Introduction
Herpes zoster is caused by reactivation of latent
varicella-zoster virus (VZV) in ganglia and usually occurs decades
after the primary infection. It is mainly characterized by
vesicular rash distributed in unilateral skin and is often
accompanied with pain (1,2). Herpes zoster is common in individuals
aged over 50 years because of their declined immune function.
Approximately 90% of adults are infected with VZV and are at risk
of developing herpes zoster. The overall incidence rate of herpes
zoster is 2.0–4.6 cases/1,000 person/year, and that in patients
aged 80 or above is 10.0–12.8 cases/1,000 person/year. Moreover,
the incidence rate of post herpetic neuralgia increases and the
symptoms are more severe with age (3–7).
Currently, corticosteroid-containing regimens are
the main treatments for herpes zoster. Corticosteroids, with strong
anti-inflammatory effects, may effectively improve the levels of
inflammatory factors such as IL-6 and IL-10, reduce nerve injury,
and promote the regression of blisters as well as improve nerve
pain (8). However, the dosage has
been controversial among clinical workers in recent years. Levin
(9) reported that corticosteroids
caused immunosuppression, thus increasing or even aggravating the
infection of patients with herpes zoster. A study also questioned
that corticosteroids had no capacity to prevent post-herpetic
neuralgia in patients with acute infective herpes zoster (10). However, in the study by Harpaz et
al (1), it was found that the
use of low to middle doses of short-acting systemic corticosteroids
did not cause immunosuppression. A study of the respiratory tract
also found that the use of high-dose glucocorticoid combined with
cytochrome P450 enzyme inhibitor was not an important risk factor
for the occurrence of herpes zoster, suggesting that
glucocorticoid-induced immunosuppression was not severe (11).
In order to solve the above contradictions, a
prospective analysis was carried out to explore the therapeutic
effect and safety of different doses of prednisone in herpes
zoster, so as to provide reference for clinical treatment.
Patients and methods
Study objects
A total of 125 patients (aged 40–70 years) with
acute infective herpes zoster who were admitted to Daqing Oilfield
General Hospital from June 2016 to September 2017 were selected and
divided into 3 groups according to different treatment methods:
low-dose (n=44), middle-dose (n=42) and high-dose (n=39) groups.
Inclusion criteria were: Patients meeting the diagnostic criteria
for herpes zoster as per the Expert Consensus on Diagnosis and
Treatment of Neuropathic Pain, established by the International
Association for the Study of Pain (IASP) in 2008; patients with
complete clinical data; patients with no past or present tumors;
patients with no clinical features of motor nerve involvement;
patients with no corticosteroid use within 3 months. Exclusion
criteria were: patients with: history of drug allergy and abnormal
bleeding; hypertension, diabetes, systemic inflammatory response,
autoimmune diseases; severe defects of liver, kidney and heart lung
functions; pregnant or nursing women; and patients with mental and
cognitive dysfunctions. This study was approved by the Ethics
Committee of the Daqing Oilfield General Hospital, and the patients
and their families signed informed consent forms.
Methods
The patients were treated with famciclovir at a dose
of 0.25 g/time, three times a day. In addition, according to
medication guidelines for glucocorticoids and instructions for
prednisone, 10–40 mg/day is generally recommended as the dose of
prednisone, and it can be increased to 60 mg/day, if necessary.
Therefore, we divided the patients into three groups: low-, middle-
and high-dose groups. The patients in the low-dose group were given
prednisone 15 mg/day, orally, three times a day, and the patients
in the the middle-dose group were given prednisone 25 mg/day, 5
times a day, whereas those in the high-dose group were given
prednisone 40 mg/day, 4 times a day, for two weeks.
Outcome measures
The therapeutic effect, visual analogue scale (VAS)
pain score, pain relieving and disappearing time, herpes stopping
and disappearing time, incrustation and decrustation time, and
incidence of adverse reactions in the three groups were recorded.
The changes of IL-6 and IL-10 levels in the peripheral blood of
patients before and after treatment were detected by enzyme-linked
immunosorbent assay (ELISA) in order to analyze their relationship
with pain degree and the time of symptom remission and
subsidence.
Efficacy evaluation criteria
Referring to Clinical Dermatology (12): cure: Herpes scab shedding area was
>90%, and the pain did not affect life and sleep quality;
significant effective: Herpes scab shedding area was ≥60% -
<90%, VAS pain score decreased by >50% compared with that
before treatment; effective: Herpes scab shedding area was ≥30% -
<60%, and the pain score of VAS decreased by >25% compared
with that before treatment; ineffective: Herpes scab shedding area
was <30%, VAS pain score had no obvious change or even increased
compared with that before treatment. The total effective rate of
treatment was calculated as: (cure + significant effective +
effective)/total number ×100%.
