Breast cancer (BC) is one of the most common
malignant diseases and has become the second leading cause of death
in females (1-3).
According to statistics from the World Health Organization and
American Cancer Society, 2.09 million new cases of BC were
diagnosed worldwide in 2018. BC has reached the highest
age-standardized frequency (46.3 per 100,000) compared with other
types of cancer. In addition, the prognosis of BC ranks third after
thyroid and prostate cancer, with a 5-year survival rate of 80-85%
(4). The incidence of BC, which
seriously affects the quality of life of patients, is gradually
increasing owing to an unhealthy lifestyle, enhanced awareness and
new screening technologies (5,6).
Although certain advances in therapeutic strategies against BC have
been made, it remains a major public health problem. Clinically, it
is difficult to observe the early symptoms of patients with cancer
due to the pathophysiological characteristics of the tumor, which
results in the majority of patients with BC being diagnosed at an
advanced stage (7). Therefore, it is
important to identify novel and valuable biomarkers for the
diagnosis and prognosis of BC, and/or to provide novel therapeutic
targets.
Pre-B-cell leukemia transcription factor (PBX)
proteins are atypical homeodomain-containing transcription factors,
which belong to the family of three amino-acid loop extension-class
homeodomain proteins (8). They have
an integral role in the development of multiple organs, including
the appendicular skeleton, lung, heart, pancreas, spleen and
kidney, by regulating the expression of crucial target genes
(9). The mammalian system consists
of four members, namely PBX1, PBX2, PBX3, and PBX4(10). PBX1 was initially identified due to
its involvement in a chromosomal translocation in human pre-B cell
acute lymphoid leukemia (11). PBX2
and PBX3 were subsequently identified based on their sequence
homology with PBX1(12). PBX4 was
first identified in the developing zebrafish brain (13). A large number of studies have
demonstrated that the deregulation of PBX proteins is closely
associated with the development and progression of numerous
diseases, including cancer (14-18).
PBX proteins have also been reported to be overexpressed in a
variety of solid tumor types, including colorectal, gastric and
ovarian cancers, as well as glioma (19-21).
