Introduction
Liver cancer was estimated to be the sixth most
common cancer type and the fourth leading cause of
cancer-associated death worldwide in 2018, and it has been reported
that hepatocellular carcinoma (HCC) is the most common subtype,
accounting for 75–85% of all primary cases of liver cancer
(1). However, the underlying
molecular mechanisms of HCC development remain to be fully
elucidated, and the high incidence of metastasis and recurrence
frequently results in poor prognosis for patients with HCC, even
following curative therapy (2).
Therefore, it is imperative to identify novel diagnostic and
prognostic biomarkers and efficacious therapeutic targets.
Abnormalities in gene transcription and translation
serve important roles in the development and progression of HCC.
Developments in biotechnology, including genome sequencing and
bioinformatics, have facilitated the study of the tumor
transcriptome and proteome, which has advanced the current
understanding of the underlying molecular mechanisms of HCC. Genome
sequencing has indicated that the human genome is comprised of
<2% protein-coding genes and >90% of the genome is
transcribed as non-coding RNA (ncRNA) (3). Numerous classes of ncRNA have been
identified and demonstrated to be associated with cancer, including
long-ncRNAs (lncRNAs), microRNAs (miRNAs), small nucleolar RNAs and
PIWI-interacting RNAs (4–7), among which lncRNAs and miRNAs have been
most extensively studied. lncRNAs are >200 nucleotides long and
participate in multiple biological functions, including
epigenetics, nuclear import, alternative splicing, RNA decay and
translation (5). miRNAs are small
ncRNAs of 18–25 nucleotides in length that regulate the expression
of multiple mRNAs by reducing the stability and inhibiting the
translation of mRNAs at the post-transcriptional level (8). miRNAs restrain the expression of target
genes, whereas lncRNAs may competitively combine with miRNAs to
promote the expression of target genes; in this interaction, they
are commonly referred to as competing endogenous RNAs (ceRNAs)
(9). Although previous studies have
reported on the roles of lncRNAs and miRNAs in HCC (10–14), the
specific underlying mechanisms associated with the initiation and
progression of HCC remain to be fully elucidated (15).
In the present study, profiles of differentially
expressed lncRNAs between HCC tissues and adjacent normal tissues
were determined from high-throughput RNA sequencing data.
Bioinformatics and survival analysis using
integrated mining of data from The Cancer Genome Atlas (TCGA) was
performed (16). Simultaneously, the
regulatory mechanisms and signaling pathways in HCC were predicted
and their differential diagnostic performance was analyzed to
provide a potentially valuable reference for the early diagnosis,
effective treatment and prognosis of patients with HCC.
Materials and methods
RNA-sequencing (seq) data retrieval
and processing
The GSE94660 dataset was obtained from the Gene
Expression Omnibus (GEO), which contains the RNA sequencing data
from 21 pairs of tumor and non-neoplastic liver tissues from
patients with hepatitis B virus-associated HCC. The normalized gene
expression dataset GSE94660 was downloaded from the GEO database
(https://www.ncbi.nlm.nih.gov/geo/)
(17). To identify the
differentially expressed genes between tumor and non-tumor tissues,
threshold values of a fold change of |1.5| and P<0.05 were
used.
Expression level analysis
The expression of lncRNA and mRNA in patients were
analyzed using either Gene Expression Profiling Interactive
Analysis (GEPIA) (18) or from
expression data downloaded from TCGA (16). Expression data of lncRNA in normal
tissues were obtained from BioProject (accession no. PRJEB4337;
linc01093) (19).
Target prediction and network
establishment
To predict potential miRNAs that target candidate
lncRNAs, lncBase version 2 (http://carolina.imis.athena-innovation.gr/diana_tools/web/index.php?r=lncbasev2/index)
(20) was used with a prediction
score of ≥0.95 as the threshold value. The miRNAs identified were
further inputted into TargetScan (http://www.targetscan.org/vert_72/) (21) to identify their potential target
genes. Targets with a cumulative weighted context score ≤-0.6 that
interacted with a differentially expressed gene identified in the
GSE94660 dataset were selected to establish an interaction network
with candidate miRNAs and lncRNA using Cytoscape 3.40 (22).
Interactome identification and
functional enrichment analysis
Proteins that physically interacted with zinc finger
AN1-type containing 5 (ZFAND5) were identified from BioGRID
(https://thebiogrid.org/) (23), an interaction repository containing
1,623,645 protein and genetic interactions in a number of species.
Gene ontology (GO) (24) and
reactome pathway enrichment analyses of the ZFAND5 interactome were
performed using the Database for Annotation, Visualization and
Integrated Discovery (DAVID; http://david.abcc.ncifcrf.gov/) (25), which provides a comprehensive set of
functional annotation tools for researchers to understand the
biological contexts surrounding a large number of interacting
genes.
Survival analysis
To assess the prognostic value of the lncRNAs and
genes identified, GEPIA and Kaplan-Meier plotter (http://kmplot.com/analysis/) (26) were used. Overall survival of patients
with HCC was analyzed using a Kaplan-Meier plot based on the median
expression levels of each gene. The hazard ratio (HR) and 95%
confidence intervals were calculated and a log-rank test was used
to determine whether survival was significantly different between
patients with high and low expression of each gene in their HCC
tissues.
