Introduction
Subarachnoid hemorrhage (1) (SAH) refers to the rupture of diseased
blood vessels at the bottom or surface of the brain. Blood directly
flows into the subarachnoid cavity to cause a clinical syndrome,
also known as primary subarachnoid hemorrhage, accounting for
approximately 10% of acute stroke. It is a very serious and common
disease. It is reported that secondary subarachnoid hemorrhage is
caused by cerebral parenchyma, intraventricular hemorrhage,
epidural or subdural vascular rupture, and vascular perforation of
brain tissue into subarachnoid space (2).
Nimodipine used to be a common drug for the
treatment of subarachnoid hemorrhage and the prevention of cerebral
vasospasm, but clinical treatment found that this drug can increase
intracranial pressure of patients, causing cerebral rebleeding, and
cannot well protect brain tissue (3). Cinepazide maleate injection (CMI)
(4) is a new type of drug with brain
protection, which can effectively protect central nerve cells,
expand cerebral vessels and improve brain microcirculation
(5). Edaravone (6) is a free radical scavenger listed in
2001. It can effectively regulate the expression of apoptosis
genes, inhibit lipid peroxidation, prevent oxidative damage of the
body, and play a role in vascular protection. It is widely used in
the treatment of cerebral infarction, acute cerebral hemorrhage and
other diseases (7).
Studies have found that autophagy participates in
the pathological process of early brain injury (EBI) of
subarachnoid hemorrhage and determines the outcome of nerve cells
in EBI stage: recovery or repair, aggravation or death. At present,
there is scarce research on the protective mechanism of edaravone
and cinepazide maleate on SAH (8,9).
Therefore, the SAH animal model was established in this study.
Edaravone combined with cinepazide maleate was used for
intervention, and the effects of expression and neurological
function in key autophagy factor Beclin-1 and micro-related protein
1 (light chain LC3)-II were observed to analyze the protective
mechanism of edaravone and cinepazide maleate on SAH nerve.
Materials and methods
Animal grouping
Eighty clean grade male SD rats weighing 350–450 g
were purchased from Nanjing Junke Bioengineering Co., Ltd. They
were divided into sham operation group, SAH group, CMI group and
edaravone combined with CMI group (combined group). Each group was
divided into four time subgroups of 24, 48, 72 and 144 h.
The study was approved by the Ethics Committee of
Daqinglongnan Hospital (Daqing, China).
Model making and treatment
The model of subarachnoid hemorrhage in rats was
prepared by classic secondary blood injection method (0.3 ml of
autologous arterial blood was injected into occipital cistern with
a secondary interval of 48 h). The sham operation group was
injected by 0.3 ml of normal saline into the occipital cistern
twice, and the other operations were consistent with the SAH group.
Criteria for model success: i) When secondary occipital cistern
injection was performed, a small amount of hemorrhagic
cerebrospinal fluid exuded from the puncture site; ii) when peeling
off the brain, it was obvious that bloody liquid was scattered in
the basal cistern of the brain floor. CMI group: 100 µg/kg was
injected intraperitoneally immediately after SAH model was
successfully prepared, and the drug was given once every 24 h. The
combined group received edaravone 10 mg/kg via tail vein on the
basis of CMI, once every 24 h.
Observation on morphology and
structure of hippocampus in brain tissue
Two rats in each group were taken at each time
point. After routine anesthesia treatment, the animals were
sacrificed. After perfusion and fixation with 4% paraformaldehyde,
paraffin embedding, coronal section, hematoxylin and eosin
(H&E) staining a 400-fold optical microscope was used to
observe the morphological structure of hippocampal neurons. The
number of viable nerve cells in each field of vision were counted,
the specific method was as follows: Four slices of rats in
hippocampus of each group were taken, 5 non-repeated fields of
vision (100 fields in total) were selected from each slice, and
Motic-6.0 image acquisition and analysis system were used for
analysis, which was expressed by the ratio of the number of viable
cells in CA1 area to the total number of cells under the average
cell percentage (400×) in each field of vision.
