Introduction
Stress is triggered when various psychological,
physiological or environmental stressors induce a state of
threatened homeostasis (1). Stress
has be en identified as a risk factor in 75–90% of all diseases,
including those that lead to morbidity and mortality (2). The production of reactive oxygen
species (ROS), which are neutralized by enzymatic and non-enzymatic
anti-oxidant defense mechanisms, has been recognized as a key
mechanism of stress (3). Among the
several types of stress (including acute and episodic acute),
chronic stress can affect the balance between the production and
scavenge of ROS in cells, thereby disrupting metabolic regulation
and causing oxidative damage (4).
Chronic stress can be classified into several types (including
restraint stress and chronic heterotypic stress), with some leading
to oxidative stress in several tissues, including the brain, lungs,
heart, kidney and liver (5–8).
Gastroesophageal reflux associated tissue fibrosis
can lead to esophageal stiffness, reduced esophageal compliance,
increased smooth muscle mass and reduced esophageal diameter,
leading to smooth muscle dysfunction, esophageal strictures and
ultimately a decreased quality of life (9). Some studies have demonstrated that
chronic stress can regulate the expression of gene-regulating
anti-oxidant systems and NADPH oxidase (NOX), a major driver of ROS
production in various types of cells (10,11).
NOX-induced ROS have been identified as main sources of oxidative
stress, which can accelerate the progression of various fibrotic
diseases, including skin fibrosis (12), idiopathic pulmonary fibrosis
(13), liver fibrosis (14), cardiovascular fibrosis (15) and kidney fibrosis (16).
Previous studies reported that the
psychological-induced oxidative stress can be observed in different
cells or tissues, including white adipose and intestinal tissues
(17,18). Given the lack of research on
esophageal fibrosis, the aim of the current study was to
investigate the effect of chronic stress-induced esophageal
fibrosis using a chronic restraint stress mouse model.
Materials and methods
Experimental animals
A total of 30 male C57BL/6J mice (age, 8 weeks;
weight, 25.6±2.52 g) were obtained from the Animal Center of
Xinjiang Medical University (Urumqi, China) and used in subsequent
experiments. Mice were placed in cages and housed in a
viral-pathogen-free facility at the Research Institute of Uygur
Pharmaceutics (Urumqi, China) under standard conditions
(temperature, 21–25°C and humidity, 50±5%) with a 12 h light/dark
cycle. All mice received free access to water and a normal chow
diet (Teklad Diet; 18% fat, 24% protein, 58% carbohydrates). All
animal experiments were approved by the Animal Care and Use
Committee of the People's Hospital of Xinjiang Uygur Autonomous
Region (protocol no. KY201803703), and all experimental procedures
complied with the Guidelines for the Care and Use of Laboratory
Animals published by the National Institute of Health.
Chronic restraint stress protocol
Mice were randomly divided into two groups: A
control and a chronic restraint stress group. Mice in the control
group were housed in the individual cages and were left
undisturbed. Mice in the chronic restraint stress group were
subjected to restraint stress using a ventilated plastic 50 ml tube
that allowed for a close fit to mice. Subsequently, mice were
submitted to immobilization stress for 2 h per day for period of 14
consecutive days using a self-made restraint device (a 50 ml
centrifugal tube with a number of ventilation holes ~5 mm in
diameter and a small hole for the tail) (17,18).
During the stress period, mice were not allowed access to food and
water. Following chronic restrained stress, mice were maintained in
individual cages and allowed free access to food and water. Body
weight and food intake were monitored every two days during the
stress period.
Sample collection
All mice underwent a 16–18 h fasting period and were
euthanized by intraperitoneal injection of 150 mg/kg sodium
pentobarbital. Blood samples were collected from the inferior vena
cava for biological analysis. Esophageal tissue samples were
collected to examine chronic restraint stress-induced pathology, as
well as the expression levels of specific biological markers.
Histological analysis
Esophageal tissue was collected, weighed, fixed in
10% formalin for 24 h at room temperature and dehydrated by a
descending series of ethanol at room temperature for 4 h. Tissue
samples were embedded in paraffin and cut into 4-µm-thick sections.
Tissue sections were subsequently stained (all at room temperature
for ~2.5 h) with hematoxylin and eosin (H&E), Sirus red or
Masson's trichrome (MT) and observed under a light microscope
(magnification, ×200) and imaged using a digital camera (Eclipse
E200; Nikon Corporation). Staining was observed in 10 randomly
selected fields and analyzed using Adobe Photoshop (Adobe, Inc.)
and ImageJ (version 1.62; National Institutes of Health).
Histological sections were examined for stress-induced inflammatory
changes and scored by a ‘blinded’ observer for three parameters
(19): i) Epithelial damage (0,
normal morphology; 1, mild surface lifting; 2, intraepithelial
separation and surface lifting; and 3, epithelial cell loss to
basal cell layer or deeper); ii) submucosal edema (0, normal; 1,
mild focal edema; 2, moderate diffuse edema; and 3, severe edema);
iii) submucosal inflammation (0, 0–5/high power field (HPF); 1,
5–10/HPF; 2, 10–15/HPF; 3, ≥15/HPF). Each individual score
represented the mean of the three sections.
Immunohistochemistry
The streptavidin-biotinylated peroxidase complex
method was performed as previously described (17,18).
Briefly, esophageal tissue sections were deparaffinized in xylene
and rehydrated with a descending ethanol series at room
temperature. Endogenous peroxidase activity was inhibited using
0.3% H2O2 in methanol for 10 min in room
temperature. Samples were then rinsed with PBS and incubated with
10% goat normal serum (cat. no. 414322F; Nichirei Biosciences,
Inc.) for 30 min at room temperature. Sections were then treated
with the following primary antibodies at 4°C overnight: NAPDH
oxidase 4 (Nox4; cat. no. ab195524; 1:100; Abcam), malondialdehyde
(MDA; cat. no. ab6463; 1:100; Abcam), nuclear factor erythroid
2-related factor 2 (Nrf-2; cat. no. ab62352; 1:100; Abcam), heme
oxygenase 1 (HO-1; cat. no. ab13248; 1:100; Abcam), collagen type I
(cat. no. ab34710; 1:100; Abcam), transforming growth factor β-1
(TGF-β-1; cat. no. sc-130348; 1:100; Santa Cruz Biotechnology,
Inc.), α-smooth muscle actin (SMA; cat. no. A5228; 1:100;
Sigma-Aldrich; Merck KGaA), SMAD-3 (cat. no. #8685; 1:100; Cell
Signaling Technology, Inc.) and F4/80 (cat. no. ab240946; 1:100;
Abcam). Sections were subsequently incubated with horseradish
peroxidase (HRP)-conjugated anti-rabbit IgG (1:200; cat. no.