Results
Downregulated expression of LINC01093
in HCC tissues is associated with poor prognosis
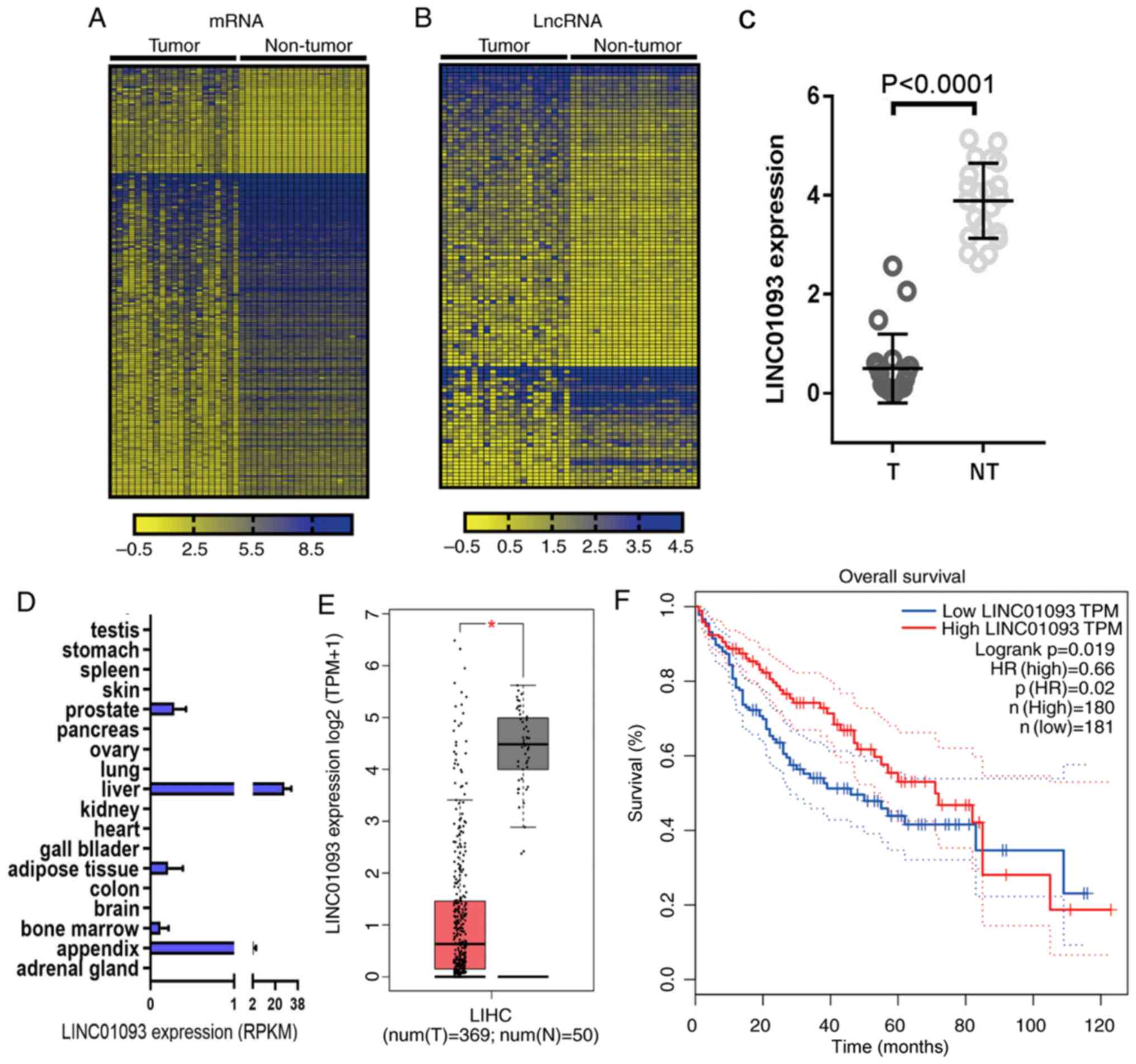
The GSE94660 dataset from GEO database was analyzed,
and heatmaps were generated to present the differentially expressed
genes (Fig. 1A) and lncRNAs
(Fig. 1B) with a fold change ≥|1.5|
and P<0.05. LINC01093 (27,28) was
the most significantly downregulated lncRNA (Fig. 1C). The expression profile of
LINC01093 in normal tissues was obtained from BioProject, and
LINC01093 was almost exclusively expressed in normal liver tissue
in humans (Fig. 1D). To validate
these results, the expression of LINC01093 in HCC tissues and
adjacent non-tumor tissues was analyzed using the GEPIA database.
The results indicated that LINC01093 was significantly
downregulated in tumor tissues compared with that in normal tissues
(Fig. 1E). Survival analysis using
Kaplan-Meier plotter suggested that low expression levels of
LINC01093 in patients with HCC predicted a less favorable prognosis
(Fig. 1F).
LINC01093 is a potential target of
miR-96-5p
lncRNAs primarily function as ceRNAs; thus, low
expression levels of LINC01093 in tumor tissues lead to high
expression of target miRNAs. To identify potential miRNAs that
interact with LINC01093, the lncBase database was used. By
implementing a cutoff prediction score of ≥0.95, five potential
miRNAs were identified: miR-30b-3p, miR-627-3p, miR-676-5p,
miR-3689d and miR-96-5p. The potential target genes of these miRNAs
were predicted using TargetScan and screened with a total context
score of ≤0.6 used as the cutoff. Genes identified in TargetScan,
which were also differentially expressed in the GSE94660 dataset,
were selected as candidate targets. A lncRNA-miRNA-gene network was
established (Fig. 2A) with the
aforementioned diverse bioinformatics tools (Fig. 2B). The expression levels of the five
aforementioned miRNAs were validated using data from TCGA,
revealing that three of the miRNAs were significantly
differentially expressed between tumor tissues and adjacent
non-tumor tissues; among these miRs, miR-96-5p was upregulated in
tumor tissues, whereas miR-30b-3p and miR-627-3p were significantly
downregulated in tumor tissues (Fig.
