Introduction
Vascular smooth muscle cells (VSMCs) are vital cells
that maintain normal physiological functions of blood vessels.
Under normal conditions, VSMCs are non-proliferative contractile
type. However, they are stimulated to proliferate in the presence
of vascular injury and some bioactive substances (i.e. nitric oxide
products, angiotensin II and platelet growth factor). Proliferative
VSMCs synthesize and secret vasoactive substances and growth
factors, thus leading to thickening of blood vessels, luminal
stenosis and vascular remodeling (1). Phenotype conversion and proliferation
stimulation of VSMCs are the key factors in the development of
vascular proliferative diseases, such as hypertension and
atherosclerosis (2,3).
Long non-coding RNA (lncRNA) is a class of ncRNAs
synthesized by RNA polymerase II over 200 nucleotides long. In
generally, lncRNAs are classified into five subtypes, namely
antisense lncRNAs, intronic transcripts, large intergenic noncoding
RNAs, promoter-associated lncRNAs and UTR-associated lncRNAs
(4,5). It is reported that certain lncRNAs are
able to influence the phenotypes of VSMCs and further affect the
occurrence of atherosclerosis (6,7). LncRNA
UCA1 (urothelial carcinoma antigen 1) was initially discovered by
Wang et al (8). UCA1 locates
on 19p13.12, and is commonly expressed in embryonic tissues. Han
et al (9) found that UCA1 is
highly expressed in colorectal cancer tissues, which is closely
related to tumor size, depth of invasion and poor tissue
differentiation. A recent study demonstrated the ability of UCA1 in
mediating the proliferative and migratory capacities of VSMCs
(10).
Matrix metalloproteinases (MMPs), known as matrix
metalloproteinases, are calcium-dependent zinc-containing
endopeptidases. They are capable of degrading components of the
extracellular matrix (ECM), including laminin, collagen, and
fibronectin (11). Currently, at
least 26 members of the MMPs family have been discovered. Among
them, MMP9 is closely related to cerebrovascular system (12). MMP9, also known as gelatinase B or 92
kDa gelatinase, locates on 16q 11.2-13.1 and contains 13 exons. The
basic structure of MMP9 consists of a signal peptide region,
amino-terminal propeptide, the zinc-binding catalytic domain, the
carboxyl-terminal hemopexin-like domain and the hinge region
(13). A relevant study has
demonstrated that MMP9 downregulation suppressed chlamydia
pneumonia infection-induced migration of VSMCs (14). This study mainly investigated the
potential function of UCA1 in ox-LDL-treated cellular phenotype
changes of VSMCs through regulating MMP9, thus providing novel
directions in the treatment of vascular diseases.
Materials and methods
Cell culture and induction
VSMCs were provided by Cell Bank (Shanghai, China).
Cells were cultured in Roswell Park Memorial Institute 1640
(RPMI-1640) (HyClone) containing 10% fetal bovine serum (FBS)
(Gibco; Thermo Fisher Scientific, Inc.), 100 µg/ml penicillin and
0.1 µg/ml streptomycin, at 37˚C, in a 5% CO2 incubator.
Fourth to fifth generation VSMCs were selected for treatment with
ox-LDL.
Cell transfection
Cells were inoculated in 6-well plates with
2x105 cells per well. At 80% confluence, cells were
transfected using Lipofactamine 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.). Medium containing 2 µg/ml puromycin was replaced
48 h later, and continued for 72 h of culture. Positive colonies
were selected and amplified for in vitro experiments.
Quantitative real-time polymerase
chain reaction (qRT-PCR)
Extraction of total RNA in cells was performed using
TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) and
subjected to reverse transcription. The extracted complementary
deoxyribose nucleic acid (cDNA) was applied for PCR using SYBR
Green method. Primer sequences were as follows: UCA1, forward:
5'-CTCTCCATTGGGTTCACCATTC-3' and reverse:
5'-GCGGCAGGTCTTAAGAGATGAG-3'; MMP9, forward:
5'-CGATGCCTGCAACGTGAAC-3' and reverse: 5'-AGAGCCGCTCCTCAAAGACC-3';
Glyceraldheyde 3-phosphate dehydrogenase (GAPDH), forward:
5'-TGAAGGTCGGAGTCAACGG-3' and reverse:
5'-CCTGGAAGATGGTGATGCG-3'.