The estradiol acetate (E2A)-PBX1 fusion protein contributes to
pre-B cell transformation in leukemogenesis by enhancing the
transactivation activity of homeobox (HOX) family members (22). The HOXB7/PBX2 complex promotes the
progression of cutaneous melanoma by transcriptionally activating
microRNA (miR)-221 and -222(18).
Furthermore, PBX4 was reported to be downregulated in leukemic cell
lines and the samples from patients with acute lymphoblastic
leukemia (ALL) (23). PBX3 has been
indicated to be a key driver of mesenchymal transition and a
potential therapeutic target in glioblastoma (24). In addition, it has been reported to
be a prognostic biomarker in prostate cancer (25). Although PBX proteins have been
demonstrated to act as crucial transcription factors in numerous
malignancies (26-29),
the roles of different PBX family members in the progression of BC
have remained elusive.
The present study extensively analyzed the
expression of distinct PBX family members in patients with BC based
on publicly available clinical datasets in order to determine their
expression patterns and prognostic value in BC. It was demonstrated
that PBX1 is highly expressed in patients with BC and is associated
with poor clinical outcomes in patients with estrogen receptor
(ER)-positive, luminal A and luminal B subtypes of BC.
Informed consent for publication was not required,
as all patient data used in the study were obtained from publicly
available databases.
To date, four PBX members have been identified in
mammalian cells. However, little is known about their roles in BC
progression. To determine whether PBX family members are involved
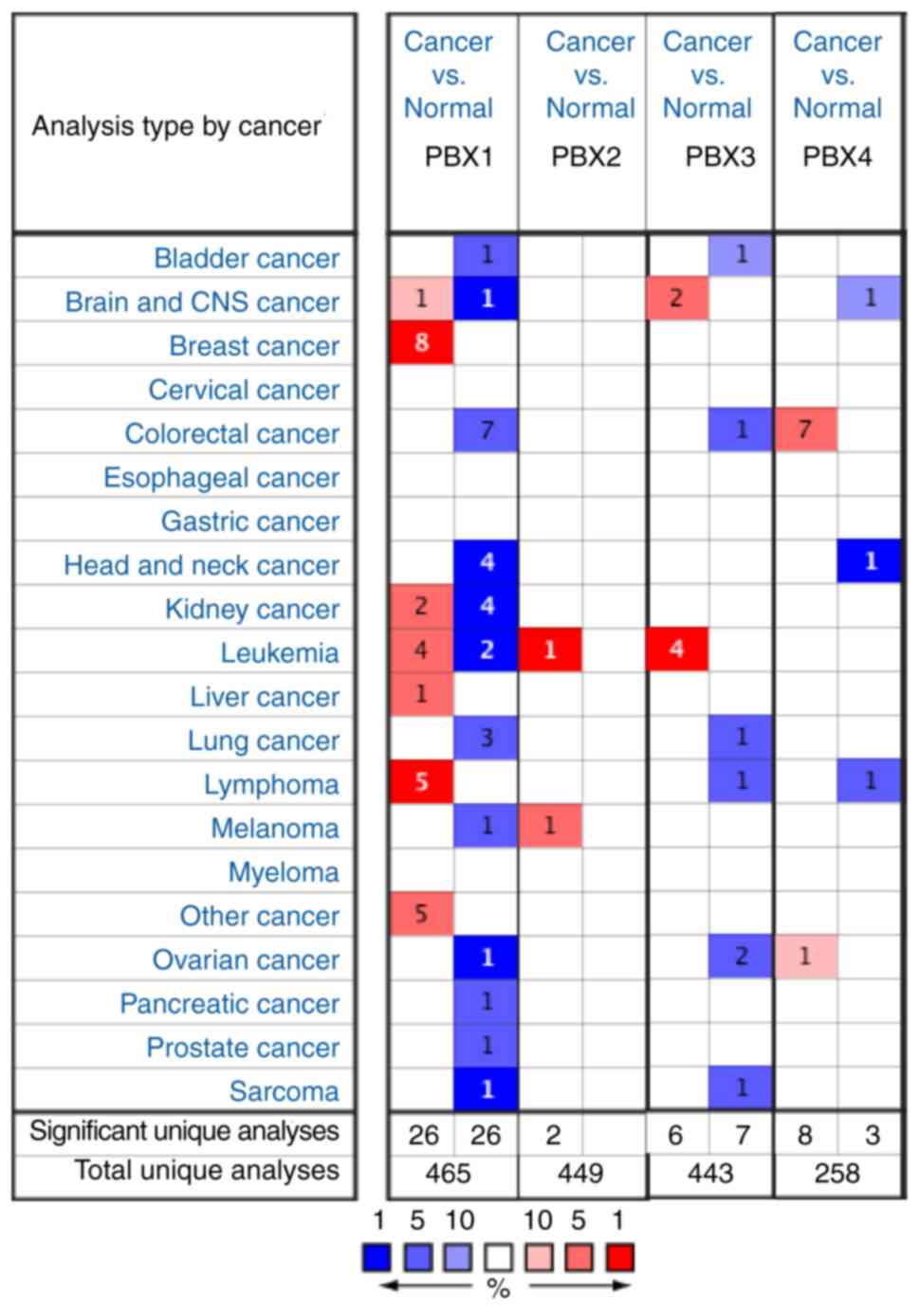
in BC progression, the expression of PBXs was determined in BC
tissues and control tissues using the Oncomine database, which
altogether includes 465, 449, 443 and 258 unique datasets for PBX1,
PBX2, PBX3 and PBX4, respectively. The mRNA level of PBX1 in
patients with BC was significantly upregulated in 8 datasets,
whereas no significant difference in PBX2, PBX3 or PBX4 mRNA levels
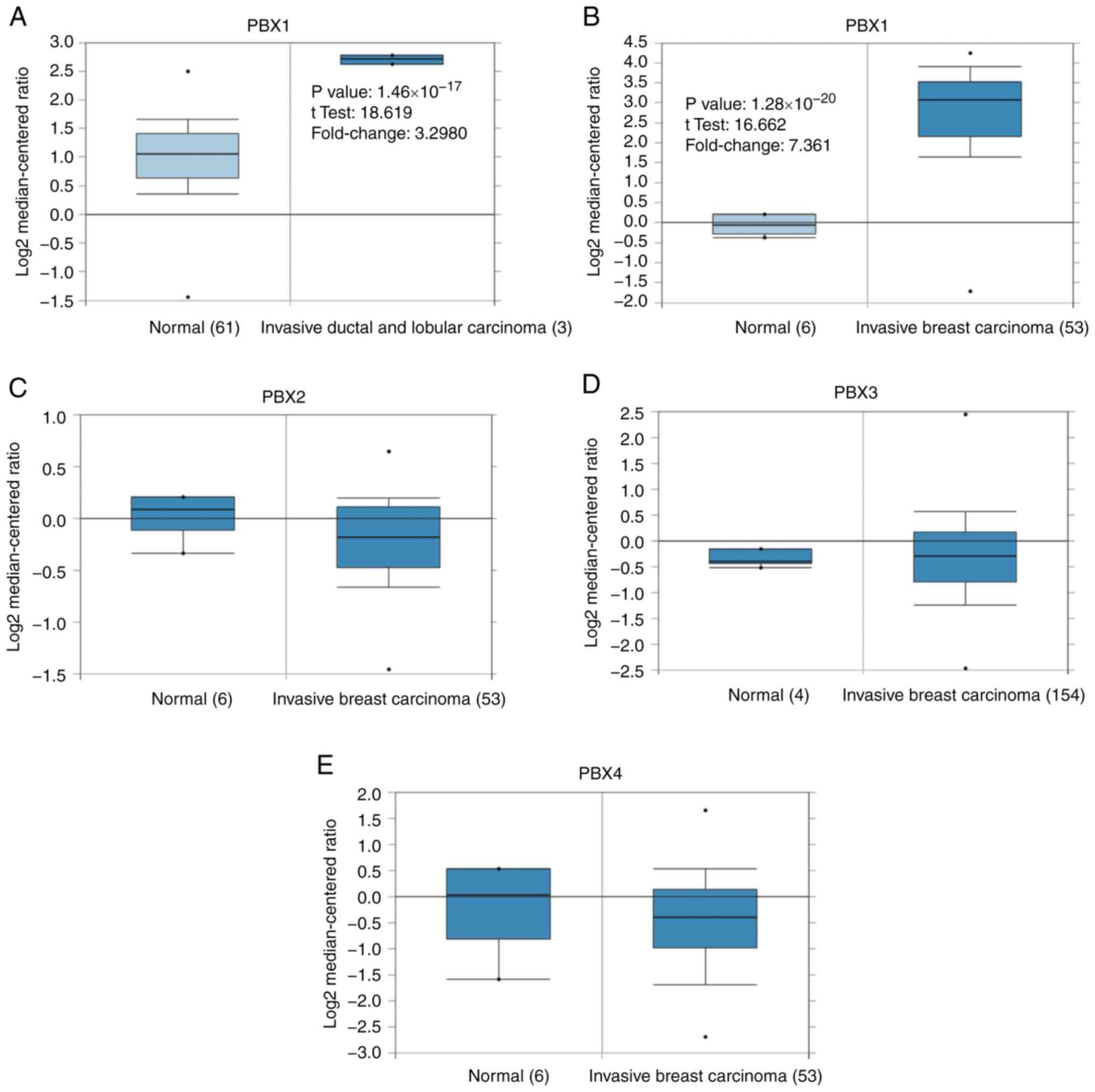
was observed between BC tissues and control tissues (Fig. 1). The PBX1 mRNA level was 3.298-fold
elevated in BC tissues compared with control samples in an analysis
of 593 specimens from The Cancer Genome Atlas (http://cancergenome.nih.gov) database
(P=1.46x10-17; Fig.