ELISA
Both IL-6 and IL-10 were detected by ELISA, and the
detection kits were purchased from Wuhan Elabscience Biotechnology
Co., Ltd., with the item numbers of E-EL-H0102c and E-EL-H0103c,
respectively. Refer to the kit instructions for specific detection
steps.
Statistical analysis
SPSS 19.0 (Asia Analytics Formerly SPSS) was used to
analyze the data of this study. Enumeration data were expressed as
rates, and the comparison of rates was carried out using the
χ2 test. Measurement data were expressed as mean ± SD.
Comparisons of the three groups were carried out using analysis of
variance (ANOVA) and the least significant difference (LSD) test
was used for pairwise comparison afterwards. Comparisons before and
after treatment in the group employed the paired t-test. Pearson
correlation analysis was used to analyze the correlation between
the levels of IL-6 and IL-10 before and after treatment and VAS
pain score, as well as the difference between IL-6 and IL-10 before
and after treatment and the pain relieving and disappearing time,
herpes stopping and disappearing time, the time of incrustation and
decrustation. P<0.05 indicated statistical significance.
Results
General information
The study comprised 44 patients in the low-dose
group, including 25 males and 19 females, with an age of 52.25±8.36
years; 42 patients in the middle-dose group, including 23 males and
19 females, with an age of 53.71±7.83 years; 39 patients in the
high-dose group, including 21 males and 18 females, with an age of
50.62±8.04 years. Therefore, there was no statistical difference in
sex ratio and age among the three groups (P>0.05). Moreover,
there were no statistical differences in the course of disease,
herpes distribution, and other basic data among the three groups
(P>0.05) (Table I).
 | Table I.General information. |
Table I.
General information.
| Characteristics | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | χ2/t
value | P-value |
|---|
| Sex [n (%)] |
|
|
| 0.079 | 0.961 |
| Male | 25 (56.82) | 23 (54.76) | 21 (53.85) |
|
|
|
Female | 19 (43.18) | 19 (45.24) | 18 (46.15) |
|
|
| Age (years) | 52.25±8.36 | 53.71±7.83 | 50.62±8.04 | 1.477 | 0.232 |
| Course of disease
(days) | 3.83±1.91 | 3.95±2.32 | 4.02±2.11 | 0.086 | 0.918 |
| Distribution [n
(%)] |
|
|
| 2.907 | 0.940 |
|
Limbs | 4 (9.09) | 3 (7.14) | 4 (10.26) |
|
|
| Waist and
abdomen | 10 (22.73) | 9 (21.43) | 8 (20.51) |
|
|
| Chest and
back | 19 (43.18) | 17 (40.48) | 15 (38.46) |
|
|
| Head and
face | 10 (22.73) | 13 (30.95) | 12 (30.77) |
|
|
|
Perineum | 1 (2.27) | 0 (0.00) | 0 (0.00) |
|
|
| Vaccination rate | 4 (9.09) | 5 (11.90) | 4 (10.26) | 0.184 | 0.912 |
| Immune function [n
(%)] |
|
|
| 0.709 | 0.701 |
|
Normal | 30 (68.18) | 28 (66.67) | 27 (69.23) |
|
|
|
Suppressed | 14 (31.82) | 14 (33.33) | 12 (30.77) |
|
|
Clinical efficacy
The cure rate, significant effective rate, effective
rate and ineffective rate in the three groups had no statistical
difference (P>0.05), neither the total effective rate
(P>0.05) (Table II).
 | Table II.Clinical efficacy. |
Table II.
Clinical efficacy.
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | χ2/t
value | P-value |
|---|
| Cure | 10 (22.73) | 8 (19.05) | 8 (20.51) | 0.180 | 0.914 |
| Significant
effective rate | 12 (27.27) | 13 (30.95) | 10 (25.64) | 0.301 | 0.860 |
| Effective rate | 15 (34.09) | 12 (28.57) | 12 (30.77) | 0.310 | 0.857 |
| Ineffective
rate | 7 (15.91) | 9 (21.43) | 9 (23.08) | 0.745 | 0.689 |
| Total effective
rate | 37 (84.09) | 33 (78.57) | 30 (76.92) | 0.745 | 0.689 |
VAS pain score
There were no significant differences in VAS pain
scores among the three groups before treatment (P>0.05), while
the scores were all decreased after treatment (P<0.05).