414181F; Nichirei Biosciences, Inc.) or HRP-conjugated anti-mouse
IgG secondary antibodies (1:200; cat. no. 414191F; Nichirei
Biosciences, Inc.) at room temperature for 30 min. Samples were
then rinsed with PBS and treated with peroxidase-conjugated
streptavidin (Nichirei Biosciences, Inc.) at 37°C for 30 min.
Tissue sections were subsequently stained with 3,3-diaminobenzidine
tetra-hydrochloride (DAB; Sigma-Aldrich; Merck KGaA) with 0.03%
H2O2 at room temperature for 15 min to
visualize the localization of Nox4, MDA, Nrf-2, HO-1, collagen type
I, TGFβ-1, α-SMA and SMAD-3. esophageal tissue sections were
counterstained with methylene green, observed under a light
microscope (magnification, ×200) and imaged using a digital camera
(Eclipse E200; Nikon Corporation). Nox4, MDA, Nrf-2, HO-1, collagen
type I, TGFβ-1, α-SMA and SMAD-3 positive cells were observed in 10
randomly selected fields/section and analyzed using Adobe Photoshop
(Adobe, Inc.) and quantified using ImageJ (version 1.45S; National
Institutes of Health).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from esophageal tissues
using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). Total RNA (1 µg) was then reverse transcribed
into cDNA using the RT system (Qiagen GmbH) in accordance with the
manufacturer's protocol and qPCR was subsequently performed under
the following thermocycling conditions: Initial denaturation for 2
min at 95°C, followed by 40 cycles for 12 sec at 95°C and 60 sec at
60°C. The Bio-Rad CFX96 RT-PCR Detection System (Bio-Rad
Laboratories, Inc.) and Power SYBR Green PCR Master mix (Applied
Biosystems; Thermo Fisher Scientific, Inc.) were utilized for PCR.
The primer pairs used for qPCR are presented in Table I. Serial dilutions of a control cDNA
sample were taken and used as the standard curve for each reaction.
mRNA levels were quantified using the 2−ΔΔCq method
(20) and normalized to the internal
reference gene β-actin. Each experiment was performed in
triplicate.
 | Table I.Primer sequences used in reverse
transcription-quantitative PCR. |
Table I.
Primer sequences used in reverse
transcription-quantitative PCR.
| Gene | Primer sequence
(5′-3′) | Size (bp) |
|---|
| Nox4 | F:
CACCTCTGCCTGCTCATTTGG | 153 |
|
| R:
AGTTGAGGTTCAGGACAGATGC |
|
| Nrf-2 | F:
CGAGATATACGCAGGAGAGGTA AGA | 79 |
|
| R:
GCTCGACAATGTTCTCCAGCTT |
|
| HO-1 | F:
CAGCCCCACCAAGTTCAAAC | 101 |
|
| R:
AGGCGGTCTTAGCCTCTTCTG |
|
| Collagen type
I | F:
GGAATGAAAGGGACACAGAGG | 197 |
|
| R:
TAGCACCATCATTTCCACGA |
|
| TGF-β1 | F:
GGACTCTCCACCTGCAAGAC | 100 |
|
| R:
GACTGGCGAGCCTTAGTTTG |
|
| SMAD-3 | F:
CATCGAGCCCCAGAGCAATA | 88 |
|
| R:
GTGGTTCATCTGGTGGTCACT |
|
| α-SMA | F:
TGCTGACAGAGGCACCACTGAA | 138 |
|
| R:
CAGTTGTACGTCCAGAGGCATA |
|
| β-actin | F:
TATTGGCAACGAGCGGTTC | 75 |
|
| R:
ATGCCACAGGATTCCATACCC |
|
Western blot analysis
Total protein was extracted from esophageal tissue
(~30 mg) using lysis buffer [65 mmol/l Tris-HCl (pH 6.8), 3.3% SDS,
10% glycerol, 2.2% bromophenol blue]. Protein concentration was
subsequently determined using a BCA protein assay kit (Pierce;
Thermo Fisher Scientific, Inc.). Equal quantities of protein (50
µg) were separated via SDS-PAGE on a 10–15% polyacrylamide gel. The
separated proteins were transferred onto polyvinylidene difluoride
membranes and blocked for 1 h at room temperature with 5% bovine
serum albumin (cat. no. 10735078001; Sigma-Aldrich; Merck KGaA) in
Tris-buffered saline containing Tween®−20 (TBS-T).
Membranes were washed with TBS-T, and incubated with the following
primary antibodies (all, 1:1,000): Nrf-2, phosphoNrf-2 (cat. no.
ab76026; 1:100; Abcam), Kelch-like ECH-associated protein 1
(keap-1; cat. no. PAL648Mu01; Cloud-Clone Corp.), HO-1, collagen
type I, TGFβ-1, α-SMA, SMAD-3 and normalized to proliferating cell
nuclear antigen (cat. no. M0879; Dako; Agilent technologies, Inc.)
and β-actin (cat. no. #3700; Cell Signaling Technology, Inc.).
Following primary antibody incubation, membranes were further
incubated with HRP-conjugated anti-mouse IgG (cat. no. #7076; Cell
Signaling Technology, Inc.) and HRP-conjugated anti-rabbit IgG
secondary antibodies (cat. no. #7074; Cell Signaling Technology,
Inc.; each, 1:10,000) for 1 h at room temperature. Membranes were
then washed three times with TBS-T. Protein bands were visualized
using the enhanced Chemi-Lumi One System (Nacalai Tesque,
Inc.).
ELISA
Plasma samples were taken from all mice and
processed as previously described (17,18).
Plasma Nox4 (cat. no. SEB924Mu; Cloud-Clone Corp.), MDA (cat. no.
KGE013; R&D Systems, Inc.), total cholesterol (cat. no.
ab65390; Abcam), tryglycerides (cat. no. ab178780; Abcam) and free
fatty acids (FFA; cat. no. ab65341; Abcam) expression was detected
using competitive ELISA kits, according to the manufacturer's
protocol.