ELISA detection of protein expression
of related factors in rats
Blood was collected from vena cava, left in the
greenhouse for 4 h, centrifuged at 4°C and 3000 rpm for 10 min, and
serum was collected and split-packed. ELISA kit was used to detect
the relative expression levels of protein factors Beclin-1 and
LC3-II. Finally, the absorbance (A) value of each sample at 450 nm
wavelength was determined by using an enzyme-labeled instrument (to
ensure that no water drops existed in the enzyme-labeled bottom
plate and no bubbles were generated in the drop holes). After
subtracting the A values of TMB blank color development holes from
A values of all standard products and samples, the standard curve
was drawn with the standard product concentration as the abscissa
and the zero-adjusted A value as the ordinate, and the actual
concentration of each sample was calculated. Beclin-1 ELISA kit was
purchased from R&D Systems, Inc. and LC3-II ELISA kit was
purchased from Jianglai Biology.
Neurobehavioral scoring criteria and
inclusion criteria
On the 14th and 28th day after administration, Longa
nerve function score and muscle strength score were respectively
performed on rats in each group, and the scores were statistically
analyzed.
Longa score (10)
Neurological examination was divided into 5 grades:
0: normal, no neurological impairment; 1 point: left anterior paw
could not be fully extended, with slight neurological impairment; 2
points: When walking, the rat turned to the left (paralyzed side)
with moderate neurological deficits; 3 points: When walking, the
body of the rat toppled to the left (paralyzed side), resulting in
severe neurological deficits; 4 points: unable to walk
spontaneously, loss of consciousness.
Muscle strength assessment (11)
In order to reduce the error, we conducted two
inspections and recorded the best results. The interval between the
two inspections was 5 min. The specific method was to add a
diameter of 0. A 6 cm nylon rope with a length of 1.0 meters was
horizontally fixed at a height of 80.0 cm, and a thick sponge was
placed below to prevent rats from falling. Then, the tail was
lifted and the forepaws of animals hang in the middle of the rope
and the suspension time was recorded to indicate the strength of
the forelimbs. The score was divided into 4 grades: 0: hanging on
the rope for 0–2 sec; 1 point: hanging on the rope for 3–4 sec; 2
points: hanging on the rope for more than 5 sec; 3 points: hanging
on the rope for more than 5 sec and the hind legs could be put on
the rope.
Statistical analysis
This study used SPSS18.0 (Bizinsight (Beijing)
Information Technology Co., Ltd.) to carry out statistical analysis
on the data. GraphPad Prism 6 software was used to draw all the
pictures in this experiment. Chi-square test was used to compare
the enumeration data, mean ± standard deviation was used to express
the measurement data, T test was used to analyze the two groups,
variance analysis was used to compare the multiple groups, and
Pearson's correlation analysis to analyze the relationship between
variables. P<0.05 was considered statistically significant.
