Introduction
Parkinson's disease (PD) is a degenerative
neurological disease affected by a variety of comprehensive
factors. Its main manifestations include motor symptoms represented
as sluggish movement, tremor and paralysis, postural gait disorder
and other non-motor symptoms such as depression, which can
seriously threaten the quality of life of middle-aged and elderly
patients (1). Pathologically, PD is
characterized by a severe loss of dopaminergic neurons in the
striatum nistriatum, a change that is caused by the accumulation
and spread of characteristic Lewy bodies and nerve sheath
inclusions due to misfolded fibers (2). These misfolded fibers, mainly derived
from a neurotoxic form of α-synuclein, can accumulate spontaneously
at the junction of brain regions and cause degeneration of
dopaminergic neurons (3,4). In recent years, a large number of
studies have shown that α-synuclein plays an important regulatory
role in non-specific immunity of the body. Among them, A53T mutant
α-synuclein can significantly activate the key transcription factor
of inflammatory response, NF-κB, thereby activating microglia and
astrocytes (5). Apart from that,
α-synuclein can also promote the expression of toll-like receptors
(TLRs), such as TLR2, TLR3 and TLR7 (6). As an important component of
non-specific immunity, TLR is regularly localized on the surface of
cells or internal vesicles, recognizing molecules with conserved
structures and stimulating the body to produce an immune response
that causes an inflammatory response. Therefore, we highly
hypothesized that mutations and abnormal aggregation of α-synuclein
might have a certain regulatory effect on the inflammatory state of
the body.
At present, emerging evidence exhibits that systemic
low-level inflammation can accelerate the development of
degenerative changes in the brain with increasing age (7), and most men and women aged over 65
years have increased serum IL-6, TNF and IL-18 levels (8,9). NLRP3
inflammasome, as a polymeric complex, can bind to caspase-1 and
affect the secretion of the inflammatory factor IL-1β. Many
experiments have proved that its expression is closely related to
the onset of age-related diseases, however, its role in the
pathogenesis of PD remains poorly understood. While autophagy, acts
as an intracellular, homeostatic mechanism, mediates the
inactivation or selective clearance of harmful substrates or
secondary material signals, such as damaged mitochondria. There is
increasing evidence that autophagy exerts vital effect on the
innate immune response. In addition, autophagy-related pathways are
implicated in the regulation of NLRP3 inflammasome activation
(10–12). It has been reported in the literature
that α-synuclein can promote immune activation in PD patients.
Therefore, this study aimed to investigate the mechanism of
α-synuclein in affecting the progression of PD through
autophagy-related pathways.
Materials and methods
Study subjects
This study was approved by the Medical Ethics
Committee of the Third Affiliated Hospital of Qiqihar Medical
University (Qiqihar, China), and the experiment was conducted in
accordance with the Guide for the Care and Use of Experimental
Animals. Peripheral blood samples of 40 PD patients admitted to the
department of neurology of the hospital from March 2018 to January
2019 were collected. Inclusion criteria: Patients with the
characteristic progressive progression of the disease and 2 or more
typical symptoms (including static tremor, sluggish movement,
rigidity, postural instability) were included, while those with
other neurological tumors were excluded. Further 40 healthy normal
people who participated in physical examination were enrolled in
the control group. They were matched with PD patients according to
age, sex and education level, and those with traumatic brain
injury, suspected degenerative disease with Parkinson's disease,
Parkinson-related diseases (such as Lewy body dementia and
Alzheimer's disease), or severe organic or systemic diseases were
excluded. All the study subjects were informed of the purpose and
the protocol of the study before the blood was drawn, and written
informed consent was obtained.
Enzyme-linked immunosorbent assay
(ELISA) for the detection of plasma oligomerized, phosphorylated
α-synuclein and IL-1β
Detection method of oligomerized α-synuclein: The
detection method was carried out in accordance with the established
detection method (13). An amount of
100 µl of 3D5 monoclonal antibody (Santa Cruz Biotechnology, Inc.,
sc-47696) with a final concentration of 1 µg/ml was added to each
well of the 96-well plate, incubated at 37°C for 2 h for coating,
and washed with PBST after culturing overnight at 4°C. Then each
well was incubated at 37°C for 2 h with 200 µl of 10% BSA sealant
and then washed with PBST. Then recombinant human α-synuclein
(rPeptide, s-1005-2) diluted by multiple ratio (concentrations of
0.5, 0.25, 0.125, 0.0625, 0.03125, and 0 mol/l, respectively) was
added with the sample to be tested into each well, incubated at
37°C for 2 h before rinsing with PBST. Then, 100 µl of 1 mg/l
avidin (Solarbio, A8280) was added, and the cells were incubated
for 2 h at 37°C and washed with PBST. Diluted human avatin-labeled
alkaline phosphatase (Solarbio, k0068r-ap) was added, incubated at
37°C for 1 h and then washed with PBST. Finally, 100 µl pNPP was
added to each well, and the color was developed at 37°C for 30 min,
and the absorbance was measured using a 405 nm wavelength
microplate reader (Thermo Fisher Scientific, Inc., Multiskan FC).
Three replicate wells were set for each sample and the experiment
was performed three times.
Detection method of phosphorylated α-synuclein:
α-synuclein (p-Ser129) (Abcam, ab51253) monoclonal antibody (100
µl) with a final concentration of 0.1 mg/l was added to each well
of the 96-well plate, and the remaining steps were the same as
stated above.
Detection method of IL-1β, IL-6 and TGF-α: The
collected peripheral blood of the patient was allowed to stand at
room temperature for 40 min, and the serum was precipitated and
centrifuged at 1,000 × g for 10 min at 4°C. After centrifugation,
the light yellow transparent serum from the upper layer was
collected and stored frozen at −80°C in a refrigerator for later
use. Then the expression levels were measured using IL-1β
(ProteinTech Group, Inc., KE00021), IL-6 (ProteinTech Group, Inc.,
KE00007) and TGF-α (Qiaoyu Biotechnology, QY-MB10203) ELISA kits,
and the procedures were in accordance with the kit
instructions.