Statistical analysis
Data presented as the mean ± standard deviation and
were analyzed using GraphPad Prism 5.01 software (GraphPad
Software, Inc.). A Student's t-test was performed to analyze the
differences between the chronic restraint stress and control groups
with SPSS 19 software (IBM, Corp.). One-way analysis of variance
followed by a Fisher's protected least significant differences test
was performed to analyze the quantitative data collected from both
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
Chronic restraint stress increases
fibrotic biomarker expression in esophageal tissue
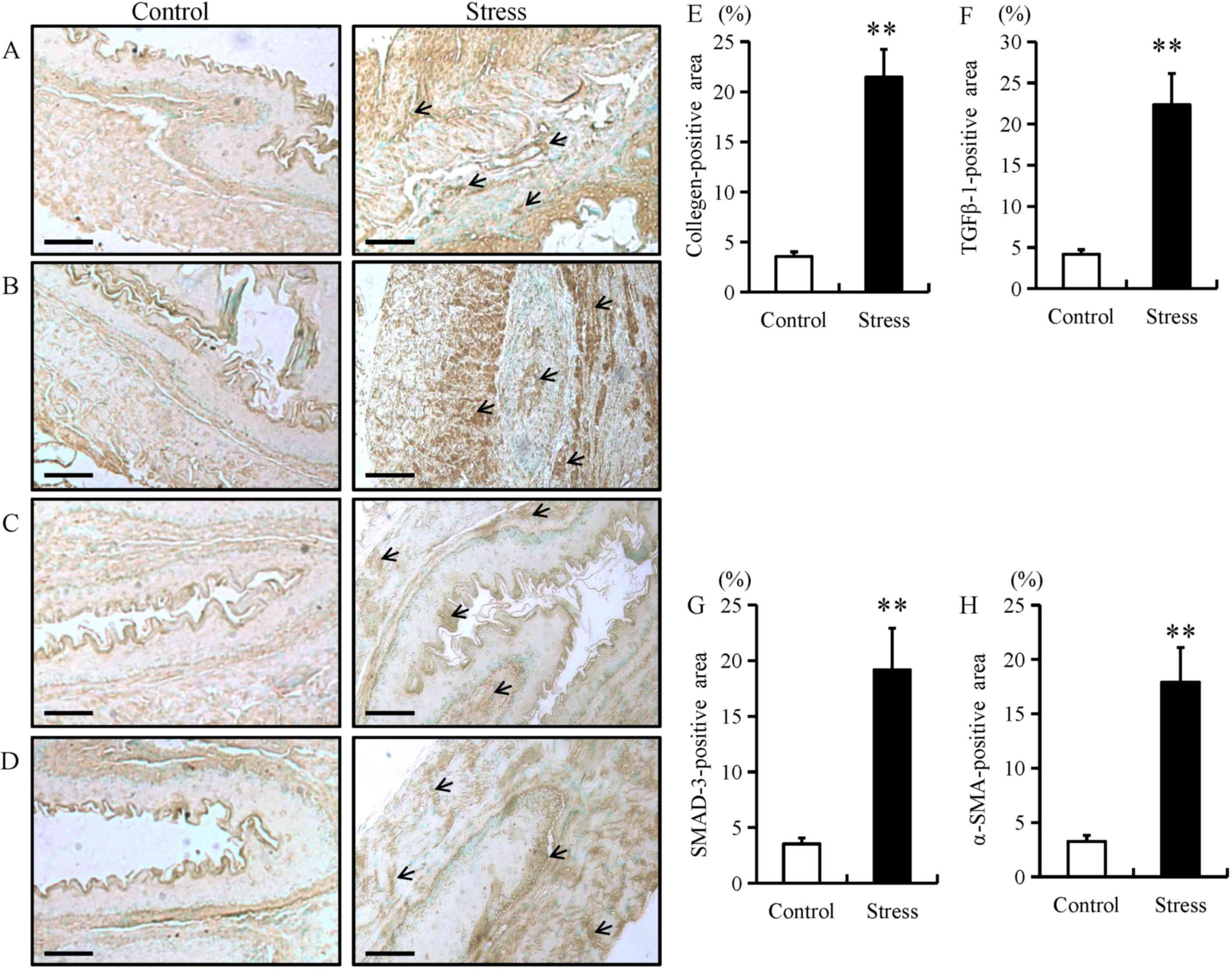
The expression of fibrotic proteins, including
collagen type I, TGF-β1, SMAD-3 and α-SMA were examined in
esophageal tissue using immunohistochemistry, RT-qPCR and western
blotting. Compared with control mice, the expression of fibrotic
proteins in stressed mice was predominantly located in the mucosal
and epithelial layers of the esophagus (as indicated by arrows;
Fig. 1A-D). In addition, chronic
restraint stress significantly increased the expression of collagen
type I, TGF-β1, SMAD-3 and α-SMA in the mucosal and epithelial
layers of the esophagus when compared with the control group
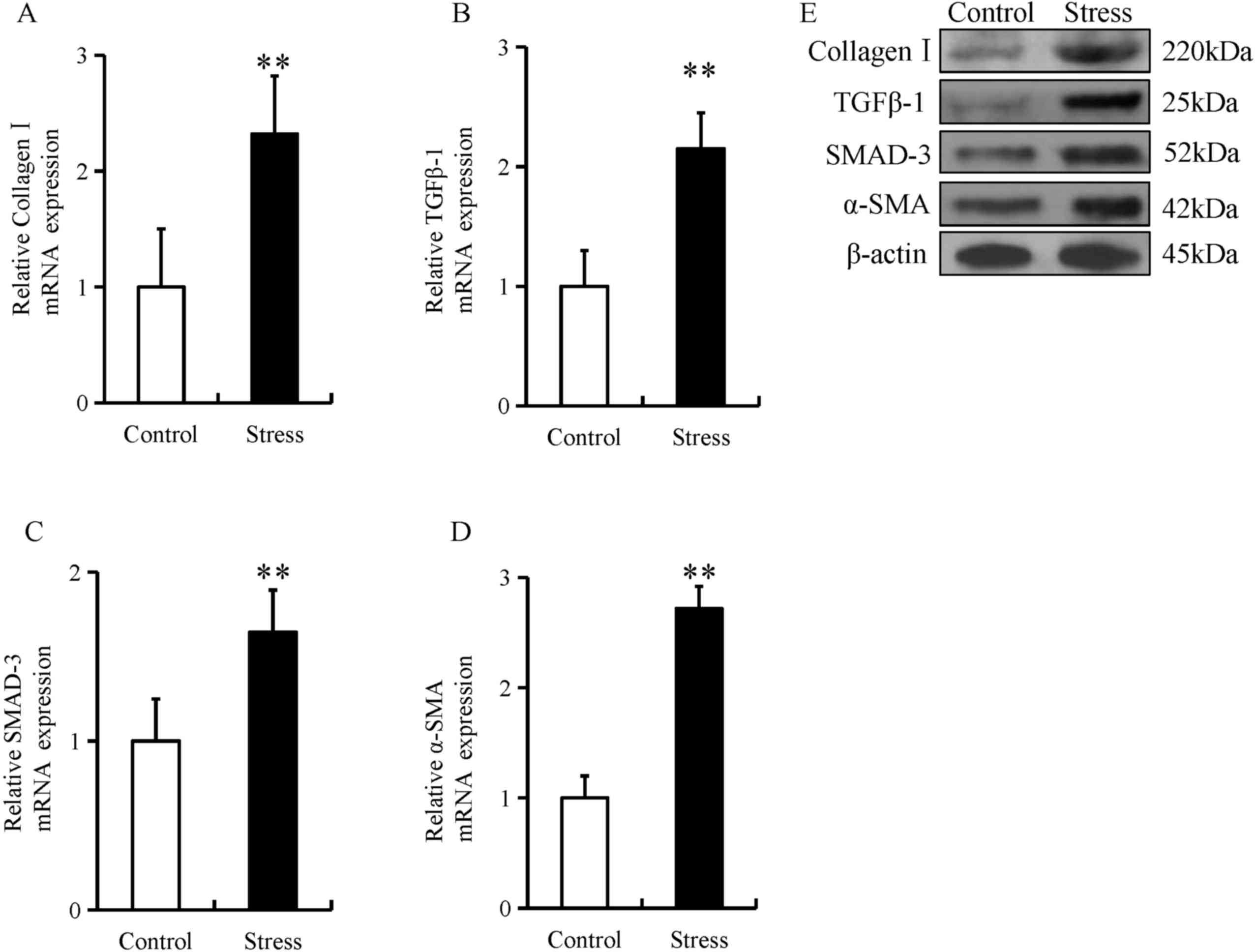
(Fig. 1A-H). Furthermore, the mRNA
and protein expression of these fibrotic proteins were
significantly increased in the esophageal tissue of mice in the
chronic restraint stress group compared with the control group
(Fig. 2A-E).