Moreover, the score in the middle-dose group was lower than that in
the low-dose group and the high-dose group (P<0.05), and there
was no significant difference between the low-dose group and the
high-dose group (P>0.05) (Table
III).
 | Table III.VAS pain score. |
Table III.
VAS pain score.
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | F value | P-value |
|---|
| Before
treatment | 7.16±1.25 | 7.33±1.33 | 7.66±1.35 | 1.543 |
0.217 |
| After
treatment |
3.42±1.42a | 2.26±1.15 |
3.38±1.52a | 9.695 | <0.001 |
| t value | 12.565 | 17.376 | 12.896 |
|
|
| P-value | <0.001 | <0.001 | <0.001 |
|
|
Pain relieving and disappearing
time
The pain relieving and disappearing time in the
middle-dose group were shorter than those in the low-dose group and
the high-dose group (P<0.05). There were no statistical
differences in pain relieving time between the low-dose group and
the high-dose group (P>0.05), while the pain disappearing time
in the high-dose group was shorter than that in the low-dose group
(P<0.05) (Table IV).
 | Table IV.Pain relieving and disappearing time
(days). |
Table IV.
Pain relieving and disappearing time
(days).
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | F value | P-value |
|---|
| Pain relieving
time |
3.1±1.5a | 2.2±1.1 |
2.8±1.3a | 5.193 | 0.007 |
| Pain disappearing
time |
13.7±3.8a | 9.32±3.2 |
11.4±2.7a,b | 19.286 | <0.001 |
Herpes stopping and disappearing
time
The herpes stopping and disappearing time in the
middle-dose group were shorter than those in the low-dose group and
the high-dose group (P<0.05), and there were no statistical
differences between the low-dose group and the high-dose group
(P>0.05) (Table V).
 | Table V.Herpes stopping and disappearing time
(days). |
Table V.
Herpes stopping and disappearing time
(days).
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | F value | P-value |
|---|
| Herpes stopping
time |
3.6±1.1a | 2.9±1.7 |
3.3±1.1a | 3.519 | 0.033 |
| Herpes disappearing
time |
13.4±1.3a | 10.5±2.1 |
14.0±1.8a | 49.832 | <0.001 |
Incrustation and decrustation
time
The incrustation time in the middle-dose group was
shorter than that in the low-dose group and high-dose group
(P<0.05), and there were no statistical differences between the
low-dose group and the high-dose group (P>0.05). The
decrustation time in the low-dose group and the middle-dose group
was shorter than that in the high-dose group (P<0.05), and there