Cell Counting Kit-8 (CCK-8)
Cells were seeded in a 96-well plate and cultured
overnight. Absorbance (A) at 490 nm was recorded at the appointed
time points using the CCK-8 kit (Dojindo Laboratories) for
depicting the viability curves.
Transwell migration assay
Cells transfected for 48 h were adjusted to the dose
of 1.0x105 cells/ml and subjected to serum starvation
for 12 h. Then, 200 µl/well suspension was applied to the upper
Transwell chamber (Merck KGaA). In the lower chamber, 700 µl of
medium containing 10% FBS was applied. After 48 h of incubation,
cells migrated to the lower chamber were subjected to fixation in
methanol for 15 min, crystal violet staining for 20 min and cell
counting using a microscope. Penetrating cells were counted in 5
randomly selected fields per sample.
Western blotting
Total protein was extracted from cells using
radioimmunoprecipitation assay (RIPA) and quantified by
bicinchoninic acid (BCA) method (Pierce; Thermo Fisher Scientific,
Inc.). Protein sample was loaded for electrophoresis and
transferred on polyvinylidene fluoride (PVDF) membranes (Merck
KGaA). Membranes were blocked in 5% skim milk for 2 h, and
subjected to incubation with primary and secondary antibodies.
Bands were exposed by electrochemiluminescence (ECL) and analyzed
by Image Software (National Institutes of Health).
Determination of subcellular
distribution
Cytoplasmic and nuclear RNAs were extracted using
the PARIS kit (Invitrogen; Thermo Fisher Scientific, Inc.) and
subjected to qRT-PCR. 18s was the internal reference of nucleus and
U1 was that of the cytoplasm.
RNA immunoprecipitation (RIP)
Cells were treated according to the procedures of
Millipore Magna RIPTM RNA-Binding Protein
Immunoprecipitation kit. Cell lysate was incubated with anti-EZH2
(enhancer of zeste homolog 2), or anti-IgG antibody at 4˚C for 6 h.
A protein-RNA complex was captured and digested with 0.5 mg/ml
proteinase K containing 0.1% SDS to extract RNA. The magnetic beads
were repeatedly washed with RIP washing buffer to remove
non-specific adsorption as much as possible. Finally, the extracted
RNA was subjected to mRNA level determination using qRT-PCR.
Chromatin immunoprecipitation
(ChIP)
Cells were subjected to 10 min cross-link with 1%
formaldehyde at room temperature into small fractions with 200-1000
bp. Subsequently, cells were lysed and sonicated for 30 min.
Finally, the sonicated lysate was immuno-precipitated with
anti-EZH2, anti-H3K27me3 or anti-IgG. Purified immunoprecipitated
chromatins were subjected to qRT-PCR.
Statistical analysis
Statistical Product and Service Solutions (SPSS)
20.0 (IBM Corp.) was used for data analyses. Data were expressed as
mean ± standard deviation. Intergroup differences were analyzed by
t-test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Downregulation of UCA1 in VSMCs
undergoing ox-LDL treatment
QRT-PCR data showed that UCA1 level was gradually
reduced after 50 and 100 mg/l ox-LDL treatment in VSMCs for 48 h
(Fig. 1A). With the prolongation of
100 mg/l ox-LDL treatment, UCA1 was downregulated at 24 and 48 h
(Fig. 1B). It is indicated that UCA1
was dose-dependently and time-dependently downregulated by ox-LDL
treatment. Transfection of pcDNA-UCA1 sufficiently upregulated UCA1
level in VSMCs, showing great transfection efficacy (Fig. 1C). CCK-8 assay showed increased
viability in VSMCs undergoing 100 mg/l ox-LDL treatment for 48 h,
which was reversed by transfection of pcDNA-UCA1 (Fig. 1D). Similarly, relative number of
migratory VSMCs increased by 100 mg/l ox-LDL treatment for 48 h,
and was further reduced after overexpression of UCA1 (Fig. 1E). It is suggested that UCA1
suppressed the proliferative and migratory abilities of VSMCs.