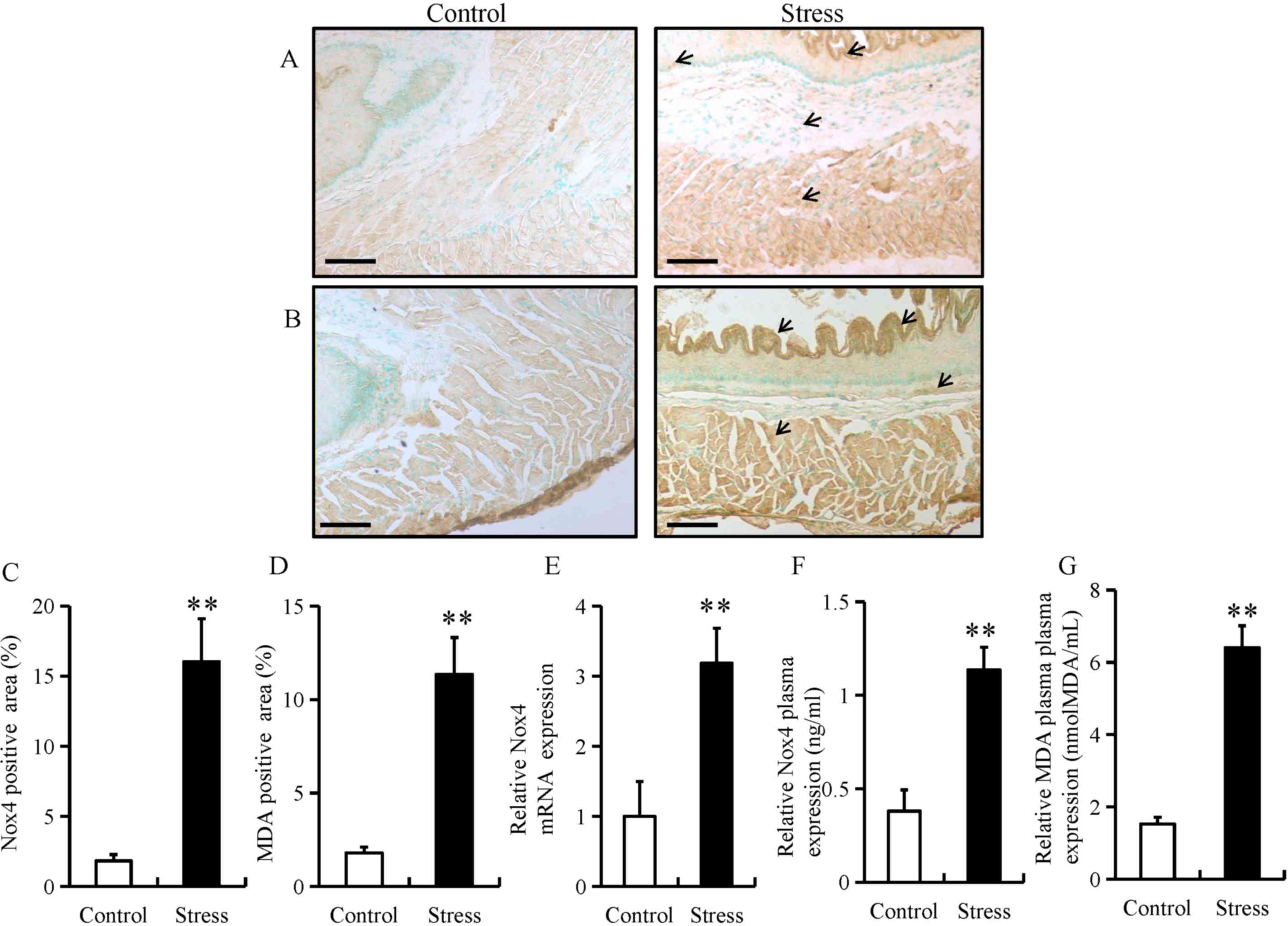
Chronic restraint stress induces ROS
generation in the esophagus
Chronic stress has been previously reported to
trigger ROS production in adipose (18) and colon tissue (21). To determine whether chronic stress
triggers the generation of ROS in the esophagus, the expression of
Nox4 and MDA were determined using immunohistochemistry in the
esophageal tissue of mice. The expression of Nox4 and MDA were
predominantly located in the mucosal and epithelial layers of the
esophagus (as indicated by arrows; Fig.
3A and B). In addition, chronic restraint stress significantly
increased the expression of Nox4 and MDA in the mucosal and
epithelial layers of the esophagus (Fig.
3C and D). Furthermore, chronic restraint stress significantly
upregulated Nox4 mRNA levels, as well as Nox4 and MDA plasma
expression compared with the control group (Fig. 3E-G).