Primary cell isolation and
culture
Neonatal 3-4d C57BL/6 mice were sacrificed by spinal
dislocation, and the heads of the mice were placed on ice to
isolate the cerebral cortex. The primary cell separation method was
established as previously described (13). After the cerebral cortex was removed,
0.25% trypsin (Beyotime, C0201) was used for digestion and cell
suspension. Then, 90% Dulbecco's modified Eagle's medium: Nutrient
Mixture F-12 (DMEM/F-12) (HyClone, sh30023.01b) +10% fetal bovine
serum (FBS) (HyClone, Sv30087.01) +100 µ/ml penicillin +100 µ/ml
streptomycin (Beyotime, C0222) was cultured in a polylysine-coated
culture flask for 16 h. According to the different adhesion ability
between glial cells, the culture flask was put into a thermostatic
oscillator, oscillated at 37°C, 180 rpm, for 5 h, and the bottom
layer cells were collected as astrocytes with higher purity.
Finally, the cells were digested with 0.25% trypsin, subcultured in
a plurality of flasks at 1:2, and cultured to a density of 90%.
Drug treatment of cells
The number of astrocytes was adjusted to
1×107/ml, inoculated in the 6-well plate, with 3 ml per
well. The astrocytes were divided into control group and drug
treatment group when they grew to 80% density. The drug treatment
group was divided into low, medium and high concentration groups
according to different concentrations of the added drugs, which
were 5, 10 and 20 µg/ml of oligomerized α-synuclein. Judged by
whether autophagy inhibitors were added or not, the cells were
divided into a drug treatment group and an autophagy inhibition
group. The drug treatment group was treated with 10 µg/ml
oligomerized α-synuclein, while the autophagy inhibition group was
added with 10 µg/ml oligomerized α-synuclein and 5 mM 3-MA (MCE,
hy-19312), and the culture was continued for 24 h.
Western blot for protein
detection
RIPA lysate (Beyotime, P0013C) was applied for cell
lysis and centrifugation at 15,000 × g for 15 min at 4°C to extract
cell protein. The protein concentration was detected by BCA assay
(Beyotime, P0012S) and gel electrophoresis was performed in 15%
SDS-PAGE. Then, the protein was transferred to PVDF membrane by
semi-dry transfer method, and 5% skim milk powder was adopted to
seal the membrane for 1 h. Then, NLRP3 (ProteinTech Group, Inc.,
19771-1-AP, 1:1000), caspase-1 (ProteinTech Group, Inc.,
22915-1-AP, 1:500), IL-1β (ProteinTech Group, Inc., 16806-1-AP,
1:1000) and Atg5 (ProteinTech Group, Inc., 10181-2-AP, 1:1000) were
added, respectively, with GAPDH (ProteinTech Group, Inc.,
60004-1-Ig, 1:5000) as the internal reference, triple rinsed with
0.1% PBST after incubation overnight at 4°C, adding corresponding
HRP-labeled goat anti-rabbit (or mouse) immunological secondary
antibody (ProteinTech Group, Inc., SA00001-1/2, 1:3000), incubated
for 1 h at room temperature, and washed 3 times with 0.1% PBST.
Finally, ECL chemiluminescence reagent (Beyotime, P0018FS) was
employed to develop, fix and photograph (Odyeesy, LI-COR) in the
dark, and then it was analyzed using the Corning Axygen gel imaging
system.
Statistical analysis
All the collected data were statistically analyzed
by SPSS 23.0. The measurement data were expressed as mean ±
standard deviation. Independent t-test was applied for comparison
between the two groups, and one-way ANOVA was used for comparison
among multiple groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
Elevated plasma inflammation-related
factor IL-1β in PD patients
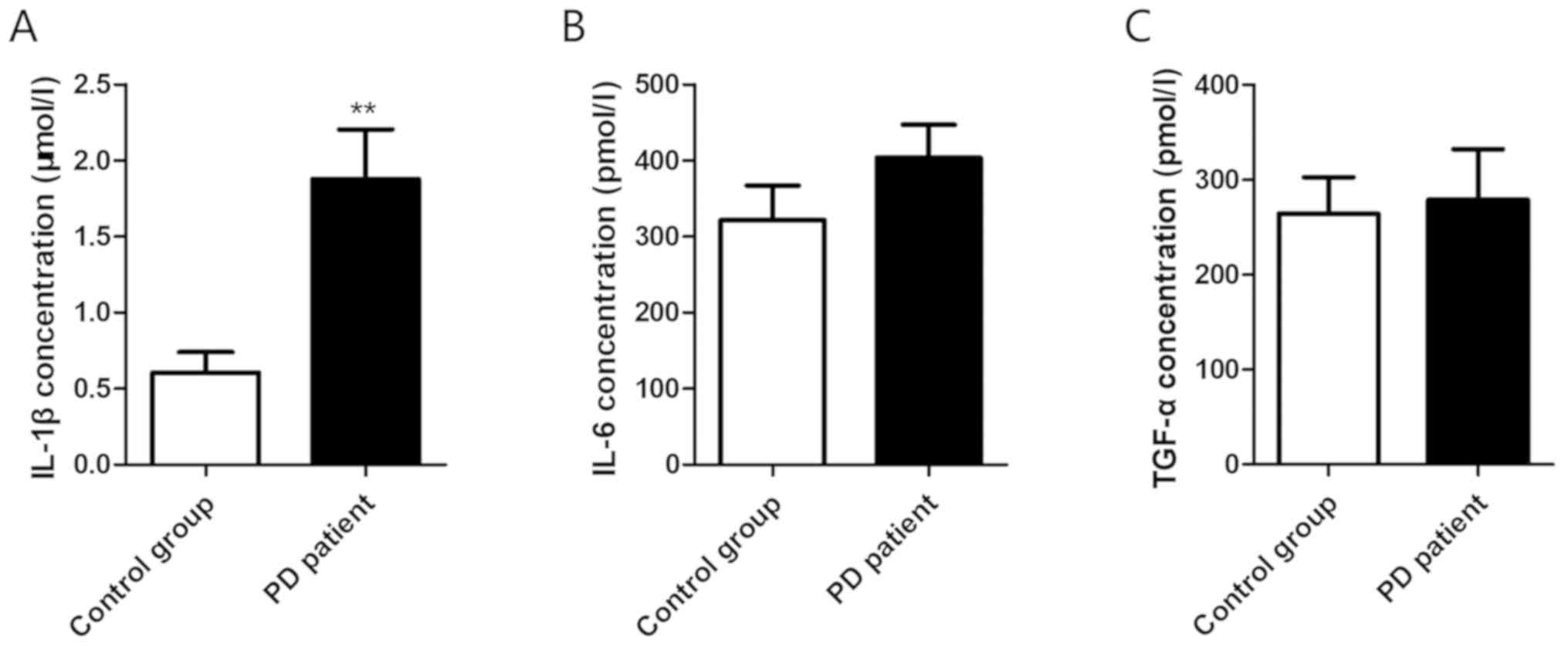
The levels of inflammatory factors in the plasma of
PD group and control group were detected by ELISA. It was found
that IL-1β was significantly increased in the patient group
(0.604±0.136 µmol/l vs. 1.876±0.327 µmol/l, P=0.002) (Fig. 1A), while the expression levels of
IL-6 (321.4±45.7 pmol/l vs. 403.8±43.19 pmol/l, P=0.206) (Fig. 1B) and TGF-α (264.3±38.2 pmol/l vs.