2A). In Finak's dataset (33),
PBX1 expression was 7.361-fold elevated in BC tissues compared with
control samples (P=1.28x10-20), whereas no
significant difference in PBX2, PBX3 or PBX4 mRNA levels was
observed between BC samples and normal controls (Fig. 2B-E).
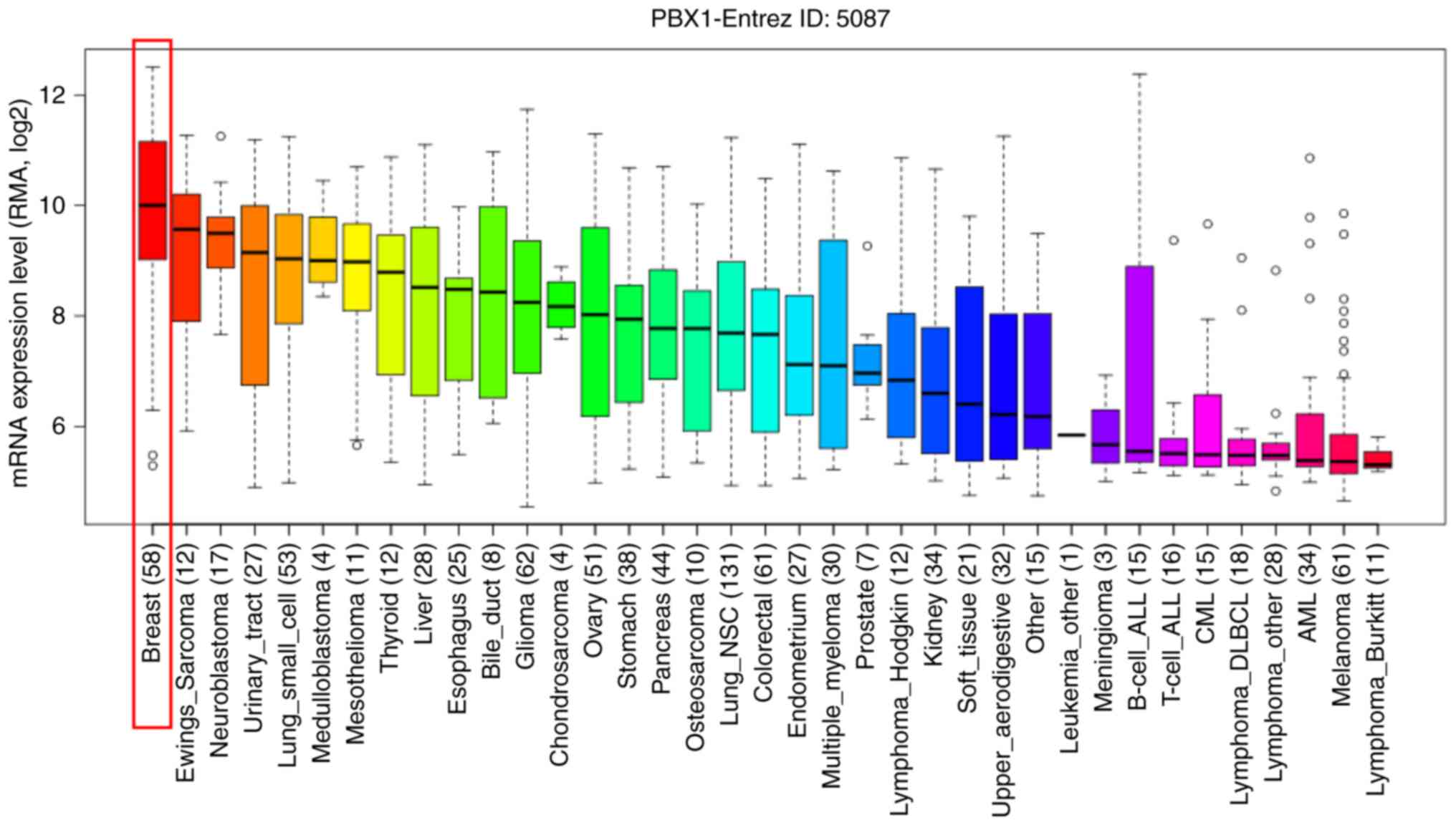
To further determine the exact expression pattern of
PBX1, the mRNA expression of PBX1 in different cancer cell lines
was examined using the CCLE database, which contains a large number
of cancer cell lines and provides extensive information on the
expression of PBX1 in numerous cancer subtypes with different
tissue types of origin. As indicated in Fig. 3, the transcription level of PBX1 in
BC cell lines was the highest among all of the cancer types. This
result was consistent with the data from the Oncomine analysis,
suggesting that PBX1 may have a crucial role in the carcinogenesis
of BC.
Since the above evidence indicated that PBX1 is
highly expressed in BC tissues and cell lines, a pooled
meta-analysis was performed to confirm the high expression of PBX1
in BC using the Oncomine database. A total of 13 analyses,
including 939 samples, were included in the meta-analysis. Overall,
the pooled analysis demonstrated that the mRNA level of PBX1 was
significantly increased in the 13 clinical cohorts of patients with
BC (Fig. 4). This result provided
strong evidence that the expression of PBX1 is significantly
increased in BC.