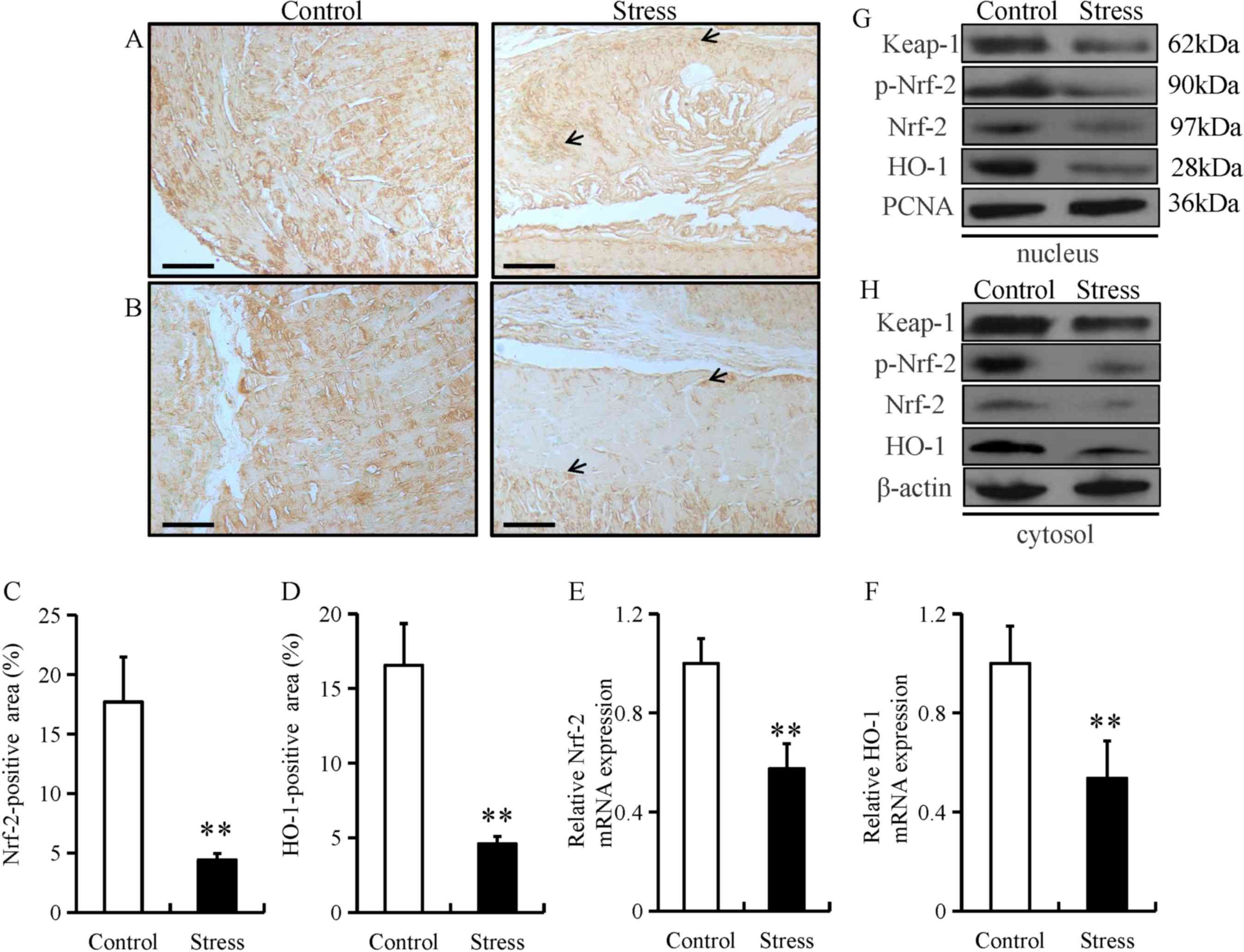
Chronic restraint stress reduces
esophageal expression of anti-oxidant proteins
Under normal physiological conditions, Nrf-2 remains
in an inactive form in the cytoplasm by Keap1 (22). Nrf-2 can be activated by diverse
stimuli, including oxidants, pro-oxidants and antioxidants
(22). Both Nrf-2 and its downstream
target gene, HO-1, serve as major regulators in the protection
against oxidative stress in the esophagus (23). Oxidative stress can damage the
function of the epithelial barrier in the gastrointestinal system
(24). Thus, to investigate the
potential role of Nrf-2 in esophageal fibrosis, the expression of
Nrf-2 and HO-1 was examined in in esophageal tissue from mice
subjected to chronic restraint stress. The expression of Nrf-2 and
HO-1 were predominantly located in the mucosal and epithelial
layers of the esophagus (as indicated by arrows; Fig. 4A and B). In addition, chronic
restraint stress significantly decreased the expression of Nrf-2
and HO-1 in the mucosal and epithelial layers of the esophagus
(Fig. 4C and D). Chronic restraint
stress also significantly decreased the mRNA level of Nrf-2 and
HO-1 mRNA compared with the control group (Fig. 4E and F). Furthermore, Keap-1,
p-Nrf-2, Nrf-2 and HO-1 protein levels were downregulated in both
the nucleus and cytoplasm fractions from the chronic restraint
stress group compared with the control group (Fig. 4G and H).
Chronic restraint stress induces
weight loss and FFA release in mice
Consistent with prior studies (17,18),
subjecting the mice to chronic restraint stress for two weeks
significantly reduced their body weight compared with the mice in
the control group (Table II). In
addition, there were no significant differences in the food intake,
cholesterol or triglyceride levels of mice in the chronic restraint
stress group compared with the control group (Table II). However, the concentration of
FFA was significantly increased in the chronic restraint stress
group compared with the control group (Table II).
 | Table II.Chronic restraint stress-induced
weight loss and FFA release in mice. |
Table II.
Chronic restraint stress-induced
weight loss and FFA release in mice.
|
| Control | Stress | P-value |
|---|
| BW gain (g) |
1.37±0.02 |
1.04±0.04 | <0.001 |
| Food intake
(mg) | 133.5±2.46 | 130.7±2.46 |
0.441 |
| TC (mg/dl) |
64±2.42 |
70.6±3.23 |
0.123 |
| TG (mg/dl) |
8.19±0.39 |
8.95±0.62 |
0.324 |
| FFA (mEq/l) |
0.36±0.03 |
0.91±0.05 | <0.001 |
Chronic restraint stress induces
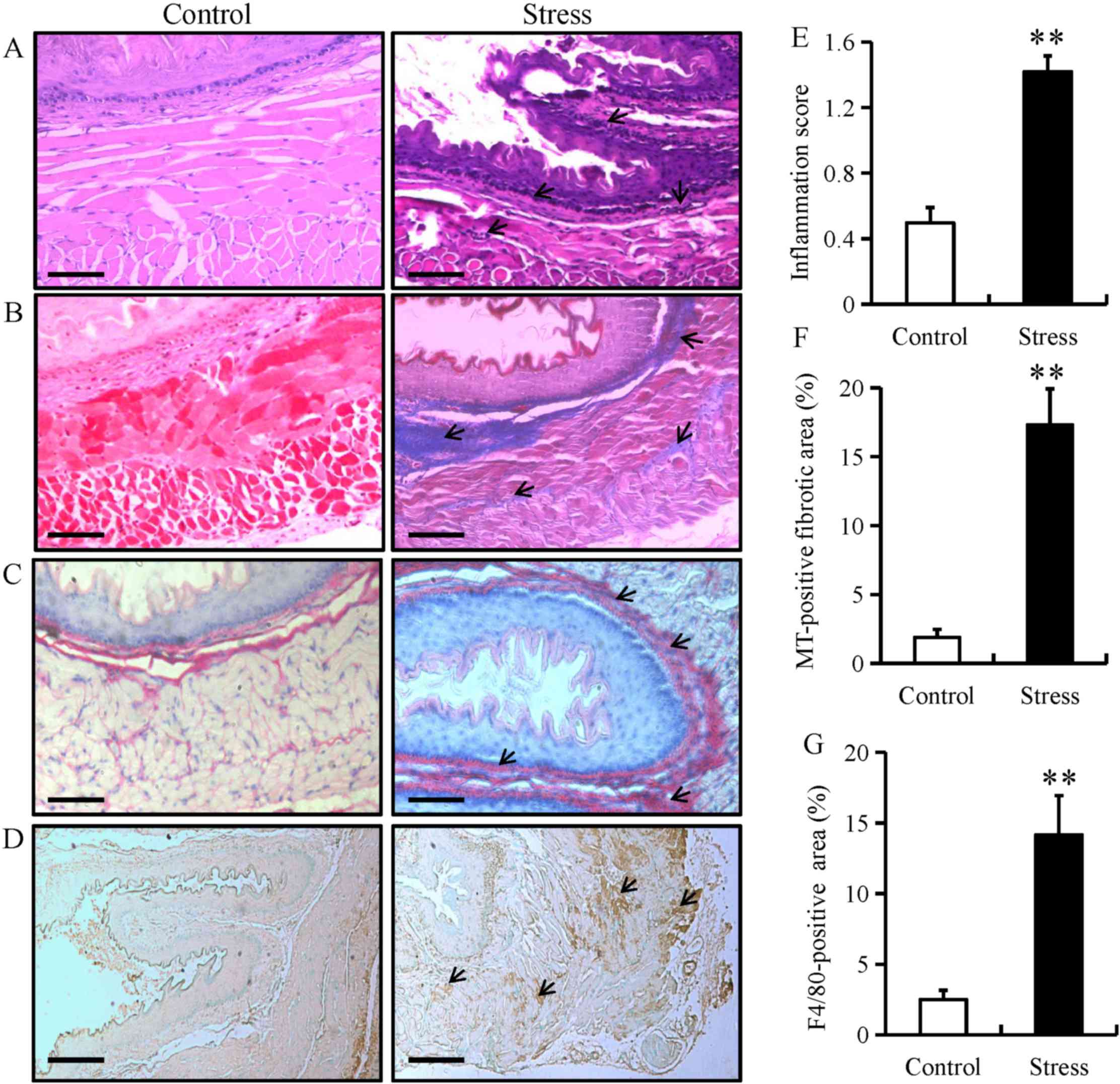
esophageal fibrosis in mice
To examine stress-induced histopathological changes
and fibrosis, esophageal tissue from mice in the chronic restraint
stress and control groups were stained with H&E, Sirus red or
MT. H&E staining demonstrated that chronic restraint stress
increased lymphocytic infiltration (as indicated by arrows) and the
degree of inflammation within the submucosa of the esophagus, while
there were no distinct changes observed in the control group
(Fig. 5A and E). Furthermore, MT and
sirus red staining highlighted the occurrence of fibrosis (as
indicated by arrows) in the esophagus of the stressed mice
(Fig. 5B, C and F). The MT-positive
fibrotic areas in esophageal tissue from mice in the chronic
restraint stress group were increased compared with the control
group (Fig. 5F). IHC for F4/80
(specific for monocytes/macrophages, respectively) was performed in
esophageal tissue from mice in the chronic restraint stress and
control groups. The results demonstrated that F4/80 expression was
predominantly located in the mucosal and epithelial layers of the
esophagus (as indicated by arrows), and that expression levels
significantly increased in the chronic restraint stress group
compared with the control group (Fig. 5D
and G).
Discussion
The current study identified several key findings.
The expression of fibrotic proteins, including collagen type I,