were no statistical differences between the low-dose group and the
middle-dose group (P>0.05) (Table
VI).
 | Table VI.Incrustation and decrustation time
(days). |
Table VI.
Incrustation and decrustation time
(days).
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | F value | P-value |
|---|
| Incrustation
time |
4.8±0.7a | 3.2±1.1 |
4.2±0.8a | 35.889 | <0.001 |
| Decrustation
time | 9.6±5.7 | 9.7±2.9 |
12.4±3.4a,b | 5.686 | 0.004 |
Incidence of post herpetic
neuralgia
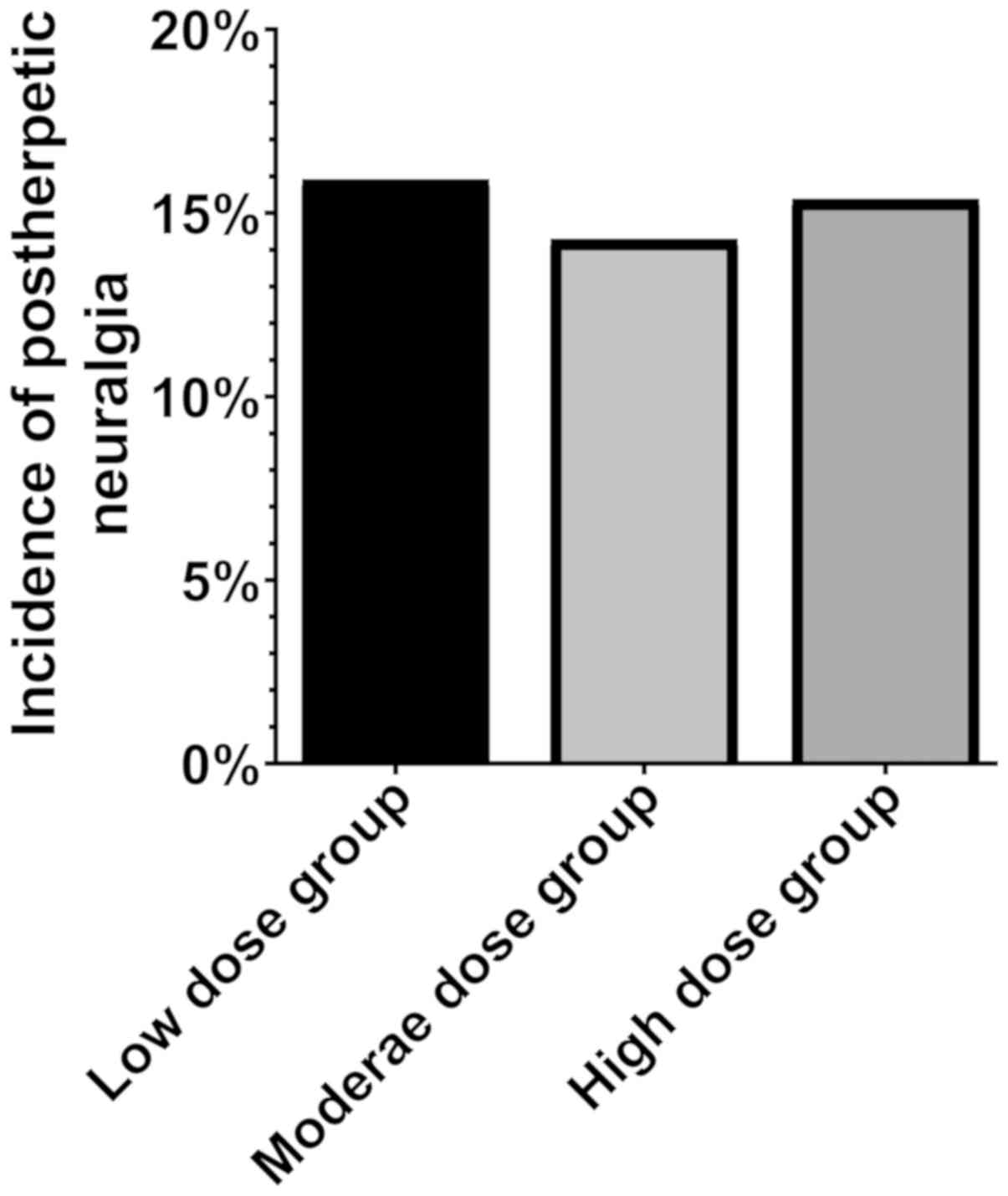
The incidence of post herpetic neuralgia in the
low-, middle- and high-dose groups was 15.91% (7 cases), 14.29% (6
cases), 15.38% (6 cases), respectively. Therefore, there were no
statistical differences in the incidence of post-herpetic neuralgia
among the three groups (P>0.05) (Fig.
1).
Incidence of adverse reactions
There were no statistical differences in the total
incidence of adverse reactions among the three groups (P>0.05).
However, the incidence of mild edema in the high-dose group was
higher than that in the low-dose group and the middle-dose group
(P<0.05), and there were no statistical differences in the
incidence of other adverse reactions (P>0.05) (Table VII).
 | Table VII.Incidence of adverse reactions [n
(%)]. |
Table VII.
Incidence of adverse reactions [n
(%)].
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | χ2
value | P-value |
|---|
| Dizziness | 1 (2.27) | 1 (2.38) | 1 (2.56) | 0.008 | 0.996 |
| Sleepiness | 0 (0.00) | 1 (2.38) | 2 (5.13) | 2.321 | 0.313 |
| Mild edema | 0 (0.00) | 0 (0.00) | 3
(7.69)a,b | 6.778 | 0.034 |
| Stomach
discomfort | 1 (2.27) | 2 (4.76) | 1 (2.56) | 0.504 | 0.777 |
| Total adverse
reactions | 2 (4.55) | 4 (9.52) | 7 (17.95) | 4.038 | 0.133 |
Changes of IL-6 and IL-10 levels
The levels of IL-6 and IL-10 showed no statistical
difference in the three groups before treatment (P>0.05). After
treatment, the level of IL-6 decreased in the three groups
(P<0.05), the middle-dose group and the high-dose group were
lower than the low-dose group (P<0.05), and the high-dose group
was lower than middle-dose group (P<0.05). By contrast, the
level of IL-10 increased in the three groups (P<0.05), the
middle-dose group and the high-dose group were higher than low-dose
group (P<0.05), and the high-dose group was higher than the
middle-dose group (P<0.05) (Table
VIII).
 | Table VIII.Changes of IL-6 and IL-10 levels. |
Table VIII.