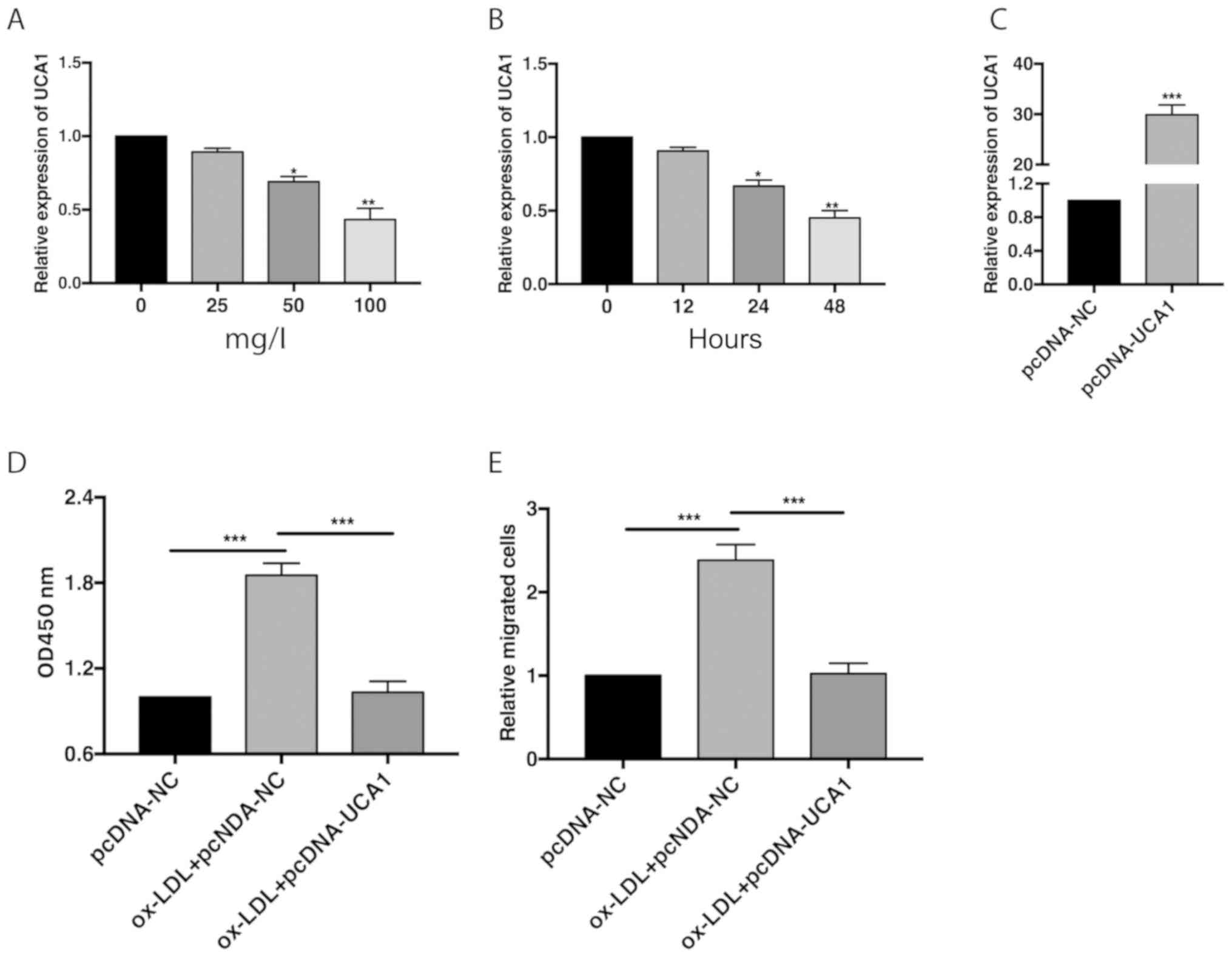 | Figure 1Downregulation of UCA1 in VSMCs
undergoing ox-LDL treatment. (A) Relative level of UCA1 in VSMCs
induced with 0, 25, 50 and 100 mg/l ox-LDL for 48 h. (B) Relative
level of UCA1 in VSMCs induced with 100 mg/l ox-LDL for 0, 12, 24
and 48 h. (C) Transfection efficacy of pcDNA-UCA1 in VSMCs. (D)
CCK-8 assay showed viability in VSMCs transfected with pcDNA-NC,
ox-LDL + pcDNA-NC or ox-LDL + pcDNA-UCA1. (E) Relative number of
migratory VSMCs transfected with pcDNA-NC, ox-LDL + pcDNA-NC or
ox-LDL + pcDNA-UCA1. UCA1, urothelial carcinoma antigen 1; VSMCs,
vascular smooth muscle cells. *P<0.05,
**P<0.01, ***P<0.001. |
UCA1 negatively regulates MMP9
level
Subcellular distribution analysis indicated that
UCA1 was mainly enriched in the nucleus (Fig. 2A). Treatment of ox-LDL in VSMCs
gradually upregulated protein level of MMP9 in a
concentration-dependent manner (Fig.