Results
Comparison of survival rate of
hippocampal nerve cells in each group
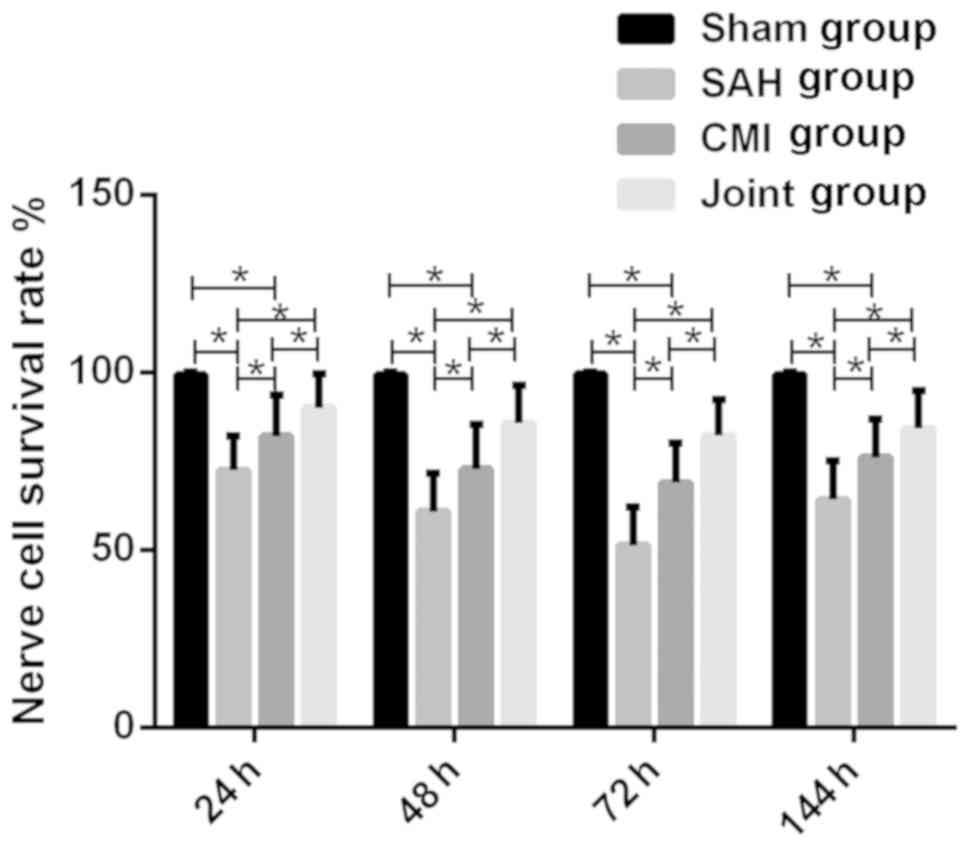
Compared with the sham operation group, the survival
rate of nerve cells in SAH group, CMI group and combined group at
each time point was significantly reduced (P<0.05). The
morphological and structural damage of nerve cells in CMI group was
alleviated. Compared with SAH group, the number of viable nerve
cells in the visual field was significantly increased (P<0.05).
The morphological and structural damage of nerve cells in the
combined group was further alleviated, and their survival rate
significantly increased at all time points (P<0.05) (Fig. 1).
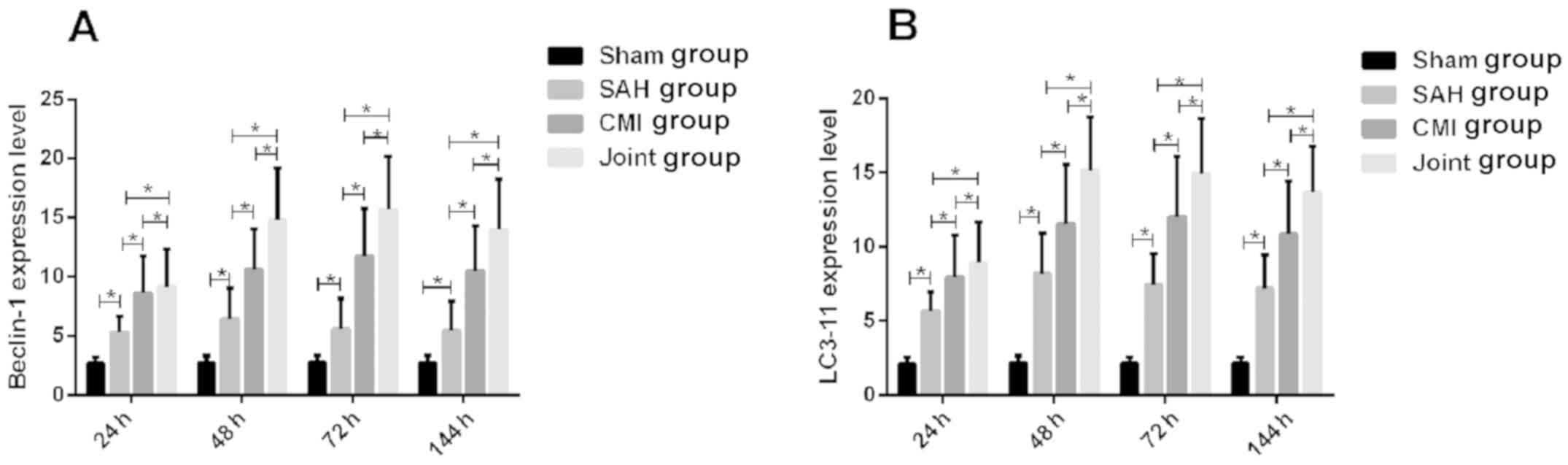
Expression of Beclin-1 and LC3-11 in
rats of each group
The expression levels of Beclin-1 and LC3-11 in SAH
group was significantly higher than those in sham operation group
(P<0.05). The expression levels of Beclin-1 and LC3-11 in CMI
group was significantly higher than those in SAH group (P<0.05).