278.8±53.3 pmol/l, P=0.827) (Fig.
1C) did not identify any significant increase.
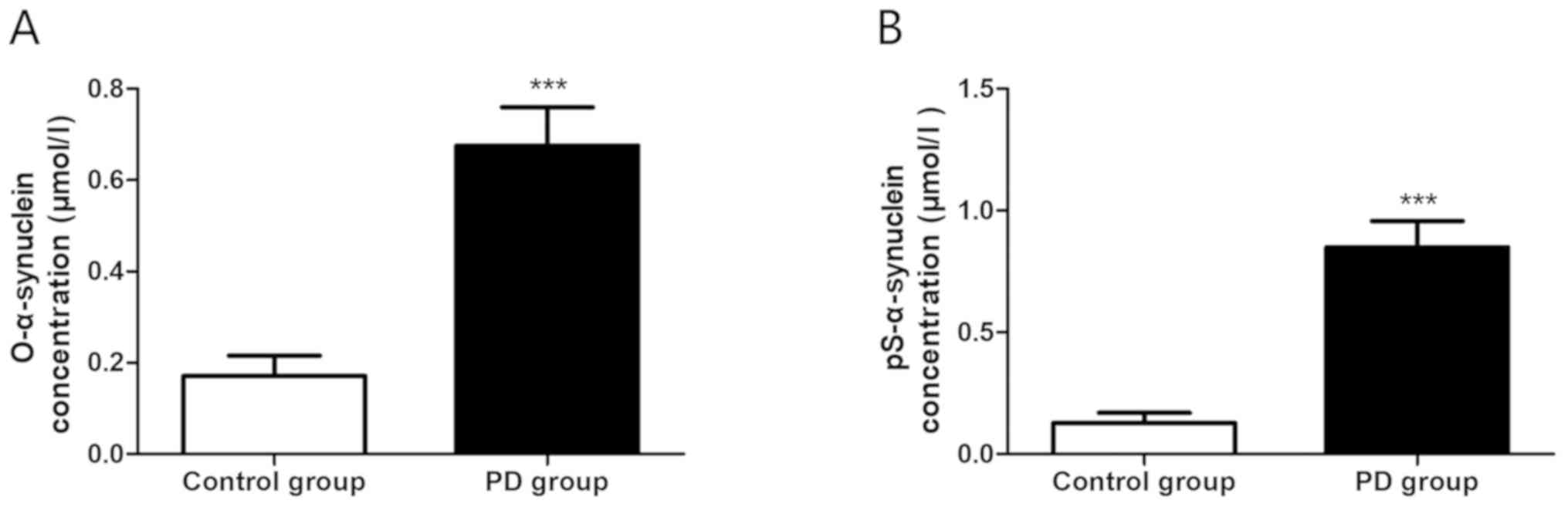
Increased plasma α-synuclein
expression in PD patients
Plasma α-synuclein levels in the PD group and the
control group were detected by ELISA, and it was observed that both
the expression levels of oligomerized α-synuclein (0.171±0.044
µmol/l vs. 0.676±0.083 µmol/l, P<0.0001) (Fig. 2A) and phosphorylated α-synuclein
(0.128±0.041 µmol/l vs. 0.849±0.108 µmol/l, P<0.0001) (Fig. 2B) in the PD group were markedly
increased, with statistically significant differences.