TGF-β1, SMAD-3 and α-SMA, demonstrated that chronic restraint
stress may induce esophageal fibrosis in mice. In addition, chronic
restraint stress may induce oxidative stress as the expression
levels of Nox4 and MDA were significantly increased in mice.
Furthermore, chronic restraint stress reduced the expression of
several anti-oxidative proteins, including Keap-1, p-Nrf-2, Nrf-2
and HO-1 in the esophageal tissue of mice. In conclusion, the
current study demonstrated that chronic stress may trigger
esophageal fibrosis by enhancing oxidative stress and suppressing
the anti-oxidative system.
Oxidative stress is an essential factor in the
pathogenesis of esophageal injury/repair and in esophageal barrier
dysfunction (25). NADPH oxidases,
including Nox4, are the main producers of ROS in the esophagus and
serve key roles in esophageal remodeling, esophageal barrier
dysfunction and inflammation (26,27).
Previous studies have demonstrated that two weeks of chronic
restraint stress can trigger inflammation and ROS accumulation in
different types of tissues (18,20).
Oxidative stress also leads to esophageal fibrosis by increasing
the expression of TGF-β1, which enhances the synthesis of
esophageal collagen and suppresses the degradation of collagen in
the gastroesophageal reflux disease (GERD) model (25,28). The
current study demonstrated that chronic restraint stress enhanced
the in vivo esophageal expression of Nox4 and MDA, a
biomarker of oxidative stress.
Chronic psychological stress can induce oxidative
stress in different tissues, including the brain and peripheral
blood cells, and these adverse effects can be partially reversed by
anxiolytic agents (29). A previous
study demonstrated that two weeks of chronic restraint stress in
mice caused an accumulation of ROS and inflammation in several
types of tissue, including visceral adipose tissue (VAT) as well as
liver and intestine (21).
Suppressed chronic stress-induced ROS production and VAT
inflammation were identified as potential therapeutic targets for
stress-associated disorders (18).
Increased ROS accumulation in VAT is accompanied by increased NADPH
oxidase (NOX) subunits and decreased antioxidant enzymes and has
been recognized as an early marker and potential therapeutic target
of metabolic syndrome (16).
Activated myofibroblasts are key effector cells in
all models of fibrosis. In wound healing, tissue strain and
cytokine release activate myofibroblasts, which initiate migration,
extracellular matrix (ECM) deposition and tissue contraction,
thereby maintaining tissue homeostasis (30). However, in fibrosis, an exaggerated
myofibroblast response results in inappropriate ECM deposition,
increased tissue stiffness and organ dysfunction (31). As epithelial cells are capable of
transdifferentiation under these conditions, it has been recognized
that during chronic inflammation, epithelial cells undergo
epithelial-to-mesenchymal transition in fibrosis (32). As the epithelium is often the site of
primary injury and inflammation, epithelial cells may also function
as effector cells in fibrogenesis.
Oxidative stress is closely associated with the
pathogenesis of GERD, which leads to increased ROS production
(33). Long-term exposure to
oxidative stress in GERD induces chronic inflammation and fibrosis
in the esophagus, which leads to the formation and progression of
disease states in esophageal tissue (25). In addition, markers for oxidative
stress are overexpressed in patients with GERD, which indicates
that increased ROS may be primarily responsible for the development
of GERD (34). ROS also leads to
esophageal fibrosis by increasing the expression of TGF-β1, which
enhances the synthesis of esophageal collagen and suppresses the
degradation of collagen in the GERD model (35). In the present study, chronic restrain
stress upregulated subunits of NOX, a major source of ROS and
downregulated antioxidant proteins in the esophagus.