The expression levels of Beclin-1 and LC3-11 in the combined group
was significantly higher than those in SAH group and CMI group
(P<0.05) (Fig. 2).
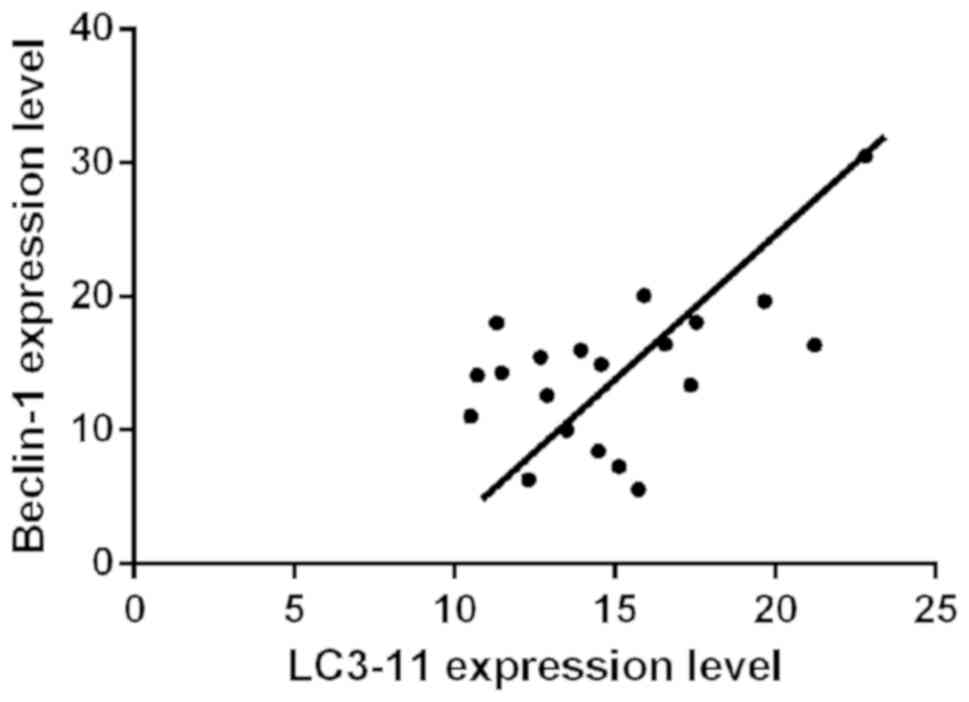
Correlation analysis between Beclin-1
and LC3-11
Pearson's correlation analysis showed that there was
a correlation between Beclin-1 and LC3-11 (P<0.05, r=0.9454).
The data of Beclin-1 and LC3-11 were taken from the index level
measured in the Joint group for 72 h (Fig. 3).