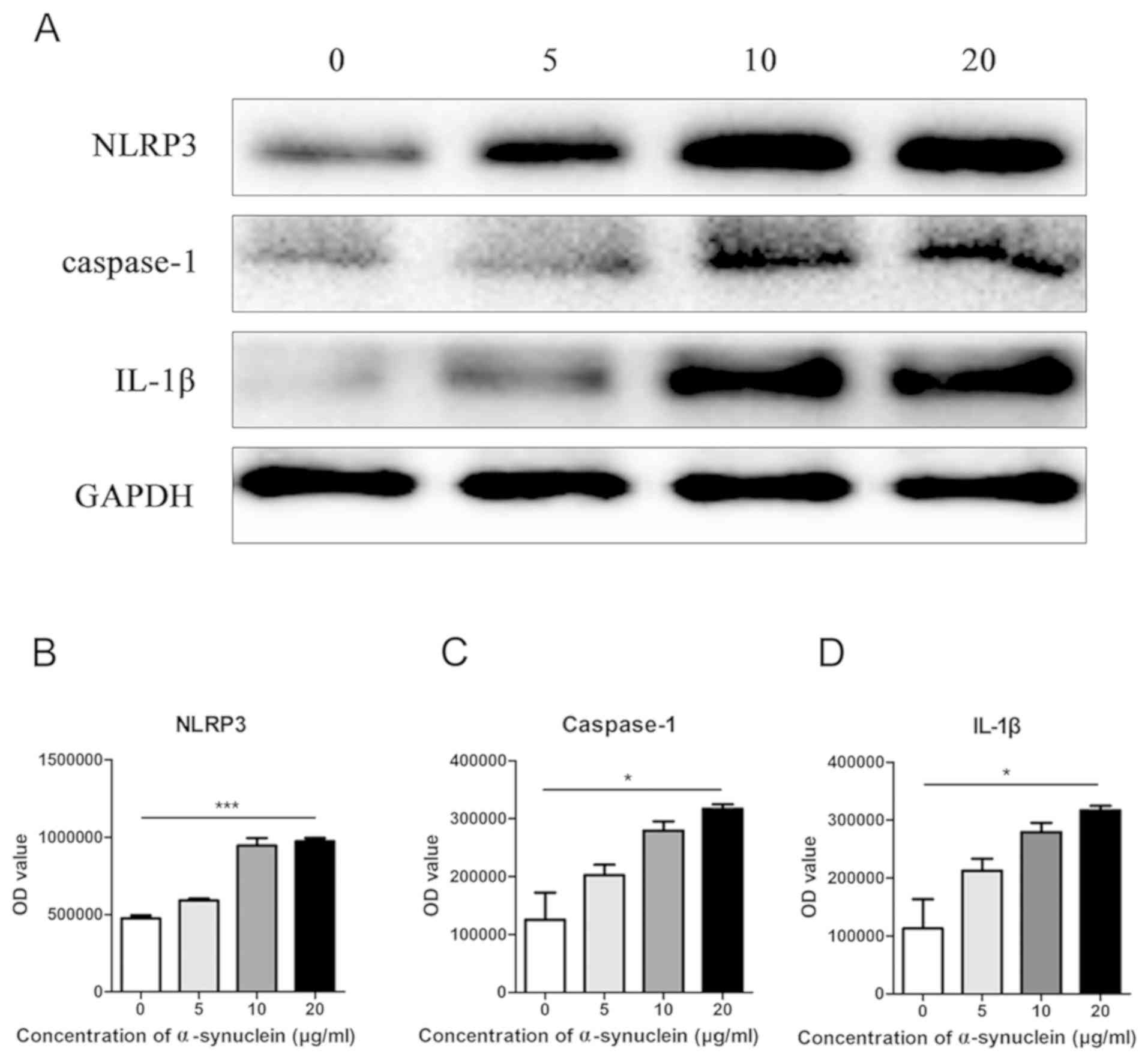
Oligomerized α-synuclein induction of
NLRP3 inflammasome and expression of related molecules in
astrocytes
Oligomerized α-synuclein with different
concentrations of 0, 5, 10, and 20 µg/ml were added to the
astrocytes of primary mice, respectively, and the expression level
of NLRP3 was detected by western blot. It was found that NLRP3
expression was elevated with the increase of drug concentration in
the cells (Fig. 3A and B). Further
detection of other inflammasome related molecular proteins revealed
a similar trend in caspase-1 and IL-1β (Fig. 3C and D).
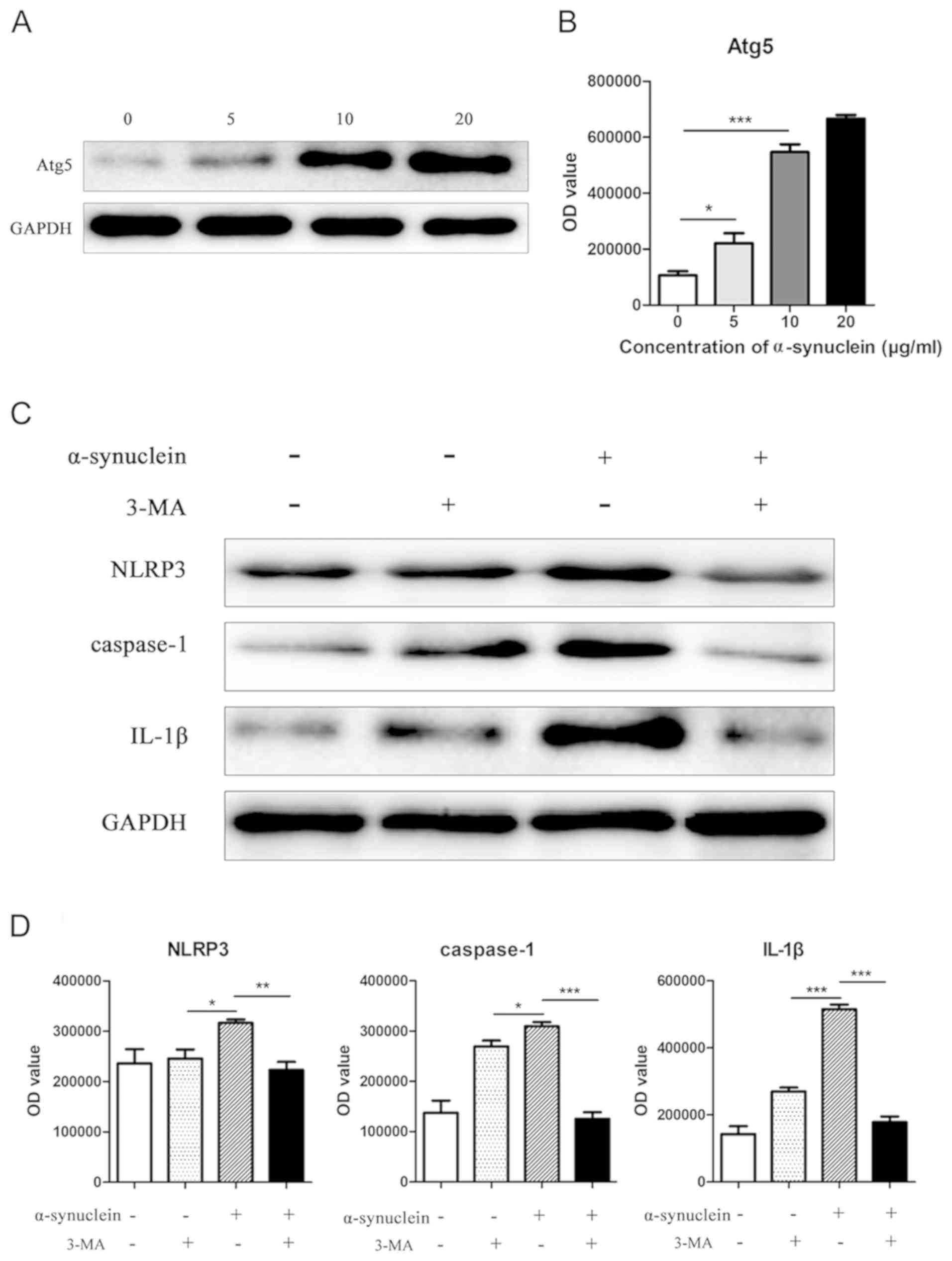
Oligomerized α-synuclein affects NLRP3
inflammation by affecting autophagy
After the addition of 0, 5, 10, and 20 µg/ml
oligomerized α-synuclein, the autophagy-related molecule Atg5 was
upregulated in astrocytes with increasing drug concentration
(Fig. 4A and B). To further explore
whether oligomerized α-synuclein causes NLRP3-related inflammatory
responses by affecting autophagy, we added oligomerized
α-synuclein/oligomerized α-synuclein + autophagy inhibitors (3-MA)
to cells. Western blot results showed that the expression levels of
NLRP3, caspase-1 and IL-1β expression were inhibited by 3-MA
(Fig. 4C and D), indicating that the
activation of NLRP3 inflammatory pathway by oligomerized
α-synuclein was mediated by autophagy.
Discussion
As a neurodegenerative disease, Parkinson's disease
has been extensively confirmed to be significantly associated with
age, but its specific pathogenesis remains a subject of
investigation. In recent years, studies have found a positive
correlation between age and inflammatory status (7), as serum levels of IL-6, TNF, and IL-18
are elevated in older people over the age of 65 (8,9). A
growing number of studies have found that with the increase of age,
a systemic mild inflammatory state can lead to the progression of
degenerative changes in the brain, thus forming the ‘hypothalamic
microinflammation’ theory, which mainly believes that under the
condition of aging and metabolic syndrome, the abnormal stimulation
of the hypothalamus activates the inflammatory signal pathway,
leading to the formation of chronic and stable inflammatory
background (14,15). The NLRP3 inflammasome acts as a
multimeric complex in the cytoplasm and serves as a platform for
caspase-1 activation and promotion of IL-1β maturation (16). It has been found that overexpression
is associated with many age-related inflammatory disorders
(10). The role of NLRP3
