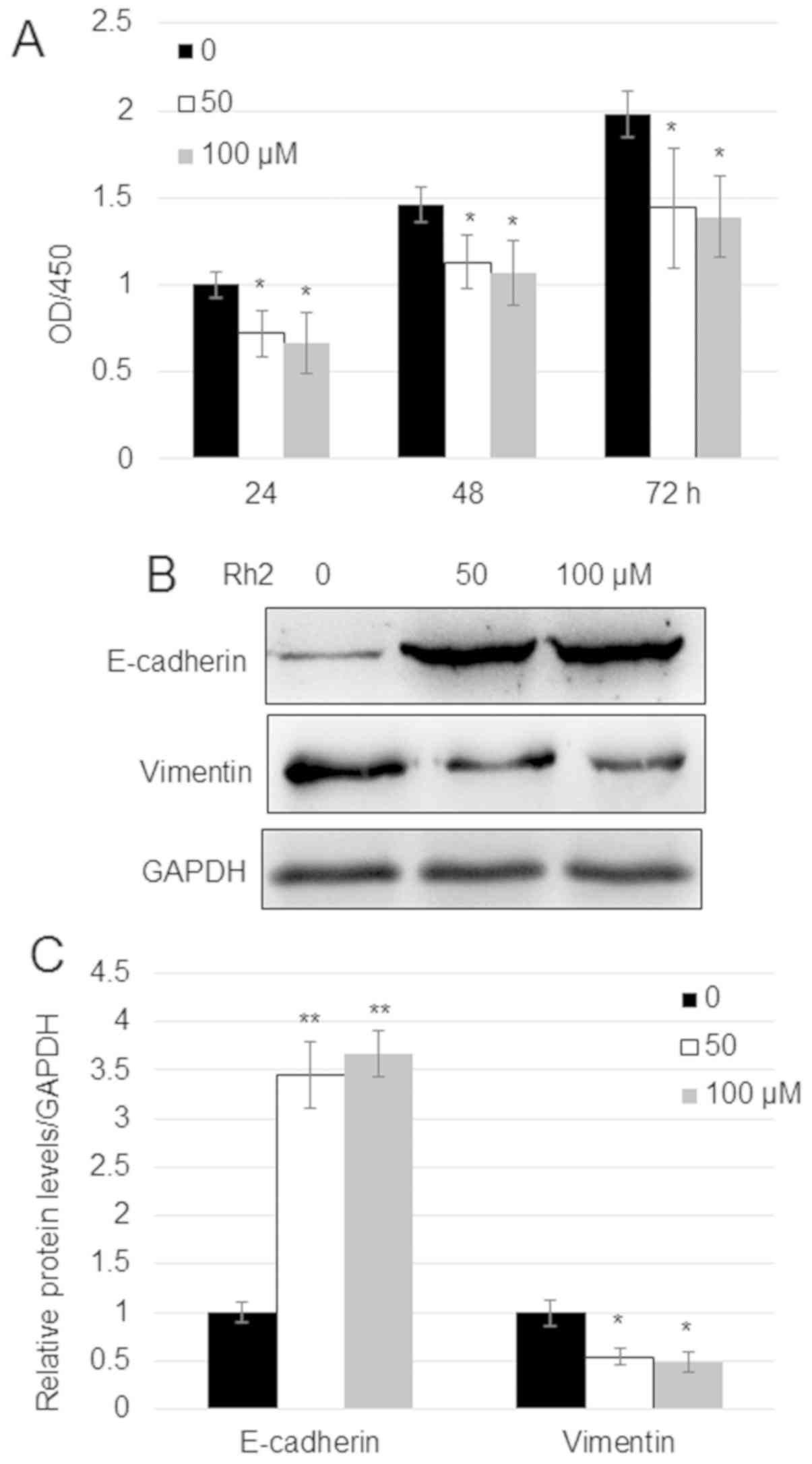
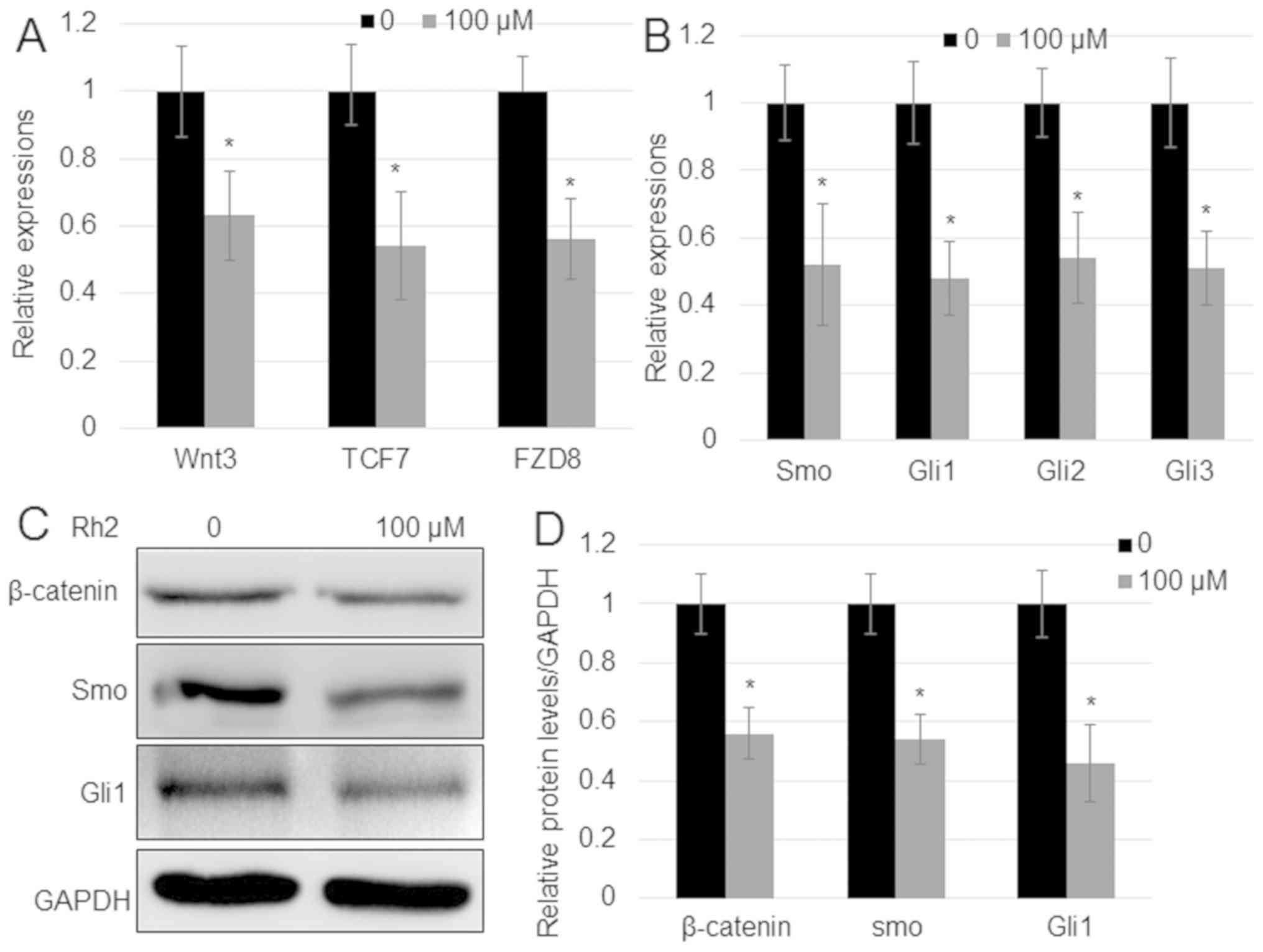
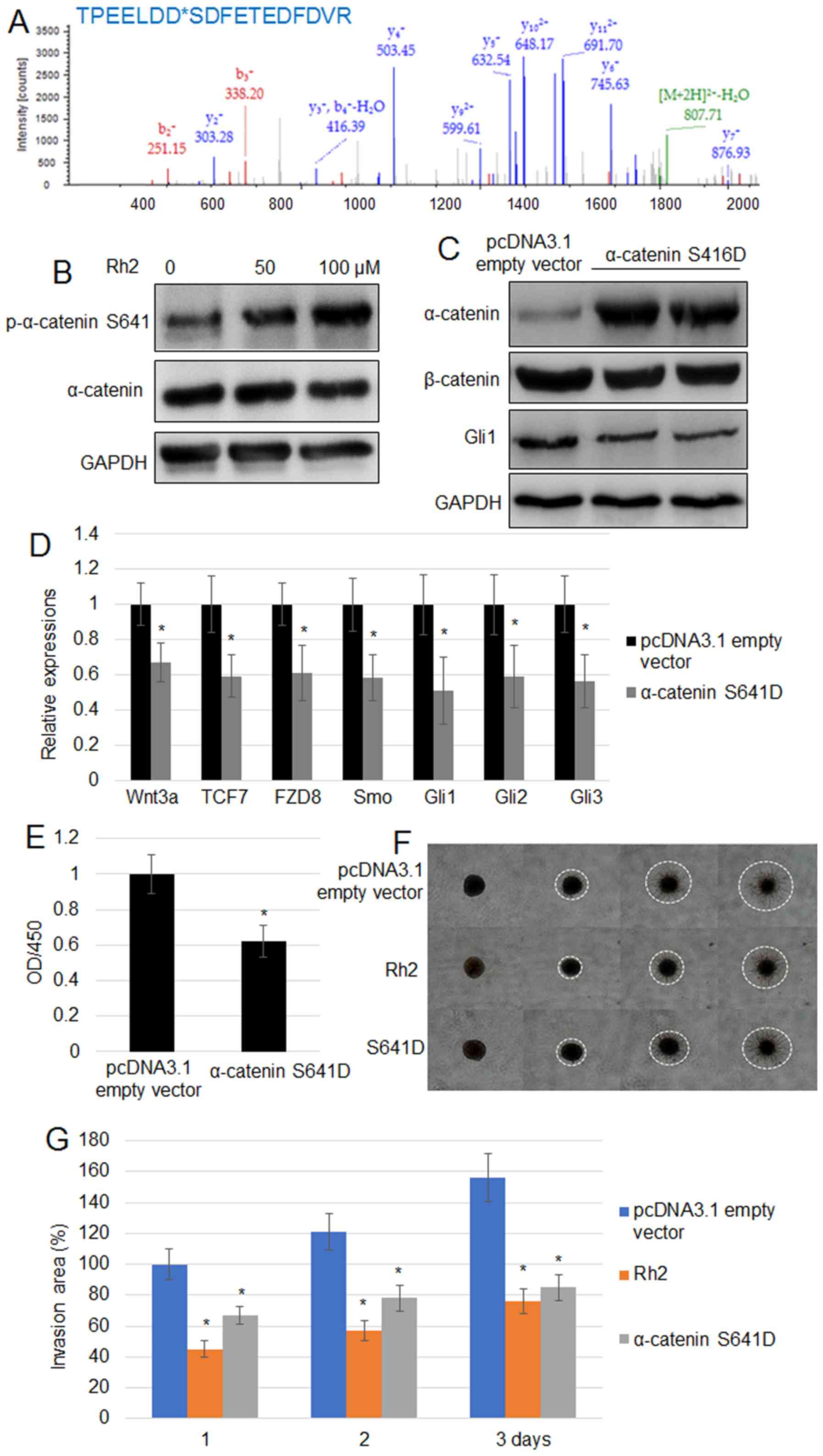
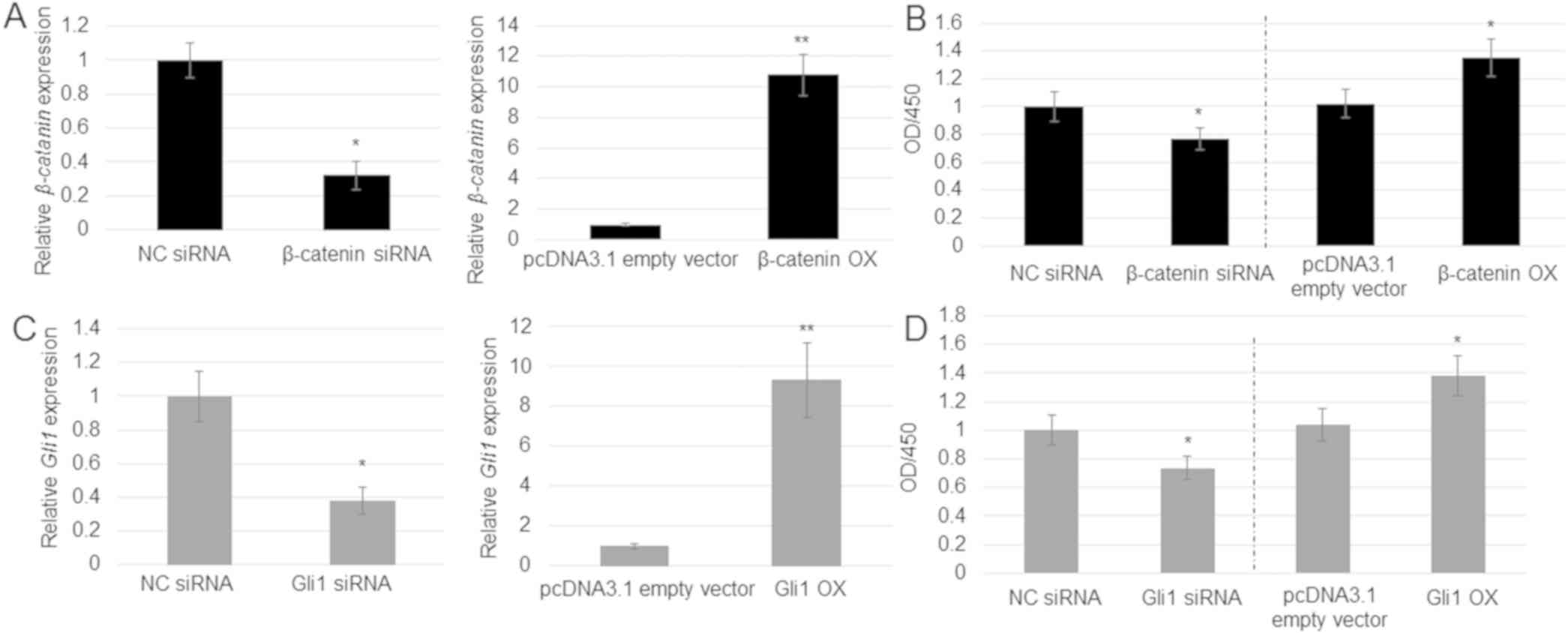
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Chan BA and Hughes BG: Targeted therapy
for non-small cell lung cancer: Current standards and the promise
of the future. Transl Lung Cancer Res. 4:36–54. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Roberts PJ, Stinchcombe TE, Der CJ and
Socinski MA: Personalized medicine in non-small-cell lung cancer:
Is KRAS a useful marker in selecting patients for epidermal growth
factor receptor-targeted therapy? J Clin Oncol. 28:4769–4777.
2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
de Looff M, de Jong S and Kruyt FA: The