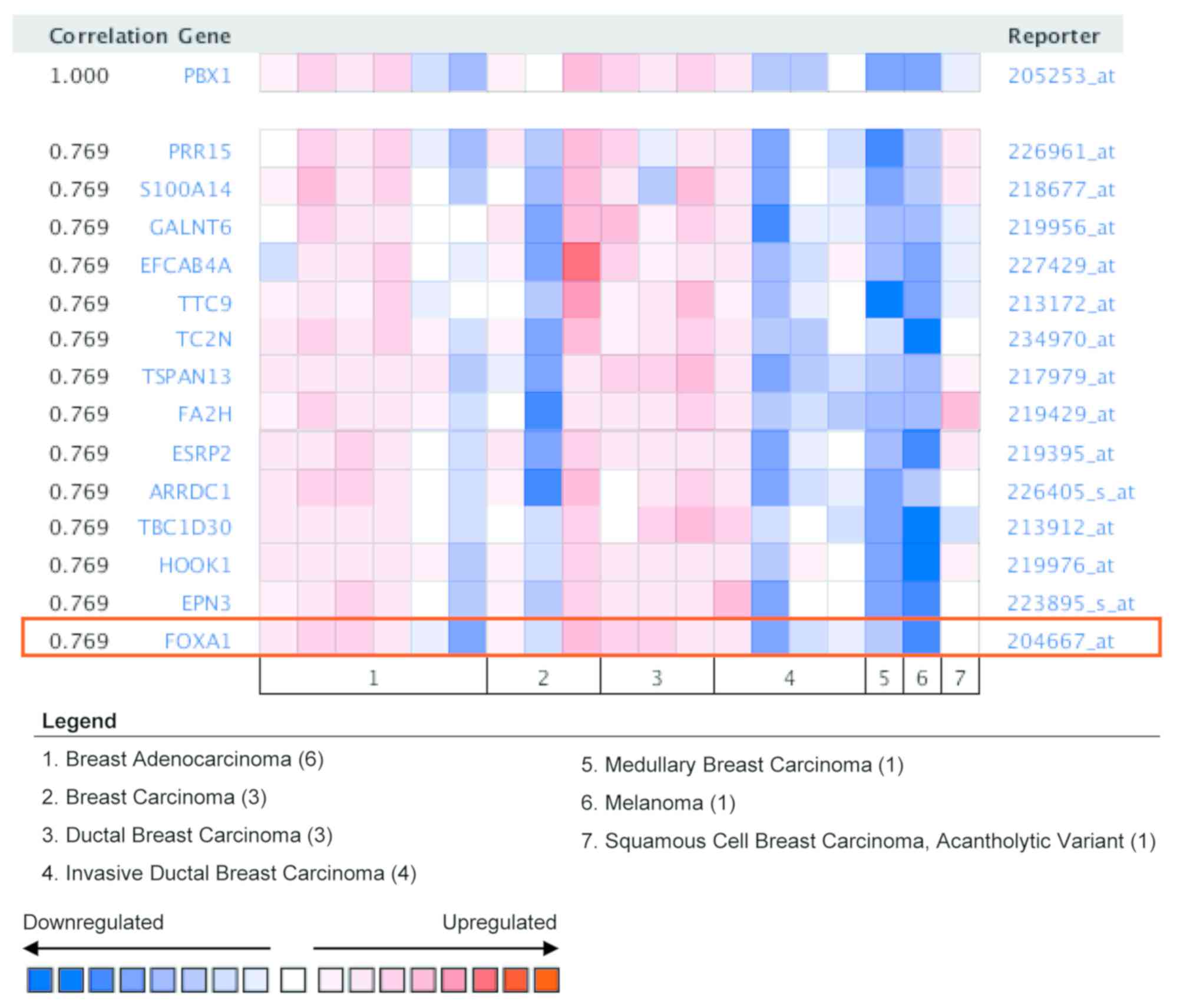
To gain further insight into the potential roles of
PBX1 in BC, co-expression analysis of PBX1 mRNA was performed using
the Oncomine database. The co-expression profile of PBX1 was
examined using a cluster of 19,574 genes in 19 BC samples (34). As presented in Fig. 5, PBX1 expression was significantly
correlated with FOXA1 (r=0.769). FOXA1 is a member of the FOX
family of transcription factors, which has been reported to promote
BC progression by enhancing the expression and activity of ERα
(35,36). This result suggested that PBX1 might
have a role in BC carcinogenesis by interacting with the FOXA1-ERα
axis.
