induced leukopenia in
Hong Kong. Hong Kong Med J. 22:185–187. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Azimi F, Jafariyan M, Khatami S, Mortazavi
Y and Azad M: Assessment of Thiopurine-based drugs according to
Thiopurine S-methyltransferase genotype in patients with acute
lymphoblastic leukemia. Iran J Ped Hematol Oncol. 4:32–38.
2014.PubMed/NCBI
|
|
23
|
Yin D, Xia X, Zhang J, Zhang S, Liao F,
Zhang G, Zhang Y, Hou Q, Yang X, Wang H, et al: Impact of NUDT15
polymorphisms on thiopurines-induced myelotoxicity and thiopurines
tolerance dose. Oncotarget. 8:13575–13585. 2017.PubMed/NCBI
|
|
24
|
Liang DC, Yang CP, Liu HC, Jaing TH, Chen
SH, Hung IJ, Yeh TC, Lin TH, Lai CL, Lai CY and Shih LY: NUDT15
gene polymorphism related to mercaptopurine intolerance in Taiwan
Chinese children with acute lymphoblastic leukemia.
Pharmacogenomics J. 16:536–539. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yi ES, Choi YB, Choi R, Lee NH, Lee JW,
Yoo KH, Sung KW, Lee SY and Koo HH: NUDT15 variants cause
hematopoietic toxicity with low 6-TGN levels in children with acute
lymphoblastic leukemia. Cancer Res Treat. 50:872–882. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Moriyama T, Nishii R, Perez-Andreu V, Yang
W, Klussmann FA, Zhao X, Lin TN, Hoshitsuki K, Nersting J, Kihira
K, et al: NUDT15 polymorphisms alter thiopurine metabolism and
hematopoietic toxicity. Nat Genet. 48:367–373. 2016. View Article : Google Scholar : PubMed/NCBI
|