2C). The Kaplan-Meier plotter was used to perform survival
analysis, and the results indicated that low expression levels of
miR-96-5p predicted a favorable prognosis for patients with HCC
(Fig. 2D). Although the expression
levels were negatively associated with patient prognosis,
miR-627-3p was not statistically considered a potential candidate
miRNA (Fig. 2E). There was no
significant association between the expression of miR-30b-3p and
survival outcome (Fig. 2F). These
results indicate that miR-96-5p potentially targets LINC01093. The
results indicated that miR-96-5p has a tumor suppressor role in HCC
by inhibiting LINC01093, and its upregulation is associated with a
favorable prognosis regarding survival.
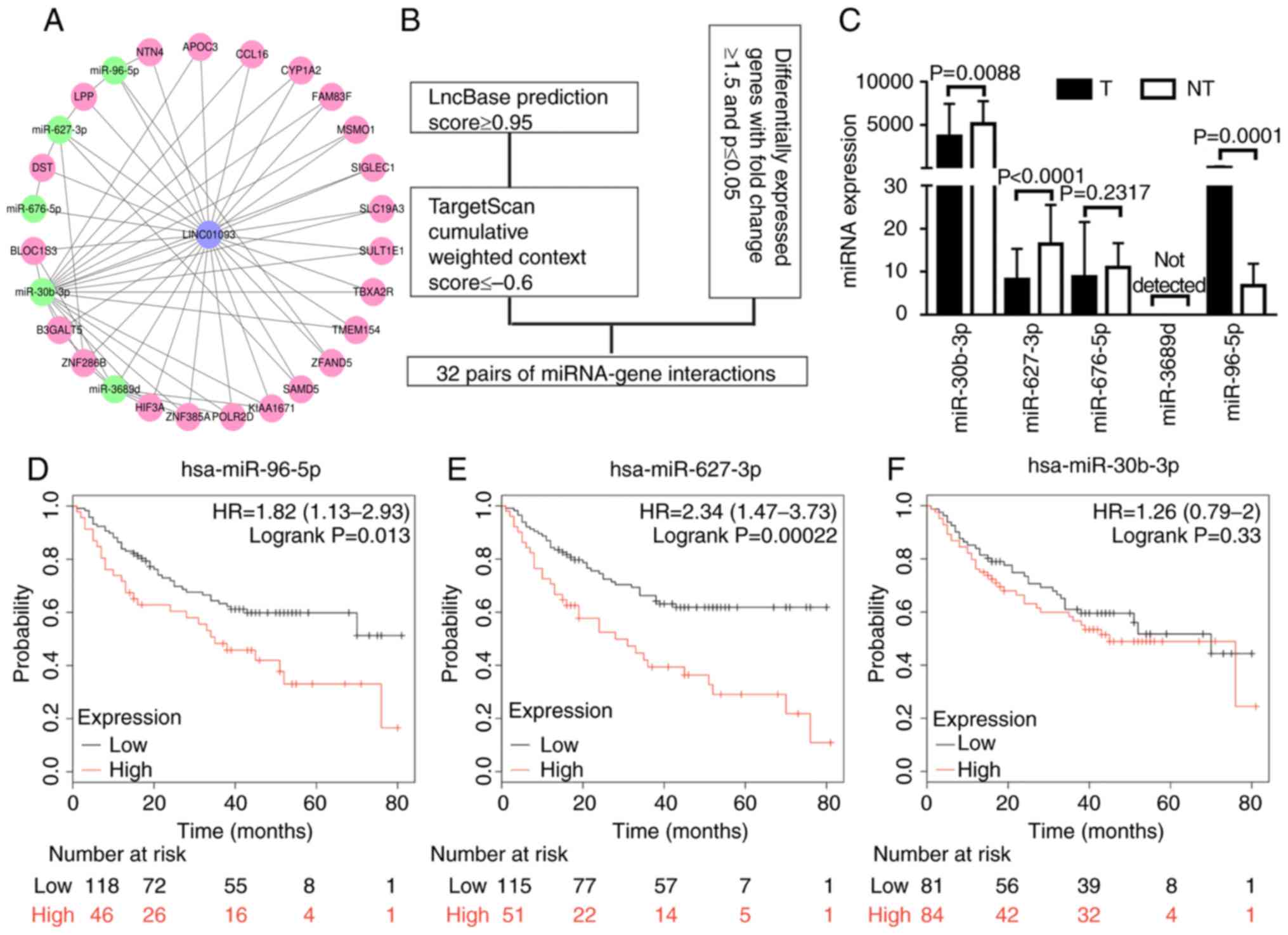 | Figure 2.LINC01093 is a potential target of
miR-96-5p. (A) Regulatory network of LINC01093, miRNAs and mRNAs:
Blue nodes, LINC01093; green nodes, miRNA; pink nodes, mRNA. (B)
Flow chart for the in silico prediction of
LINC01093-miRNA-mRNA pairs. (C) Validation of miRNA expression
using data from The Cancer Genome Atlas. Kaplan-Meier analysis of
overall survival for patients with hepatocellular carcinoma with
high vs. low expression of (D) miR-96-5p, (E) miR-627-3p and (F)
miR-30b-3p. miRNA, microRNA; hsa, Homo sapiens; LINC01093,
long intergenic non-protein coding RNA 1093; T, tumor samples; NT,
non-tumor tissue samples; HR, hazard ratio. NTN4, nerve guidance
factor 4; APOC3, apolipoprotein C3; CCL16, C-C motif chemokine