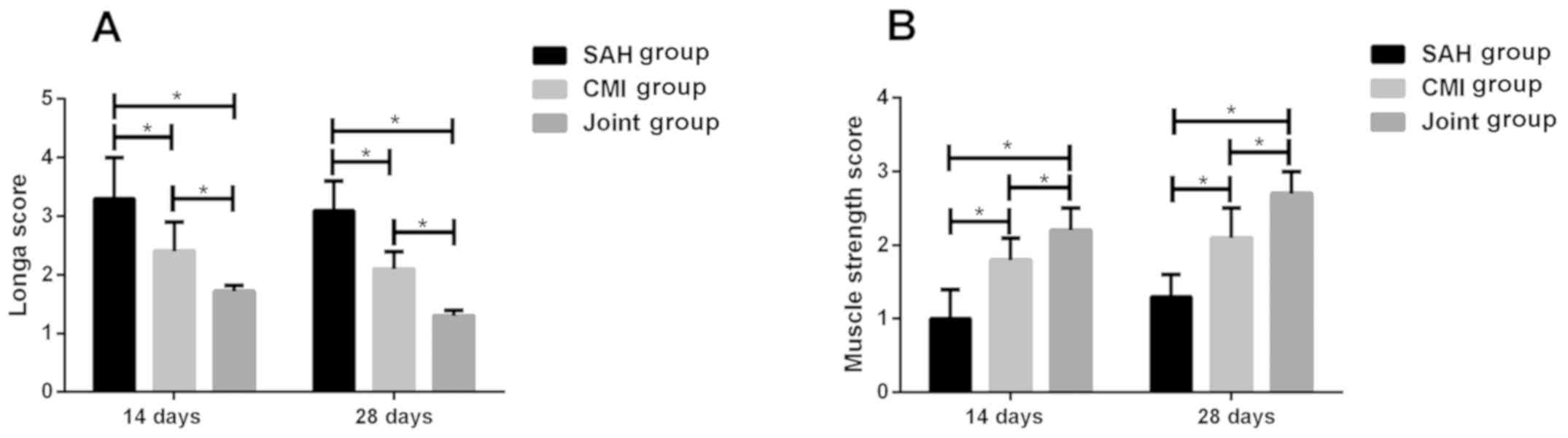
Score of neurological function of rats
in each group
Longa score and muscle strength score were performed
at days 14 and 28, respectively, of rats in the three groups. The
results showed that Longa score in SAH group was significantly
higher than CMI group and Joint group, while that in combined group
was significantly lower than CMI group (P<0.05). The muscle
strength score of SAH group was significantly lower than that of
CMI group and Joint group, and the combined group was significantly
higher than that of CMI group (P<0.05) (Fig. 4).
Discussion
Subarachnoid hemorrhage is a common brain disease in
clinical practice, and it is also a critical disease for life
safety of crisis patients (12). The
main causes of the disease are smoking, drinking, hypertension,
trauma, hematological diseases, arterial angioma and other brain
diseases (13). Patients often
suffer from severe headache accompanied by dizziness, nausea,
vomiting, pale complexion and cold sweat all over the body. With
the aggravation of the disease, headache can radiate to the waist,
back, neck and other parts, and even vascular spasm, cerebral
infarction and limb hemiplegia in severe cases (14,15). The
key to treat the disease is to inhibit hemorrhage, prevent spasm
and reduce the occurrence of complications. Cranial nerve autophagy
is also an important reason for subarachnoid hemorrhage to harm
cranial nerves.
It was found that CMI can reduce neuronal death in
hippocampus of SAH rats and reduce neurological recovery disorders
in animals, suggesting that CMI has a certain protective effect on
EBI of SAH (16). Guidolzite maleate
is a new type of ophthalmic calcium channel blocker, which can
effectively prevent calcium ions from entering cells, relax
vascular smooth muscle and dilate blood vessels, thus relieving
vascular healing twins, reducing blood flow resistance and
increasing blood flow. It can also enhance the content of adenosine
and adenosine cyclophosphosphate, reduce oxygen consumption,
improve erythrocyte deformability, reduce blood stalk property,
increase cerebral blood flow and improve cerebral metabolism
(4). Previous studies have shown
that CMI has a certain neuroprotective effect after treatment of
ischemic encephalopathy (17,18). In
the state of brain injury, autophagy activation can clear damaged
organelles in cells, regulate cell energy metabolism and reduce the
degree of cell injury. However, excessive autophagy promotes
caspase-3 lysis and aggravate the death of nerve cells (19). In this study, the expression levels
of autophagy marker proteins Beclin-1 and LC3-11 in CMI group
increased. At this time, the survival rate of nerve cells
increased. On the one hand, it showed that autophagy activation in
EBI stage of SAH had neuroprotective effect, and on the other hand,
CMI played neuroprotective effect on SAH by enhancing autophagy
degree in EBI stage.