inflammasome in degenerative diseases may be related to their
responses to various danger-associated molecule patterns (DAMP),
including α-synuclein, extracellular adenosine triphosphate (ATP),
excessive glucose, ceramide, amyloid, urate and cholesterol
crystals (17). In this study, the
plasma levels of oligomerized and phosphorylated α-synuclein in
peripheral blood of patients with PD were both higher than those in
normal subjects. ELISA method applied to analyze the expression
level of inflammatory factors in plasma of PD patients, revealed
that the expression level of IL-1β was significantly increased,
while there was no significant difference in IL-6 and TGF-α. These
result suggests that α-synuclein in PD patients may activate NLRP3
inflammatory factors to release excess IL-1β, thus leaving the
patient in a state of chronic mild inflammation.
The characteristic pathological changes of PD are
Lewy body formation and dopaminergic neuropathy, of which the
latter often occurs in the late stage of the disease, when the
neurodegeneration rate can be as much as 70% (18). The formation of Lewy body is usually
much earlier than that of dopaminergic neuropathy, so the early
Lewy body formation is the best target for disease occurrence and
intervention. The main component of Lewy body is abnormally
aggregated α-synuclein. Studies have shown that α-synuclein can be
released from neurons into extracellular fluids, including
cerebrospinal fluid and plasma. In the current study, ELISA was
used to find a significant increase in plasma α-synuclein
expression in PD patients. α-synuclein normally exists in
presynaptic nerve terminals and mediates normal physiological
functions (19). In special cases,
its neurotoxic abnormal metabolites are released into the
extracellular matrix, which can activate glial cells and innate
immune responses, leading to neuronal damage (20–22).
While in turn, neurons can be internalized by neurotoxic
α-synuclein through endocytosis to avoid extensive damage, but
long-term pathological accumulation of α-synuclein will result in
apoptosis or degeneration of neuronal cells (21). A study demonstrated that the
overexpression of α-synuclein in rodent models (23) without obvious toxicity could impair
the release of the neurotransmitters glutamate and dopamine.
Through direct imaging of the synaptic vesicle circulation process,
it was found that the synaptic vesicle circulation pool was
significantly reduced, and ultrastructural analysis exhibited that
the density of synaptic vesicles in the active area was decreased.