2B). In addition, transfection of pcDNA-UCA1 markedly
downregulated MMP9 level (Fig. 2C).
RIP assay pointed out higher enrichment of UCA1 in anti-EZH2
relative to anti-IgG (Fig. 2D).
Transfection of si-EZH2 markedly upregulated MMP9 level in VSMCs
(Fig. 2E). Furthermore, higher
immunoprecipitants of EZH2 and H3K27me3 were shown in VSMCs
overexpressing UCA1 (Fig. 2F). It is
suggested that UCA1 recruited EZH2 to negatively mediate the PTEN
level.
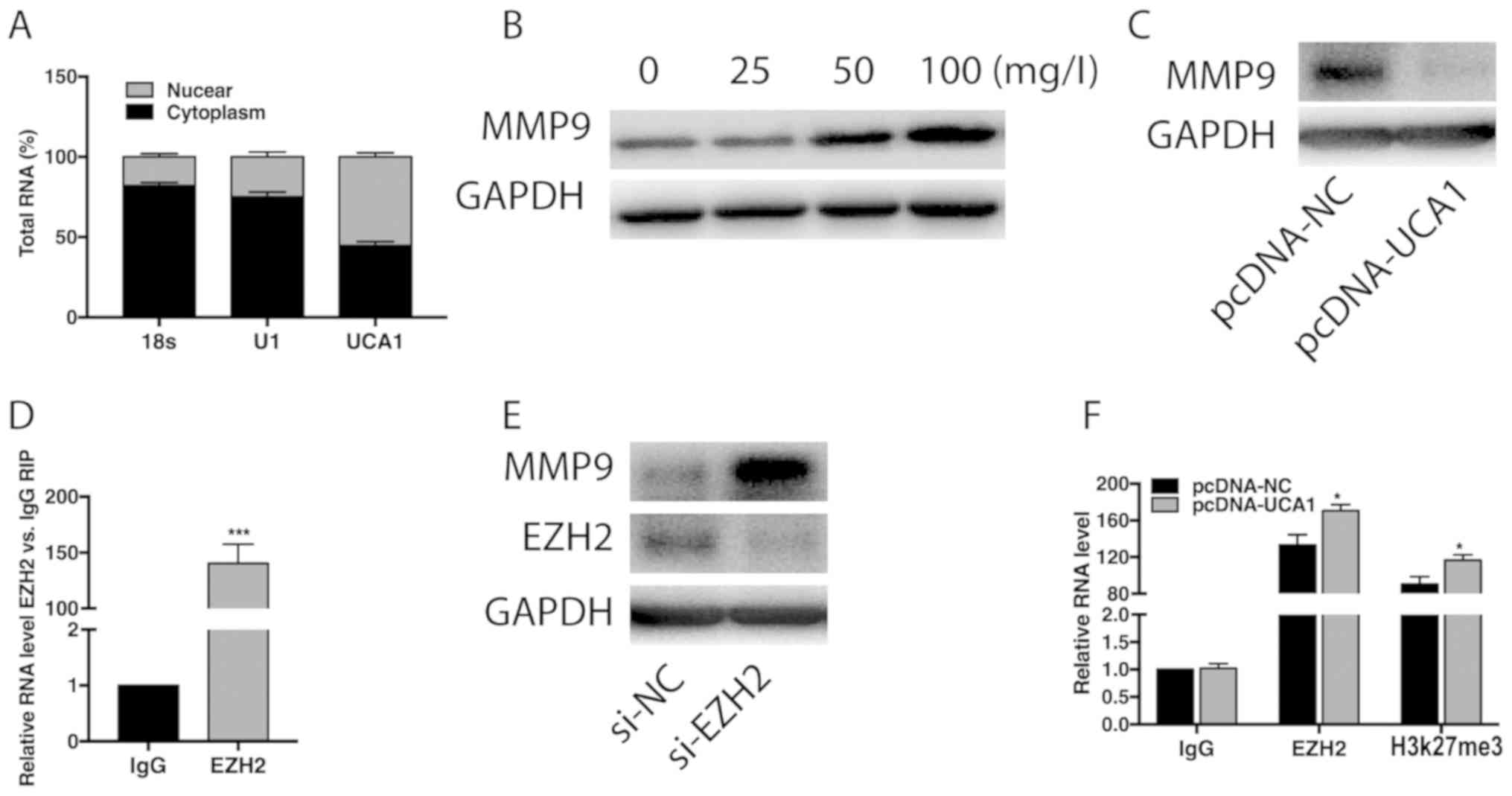 | Figure 2UCA1 negatively regulates MMP9 level.
(A) Subcellular distribution of UCA1 in nuclear and cytoplasmic
fractions of VSMCs. 18s and U1 are internal reference for cytoplasm
and nucleus, respectively. (B) Relative level of MMP9 in VSMCs
induces with 0, 25, 50 and 100 mg/l ox-LDL for 48 h. (C) Relative
level of MMP9 in VSMCs transfected with pcDNA-NC or pcDNA-UCA1. (D)
RIP assay showed the enrichment of UCA1 in anti-IgG or anti-EZH2.