Changes of IL-6 and IL-10 levels.
| Variable | Low-dose group
(n=44) | Middle-dose group
(n=42) | High-dose group
(n=39) | F value | P-value |
|---|
| IL-6 (pg/ml) |
| Before
treatment | 482.4±36.5 | 483.8±37.2 | 486.7±38.8 | 0.140 | 0.870 |
| After
treatment | 308.7±28.3 |
226.3±21.8b |
118.7±21.7a,b | 635.062 | <0.001 |
| t value | 26.807 | 41.076 | 55.450 |
|
|
| P-value | <0.001 | <0.001 | <0.001 |
|
|
| IL-10 (µg/ml) |
| Before
treatment | 124.6±11.8 | 126.5±10.9 | 123.4±14.5 | 0.644 | 0.527 |
| After
treatment | 142.5±21.2 |
168.9±20.8b |
211.6±24.5a,b | 101.621 | <0.001 |
| t value | 4.262 | 9.987 | 17.961 |
|
|
| P-value | <0.001 | <0.001 | <0.001 |
|
|
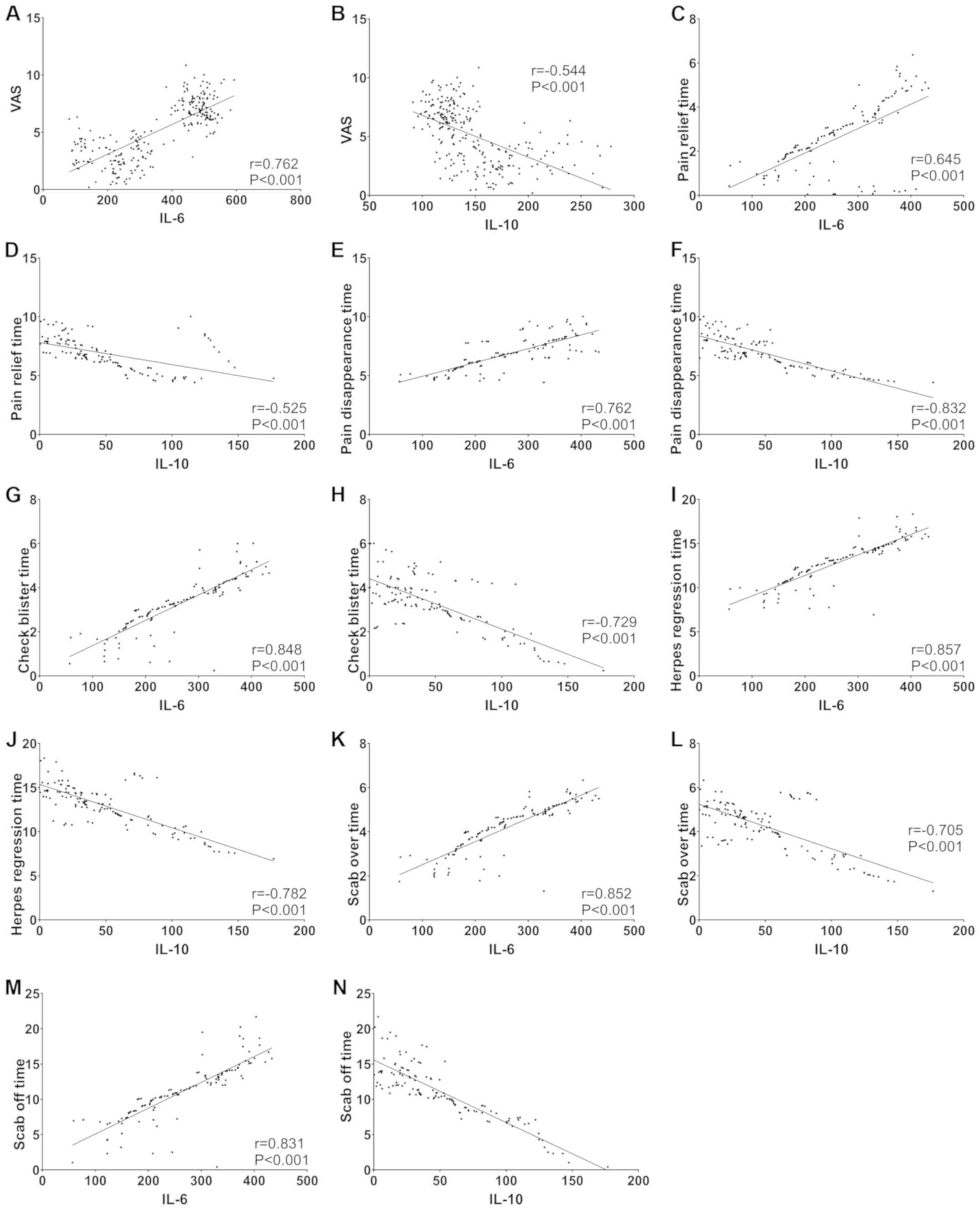
Correlation analysis
Pearson correlation analysis showed that IL-6 was
positively correlated with VAS pain score, pain relieving and
disappearing time, herpes stopping and disappearing time, and
incrustation and decrustation time (all P<0.05), while IL-10 was
negatively correlated with the above indicators (all P<0.05)
(Fig. 2).