Imaging further revealed the defect of reaggregation of synaptic
vesicles after endocytosis. Therefore, the increased level of
α-synuclein in PD patients will lead to a special physiological
defect in the process of synaptic vesicle regeneration, which
precedes the detectable neuropathology and provides a solid
theoretical basis for further exploration of the influence of
α-synuclein on the progress of PD disease. In the present study,
mouse astrocytes were extracted in the primary generation, and
different concentrations of oligomerized α-synuclein were added to
the culture medium. The activation of NLRP3-related signaling
pathway was detected by western blot, and significant activation of
NLRP3, caspase-1 and IL-1β was observed. Combined with a large
number of literature and the results of this study, it can be
concluded that α-synuclein plays an important regulatory role in
non-specific immune responses and is of great significance in the
pathogenesis of neurodegenerative diseases.
Some studies have analyzed the autopsy of brain
tissue of PD patients, and the results showed that there was
obvious microglial cell activation and glial cell invasion in the
regions with degeneration of dopaminergic neurons (24). The reason is that abnormal lipids,
proteins and metabolic stress factors accumulated in tissues and
neurons of PD patients (such as α-synuclein or mitochondrial
dysfunction) could send stress signals to small glial cells and
astrocytes in the environment (23).
Others (25) have reported that when
severe metabolic and lipid disorders occur in neurons, fatty acids
and other molecules can directly participate in the activation of
neuronal inflammation in brain tissue. In recent years, researchers
have found that α-synuclein activates microglia activation and
produces neuroinflammation in neurodegenerative diseases such as PD
and other related diseases. A study (26) displayed that the combined application
of wild-type α-synuclein and toll-like receptor stimulation (TLR)
in the microglia of primary mice could significantly increase the
secretion levels of IL-6, McP-1 and CXCL10. In addition, it showed
that microglia stimulated by α-synuclein could simultaneously
differentiate into M1/M2 intermediate phenotype. According to some
other studies, α-synuclein upregulates the expression of
immunosome-regulating receptors TLR2, TLR3, and TLR7 (5,6), and
activates NF-κB, a key transcription factor for inflammation.
Previous studies mainly focused in microglia and neuronal cells. In
this study, astrocytes from mouse brain tissue were extracted for
testing, and we found that α-synuclein could activate NLRP3
inflammasome through autophagy signaling pathway and promote the
occurrence of chronic inflammatory state, which was also consistent
with the previous results. Moreover, a study on traumatic brain
injury also revealed that trauma could cause severe brain
inflammation and significantly increase the level of α-synuclein in
the brain (27), increasing the risk
of PD, further validating the relationship between inflammation and
Parkinson's disease. Most of the studies were designed to
investigate the promotion of normal or abnormal structural
α-synuclein levels by different stimuli. However, there is still
scarce research on the influence of abnormally high expression of
oligomerized α-synuclein on brain tissue. In this study, combined
with previous literature reports, we found that there was a mutual
regulatory relationship between inflammation and α-synuclein.
The relationship between inflammation and PD has
long been elucidated. The 1918 influenza pandemic caused
encephalitis, and some patients further developed PD, which was
then called Von Ikonomo disease (28). Since then, a significant link between
inflammation and Parkinson's disease has been found by studying
other viral types, in which mice with H5N1 infection showed that
the virus caused microglia activation from peripheral infection to
the central nervous system, and α-synuclein polymerization and
death of dopaminergic neurons in the substantia nigra. Whereas,
previous studies believed that inflammatory response and activation
of immune cells are secondary processes in PD. In recent years,
studies have clarified that inflammation can directly participate
in the pathogenesis of PD. First, the expression levels of
pro-inflammatory factors, including TNF-α, IL-1β, IL-6 and TLR,
were increased in PD brain tissue. Second, injection of
PD-associated toxins (MPTP and 6-hydroxydopamine) in a mouse
experimental model could cause significant inflammation in the
brain tissue after injection and lead to degeneration of
dopaminergic neurons (29,30). However, intracerebral injection of
lipopolysaccharide (LPS) can induce intracellular accumulation of
insoluble polymeric α-synuclein (31), reducing the resistance of dopamine
neurons to MPTP and 6-hydroxydopamine (32), which is mainly related to the
activation of related proinflammatory factors, especially IL-1β
(33). Accorded with previous
results, the addition of IL-1β receptor antagonists significantly
reduced TNF-α and IFN-γ expression levels and abolished
LPS-mediated dopamine neuronal death below toxic doses (34). In another model of inflammation,
injection of the TLR3 agonator polyglucoside:polycysteine
[poly(I:C)] into the substantia nigra of mice led to persistent
inflammation, increased sensitivity to low doses of
6-hydroxydopamine, and interfered with proteins involved in
synaptic transmission and axonal transport (35). Compared with the normal control
group, the expression of TLR2 and TLR9 in the brain of PD patients
was increased, and the expression of TLR2 was positively correlated
with the accumulation of α-synuclein (36) in the body.
Furthermore, it has been reported that α-synuclein
stimulation can significantly activate inflammatory monocytes and
microglia in mice, and promote PD disease progression by producing
excessive inflammatory responses (5,6).
Moreover, studies have indicated that autophagy is closely related
to immune cells and may be involved in the pathogenesis of many
diseases, including neurodegeneration, metabolic disorders and
aging-related diseases. For example, autophagy is reported to
participate in the negative regulation of NLRP3 related signaling
pathways, and the product of mitochondrial reactive oxygen species
(37) and autophagosome dysfunction,
cathepsin B (38), can be used as a
‘second signal’ to stimulate the activation of NLRP3 inflammasome.
Previously, it was exhibited that the exposure of methamphetamine
(METH) to α-synuclein significantly reduced chaperon-mediated
autophagy activity, and the decreased lamp-2a expression could
alleviate neurotoxic reactions induced by α-synuclein (39). On the contrary, overexpression of the
heat shock protein 70 (HSP70) reduced abnormally aggregated
α-synuclein and inhibited apoptosis induced by METH exposure. A
study on PD (40) suggested that the
addition of autophagy small molecule inhibitors in cell experiments
and animal models could inhibit the formation of NLRP3-PYCARD-CASP1
complex, thereby delaying the progression of PD. In line with
previous studies, we found that the expression of autophagy
associated molecule Atg5 was significantly increased when
stimulated by α-synuclein, and autophagy inhibitor 3-MA inhibited
α-synuclein-mediated activation of NLRP3-related molecules.