It was found that edaravone had a good protective
effect on SAH (20,21). Intracranial blood accumulation after
SAH, compression of coagulated blood clots, resulting in ischemia
and anoxia, lysis of accumulated red blood cells, further release
of superoxide anion and generation of hydroxyl radicals by
oxyhemoglobin and increase of generation of oxygen radicals, induce
strong oxidative stress reaction and aggravate nerve cell damage
(22). In this study, Longa score
and muscle strength score of the combined treatment group with
added edaravone were significantly higher than those of CMI group
and SAH group in neurological function score. Neuroprotective agent
edaravone mainly prevents and treats the continuous progress of
brain injury by scavenging oxygen free radicals and alleviating a
series of cascade reactions caused by oxidative stress, which is
the main reason for further improvement of the number of surviving
nerve cells and nerve function in the combined group. The
expression levels of Beclin-1 and LC3-11 in combined group
increased further, suggesting that edaravone can strengthen CMI's
regulation of autophagy activity after SAH. Edaravone can remove a
large amount of oxygen free radicals, and the level of collective
oxygen-free radicals is kept at a certain level, thus maintaining
the moderate expression of autophagy level after SAH.
Collectively, edaravone combined with cinepazide
maleate can effectively increase the survival rate of brain cells
and promote the volatilization of nerve function in the treatment
of hemorrhage in the subretinal space of the omentum, which is
worthy of popularization and application.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
ZC wrote the manuscript. ZC and HZ conceived and
designed the study. ZC and HS were responsible for the collection
and analysis of the experimental data. YP and XZ interpreted the
data and drafted the manuscript. ZC and YP revised the manuscript
critically for important intellectual content. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Daqinglongnan Hospital (Daqing, China).
Patient consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no competing
interests.
References
|
1
|
Feigin VL, Rinkel GJ, Lawes CM, Algra A,
Bennett DA, van Gijn J and Anderson CS: Risk factors for
subarachnoid hemorrhage: An updated systematic review of
epidemiological studies. Stroke. 36:2773–2780. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Locksley HB: Natural history of
subarachnoid hemorrhage, intracranial aneurysms and arteriovenous
malformations. Based on 6368 cases in the cooperative study. J
Neurosurg. 25:219–239. 1966. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kotwica Z and Persson L: Effect of
intravenous infusion of nimodipine on intracranial pressure.
Experimental study of ICP measurement conducted in a rat model.
Neurol Neurochir Pol. 23:227–230. 1989.(In Polish). PubMed/NCBI
|
|
4
|
Akashi A, Hirohashi M, Suzuki I, Shibamura
S and Kasahara A: Cardiovascular pharmacology of cinepazide, a new
cerebral vasodilator (author's transl). Nippon Yakurigaku Zasshi.
75:507–516. 1979.(In Japanese). View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yi ZM, Liu F and Zhai SD: Cinepazide
maleate injection for cerebral infarction: a systematic review.