Discussion
At present, there are few studies on the therapeutic
effect and safety of different doses of corticosteroids in herpes
zoster. However, previous studies found that the use of
corticosteroids increased the risk of herpes zoster, with a risk
ratio of 1.75 (13,14). Moreover, the risk increased with the
increase of the dose, especially for patients whose dose exceeded
10 mg/day (13,14). Therefore, it is speculated that the
immunosuppression of corticosteroids may affect the therapeutic
effect and safety of patients with herpes zoster.
This study established three groups of patients
receiving different doses of prednisone. We grouped the subjects by
dose, and found that there were more patients treated with doses of
15, 25 and 40 mg/time. Relevant literature was also reviewed. The
administered dose of prednisone was 30 mg/time in the study of
Anderson and Janoff (15). By
contrast, in the study of Clemmensen and Andersen (16), the dose was 60 mg/time initially, 30
mg/time after one week and 15 mg/time in the third week. Whitley
et al (17) set the initial
dose of prednisone at 45 mg/time, which was reduced to 30 mg/time
after one week, and was discontinued in the third week. Therefore,
wedivided the patients into the low-dose, middle-dose and high-dose
groups according to relevant statistics. The results of the present
study showed that there were no significant differences in the
therapeutic efficacy among the three groups. However, the VAS score
demonstrated that, the higher the dose of prednisone, the lower the
VAS pain score of the patients after two weeks of treatment, and
the greater the improvement in pain. Interestingly, we found that
among the three doses, the middle-dose prednisone had the shortest
pain relieving and disappearing time, herpes stopping and
disappearing time, and incrustation and decrustation time. Only
pain disappearing time and decrustation time in the high-dose group
were shorter than those in the low-dose group. The high-dose group
had the highest incidence of mild edema, while there was no mild
edema in the low-dose group and the middle-dose group. Moreover,
there was no difference for the incidence of post-herpetic
neuralgia in the three groups of patients with herpes zoster, which
was similar to the results reported in previous studies that
corticosteroids had no ability to prevent the occurrence of
post-herpetic neuralgia (18,19).
From the above results, middle-dose prednisone can
achieve the best therapeutic effect and higher safety in treating
herpes zoster. In studies on different doses of corticosteroid
therapy, Rygård et al (20)
revealed that small-dose corticosteroid effectively reduced the
shock duration, mechanical ventilation and ICU stay in treating
septic shock. Despite increased adverse reactions, it did not
affect the short-term and long-term mortality of patients. Sugimoto
et al (21) reported that for
patients who need lung transplantation after hematopoietic stem
cell transplantation, preoperative low-dose corticosteroid was more
effective than high-dose corticosteroid in reducing postoperative
complications. Izquierdo and Cosio (22) demonstrated a similar point of view;
that for patients with chronic obstructive pulmonary diseases,
low-dose corticosteroid showed significantly higher efficacy and
lower incidence of corticosteroid-related pneumonia than high-dose
corticosteroid. The above studies reported that it was beneficial
to reduce the use of corticosteroids in the treatment of the
diseases. It is well known that inhaled corticosteroids are more
effective than oral ones. In the study by Daley-Yates (23), it was reported that the binding
affinity of glucocorticoid receptor was exponentially related to
the dose of inhaled glucocorticoids, suggesting that low doses of
inhaled corticosteroids also provided the same effective molecules
to achieve therapeutic effects similar to those at high doses, and
reduced systemic exposure and improved the therapeutic index.
However, this treatment is not advocated in the treatment of herpes
zoster, but a historical equivalent dose method.
Corticosteroids are widely used for their powerful
anti-inflammatory effects (24).