(E) Protein levels of MMP9 and EZH2 in VSMCs transfected with si-NC
or si-EZH2. (F) ChIP assay shows the immunoprecipitants of IgG,
EZH2 and H3K27me3 in VSMCs transfected with pcDNA-NC or pcDNA-UCA1.
UCA1, urothelial carcinoma antigen 1; VSMCs, vascular smooth muscle
cells; MMP9, matrix metalloproteinase-9; EZH2, enhancer of zeste
homolog 2; RIP, RNA immunoprecipitation; ChIP, Chromatin
immunoprecipitation. *P<0.05,
***P<0.001. |
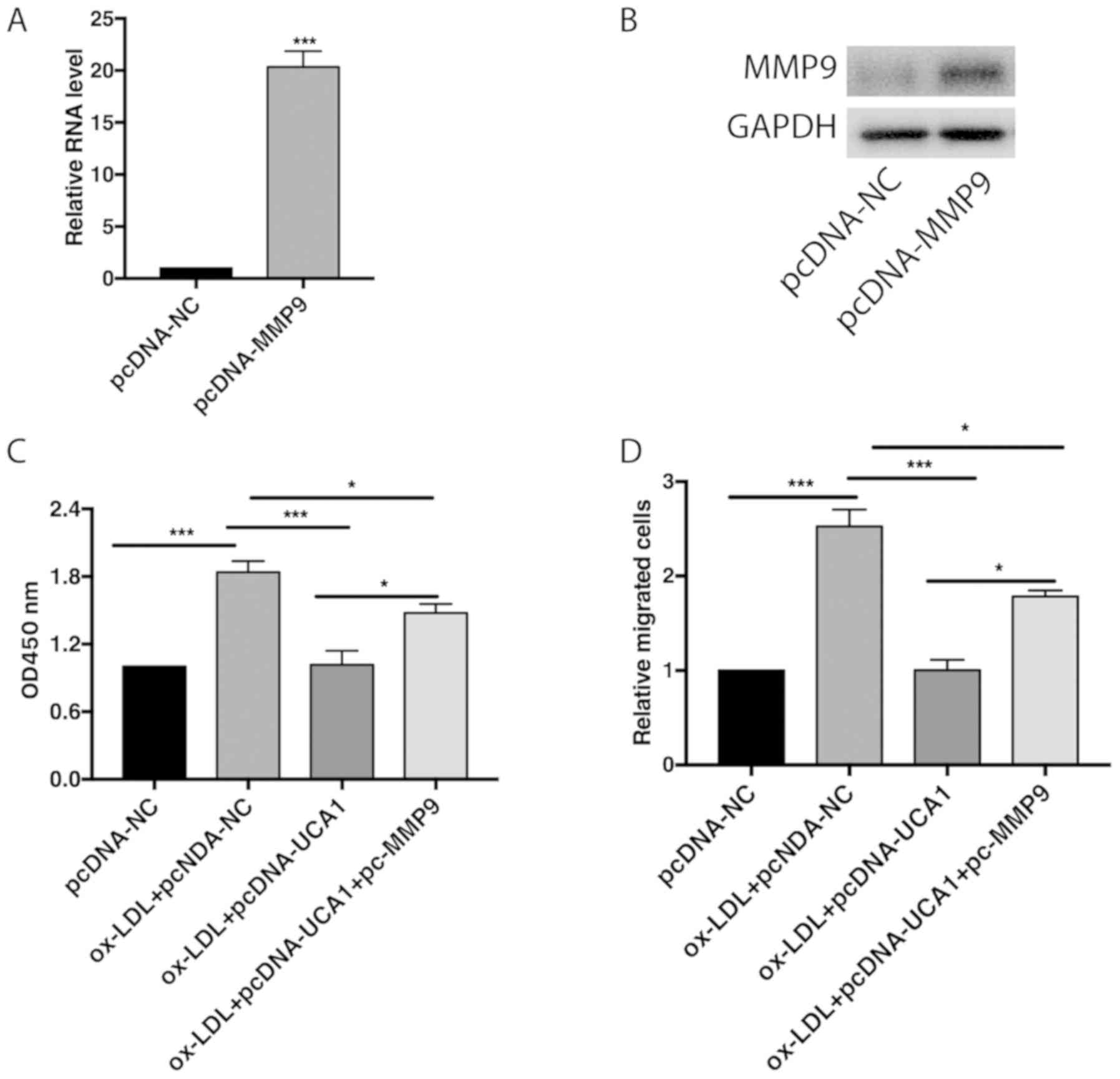
MMP9 partially reverses the biological
role of UCA1
Transfection of pcDNA-MMP9 remarkably upregulated
mRNA and protein level of MMP in VMSCs (Fig. 3A and B). Overexpression of UCA1 in ox-LDL-treated
VSMCs attenuated their proliferative and migratory abilities, but
were further reversed by MMP overexpression (Fig. 3C and D). Hence, it is believed that UCA1
suppressed proliferative and migratory abilities of VSMCs by
negatively regulating the MMP9 level.
Discussion
Dysfunction of VSMCs contributes to the occurrence
and development of cardiovascular diseases (15,16). In
recent years, the morbidity and mortality of cardiovascular
diseases, including hypertension, atherosclerosis and ischemic
encephalopathy have been enhanced each year. VSMCs and vascular
endothelial cells are important components of blood vessels. The
former are located in the tunicae media vasorum and the latter are
distributed in the tunicae intima vasorum. Under normal
circumstances, VSMCs are differentiated and mature (contractile
type), which maintains the normal contractile function of the
arterial wall and regulates blood pressure. After vascular
endothelium damage or surrounding microenvironment changes,
multiple activated pathways stimulate the contractile type of VSMCs
into synthetic type. At this time, VSMCs are prone to proliferate
and migrate, which accelerate the deposition of ECMs in blood
vessels and lead to vascular remodeling (17,18).