ligand 16; CYP1A2, cytochrome P450 1A2; FAM83F, family with
sequence similarity 83; MSMO1, methylsterol monooxygenase 1;
SIGLEC1, sialic acid binding Ig like lectin 1; SLC19A3, solute
carrier family 19 member 3; SULT1E1, sulfotransferase family 1E
member 1; TBXA2R, thromboxane A2 receptor; TMEM154, transmembrane
protein 154; ZFAND5, zinc finger AN1-type containing 5; SMAD5, SMAD
family member 5; POLR2D, RNA polymerase II subunit D; ZNF385, zinc
finger protein 385A, HIF3A, hypoxia inducible factor 3 subunit α;
ZNF286B, zinc finger protein 286B; B3GALT5,
β-1,3-galactosyltransferase 5; BLOC1S3, biogenesis of lysosomal
organelles complex 1 subunit 3; DST, dystonin; LPP, LIM domain
containing preferred translocation partner in lipoma. |
ZFAND5 is a candidate target of
miR-96-5p
miRNA regulates gene expression in a negative
manner; thus, high expression levels of miR-96-5p in the tumor
tissue theoretically result in decreased expression of the target
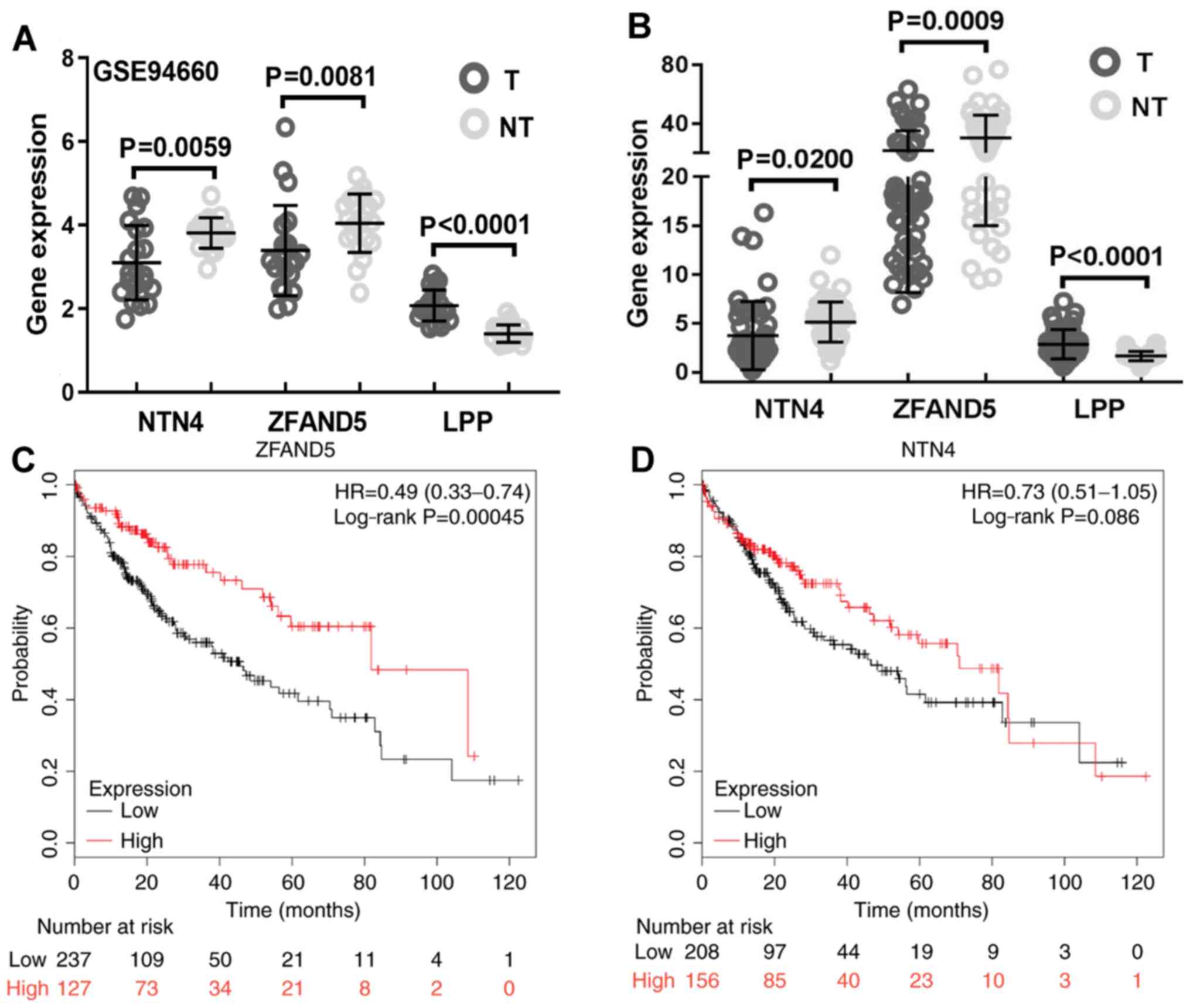
gene. To identify the potential targets, expression of previously
predicted miR-96-5p target genes were analyzed and the results
suggested that the expression levels of netrin 4 (NTN4) and ZFAND5
were lower in HCC tissues compared with those in the adjacent
non-tumor tissues (Fig. 3A). LIM
domain containing preferred translocation partner in lipoma (LPP)
was significantly differentially expressed; however, its expression
was upregulated in tumor tissues (Fig.