Inflammation and changes in cytokines accompany the process of
herpes zoster (25), and the
abnormal secretions of inflammatory cytokines and immunoglobulins
caused by humoral immunity and cellular immune response disorders,
such as IL-6 and IL-10, are related to the occurrence of post
herpetic neuralgia in acute herpes zoster (26). Our analysis revealed that after
treatment, IL-6 and IL-10 in the three groups of patients were
effectively improved, and the improvement degree was
dose-dependent. This study analyzed the relationship between IL-6,
IL-10 and the pain degree and the duration of symptom improvement,
and the results showed that IL-6 was positively correlated with VAS
pain score, pain relieving and disappearing time, herpes stopping
and disappearing time, incrustation and decrustation time, while
IL-10 was negatively correlated with them. Huang et al
(27) reported that treadmill
exercise and therapeutic ultrasound controlled pain associated with
nerve injury in rats by improving IL-6 and IL-10. In addition,
Leung et al (28) also
believed that exogenous administration of IL-10 reduced
hyperalgesia in mice. These studies verified the accuracy of some
of our results, which increased the credibility of our other
results.
There are some deficiencies in this study. Firstly,
prospective analysis was used in this study, so there were
inevitably some biases in the inclusion of patients, as well as in
the treatment methods. Secondly, the subjects included had a small
size and narrow scope, which may increase some accidental errors.
Therefore, multi-center clinical experiments should be conducted to
further confirm our results. The long-term efficacy of different
doses of prednisone in the treatment of herpes zoster needs to be
further followed up. In this study, the number of patients in each
group exceeded 30, so the sample size requirement was statistically
met. For the determination of adverse reactions, some adverse
reactions may be caused by fanciclovir. However, the statistical
difference in the incidence of adverse reactions was considered to
be caused by prednisone, because only the doses of prednisone were
different in this study.
In conclusion, middle-dose prednisone has similar
short-term efficacy to high-dose prednisone in the treatment of
herpes zoster, but with less complication and higher safety. IL-6
and IL-10 are closely related to the pain degree and the time of
symptom remission and subsidence, which may provide a reference for
clinical evaluation of the therapeutic effect of patients with
herpes zoster.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
LP conceived the study and wrote the manuscript. BD
was responsible for ELISA. LS analyzed and interpreted the
patients' data. YZ and XZ helped with statistical analysis. All the
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Daqing Oilfield General Hospital. Patients who participated in this
research, signed informed consent and had complete clinical data.
Signed informed consents were obtained from the patients or the
guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Harpaz R, Ortega-Sanchez IR and Seward JF;
Advisory Committee on Immunization Practices (ACIP) Centers for
Disease Control Prevention (CDC), : Prevention of herpes zoster:
Recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 57:1–30; quiz CE2-CE4. 2008.PubMed/NCBI
|
|
2
|
Cohen JI: Clinical practice: Herpes
zoster. N Engl J Med. 369:255–263. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bonanni P, Breuer J, Gershon A, Gershon M,
Hryniewicz W, Papaevangelou V, Rentier B, Rümke H, Sadzot-Delvaux
C, Senterre J, et al: Varicella vaccination in Europe - taking the
practical approach. BMC Med. 7:26. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Johnson RW: Herpes zoster and postherpetic
neuralgia. Expert Rev Vaccines. 9 (Suppl):21–26. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gershon AA, Gershon MD, Breuer J, Levin
MJ, Oaklander AL and Griffiths PD: Advances in the understanding of
the pathogenesis and epidemiology of herpes zoster. J Clin Virol.
48 (Suppl 1):S2–S7. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Johnson BH, Palmer L, Gatwood J, Lenhart
G, Kawai K and Acosta CJ: Annual incidence rates of herpes zoster
among an immunocompetent population in the United States. BMC
Infect Dis. 15:502. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pinchinat S, Cebrián-Cuenca AM, Bricout H
and Johnson RW: Similar herpes zoster incidence across Europe:
Results from a systematic literature review. BMC Infect Dis.
13:170. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nordgaard-Lassen I, Dahlerup J F, Belard
E, Gerstoft J, Kjeldsen J, Kragballe K, Ravn P, Sørensen IJ, Theede
K and Tjellesen L: Danish Society for Gastroenterology. Guidelines
for screening, prophylaxis and critical information prior to
initiating anti-TNF-alpha treatment. Dan Med J.
59:C44802012.PubMed/NCBI
|
|
9
|
Levin MJ: Varicella-zoster virus and virus
DNA in the blood and oropharynx of people with latent or active
varicella-zoster virus infections. J Clin Virol. 61:487–495. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Han Y, Zhang J, Chen N, He L, Zhou M and
Zhu C: Corticosteroids for preventing postherpetic neuralgia.