The present study next examined the PBX1 expression
pattern in distinct BC subtypes using the Oncomine database. The
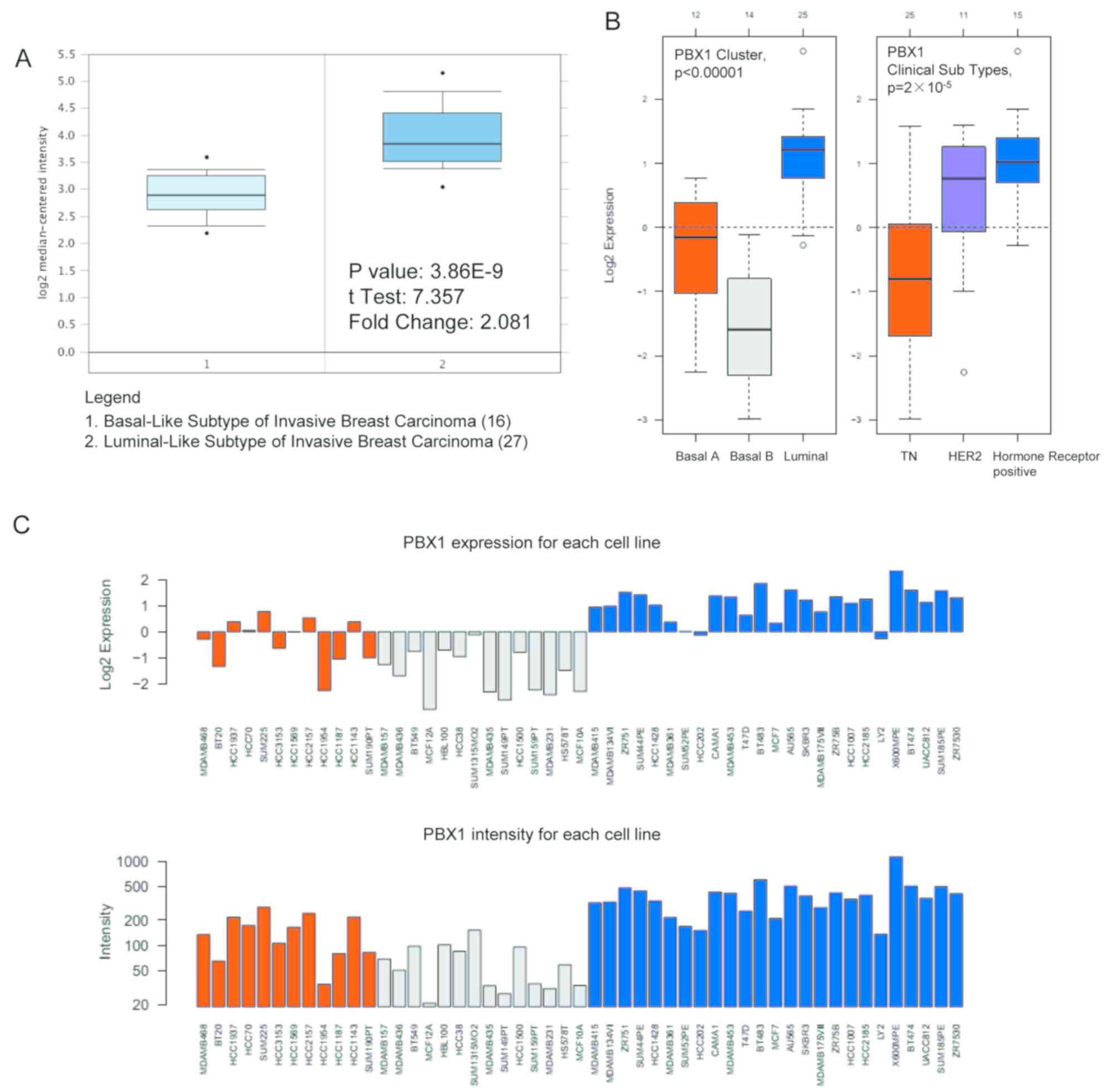
study by Farmer et al (37)
revealed that PBX1 was 2.081-fold upregulated in luminal-like BC
tissues compared with that in basal-like BC tissues
(P=3.86x10-9; Fig.
6A). GOBO analysis was used to further examine the PBX1
expression pattern in distinct BC subtypes. As presented in
Fig. 6B, PBX1 expression was
significantly upregulated in the luminal-like subtype compared with
that in the basal-A and basal-B subtypes of BC. Furthermore, the
expression of PBX1 was significantly upregulated in the hormone
receptor-sensitive subtype compared with those in the
triple-negative (TN) and human epidermal growth factor receptor 2
(Her-2) subtypes. The PBX1 mRNA expression showed differences
across 51 breast cancer cell lines (Fig.
6C).
Since elevated PBX1 expression was closely
associated with BC progression, the present study further
determined the effect of PBX1 on the long-term survival of patients
with BC using the Kaplan-Meier plotter, which is an online analysis
tool that may be used to evaluate the effect of 54,000 genes on
survival in 21 cancer types (32).
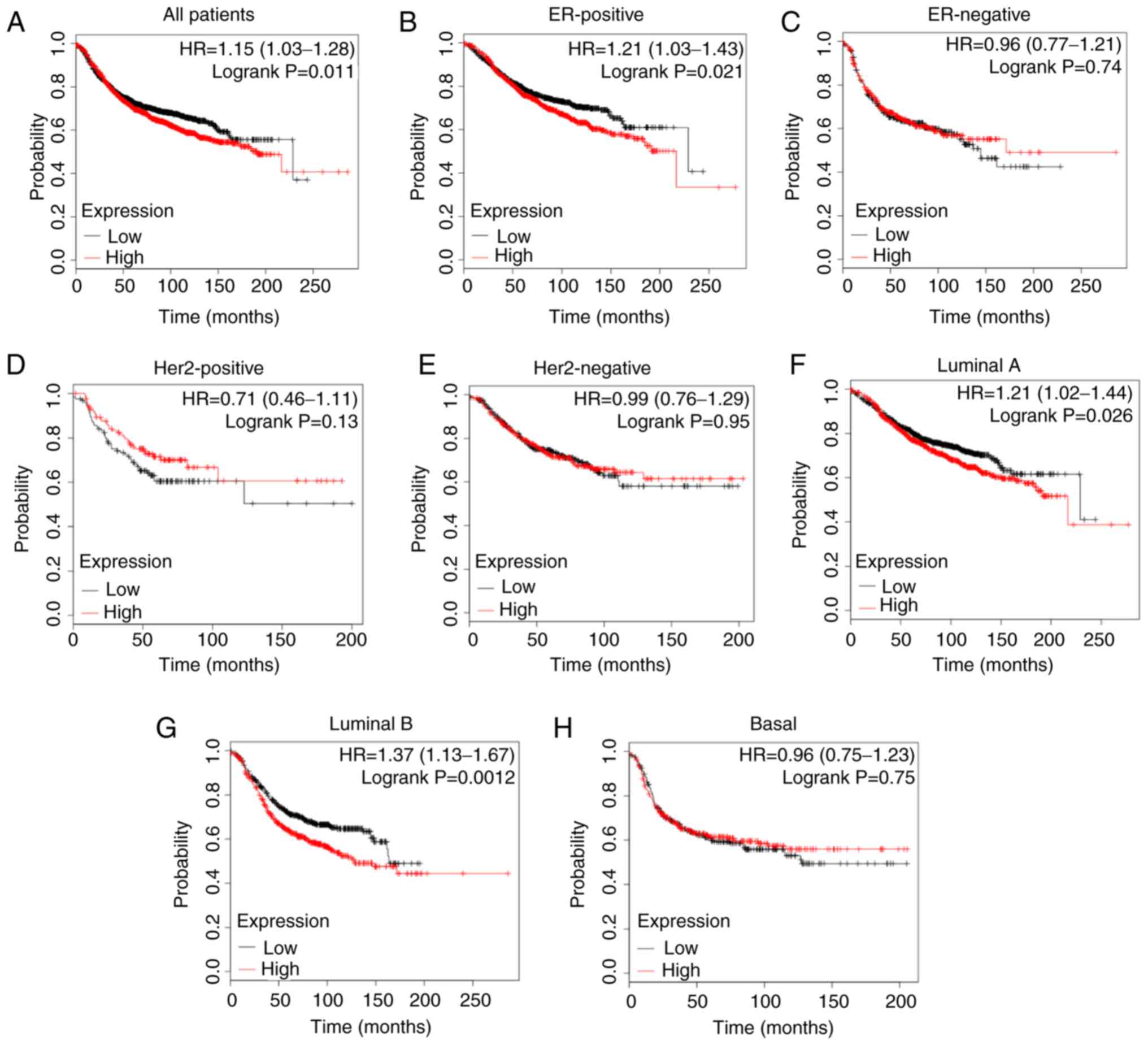
As presented in Fig. 7A, a high
expression level of PBX1 was closely associated with a shorter RFS
in all patients with BC [hazard ratio (HR)=1.15, P=0.011].
Furthermore, the analysis of BC subtypes revealed that the
overexpression of PBX1 was significantly associated with a shorter
RFS in ER-positive (HR=1.21, P=0.021), luminal A (HR=1.21, P=0.026)
and luminal B (HR=1.37, P=0.0012) subtypes, but not in ER-negative
(HR=0.96, P=0.74), Her-2-positive (HR=0.71, P=0.13), Her2-negative
(HR=0.99, P=0.95) or basal-like (HR=0.96, P=0.75) subtypes
(Fig. 7B-H). These results indicated
a crucial prognostic value of PBX1 in ER-positive, luminal A and
luminal B subtypes of BC.
BC is one of the most common types of cancer among
all malignant tumor types in females, with high incidence and
mortality rates (38-40).
Due to its pathological and anatomical features, it is difficult to
observe the early onset of BC and tumors are frequently diagnosed
in the late stages of the disease. The initiation and development
of BC are immensely complex biological processes and the disease
has a poor prognosis. Therefore, it is necessary to identify
valuable predictive and prognostic biomarkers for early diagnosis
and effective treatment of BC. In the present study, publicly
available clinical datasets were evaluated to determine
differentially expressed genes between cancer samples and control