role of Src in TRAIL signaling in non-small cell lung cancer cells.
Proceedings: AACR Annual Meeting 2018. April 14-18, 2018. Cancer
Res 78: 4377, 2018.
|
|
5
|
Hanahan D and Weinberg RA: The hallmarks
of cancer. Cell. 100:57–70. 2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Arbiser JL, Bonner MY and Gilbert LC:
Targeting the duality of cancer. NPJ Precision Oncol 1: pii: 23,
017.
|
|
7
|
Pfeffer CM and Singh ATK: Apoptosis: A
target for anticancer therapy. Int J Mol Sci.
19(448)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Rastogi V, Santiago-Moreno J and Doré S:
Ginseng: A promising neuroprotective strategy in stroke. Front Cell
Neurosci. 8(457)2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kiefer D and Pantuso T: Panax ginseng. Am
Fam Physician. 68:1539–1542. 2003.PubMed/NCBI
|
|
10
|
Helms S: Cancer prevention and
therapeutics: Panax ginseng. Altern Med Rev. 9:259–274.
2004.PubMed/NCBI
|
|
11
|
Fei XF, Wang BX, Tashiro S, Li TJ, Ma JS
and Ikejima T: Apoptotic effects of ginsenoside Rh2 on human
malignant melanoma A375-S2 cells. Acta Pharmacol Sin. 23:315–322.
2002.PubMed/NCBI
|
|
12
|
Huang Y, Huang H, Han Z, Li W, Mai Z and
Yuan R: Ginsenoside Rh2 inhibits angiogenesis in prostate cancer by
targeting CNNM1. J Nanosci Nanotechnol. 19:1942–1950.
2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chen WW, Huang YF, Hu ZB, Liu YM, Xiao HX,
Liu DB and Zhuang YZ: Microarray analysis of altered long
non-coding RNA expression profile in liver cancer cells treated by
ginsenoside Rh2. J Asian Nat Prod Res. 21:742–753. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Lee H, Lee S, Jeong D and Kim SJ:
Ginsenoside Rh2 epigenetically regulates cell-mediated immune
pathway to inhibit proliferation of MCF-7 breast cancer cells. J
Ginseng Res. 42:455–462. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li H, Huang N, Zhu W, Wu J, Yang X, Teng
W, Tian J, Fang Z, Luo Y, Chen M and Li Y: Modulation the crosstalk
between tumor-associated macrophages and non-small cell lung cancer
to inhibit tumor migration and invasion by ginsenoside Rh2. BMC
Cancer. 18(579)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang Y, Xu H, Lu Z, Yu X, Lv C, Tian Y and
Sui D: Pseudo-ginsenoside Rh2 induces A549 cells apoptosis via the
Ras/Raf/ERK/p53 pathway. Exp Ther Med. 15:4916–4924.