lncRNAs are defined as transcripts without
protein-encoding ability. They are able to influence tumorigenesis
through acting on multiple pathways. Abnormally expressed lncRNAs
can be detected in the serum, urine or tumor cells in tumor
patients. They present specific expression patterns in different
stages of tumor diseases and different types of tissues. Therefore,
lncRNAs could be utilized as diagnostic hallmarks for tumors
(19). It is indicated that
downregulation of lncRNA RNCR3 accelerates the occurrence of
atherosclerosis, elevates blood lipid levels and stimulates
inflammatory response. Moreover, the differentiation and migration
of endothelial cells and VSMCs are suppressed, while their
apoptotic abilities are enhanced (20). In this study, UCA1 was gradually
downregulated with the prolongation of increased concentrations of
ox-LDL treatment. Overexpression of UCA1 attenuated the
proliferative and migratory abilities of VSMCs.
MMPs and their tissue inhibitors are a class of
zinc-containing enzymes that degrade ECMs and remodel ECM proteins.
MMPs are mainly produced and released by smooth muscle cells,
fibroblasts, and inflammatory cells. MMP9 belongs to gelatinase,
which degrades both elastin and collagen (21). Relevant studies have shown that MMP9
influences familial aortic dissection by activating TGF-β/Smad
pathway (22). Specifically, MMP9 is
able to regulate the balance of ECM synthesis and degradation,
systolic function of VSMCs and normal function and structure of the
aortic wall. LncRNA MEG8 is reported to affect the proliferative
ability of VSMCs through targeting PPARα (23). Consistently, this study demonstrated
that UCA1 suppressed the proliferative and migratory abilities of
VSMCs through regulating MMP9. Our conclusions may lay a solid
foundation for VSMC research and the application in clinical
practice.
In conclusion, downregulated UCA1 accelerates VSMCs
to proliferate and migrate through negatively regulating the MMP9
level.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
ZX and HL designed the study and performed the
experiments, DD, ZZ and JL collected the data, YT and YG analyzed
the data, ZX and HL prepared the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kil JS, Jeong SO, Chung HT and Pae HO:
Piceatannol attenuates homocysteine-induced endoplasmic reticulum
stress and endothelial cell damage via heme oxygenase-1 expression.
Amino Acids. 49:735–745. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Ren XS, Tong Y, Ling L, Chen D, Sun HJ,
Zhou H, Qi XH, Chen Q, Li YH, Kang YM, et al: NLRP3 gene deletion
attenuates angiotensin II-induced phenotypic transformation of
vascular smooth muscle cells and vascular remodeling. Cell Physiol
Biochem. 44:2269–2280. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wei HJ, Xu JH, Li MH, Tang JP, Zou W,
Zhang P, Wang L, Wang CY and Tang XQ: Hydrogen sulfide inhibits
homocysteine-induced endoplasmic reticulum stress and neuronal
apoptosis in rat hippocampus via upregulation of the BDNF-TrkB
pathway. Acta Pharmacol Sin. 35:707–715. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kung JT, Colognori D and Lee JT: Long
noncoding RNAs: Past, present, and future. Genetics. 193:651–669.
2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Li FP, Lin DQ and Gao LY: LncRNA TUG1
promotes proliferation of vascular smooth muscle cell and
atherosclerosis through regulating miRNA-21/PTEN axis. Eur Rev Med
Pharmacol Sci. 22:7439–7447. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ballantyne MD, Pinel K, Dakin R, Vesey AT,
Diver L, Mackenzie R, Garcia R, Welsh P, Sattar N, Hamilton G, et
al: Smooth muscle enriched long noncoding RNA (SMILR) regulates
cell proliferation. Circulation. 133:2050–2065. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yao QP, Xie ZW, Wang KX, Zhang P, Han Y,
Qi YX and Jiang ZL: Profiles of long noncoding RNAs in hypertensive
rats: Long noncoding RNA XR007793 regulates cyclic strain-induced
proliferation and migration of vascular smooth muscle cells. J
Hypertens. 35:1195–1203. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wang XS, Zhang Z, Wang HC, Cai JL, Xu QW,
Li MQ, Chen YC, Qian XP, Lu TJ, Yu LZ, et al: Rapid identification
of UCA1 as a very sensitive and specific unique marker for human
bladder carcinoma. Clin Cancer Res. 12:4851–4858. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Han Y, Yang YN, Yuan HH, Zhang TT, Sui H,
Wei XL, Liu L, Huang P, Zhang WJ and Bai YX: UCA1, a long
non-coding RNA up-regulated in colorectal cancer influences cell
proliferation, apoptosis and cell cycle distribution. Pathology.