Cochrane Database Syst Rev. 1:CD0055822013.
|
|
11
|
Ernst P, Dell'Aniello S, Mikaeloff Y and
Suissa S: Risk of herpes zoster in patients prescribed inhaled
corticosteroids: A cohort study. BMC Pulm Med. 11:592011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao B: Clinical Dermatology in China.
Jiangsu Science and Technology Press; pp. 613–618. 2010
|
|
13
|
Marra F, Lo E, Kalashnikov V and
Richardson K: Risk of herpes zoster in individuals on biologics,
DMARDS and/or corticosteroids for autoimmune diseases: A systematic
review and meta-analysis. Open Forum Infect Dis. 3:ofw2052016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Long MD, Martin C, Sandler RS and
Kappelman MD: Increased risk of herpes zoster among 108,604
patients with inflammatory bowel disease. Aliment Pharmacol Ther.
37:420–429. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Anderson DJ and Janoff EN: Herpes zoster
infection in a patient on methotrexate given prednisone to prevent
post-herpetic neuralgia. Ann Intern Med. 107:783. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Clemmensen OJ and Andersen KE: ACTH versus
prednisone and placebo in herpes zoster treatment. Clin Exp
Dermatol. 9:557–563. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Whitley RJ, Weiss H, Gnann JW Jr, Tyring
S, Mertz GJ, Pappas PG, Schleupner CJ, Hayden F, Wolf J and Soong
SJ; The National Institute of Allergy and Infectious Diseases
Collaborative Antiviral Study Group, : Acyclovir with and without
prednisone for the treatment of herpes zoster. A randomized,
placebo-controlled trial. Ann Intern Med. 125:376–383. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jeon YH: Herpes zoster and postherpetic
neuralgia: Practical consideration for prevention and treatment.
Korean J Pain. 28:177–184. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heatrice A II, Alavi M, Han PP and Enciso
R: Oral corticosteroid therapy for preventing postherpetic
neuralgia: A systematic review and meta-analysis. Open J Dent Oral
Med. 5:47–57. 2017.
|
|
20
|
Rygård SL, Butler E, Granholm A, Møller
MH, Cohen J, Finfer S, Perner A, Myburgh J, Venkatesh B and Delaney
A: Low-dose corticosteroids for adult patients with septic shock: A
systematic review with meta-analysis and trial sequential analysis.
Intensive Care Med. 44:1003–1016. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sugimoto S, Miyoshi K, Kurosaki T, Otani
S, Yamane M, Kobayashi M and Oto T: Favorable survival in lung
transplant recipients on preoperative low-dose, as compared to
high-dose corticosteroids, after hematopoietic stem cell
transplantation. Int J Hematol. 107:696–702. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Izquierdo JL and Cosio BG: The dose of
inhaled corticosteroids in patients with COPD: When less is better.
Int J Chron Obstruct Pulmon Dis. 13:3539–3547. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Daley-Yates PT: Inhaled corticosteroids:
Potency, dose equivalence and therapeutic index. Br J Clin
Pharmacol. 80:372–380. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Roubille C, Richer V, Starnino T, McCourt
C, McFarlane A, Fleming P, Siu S, Kraft J, Lynde C, Pope J, et al:
The effects of tumour necrosis factor inhibitors, methotrexate,
non-steroidal anti-inflammatory drugs and corticosteroids on
cardiovascular events in rheumatoid arthritis, psoriasis and
psoriatic arthritis: A systematic review and meta-analysis. Ann
Rheum Dis. 74:480–489. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yun H, Yang S, Chen L, Xie F, Winthrop K,
Baddley JW, Saag KG, Singh J and Curtis JR: Risk of herpes zoster
in autoimmune and inflammatory diseases: Implications for
vaccination. Arthritis Rheumatol. 68:2328–2337. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shi HJ and Cui ZQ: Correlation of serum
inflammatory cytokine and immunoglobulin content with post-herpetic
neuralgia in patients with acute herpes zoster. Hainan Yixueyuan
Xuebao. 23:97–100. 2017.(In Chinese).
|
|
27
|
Huang PC, Tsai KL, Chen YW, Lin HT and
Hung CH: Exercise combined with ultrasound attenuates neuropathic
pain in rats associated with downregulation of IL-6 and TNF-α, but
with upregulation of IL-10. Anesth Analg. 124:2038–2044. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Leung A, Gregory NS, Allen LAH and Sluka
KA: Regular physical activity prevents chronic pain by altering
resident muscle macrophage phenotype and increasing interleukin-10
in mice. Pain. 157:70–79. 2016. View Article : Google Scholar : PubMed/NCBI
|