Chin J Evidence-Based Med. 10:1079–1084. 2010.
|
|
6
|
Kono H, Asakawa M, Fujii H, Maki A,
Amemiya H, Yamamoto M, Matsuda M and Matsumoto Y: Edaravone, a
novel free radical scavenger, prevents liver injury and mortality
in rats administered endotoxin. J Pharmacol Exp Ther. 307:74–82.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jin Q, Cai Y, Li S, Liu H, Zhou X, Lu C,
Gao X, Qian J, Zhang J, Ju S and Li C: Edaravone-encapsulated
agonistic micelles rescue ischemic brain tissue by tuning
blood-brain barrier permeability. Theranostics. 7:884–898. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang Z, Shi XY, Yin J, Zuo G, Zhang J and
Chen G: Role of autophagy in early brain injury after experimental
subarachnoid hemorrhage. J Mol Neurosci. 46:192–202. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee JY, He Y, Sagher O, Keep R, Hua Y and
Xi G: Activated autophagy pathway in experimental subarachnoid
hemorrhage. Brain Res. 1287:126–135. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zheng XW, Yang WT, Chen S, Xu QQ, Shan CS,
Zheng GQ and Ruan JC: Neuroprotection of catalpol for experimental
acute focal ischemic stroke: Preclinical evidence and possible
mechanisms of antioxidation, anti-inflammation, and antiapoptosis.
Oxid Med Cell Longev. 2017:50586092017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lu XY and Hao XM: Biological effects of
electrical stimulation on muscle strength and electromyogram in
rats with muscle disuse atrophy. Chin J Clin Rehabil. 10:34–36.
2006.(In Chinese).
|
|
12
|
McGirt MJ, Mavropoulos JC, McGirt LY,
Alexander MJ, Friedman AH, Laskowitz DT and Lynch JR: Leukocytosis
as an independent risk factor for cerebral vasospasm following
aneurysmal subarachnoid hemorrhage. J Neurosurg. 98:1222–1226.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hillbom M and Kaste M: Alcohol
intoxication: A risk factor for primary subarachnoid hemorrhage.
Neurology. 32:706–711. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yolas C, Ozdemir NG, Kanat A, Aydin MD,
Keles P, Kepoglu U, Aydin N and Gundogdu C: Uncovering a new cause
of obstructive hydrocephalus following subarachnoid hemorrhage:
choroidal artery vasospasm-related ependymal cell degeneration and
aqueductal stenosis-first experimental study. World Neurosurg.
90:484–491. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hartmann A, Dorndorf W and Alberti E:
Complications of subarachnoid hemorrhage. Med Klin. 72:476–482.
1977.PubMed/NCBI
|
|
16
|
Lee JY, Sagher O, Keep R, Hua Y and Xi G:
Comparison of experimental rat models of early brain injury after
subarachnoid hemorrhage. Neurosurgery. 65:331–343. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chao Y, Hu W and Geng X: The
neuroprotective strategic analysis for patients with acute
myocardial infarction combined with hypoxic ischemic encephalopathy
in ICU. Hellenic J Cardiol. 58:427–431. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zheng PU, Wang XR, Gan J and Chen QI:
Effect of cinepazide maleate preconditioning on rat models of
cerebral ischemia. J Shanghai Jiaotong Univ. 33:1064–1073. 2013.(In
Chinese).
|
|
19
|
Wang L, Wang P, Dong H, Wang S, Chu H, Yan
W and Zhang X: Ulk1/FUNDC1 prevents nerve cells from
hypoxia-induced apoptosis by promoting cell autophagy. Neurochem
Res. 43:1539–1548. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Munakata A, Ohkuma H, Nakano T, Shimamura
N, Asano K and Naraoka M: Effect of a free radical scavenger,
edaravone, in the treatment of patients with aneurysmal
subarachnoid hemorrhage. Neurosurgery. 64:423–429. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wei W, Yang G, Shu XU, Yin W-B and Ding
X-S: The brain-protective effects of edaravone in rats after
subarachnoid hemorrhage. Chin J Contemp Neurol Neurosurg.
8:544–549. 2008.(In Chinese).
|
|
22
|
Nakagomi T, Yamakawa K, Sasaki T, Saito I
and Takakura K: Effect of edaravone on cerebral vasospasm following
experimental subarachnoid hemorrhage. J Stroke Cerebrovasc Dis.
12:17–21. 2003. View Article : Google Scholar : PubMed/NCBI
|