3A). These results were further validated using data from TCGA,
confirming that NTN4 and ZFAND5 were downregulated in HCC tissues
(Fig. 3B). NTN4 and ZFAND5 were
further subjected to survival analysis, and the results
demonstrated that only high expression of ZFAND5 predicted a
favorable prognosis for patients with HCC (Fig. 3C), whereas high expression of NTN4
was not predicted to be significantly associated with improved
prognosis (Fig. 3D). Therefore,
ZFAND5 was selected for further analysis.
Functional analysis of the ZFAND5
interactome indicates the significance of NF-κB signaling
To further explore its function, the interactome of
ZFAND5 was determined using BioGRID. This led to the identification
of a total of 14 physical interactors with various experimental
systems (Fig. 4A). Functional
analysis demonstrated that the top 10 enriched GO terms were
‘Fc-epsilon receptor signaling pathway’, ‘stimulatory C-type lectin
receptor signaling pathway’, ‘nucleotide-binding oligomerization
domain containing signaling pathway’, ‘T cell receptor signaling
pathway’, ‘MyD88-dependent Toll like receptor (TLR) signaling
pathway’, ‘protein polyubiquitination’, ‘JNK cascade’, ‘inhibitor
of NF-κB kinase (IKK)/NF-κB signaling’, ‘Wnt signaling pathway’ and
‘activation of mitogen-activated protein kinase (MAPK) activity’
(Fig. 4B); while the most
significantly enriched reactome pathways were tumor necrosis factor
receptor associated factor 6 (TRAF6)-mediated NF-κB activation’;
MAPK kinase kinase 7 (TAK1) activates NF-κB by phosphorylation and
activation of IKKs complex, interleukin-1 family signaling, C-type
lectin domain containing 7A (CLEC7A) signaling, nerve growth factor
receptor (p75NTR) recruits signaling complexes, NF-κB is activated
and signals survival, neurotrophin receptor-interacting factor
(NRIF) signals cell death from the nucleus, interleukin 1 receptor
associated kinase 1 (IRAK1) recruits IKK complex upon TLR7/8 or −9
stimulation, IRAK1 recruits IKK complex, and downstream T-cell
receptor (TCR) signaling (Fig. 4C).
A network consisting of the genes involved in the top 10 GO terms
was visualized using Cytoscape (Fig.
4D). Similarly, the reactome pathway network is presented in
Fig. 4E. Colored nodes represent
different GO terms or pathways and the node color represents
enrichment significance. Combined GO and pathway analysis indicated
that NF-κB signaling had a prominent significance.
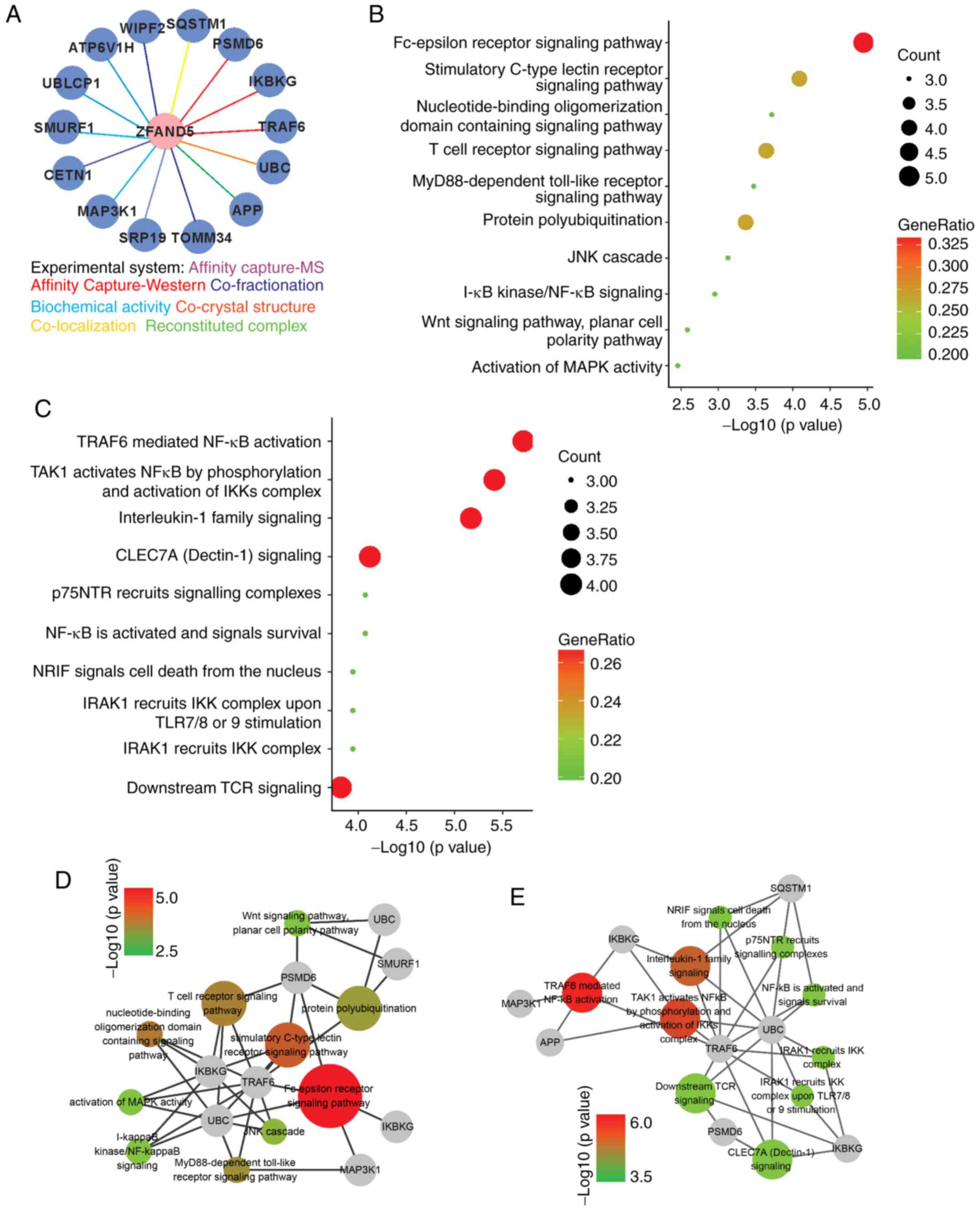 | Figure 4.Functional analyses of the ZFAND5
interactome. (A) ZFAND5 interactors identified by different
experimental systems. (B) Gene Ontology analysis of ZFAND5
interactors in the category Biological Process. (C) Reactome
pathway analysis of ZFAND5 interactors. Visualization of genes
involved in (D) Biological Process terms and (E) reactome pathways.