α-synuclein, as an immunogenic substance, is usually an abnormal
metabolite of nerve cells, which activates the NLRP3 inflammasome
pathway through the autophagy-related molecule Atg5.
We did not yet carry out in vivo experiments
in mice, nor did we explore the activation of α-synuclein and
autophagy on the inflammatory response in vivo, or the
dynamic process of the changes of Lewy body and dopaminergic
neurons in the substantia nigra. Thus, further studies are
anticipated. By examing α-synuclein and inflammation-related
factors in peripheral blood of PD patients, this study not only
verified the activation effect of α-synuclein on NLRP3-related
molecules and autophagy in cells, but also found that the
application of autophagy inhibitor 3-MA could significantly inhibit
the inflammatory pathway, providing a solid basis for autophagy
inhibitors to be potential targets for PD treatment.
Acknowledgements
Not applicable.
Funding
This study was supported by Mandatory Scientific
Research Project of Qiqihar Science and Technology Bureau
(SFGG-201949).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XW, JC, DH and LD led the conception and design of
this study. XW, JC, XZ, LJ, YY and FG were responsible for the data
collection and analysis. DH, LD and YY were in charge of
interpreting the data and drafting the manuscript. XW and DH made
revision from critical perspective for important intellectual
content. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
the Third Affiliated Hospital of Qiqihar Medical University
(Qiqihar, China). Signed informed consents were obtained from the
patients and/or the guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Scheperjans F, Aho V, Pereira PA, Koskinen
K, Paulin L, Pekkonen E, Haapaniemi E, Kaakkola S, Eerola-Rautio J,
Pohja M, et al: Gut microbiota are related to Parkinson's disease
and clinical phenotype. Mov Disord. 30:350–358. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Poewe W, Seppi K, Tanner CM, Halliday GM,
Brundin P, Volkmann J, Schrag AE and Lang AE: Parkinson disease.
Nat Rev Dis Primers. 3:170132017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brettschneider J, Del Tredici K, Lee VM
and Trojanowski JQ: Spreading of pathology in neurodegenerative
diseases: A focus on human studies. Nat Rev Neurosci. 16:109–120.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Luk KC, Kehm V, Carroll J, Zhang B,
O'Brien P, Trojanowski JQ and Lee VM: Pathological α-synuclein
transmission initiates Parkinson-like neurodegeneration in
nontransgenic mice. Science. 338:949–953. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goldberg EL and Dixit VD: Drivers of
age-related inflammation and strategies for healthspan extension.
Immunol Rev. 265:63–74. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pedersen M, Bruunsgaard H, Weis N, Hendel
HW, Andreassen BU, Eldrup E, Dela F and Pedersen BK: Circulating
levels of TNF-alpha and IL-6-relation to truncal fat mass and
muscle mass in healthy elderly individuals and in patients with
type-2 diabetes. Mech Ageing Dev. 124:495–502. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ferrucci L, Corsi A, Lauretani F,
Bandinelli S, Bartali B, Taub DD, Guralnik JM and Longo DL: The
origins of age-related proinflammatory state. Blood. 105:2294–2299.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Patel MN, Carroll RG, Galván-Peña S, Mills
EL, Olden R, Triantafilou M, Wolf AI, Bryant CE, Triantafilou K and
Masters SL: Inflammasome priming in sterile inflammatory disease.
Trends Mol Med. 23:165–180. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lu A, Magupalli VG, Ruan J, Yin Q,
Atianand MK, Vos MR, Schröder GF, Fitzgerald KA, Wu H and Egelman
EH: Unified polymerization mechanism for the assembly of
ASC-dependent inflammasomes. Cell. 156:1193–1206. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Guo C, Xie S, Chi Z, Zhang J, Liu Y, Zhang
L, Zheng M, Zhang X, Xia D, Ke Y, et al: Bile acids control
inflammation and metabolic disorder through inhibition of NLRP3
inflammasome. Immunity. 45:9442016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Matsuo K, Cheng A, Yabuki Y, Takahata I,
Miyachi H and Fukunaga K: Inhibition of MPTP-induced α-synuclein
oligomerization by fatty acid-binding protein 3 ligand in
MPTP-treated mice. Neuropharmacology. 150:164–174. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang Y, Kim MS, Jia B, Yan J,
Zuniga-Hertz JP, Han C and Cai D: Hypothalamic stem cells control
ageing speed partly through exosomal miRNAs. Nature. 548:52–57.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang G, Li J, Purkayastha S, Tang Y,
Zhang H, Yin Y, Li B, Liu G and Cai D: Hypothalamic programming of
systemic ageing involving IKK-β, NF-κB and GnRH. Nature.
497:211–216. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lamkanfi M and Dixit VM: Mechanisms and
functions of inflammasomes. Cell. 157:1013–1022. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Youm YH, Grant RW, McCabe LR, Albarado DC,
Nguyen KY, Ravussin A, Pistell P, Newman S, Carter R, Laque A, et
al: Canonical Nlrp3 inflammasome links systemic low-grade
inflammation to functional decline in aging. Cell Metab.
18:519–532. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gershanik OS: Past, present, and future of
Parkinson's disease. Mov Disord. 32:12632017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Norris EH, Giasson BI and Lee VM:
Alpha-synuclein: Normal function and role in neurodegenerative
diseases. Curr Top Dev Biol. 60:17–54. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ma QL, Chan P, Yoshii M and Uéda K:
Alpha-synuclein aggregation and neurodegenerative diseases. J
Alzheimers Dis. 5:139–148. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lee HJ, Suk JE, Patrick C, Bae EJ, Cho JH,
Rho S, Hwang D, Masliah E and Lee SJ: Direct transfer of
α-synuclein from neuron to astroglia causes inflammatory responses
in synucleinopathies. J Biol Chem. 285:9262–9272. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ma D, Jin S, Li E, Doi Y, Parajuli B, Noda
M, Sonobe Y, Mizuno T and Suzumura A: The neurotoxic effect of
astrocytes activated with toll-like receptor ligands. J
Neuroimmunol. 254:10–18. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Abdelkarim H, Marshall MS, Scesa G, Smith
RA, Rue E, Marshall J, Elackattu V, Stoskute M, Issa Y, Santos M,
et al: α-Synuclein interacts directly but reversibly with
psychosine: Implications for α-synucleinopathies. Sci Rep.