In the current study, direct measurement of ROS was
not performed and this may be considered a limitation associated
with the study, which will need to be addressed in future work. In
a previous study, chronic restraint stress markedly induced the
accumulations of ROS in adipose (18) and colon tissue (21). The present study examined the
expression levels of ROS markers including, Nox4 and MDA. The
results demonstrated that two weeks of chronic restraint stress
significantly increased the expression of Nox4 and MDA in the
mucosal and epithelial layers of the esophagus. In addition, mRNA
and plasma levels of Nox4 and MDA were significantly increased in
the esophageal tissue of mice in the chronic restraint stress group
compared with the control group. Taken together, these results
indicate that chronic stress significantly increased ROS production
in the esophagus of mice.
The Nrf-2/Keap-1 signaling pathway provides cells
with a defense mechanism against oxidative stress by regulating the
expression of enzymes that serve key roles in the anti-oxidative
stress response and detoxification (36). Esophageal hyperkeratosis in Keap-1
knockout mice was due to activation of peroxisome
proliferator-activated receptor-β/Δ and the PI3K/Akt pathway
(37). Chen et al (38) demonstrated that Nrf-2 deficiency
impairs the barrier function of mouse esophageal epithelium by
disrupting the expression of tight junction proteins. Furthermore,
as a downstream gene of Nrf-2, HO-1 prevents gastroesophageal
reflux-induced esophageal barrier dysfunction by suppressing
oxidative stress in mouse models of GERD (38). In summary, the present study
demonstrated that chronic stress reduced the esophageal expression
of several anti-oxidative proteins including, Keap-1, phopho-Nrf-2,
Nrf-2 and HO-1 in mice.
TGF-β1/SMAD-3 signaling has been recognized as a
common pathway involved in several fibrotic diseases (39). TGF-β1 overexpression can introduce
several negative impacts, which include promoting direct
transcription of pro-fibrotic factors, such as collagen type I, and
the contraction of cultured esophageal smooth muscle cells
(40). Cells secrete TGF-β1, which
binds to TGF-β1 cell surface receptors (TGF-βRI and TGF-βRII) on
fibroblasts. These receptors can activate and translocate
intracellular SMADs (a family of transcription factors that mediate
TGF-β1 signals) to the nucleus, where they regulate transcription
of collagen genes, which contributes to fibrosis (26). The phosphorylation of the
receptor-dependent SMAD2/3, along with SMAD4, creates a complex,
which can be translocated from the cytoplasm to the nucleus to
regulate collagen gene transcription (28).
Previous studies have demonstrated several types of
SMAD-3-dependent collagen gene promoters, which are activated by
TGF-β1 (41,42). Dominant-negative SMAD-3 expression
vectors specifically inhibited the activation of these promoters
(43). Cho et al (44) examined the role of SMAD-3 in a mouse
model of egg-induced eosinophilic esophagitis and revealed that
SMAD-3-deficient mice esophageal fibroblasts could not respond to
TGF-β1 to regulate the expression of collagen genes, thereby
reducing esophageal fibrosis. However, in the current study,
chronic stress increased the esophageal expression of fibrotic
genes in mice.
In conclusion, two weeks of chronic restraint stress
induced esophageal fibrosis in a murine model with enhanced
oxidative stress and reduced the anti-oxidative system.
Acknowledgements
The authors would like to thank Dr Kyosuke Takeshita
for carefully reading and editing to the manuscript.
Funding
The current study was supported by a grant from the
Xinjiang Uygur Autonomous Region Natural Science Foundation Program
(grant no. 2018D01C134).
Availability of data and materials
All data that generated or analyzed during this
study are included in this published article.
Authors' contribution
MY and KA designed the current study, performed the
experiments and prepared the manuscript. WW, AiA, YL, AzA, AlA, MA,
WZ, ZC and AM performed the experiments, collected and analyzed the
data, and prepared the manuscript. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
All animal experiments were approved by the Animal
Care and Use Committee of the People's Hospital of Xinjiang Uygur
Autonomous Region (Urumqi, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Godoy LD, Rossignoli MT, Delfino-Pereira
P, Garcia-Cairasco N and de Lima Umeoka EH: A comprehensive
overview on stress neurobiology: Basic concepts and clinical
implications. Front Behav Neurosci. 12:1272018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu YZ, Wang YX and Jiang CL:
Inflammation: The common pathway of stress-related diseases. Front
Hum Neurosci. 11:3162017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lucca G, Comim CM, Valvassori SS, Reus GZ,
Vuolo F, Petronilho F, Dal-Pizzol F, Gavioli EC and Quevedo J:
Effects of chronic mild stress on the oxidative parameters in the
rat brain. Neurochem Int. 54:358–362. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tomanek L: Proteomic responses to
environmentally induced oxidative stress. J Exp Biol.
218:1867–1879. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gutierrez J, Ballinger SW, Darley-Usmar VM
and Landar A: Free radicals, mitochondria, and oxidized lipids: The
emerging role in signal transduction in vascular cells. Circ Res.
99:924–932. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zafir A and Banu N: Modulation of in vivo
oxidative status by exogenous corticosterone and restraint stress
in rats. Stress. 12:167–177. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gumuslu S, Sarikçioğlu SB, Sahin E,
Yargiçoğlu P and Ağar A: Influences of different stress models on
the antioxidant status and lipid peroxidation in rat erythrocytes.
Free Radic Res. 36:1277–1282. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sahin E and Gümüşlü S: Immobilization
stress in rat tissues: Alterations in protein oxidation, lipid
peroxidation and antioxidant defense system. Comp Biochem Physiol C
Toxicol Pharmacol. 144:342–347. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li-Kim-Moy JP, Tobias V, Day AS, Leach S
and Lemberg DA: Esophageal subepithelial fibrosis and hyalinization
are features of eosinophilic esophagitis. J Pediatr Gastroenterol
Nutr. 52:147–153. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Linares V, Sánchez DJ, Bellés M, Albina L,
Gómez M and Domingo JL: Pro-oxidant effects in the brain of rats
concurrently exposed to uranium and stress. Toxicology. 236:82–91.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bedard K and Krause KH: The NOX family of
ROS-generating NADPH oxidases: Physiology and pathophysiology.
Physiol Rev. 87:245–313. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Babalola O, Mamalis A, Lev-tov H and
Jagdeo J: NADPH oxidase enzymes in skin fibrosis: Molecular targets
and therapeutic agents. Arch Dermatol Res. 306:313–330. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hecker L, Cheng J and Thannickal VJ:
Targeting NOX enzymes in pulmonary fibrosis. Cell Mol Life Sci.
69:2365–2371. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
De Minicis S and Brenner DA: NOX in liver
fibrosis. Arch Biochem Biophys. 462:266–272. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pennathur S, Hecker L and Thannickal VJ:
Oxidative Stress and Cardiovascular Fibrosis. Humana Press; pp.