The grey nodes indicate genes and other colored nodes represent
different GO terms or pathways. The node size and color represent
enrichment significance; the larger the node, the higher enrichment
significance of the pathway. ZFAND5, zinc finger AN1-type
containing 5. SQSTM1, sequestosome 1; PSMD6, proteasome 26S subunit
non-ATPase 6; IKBKG, inhibitor of nuclear factor κB kinase
regulatory subunit γ; TRAF6, TNF receptor associated factor 6; UBC,
ubiquitin C; APP, amyloid β precursor protein; TOMM34, translocase
of outer mitochondrial membrane 34; SRP19, signal recognition
particle 19; MAP3K1, mitogen-activated protein kinase kinase kinase
1; CETN1, centrin 1; SMURF1, SMAD specific E3 ubiquitin protein
ligase 1; UBLCP1, ubiquitin like domain containing CTD phosphatase
1; ATP6V1H, ATPase H+ transporting V1 subunit H; WIPF2,
WAS/WASL interacting protein family member 2. |
Discussion
HCC is notoriously difficult to treat successfully
and is one of the leading causes of cancer-associated death
worldwide. To date, the mechanisms underlying the initiation and
progression of HCC have remained to be fully elucidated. lncRNAs
have been indicated to regulate gene expression and have been
implicated in various biological processes and human diseases.
RNA-seq is a high-throughput sequencing technique that may reveal
the presence and quantity of specific RNAs in a sample, which
allows for the analysis of gene expression, alternative gene
splicing and gene fusion. RNA-seq has been widely used to identify
pathogenesis-associated genes, and to determine potential
therapeutic targets in various diseases.
In the present study, the mRNA and lncRNA expression
profiles between HCC tissues and adjacent non-tumor tissues from
the GSE94660 dataset were compared and it was demonstrated that
LINC01093 was significantly downregulated in tumor tissues.
Analysis with BioProject indicated that LINC01093 was almost
exclusively expressed in the normal adult liver. The expression of
LINC01093 was then validated using data from TCGA, which confirmed
its downregulation in HCC, and further survival analysis
demonstrated that high expression levels of LINC01093 in HCC
tissues were associated with a favorable clinical prognosis.
To understand the role of LINC01093 in
hepatocellular carcinogenesis, miRNAs that target LINC01093 were
predicted and screened. miR-30b-3p, miR-627-3p, miR-676-5p,
miR-3689d and miR-96-5p were identified as potential candidates.
The expression levels of these five miRNAs were validated using
TCGA, and the results demonstrated that miR-30b-3p, miR-627-3p and
miR-96-5p were significantly differentially expressed between tumor
tissues and non-tumor tissues, but only miR-96-5p was upregulated
in tumor tissues. Survival analysis of the three miRNAs was also
performed and the results demonstrated that low expression levels
of miR-96-5p predicted a favorable prognosis of patients. miR-96-5p
has previously been recognized as an oncogenic miRNA in different
types of cancer. In bladder cancer, studies revealed that miR-96-5p
promoted tumor cell migration and invasion (29). In prostate cancer, it has been
reported that miR-96-5p governed tumor progression and disease
outcome by targeting a retinoic acid receptor γ network (30). In colorectal cancer, miR-96-5p was
indicated to promote tumor invasion through inhibition of
reversion-inducing cysteine-rich protein expression (31). In the present study, the results also
suggested that miR-96-5p may promote HCC progression and to be
associated with poor disease outcome.
Target genes of miR-96-5p were then predicted and
screened with a cumulative weighted context score of ≤-0.6, and
LPP, NTN4 and ZFAND5 were the identified as target genes among the
differentially expressed genes identified in the GSE94660 dataset.
These genes were therefore selected as candidate genes. miRNA
regulates gene expression in a negative manner, and since miR-96-5p
was upregulated in tumor tissues, downregulation of target genes
was expected. The expression of the three candidate genes was
analyzed, revealing that ZFAND5 and NTN4 had lower expression in
tumor tissues compared with that in adjacent non-tumor tissues,
whereas LPP was upregulated in HCC tissues, and these results were
further confirmed in the TCGA dataset. ZFAD5 and NTN4 were
subjected to survival analysis, which revealed that high mRNA
expression levels of ZFAND5 were a favorable predictor of patient
prognosis.