2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zhan T, Rindtorff N and Boutros M: Wnt
signaling in cancer. Oncogene. 36:1461–1473. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bushman W: Hedgehog signaling in
development and cancer. In: Prostate cancer Springer, pp107-118,
2007.
|
|
19
|
Shtutman M, Zhurinsky J, Simcha I,
Albanese C, D'Amico M, Pestell R and Ben-Ze'ev A: The cyclin D1
gene is a target of the beta-catenin/LEF-1 pathway. Proc Natl Acad
Sci USA. 96:5522–5527. 1999.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Moon RT, Kohn AD, De Ferrari GV and Kaykas
A: WNT and beta-catenin signalling: Diseases and therapies. Nat Rev
Genet. 5:691–701. 2004.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Clevers H: Wnt/β-catenin signaling in
development and disease. Cell. 127:469–480. 2006.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Brack AS, Conboy MJ, Roy S, Lee M, Kuo CJ,
Keller C and Rando TA: Increased Wnt signaling during aging alters
muscle stem cell fate and increases fibrosis. Science. 317:807–810.
2007.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Reya T and Clevers H: Wnt signalling in
stem cells and cancer. Nature. 434:843–850. 2005.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Xin M, Ji X, De La Cruz LK, Thareja S and
Wang B: Strategies to target the Hedgehog signaling pathway for
cancer therapy. Med Res Rev. 38:870–913. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Beauchamp EM, Ringer L, Bulut G, Sajwan
KP, Hall MD, Lee YC, Peaceman D, Ozdemirli M, Rodriguez O,
Macdonald TJ, et al: Arsenic trioxide inhibits human cancer cell
growth and tumor development in mice by blocking Hedgehog/GLI
pathway. J Clin Invest. 121:148–160. 2011.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wang K, Pan L, Che X, Cui D and Li C:
Sonic Hedgehog/GLI1 signaling pathway inhibition restricts cell
migration and invasion in human gliomas. Neurol Res. 32:975–980.
2010.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Huangfu D and Anderson KV: Signaling from
Smo to Ci/Gli: Conservation and divergence of Hedgehog pathways
from Drosophila to vertebrates. Development. 133:3–14.
2006.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Rohatgi R, Milenkovic L and Scott MP:
Patched1 regulates hedgehog signaling at the primary cilium.
Science. 317:372–376. 2007.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Larsen MR, Thigholm TE, Jensen ON,
Roepstorff P and Jørgensen TJ: Highly selective enrichment of
phosphorylated peptides from peptide mixtures using titanium
dioxide microcolumns. Mol Cell Proteomics. 4:873–886.
2005.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Xuan YH, Huang BB, Tian HS, Chi LS, Duan
YM, Wang X, Zhu ZX, Cai WH, Zhu YT, Wei TM, et al: High-glucose
inhibits human fibroblast cell migration in wound healing via
repression of bFGF-regulating JNK phosphorylation. PLoS One.
9(e108182)2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Tripathy A, Thakurela S, Sahu MK,
Uthanasingh K, Behera M, Ajay AK and Kumari R: The molecular
connection of histopathological heterogeneity in hepatocellular
carcinoma: A role of Wnt and Hedgehog signaling pathways. PLoS One.
13(e0208194)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Neal RD, Hamilton W and Rogers TK: Lung
cancer. BMJ. 349(g6560)2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Piao HL, Yuan Y, Wang M, Sun Y, Liang H
and Ma L: α-catenin acts as a tumour suppressor in
E-cadherin-negative basal-like breast cancer by inhibiting NF-κB
signalling. Nat Cell Biol. 16:245–254. 2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wang Y, Lin P, Wang Q, Zheng M and Pang L:
Wnt3a-regulated TCF4/β-catenin complex directly activates the key
Hedgehog signalling genes Smo and GLI1. Exp Ther Med. 16:2101–2107.
2018.PubMed/NCBI View Article : Google Scholar
|