425–441. 2010
|
|
16
|
Holterman CE, Read NC and Kennedy CR: Nox
and renal disease. Clin Sci (Lond). 128:465–481. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yisireyili M, Hayashi M, Wu H, Uchida Y,
Yamamoto K, Kikuchi R, Shoaib Hamrah M, Nakayama T, Wu Cheng X,
Matsushita T, et al: Xanthine oxidase inhibition by febuxostat
attenuates stress-induced hyperuricemia, glucose dysmetabolism and
prothrombotic state in mice. Sci Rep. 7:12662017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yisireyili M, Takeshita K, Hayashi M, Wu
H, Uchida Y, Yamamoto K, Kikuchi R, Hao CN, Nakayama T, Cheng XW,
et al: Dipeptidyl peptidase-IV inhibitor alogliptin improves
stress-induced insulin resistance and prothrombotic state in a
murine model. Psychoneuroendocrinology. 73:186–195. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li Q, Kong L, Zhang S, Zhong Z, Liu X,
Wang J and Kang J: A novel external esophageal perfusion model for
reflux-associated respiratory symptoms. Pathobiology. 77:163–168.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yisireyili M, Uchida Y, Yamamoto K,
Nakayama T, Cheng XW, Matsushita T, Nakamura S, Murohara T and
Takeshita K: Angiotensin receptor blocker irbesartan reduces
stress-induced intestinal inflammation via AT1a signaling and
ACE2-dependent mechanism in mice. Brain Behav Immun. 69:167–179.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wakabayashi N, Itoh K, Wakabayashi J,
Motohashi H, Noda S, Takahashi S, Imakado S, Kotsuji T, Otsuka F,
Roop DR, et al: Keap1-null mutation leads to postnatal lethality
due to constitutive Nrf2 activation. Nat Genet. 35:238–245. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Torihata Y, Asanuma K, Iijima K, Mikami T,
Hamada S, Asano N, Koike T, Imatani A, Masamune A and Shimosegawa
T: Estrogen-dependent Nrf2 expression protects against
reflux-induced esophagitis. Dig Dis Sci. 63:345–355. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Peng D, Belkhiri A, Hu T, Chaturvedi R,
Asim M, Wilson KT, Zaika A and El-Rifai W: Glutathione peroxidase 7
protects against oxidative DNA damage in oesophageal cells. Gut.
61:1250–1260. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Oh TY, Lee JS, Ahn BO, Cho H, Kim WB, Kim
YB, Surh YJ, Cho SW and Hahm KB: Oxidative damages are critical in
pathogenesis of reflux esophagitis: Implication of antioxidantsin
its treatment. Free Radic Biol Med. 30:905–915. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Aceves SS: Remodeling and fibrosis in
chronic eosinophil inflammation. Dig Dis. 32:15–21. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wynn TA and Ramalingam TR: Mechanisms of
fibrosis: Therapeutic Translation for fibrotic disease. Nat Med.
18:1028–1040. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sziksz E, Pap D, Lippai R, Béres NJ,
Fekete A, Szabó AJ and Vannay Á: Fibrosis related inflammatory
mediators: Role of the IL-10 cytokine family. Mediators Inflamm.
2015:7646412015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Miller MW and Sadeh N: Traumatic stress,
oxidative stress and post-traumatic stress disorder:
Neurodegeneration and the accelerated-aging hypothesis. Mol
Psychiatry. 19:1156–1162. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Frangogiannis NG: Fibroblast-extracellular
matrix interactions in tissue fibrosis. Curr Pathobiol Rep.
4:11–18. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hosper NA, van den Berg PP, de Rond S,
Popa ER, Wilmer MJ, Masereeuw R and Bank RA:
Epithelial-to-mesenchymal transition in fibrosis: Collagen type I
expression is highly upregulated after EMT, but does not contribute
to collagen deposition. Exp Cell Res. 319:3000–3009. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hinz B, McCulloch CA and Coelho NM:
Mechanical regulation of myofibroblast phenoconversion and collagen
contraction. Exp Cell Res. 379:119–128. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Song JH, Han YM, Kim WH, Park JM, Jeong M,
Go EJ, Hong SP and Hahm KB: Oxidative stress from reflux
esophagitis to esophageal cancer; the alleviation with
antioxidants. Free Radic Res. 50:1071–1079. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Dandekar A, Mendez R and Zhang K: Cross
talk between ER stress, oxidative stress, and inflammation in
health and disease. Methods Mol Biol. 1292:205–214. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Maqbool A and Pauwels A: Cystic fibrosis
and gastroesophageal reflux disease. J Cyst Fibros 2 (16 Suppl).
S2–S13. 2017. View Article : Google Scholar
|
|
36
|
Jaramillo MC and Zhang DD: The emerging
role of the Nrf2-Keap1 signaling pathway in cancer. Genes Dev.
27:2179–2191. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chen H, Fang Y, Li W, Orlando RC, Shaheen
N and Chen XL: NFkB and Nrf2 in esophageal epithelial barrier
function. Tissue Barriers. 1:e274632013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen H, Hu Y, Fang Y, Djukic Z, Yamamoto
M, Shaheen NJ, Orlando RC and Chen X: Nrf2 deficiency impairs the
barrier function of mouse oesophageal epithelium. Gut. 63:711–719.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Balta C, Herman H, Boldura OM, Gasca I,
Rosu M, Ardelean A and Hermenean A: Chrysin attenuates liver
fibrosis and hepatic stellate cell activation through TGF-β/Smad
signalingpathway. Chem Biol Interact. 240:94–101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kuppan P, Sethuraman S and Krishnan UM: In
vitro co-culture of epithelial cells and smooth muscle cells on
aligned nanofibrous scaffolds. Mater Sci Eng C Mater Biol Appl.
81:191–205. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ghosh AK, Bhattacharyya S and Varga J: The
tumor suppressor p53 abrogates Smad-dependent collagen gene
induction in mesenchymal cells. J Biol Chem. 279:47455–47463. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ito S, Ogawa K, Takeuchi K, Takagi M,
Yoshida M, Hirokawa T, Hirayama S, Shin-Ya K, Shimada I, Doi T, et
al: A small-molecule compound inhibits a collagen-specific
molecular chaperone and could represent a potential remedy for
fibrosis. J Biol Chem. 292:20076–20085. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hu Z, Robbins JS, Pister A, Zafar MB,
Zhang ZW, Gupta J, Lee KJ, Newman K, Yun CO, Guise T and Seth P: A
modified hTERT promoter-directed oncolytic adenovirus replication
with concurrent inhibition of TGFbeta signaling for breast cancer
therapy. Cancer Gene Ther. 17:235–243. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cho JY, Doshi A, Rosenthal P, Beppu A,
Miller M, Aceves S and Broide D: Smad3-deficient mice have reduced
esophageal fibrosis and angiogenesis in model of egg-induced
eosinophilic esophagitis. J Pediatr Gastroenterol Nutr. 59:10–16.
2014. View Article : Google Scholar : PubMed/NCBI
|