8:124622018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mori F, Tanji K, Yoshimoto M, Takahashi H
and Wakabayashi K: Demonstration of α-synuclein immunoreactivity in
neuronal and glial cytoplasm in normal human brain tissue using
proteinase K and formic acid pretreatment. Exp Neurol. 176:98–104.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Deleidi M and Isacson O: Viral and
inflammatory triggers of neurodegenerative diseases. Sci Transl
Med. 4:121ps32012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
McGeer PL, Itagaki S, Boyes BE and McGeer
EG: Reactive microglia are positive for HLA-DR in the substantia
nigra of Parkinson's and Alzheimer's disease brains. Neurology.
38:1285–1291. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Park J, Wang Q, Wu Q, Mao-Draayer Y and
Kim CH4: Bidirectional regulatory potentials of short-chain fatty
acids and their G-protein-coupled receptors in autoimmune
neuroinflammation. Sci Rep. 9:88372019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Roodveldt C, Labrador-Garrido A,
Gonzalez-Rey E, Lachaud CC, Guilliams T, Fernandez-Montesinos R,
Benitez-Rondan A, Robledo G, Hmadcha A, Delgado M, et al:
Preconditioning of microglia by α-synuclein strongly affects the
response induced by toll-like receptor (TLR) stimulation. PLoS One.
8:e791602013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Uryu K, Chen XH, Martinez D, Browne KD,
Johnson VE, Graham DI, Lee VM, Trojanowski JQ and Smith DH:
Multiple proteins implicated in neurodegenerative diseases
accumulate in axons after brain trauma in humans. Exp Neurol.
208:185–192. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Picard F, de Saint-Martin A, Salmon E,
Hirsch E and Marescaux C: Postencephalitic stereotyped involuntary
movements responsive to L-Dopa. Mov Disord. 11:567–570. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ros-Bernal F, Hunot S, Herrero MT,
Parnadeau S, Corvol JC, Lu L, Alvarez-Fischer D, Carrillo-de
Sauvage MA, Saurini F, Coussieu C, et al: Microglial glucocorticoid
receptors play a pivotal role in regulating dopaminergic
neurodegeneration in parkinsonism. Proc Natl Acad Sci USA.
108:6632–6637. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Depino AM, Earl C, Kaczmarczyk E, Ferrari
C, Besedovsky H, del Rey A, Pitossi FJ and Oertel WH: Microglial
activation with atypical proinflammatory cytokine expression in a
rat model of Parkinson's disease. Eur J Neurosci. 18:2731–2742.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gao HM, Kotzbauer PT, Uryu K, Leight S,
Trojanowski JQ and Lee VM: Neuroinflammation and
oxidation/nitration of alpha-synuclein linked to dopaminergic
neurodegeneration. J Neurosci. 28:7687–7698. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fan LW, Tien LT, Lin RC, Simpson KL,
Rhodes PG and Cai Z: Neonatal exposure to lipopolysaccharide
enhances vulnerability of nigrostriatal dopaminergic neurons to
rotenone neurotoxicity in later life. Neurobiol Dis. 44:304–316.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pott Godoy MC, Tarelli R, Ferrari CC,
Sarchi MI and Pitossi FJ: Central and systemic IL-1 exacerbates
neurodegeneration and motor symptoms in a model of Parkinson's
disease. Brain. 131:1880–1894. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Koprich JB, Reske-Nielsen C, Mithal P and
Isacson O: Neuroinflammation mediated by IL-1beta increases
susceptibility of dopamine neurons to degeneration in an animal
model of Parkinson's disease. J Neuroinflammation. 5:82008.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Deleidi M, Hallett PJ, Koprich JB, Chung
CY and Isacson O: The Toll-like receptor-3 agonist
polyinosinic:polycytidylic acid triggers nigrostriatal dopaminergic
degeneration. J Neurosci. 30:16091–16101. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Dzamko N, Gysbers A, Perera G, Bahar A,
Shankar A, Gao J, Fu Y and Halliday GM: Toll-like receptor 2 is
increased in neurons in Parkinson's disease brain and may
contribute to alpha-synuclein pathology. Acta Neuropathol.
133:303–319. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhong Z, Umemura A, Sanchez-Lopez E, Liang
S, Shalapour S, Wong J, He F, Boassa D, Perkins G, Ali SR, et al:
NF-κB restricts inflammasome activation via elimination of damaged
mitochondria. Cell. 164:896–910. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Qiao C, Yin N, Gu HY, Zhu JL, Ding JH, Lu
M and Hu G: Atp13a2 deficiency aggravates astrocyte-mediated
neuroinflammation via NLRP3 inflammasome activation. CNS Neurosci
Ther. 22:451–460. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sun L, Lian Y, Ding J, Meng Y, Li C, Chen
L and Qiu P: The role of chaperone-mediated autophagy in
neurotoxicity induced by alpha-synuclein after methamphetamine
exposure. Brain Behav. 9:e013522019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Han X, Sun S, Sun Y, Song Q, Zhu J, Song
N, Chen M, Sun T, Xia M, Ding J, et al: Small molecule-driven NLRP3
inflammation inhibition via interplay between ubiquitination and
autophagy: Implications for Parkinson disease. Autophagy.
15:1860–1881. 2019. View Article : Google Scholar : PubMed/NCBI
|