samples. The results suggested that PBX1 was significantly
upregulated in BC samples compared with other PBX genes, indicating
its specific role in patients with BC.
PBX proteins are homeodomain-containing
transcription factors that act as HOX cofactors to regulate gene
transcription during development (41-43).
Growing evidence suggests that PBXs have oncogenic functions. For
instance, PBX1 acts as a chimeric fusion partner in the oncoprotein
E2A-PBX1 to promote the progression of pre-B cell ALL (44,45). It
also confers prostate cancer cells resistance against common
chemotherapeutic drugs, including doxorubicin and cisplatin, and
its degradation promotes the apoptosis of prostate cancer cells
(46). PBX2 has been reported to
mediate the antiapoptotic function of miR-1915-3p in lung cancer
(47). High expression of PBX2 has
been identified as a poor prognostic indicator in gingival squamous
cell carcinoma and non-small cell lung cancer (48,49).
PBX3 has been suggested to be the target of multiple miRs,
including miR-320a, miR-129-5p, and miR-526b, and mediates the
antitumor effects of these miRs (50-52).
Furthermore, PBX3 enhances the stability of Meis homeobox 1 (MEIS1)
and dimerizes with MEIS1 to efficiently promote leukemogenesis in
mouse models (53). It is also
recognized as a prognostic factor in several types of cancer,
including colorectal, cervical, gastric and prostate cancer
(17,25,54,55). Low
levels of PBX4 expression are observed in blood samples of patients
with ALL (23). However, its
function has not been reported in ALL progression. In the present
study, data from the Oncomine analysis suggested that, among all
PBX family members, only the expression level of PBX1 was higher in
BC samples than in the corresponding control samples. Consistent
with this, CCLE analysis indicated that the PBX1 expression levels
in BC were the highest among all cancer types at the cellular
level. The study further investigated the PBX1 expression pattern
in distinct subtypes of BC. GOBO analysis indicated that PBX1 mRNA
was significantly overexpressed in luminal-like subtypes of BC
compared with the basal-A and basal-B subtypes. In addition, PBX1
mRNA was significantly overexpressed in the hormone
receptor-sensitive subtype of BC compared with that in the TN and
Her-2 clinical subtypes. Furthermore, meta-analysis also revealed
that PBX1 overexpression was associated with BC. These results
demonstrated that PBX1 is overexpressed in BC tissues and BC cell
lines, and that PBX1 expression is closely associated with BC
progression, indicating its unique role in BC. However, the
detailed mechanisms of the role of PBX1 in BC remain unknown and
require further elucidation.