46:396–401. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tian S, Yuan Y, Li Z, Gao M, Lu Y and Gao
H: LncRNA UCA1 sponges miR-26a to regulate the migration and
proliferation of vascular smooth muscle cells. Gene. 673:159–166.
2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Turner RJ and Sharp FR: Implications of
MMP9 for blood brain barrier disruption and hemorrhagic
transformation following ischemic stroke. Front Cell Neurosci.
10(56)2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yang R, Zhang Y, Huang D, Luo X, Zhang L,
Zhu X, Zhang X, Liu Z, Han JY and Xiong JW: Miconazole protects
blood vessels from MMP9-dependent rupture and hemorrhage. Dis Model
Mech. 10:337–348. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Lenglet S, Montecucco F, Mach F, Schaller
K, Gasche Y and Copin JC: Analysis of the expression of nine
secreted matrix metalloproteinases and their endogenous inhibitors
in the brain of mice subjected to ischaemic stroke. Thromb Haemost.
112:363–378. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ma L and Zhang L, Wang B, Wei J, Liu J and
Zhang L: Berberine inhibits Chlamydia pneumoniae infection-induced
vascular smooth muscle cell migration through downregulating MMP3
and MMP9 via PI3K. Eur J Pharmacol. 755:102–109. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
McDonald RA, Hata A, MacLean MR, Morrell
NW and Baker AH: MicroRNA and vascular remodelling in acute
vascular injury and pulmonary vascular remodelling. Cardiovasc Res.
93:594–604. 2012.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Leeper NJ and Maegdefessel L: Non-coding
RNAs: Key regulators of smooth muscle cell fate in vascular
disease. Cardiovasc Res. 114:611–621. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ha JM, Yun SJ, Kim YW, Jin SY, Lee HS,
Song SH, Shin HK and Bae SS: Platelet-derived growth factor
regulates vascular smooth muscle phenotype via mammalian target of
rapamycin complex 1. Biochem Biophys Res Commun. 464:57–62.
2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yang F, Chen Q, He S, Yang M, Maguire EM,
An W, Afzal TA, Luong LA, Zhang L and Xiao Q: miR-22 is a novel
mediator of vascular smooth muscle cell phenotypic modulation and
neointima formation. Circulation. 137:1824–1841. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Su YJ, Yu J, Huang YQ and Yang J:
Circulating long noncoding RNA as a potential target for prostate
cancer. Int J Mol Sci. 16:13322–13338. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shan K, Jiang Q, Wang XQ, Wang YN, Yang H,
Yao MD, Liu C, Li XM, Yao J, Liu B, et al: Role of long non-coding
RNA-RNCR3 in atherosclerosis-related vascular dysfunction. Cell
Death Dis. 7(e2248)2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Wang L, Zhang J, Fu W, Guo D, Jiang J and
Wang Y: Association of smooth muscle cell phenotypes with
extracellular matrix disorders in thoracic aortic dissection. J
Vasc Surg. 56:1698–1709. 2012.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Dekkers BG, Naeimi S, Bos IS, Menzen MH,
Halayko AJ, Hashjin GS and Meurs H: L-thyroxine promotes a
proliferative airway smooth muscle phenotype in the presence of
TGF-beta1. Am J Physiol Lung Cell Mol Physiol. 308:L301–L306.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhang B, Dong Y and Zhao Z: LncRNA MEG8
regulates vascular smooth muscle cell proliferation, migration and
apoptosis by targeting PPARα. Biochem Biophys Res Commun.
510:171–176. 2019.PubMed/NCBI View Article : Google Scholar
|