ZFAND5 is a member of the ZFAND family (28), and studies have revealed that ZFAND5
enhances protein degradation by activating the ubiquitin-proteasome
system (32,33). To understand the potential role of
ZFAND5 in HCC, proteins that physically interacted with ZFAND5 were
obtained from BioGRID, which included 14 interactors identified by
7 different experimental systems. The ZFAND5-interactome was
subsequently subjected to functional enrichment analysis. GO
analysis revealed that the top 10 biological processes were
‘Fc-epsilon receptor signaling pathway’, ‘stimulatory C-type lectin
receptor signaling pathway’, ‘nucleotide-binding oligomerization
domain containing signaling pathway’, ‘T cell receptor signaling
pathway’, ‘MyD88-dependent TLR signaling pathway’, ‘protein
polyubiquitination’, ‘JNK cascade’, ‘IKK/NF-κB signaling’, ‘Wnt
signaling pathway’ and ‘activation of MAPK activity’, while the
most significantly enriched reactome pathways were ‘TRAF6 mediated
NF-κB activation’, ‘TAK1 activates NF-κB by phosphorylation and
activation of IKKs complex’, ‘interleukin-1 family signaling’,
‘CLEC7A signaling’, ‘p75NTR recruits signaling complexes’, ‘NF-κB
is activated and signals survival’, ‘NRIF signals cell death from
the nucleus’, ‘IRAK1 recruits IKK complex upon TLR7/8 or −9
stimulation’, ‘IRAK1 recruits IKK complex’ and ‘downstream TCR
signaling’. These results demonstrate that the NF-kB signaling is
an important downstream pathway of ZFAND5.
NF-κB serves a well-known function in immune
regulation and inflammatory responses, but growing evidence has
additionally demonstrated its role in tumorigenesis (34,35).
Therefore, NF-κB is frequently considered as the critical link
between inflammation and cancer (35–37). It
was reported that ZFAND5 inhibits NF-κB activation by interacting
with IKKγ (38). Therefore, the
present study indicated the role of a
LINC01093/miR96-5p/ZFAND5/NF-κB axis in the regulation of HCC
development and progression, and further investigations are
required to verify these results. Lack of experimental data is a
limitation of the present study, and in a further study, in
vitro and in vivo experiments will be performed to
verify the conclusions that were obtained through the
bioinformatics analysis. Through the use of bioinformatics analysis
and in vitro and in vivo experiments, novel insight
into the development of potential treatments for patients with HCC
may be gained.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural
Science Foundation of China (grant nos. 81372654 and 81672848).
Availability of data and materials
The datasets analyzed during the current study are
available in the GEO repository (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE94660)
and the BioProject repository (https://www.ncbi.nlm.nih.gov/bioproject?term=PRJEB4337&cmd=DetailsSearch).
Authors' contributions
JZ designed the study and revised the manuscript. YZ
and KY performed the GEO database analysis, analysed the data and
wrote the manuscript. CH and LL performed bioinformatics analysis.
HZ and MH assisted with the collection and analysis of other data.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bodzin AS and Busuttil RW: Hepatocellular
carcinoma: Advances in diagnosis, management, and long-term
outcome. World J Hepatol. 7:1157–1167. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shi X, Sun M, Liu H, Yao Y and Song Y:
Long non-coding RNAs: A new frontier in the study of human
diseases. Cancer Lett. 339:159–166. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Berindan-Neagoe I and Calin GA: Molecular
pathways: MicroRNAs, cancer cells, and microenvironment. Clin
Cancer Res. 20:6247–6253. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bartonicek N, Maag JL and Dinger ME: Long
noncoding RNAs in cancer: Mechanisms of action and technological
advancements. Mol Cancer. 15:432016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ng KW, Anderson C, Marshall EA, Minatel
BC, Enfield KS, Saprunoff HL, Lam WL and Martinez VD:
Piwi-interacting RNAs in cancer: Emerging functions and clinical
utility. Mol Cancer. 15:52016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mannoor K, Liao J and Jiang F: Small
nucleolar RNAs in cancer. Biochim Biophys Acta. 1826:121–128.
2012.PubMed/NCBI
|
|
8
|
Holoch D and Moazed D: RNA-mediated
epigenetic regulation of gene expression. Nat Rev Genet. 16:71–84.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhu C, Cheng D, Qiu X, Zhuang M and Liu Z:
Long noncoding RNA SNHG16 promotes cell proliferation by sponging
MicroRNA-205 and upregulating ZEB1 expression in osteosarcoma. Cell
Physiol Biochem. 51:429–440. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lan T, Ma W, Hong Z, Wu L, Chen X and Yuan
Y: Long non-coding RNA small nucleolar RNA host gene 12 (SNHG12)
promotes tumorigenesis and metastasis by targeting miR-199a/b-5p in
hepatocellular carcinoma. J Exp Clin Cancer Res. 36:112017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dong J, Teng F, Guo W, Yang J, Ding G and
Fu Z: lncRNA SNHG8 promotes the tumorigenesis and metastasis by
sponging miR-149-5p and predicts tumor recurrence in hepatocellular
carcinoma. Cell Physiol Biochem. 51:2262–2274. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tu J, Zhao Z, Xu M, Chen M, Weng Q, Wang J
and Ji J: LINC00707 contributes to hepatocellular carcinoma
progression via sponging miR-206 to increase CDK14. J Cell Physiol.
234:10615–10624. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang YG, Liu J, Shi M and Chen FX: lncRNA
DGCR5 represses the development of hepatocellular carcinoma by
targeting the miR-346/KLF14 axis. J Cell Physiol. 234:572–580.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen W, You J, Zheng Q and Zhu YY:
Downregulation of lncRNA OGFRP1 inhibits hepatocellular carcinoma
progression by AKT/mTOR and Wnt/β-catenin signaling pathways.