It has been reported that PBX1 is correlated with
ERα and promotes the expression of genes associated with the
aggressive progression of ERα-positive BC by guiding
estrogen-induced ERα recruitment to its target chromatin (56). Another study indicated that PBX1 also
has an important role in ERα-mediated transcriptional response to
estrogen-independent epidermal growth factor (EGF) signaling
(57). Knockdown of PBX1 blocks the
expression of a subset of EGF-ERα target genes in ERα-positive BC
cells. Furthermore, upregulation of PBX1 was associated with an
increased risk of metastatic progression of ERα-positive BC
(57). In the present study, gene
co-expression analysis revealed that PBX1 is closely associated
with FOXA1 in BC patients. FOXA1 belongs to the FOX family of
transcription factors and was first identified due to its role in
the transcriptional regulation of genes expressing liver-specific
transthyretin and α1-antitrypsin (58). FOXA1 is a prognostic marker and novel
therapeutic target in BC (59). It
has been reported that FOXA1 may act as an initiating factor to
regulate the activity of ERα by enhancing the binding of ERα to its
target genes (60). Furthermore, it
directly binds to the promoter of ER and regulates the expression
of ERα in BC cells (36). The
present results suggest that PBX1 may have a crucial role in
contributing to BC progression via the FOXA1 signaling pathway. In
addition, PBX1 has been suggested to act as an initiating factor to
guide ERα genomic activity to unique genomic regions, thus
promoting the transcription of genes involved in BC progression
(56,57).
The clinical significance of PBX1 expression in BC
remains unclear. Therefore, the present study examined the
association between the PBX1 expression pattern and the prognosis
of different molecular subtypes of BC by Oncomine analysis and
established the association of PBX1 expression with survival
outcome. The pooled results indicated that high PBX1 expression was
significantly associated with worse RFS in ER-positive, luminal A
and luminal B subtypes of BC, but not in ER-negative, Her2-positive
or basal-like BC subtypes, suggesting that PBX1 is a potential
oncoprotein in BC. The results of the present study are consistent
with those of Magnani et al (56,57).
Overall, these data demonstrate that the expression level of PBX1
may be a useful biomarker of prognosis for ER-positive, luminal A
and luminal B subtypes of BC.
In conclusion, PBX1 is significantly overexpressed
in BC tissues compared with normal tissues and its high expression
level is a predictor of poor prognosis of patients with
ER-positive, luminal A and luminal B subtypes of BC. In addition,
PBX1 is positively correlated with FOXA1. The present study
suggests that PBX1 may be an important biomarker for BC.
Large-scale and comprehensive studies are still required to further
confirm the present results and to determine the clinical
application of PBX1 in the prognostication of patients with BC.
Not applicable.
This work was supported by the National Natural
Science Foundation of China (grant nos. 81702785 and 81802822), the
Natural Science Foundation of Shandong Province (grant nos.
ZR2017PH013 and ZR2018BH017) and the Qingdao Postdoctoral
Application Research Funded Project (grant no. 2016067).
All datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
XA, WD, HG, YZ, DD and YL analyzed the data. XA and
YL designed the study and prepared the manuscript. All authors read
and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Liu Y, Ao X, Jia Z, Bai XY, Xu Z, Hu G,
Jiang X, Chen M and Wu H: FOXK2 transcription factor suppresses
ERalpha-positive breast cancer cell growth through down-regulating
the stability of ERα via mechanism involving BRCA1/BARD1. Sci Rep.
5(8796)2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wang M, Zhao F, Li S, Chang AK, Jia Z,
Chen Y, Xu F, Pan H and Wu H: AIB1 cooperates with ERα to promote
epithelial mesenchymal transition in breast cancer through SNAI1
activation. PLoS One. 8(e65556)2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ao X, Li S, Xu Z, Yang Y, Chen M, Jiang X
and Wu H: Sumoylation of TCF21 downregulates the transcriptional
activity of estrogen receptor-alpha. Oncotarget. 7:26220–26234.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Mattiuzzi C and Lippi G: Current cancer
epidemiology. J Epidemiol Glob Health. 9:217–222. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kang SY, Kim YS, Kim Z, Kim HY, Lee SK,
Jung KW and Youn HJ: Korean Breast Cancer Society: Basic findings
regarding breast cancer in Korea in 2015: Data from a breast cancer
registry. J Breast Cancer. 21:1–10. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Drukteinis JS, Mooney BP, Flowers CI and
Gatenby RA: Beyond mammography: New frontiers in breast cancer
screening. Am J Med. 126:472–479. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Liu Y, Ao X, Ding W, Ponnusamy M, Wu W,
Hao X, Yu W, Wang Y, Li P and Wang J: Critical role of FOXO3a in
carcinogenesis. Mol Cancer. 17(104)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Capellini TD, Zappavigna V and Selleri L:
Pbx homeodomain proteins: TALEnted regulators of limb patterning
and outgrowth. Dev Dyn. 240:1063–1086. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Welsh IC, Hart J, Brown JM, Hansen K,
Rocha Marques M, Aho RJ, Grishina I, Hurtado R, Herzlinger D,
Ferretti E, et al: Pbx loss in cranial neural crest, unlike in
epithelium, results in cleft palate only and a broader midface. J
Anat. 233:222–242. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Longobardi E, Penkov D, Mateos D, De
Florian G, Torres M and Blasi F: Biochemistry of the tale
transcription factors PREP, MEIS, and PBX in vertebrates. Dev Dyn.