Cancer Manag Res. 10:1817–1826. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Takahashi K, Yan I, Haga H and Patel T:
Long noncoding RNA in liver diseases. Hepatology. 60:744–753. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cancer Genome Atlas Research Network, ;
Weinstein JN, Collisson EA, Mills GB, Shaw KR, Ozenberger BA,
Ellrott K, Shmulevich I, Sander C and Stuart JM: The cancer genome
atlas pan-cancer analysis project. Nat Genet. 45:1113–1120. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Edgar R, Domrachev M and Lash AE: Gene
expression omnibus: NCBI gene expression and hybridization array
data repository. Nucleic Acids Res. 30:207–210. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tang Z, Li C, Kang B, Gao G, Li C and
Zhang Z: GEPIA: A web server for cancer and normal gene expression
profiling and interactive analyses. Nucleic Acids Res. 45:W98–W102.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ota T, Suzuki Y, Nishikawa T, Otsuki T,
Sugiyama T, Irie R, Wakamatsu A, Hayashi K, Sato H, Nagai K, et al:
Complete sequencing and characterization of 21,243 full-length
human cDNAs. Nat Genet. 36:40–45. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Paraskevopoulou MD, Vlachos IS, Karagkouni
D, Georgakilas G, Kanellos I, Vergoulis T, Zagganas K, Tsanakas P,
Floros E, Dalamagas T and Hatzigeorgiou AG: DIANA-LncBase v2:
Indexing microRNA targets on non-coding transcripts. Nucleic Acids
Res. 44:D231–D238. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lewis BP, Burge CB and Bartel DP:
Conserved seed pairing, often flanked by adenosines, indicates that
thousands of human genes are microRNA targets. Cell. 120:15–20.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Smoot ME, Ono K, Ruscheinski J, Wang PL
and Ideker T: Cytoscape 2.8: New features for data integration and
network visualization. Bioinformatics. 27:431–432. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Oughtred R, Stark C, Breitkreutz BJ, Rust
J, Boucher L, Chang C, Kolas N, O'Donnell L, Leung G, McAdam R, et
al: The BioGRID interaction database: 2019 update. Nucleic Acids
Res. 47:D529–D541. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The gene
ontology consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Huang DW, Sherman BT, Tan Q, Collins JR,
Alvord WG, Roayaei J, Stephens R, Baseler MW, Lane HC and Lempicki
RA: The DAVID gene functional classification tool: A novel
biological module-centric algorithm to functionally analyze large
gene lists. Genome Biol. 8:R1832007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hou GX, Liu P, Yang J and Wen S: Mining
expression and prognosis of topoisomerase isoforms in
non-small-cell lung cancer by using Oncomine and Kaplan-Meier
plotter. PLoS One. 12:e01745152017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mou Y, Wang D, Xing R, Nie H, Mou Y, Zhang
Y and Zhou X: Identification of long noncoding RNAs biomarkers in
patients with hepatitis B virus-associated hepatocellular
carcinoma. Cancer Biomark. 23:95–106. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dai M, Chen S, Wei X, Zhu X, Lan F, Dai S
and Qin X: Diagnosis, prognosis and bioinformatics analysis of
lncRNAs in hepatocellular carcinoma. Oncotarget. 8:95799–95809.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
He C, Zhang Q, Gu R, Lou Y and Liu W:
miR-96 regulates migration and invasion of bladder cancer through
epithelial-mesenchymal transition in response to transforming
growth factor-β1. J Cell Biochem. 119:7807–7817. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Long MD, Singh PK, Russell JR, Llimos G,
Rosario S, Rizvi A, van den Berg PR, Kirk J, Sucheston-Campbell LE,
Smiraglia DJ and Campbell MJ: The miR-96 and RARγ signaling axis
governs androgen signaling and prostate cancer progression.
Oncogene. 38:421–444. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Iseki Y, Shibutani M, Maeda K, Nagahara H,
Fukuoka T, Matsutani S, Hirakawa K and Ohira M: MicroRNA-96
promotes tumor invasion in colorectal cancer via RECK. Anticancer
Res. 38:2031–2035. 2018.PubMed/NCBI
|
|
32
|
Lee D, Takayama S and Goldberg AL:
ZFAND5/ZNF216 is an activator of the 26S proteasome that stimulates
overall protein degradation. Proc Natl Acad Sci USA.
115:E9550–E9559. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hishiya A, Iemura S, Natsume T, Takayama
S, Ikeda K and Watanabe K: A novel ubiquitin-binding protein ZNF216
functioning in muscle atrophy. EMBO J. 25:554–564. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Dolcet X, Llobet D, Pallares J and
Matias-Guiu X: NF-kB in development and progression of human
cancer. Virchows Arch. 446:475–482. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Marx J: Cancer research. Inflammation and
cancer: The link grows stronger. Science. 306:966–968. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Karin M: NF-kappaB as a critical link
between inflammation and cancer. Cold Spring Harb Perspect Biol.
1:a0001412009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Karin M: Nuclear factor-kappaB in cancer
development and progression. Nature. 441:431–436. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Huang J, Teng L, Li L, Liu T, Li L, Chen
D, Xu LG, Zhai Z and Shu HB: ZNF216 is an A20-like and IkappaB
kinase gamma-interacting inhibitor of NFkappaB activation. J Biol
Chem. 279:16847–16853. 2004. View Article : Google Scholar : PubMed/NCBI
|