243:59–75. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Nourse J, Mellentin JD, Galili N,
Wilkinson J, Stanbridge E, Smith SD and Cleary ML: Chromosomal
translocation t(1;19) results in synthesis of a homeobox fusion
mRNA that codes for a potential chimeric transcription factor.
Cell. 60:535–545. 1990.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Monica K, Galili N, Nourse J, Saltman D
and Cleary ML: PBX2 and PBX3, new homeobox genes with extensive
homology to the human proto-oncogene PBX1. Mol Cell Biol.
11:6149–6157. 1991.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Pöpperl H, Rikhof H, Chang H, Haffter P,
Kimmel CB and Moens CB: Lazarus is a novel pbx gene that globally
mediates hox gene function in zebrafish. Mol Cell. 6:255–267.
2000.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Heidet L, Moriniére V, Henry C, De Tomasi
L, Reilly ML, Humbert C, Alibeu O, Fourrage C, Bole-Feysot C,
Nitschké P, et al: Targeted exome sequencing identifies PBX1 as
involved in monogenic congenital anomalies of the Kidney and
urinary tract. J Am Soc Nephrol. 28:2901–2914. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Villaescusa JC, Li B, Toledo EM, Rivetti
di Val Cervo P, Yang S, Stott SR, Kaiser K, Islam S, Gyllborg D,
Laguna-Goya R, et al: A PBX1 transcriptional network controls
dopaminergic neuron development and is impaired in Parkinson's
disease. EMBO J. 35:1963–1978. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yu B, Tian X, Zhang L and Feng R:
Hematopoietic PBX-interaction protein promotes breast cancer
sensitivity to paclitaxel through a microtubule-dependent
mechanism. DNA Cell Biol. 35:740–745. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ma YY, Zhang Y, Mou XZ, Liu ZC, Ru GQ and
Li E: High level of homeobox A9 and PBX homeobox 3 expression in
gastric cancer correlates with poor prognosis. Oncol Lett.
14:5883–5889. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Errico MC, Felicetti F, Bottero L, Mattia
G, Boe A, Felli N, Petrini M, Bellenghi M, Pandha HS, Calvaruso M,
et al: The abrogation of the HOXB7/PBX2 complex induces apoptosis
in melanoma through the miR-221&222-c-FOS pathway. Int J
Cancer. 133:879–892. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Han HB, Gu J, Ji DB, Li ZW, Zhang Y, Zhao
W, Wang LM and Zhang ZQ: PBX3 promotes migration and invasion of
colorectal cancer cells via activation of MAPK/ERK signaling
pathway. World J Gastroenterol. 20:18260–18270. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jung JG, Shih IM, Park JT, Gerry E, Kim
TH, Ayhan A, Handschuh K, Davidson B, Fader AN, Selleri L and Wang
TL: Ovarian cancer chemoresistance relies on the stem cell
reprogramming factor PBX1. Cancer Res. 76:6351–6361.
2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Xu X, Cai N, Bao Z, You Y, Ji J and Liu N:
Silencing Pre-B-cell leukemia homeobox 3 decreases the
proliferation of human glioma cells in vitro and in vivo. J
Neurooncol. 135:453–463. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Aspland SE, Bendall HH and Murre C: The
role of E2A-PBX1 in leukemogenesis. Oncogene. 20:5708–5717.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Rosales-Aviña JA, Torres-Flores J,
Aguilar-Lemarroy A, Gurrola-Díaz C, Hernández-Flores G,
Ortiz-Lazareno PC, Lerma-Díaz JM, de Celis R, González-Ramella Ó,
Barrera-Chaires E, et al: MEIS1, PREP1, and PBX4 are differentially
expressed in acute lymphoblastic leukemia: Association of MEIS1
expression with higher proliferation and chemotherapy resistance. J
Exp Clin Cancer Res. 30(112)2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Xu X, Bao Z, Liu Y, Jiang K, Zhi T, Wang
D, Fan L, Liu N and Ji J: PBX3/MEK/ERK1/2/LIN28/let-7b positive
feedback loop enhances mesenchymal phenotype to promote
glioblastoma migration and invasion. J Exp Clin Cancer Res.
37(158)2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ramberg H, Grytli HH, Nygård S, Wang W,
Ögren O, Zhao S, Løvf M, Katz B, Skotheim RI, Bjartell A, et al:
PBX3 is a putative biomarker of aggressive prostate cancer. Int J
Cancer. 139:1810–1820. 2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Alharbi RA, Pandha HS, Simpson GR,
Pettengell R, Poterlowicz K, Thompson A, Harrington K, El-Tanani M
and Morgan R: Inhibition of HOX/PBX dimer formation leads to
necroptosis in acute myeloid leukemia cells. Oncotarget.
8:89566–89579. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ramberg H, Alshbib A, Berge V, Svindland A
and Taskén KA: Regulation of PBX3 expression by androgen and Let-7d
in prostate cancer. Mol Cancer. 10(50)2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Dardaei L, Longobardi E and Blasi F: Prep1
and Meis1 competition for Pbx1 binding regulates protein stability
and tumorigenesis. Proc Natl Acad Sci USA. 111:E896–E905.
2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Ando H, Natsume A, Senga T, Watanabe R,
Ito I, Ohno M, Iwami K, Ohka F, Motomura K, Kinjo S, et al:
Peptide-based inhibition of the HOXA9/PBX interaction retards the
growth of human meningioma. Cancer Chemother Pharmacol. 73:53–60.
2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lin HY, Zeng Liang YK, Wei XL and Chen CF:
GATA3 and TRPS1 are distinct biomarkers and prognostic factors in
breast cancer: Database mining for GATA family members in
malignancies. Oncotarget. 8:34750–34761. 2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Jin Z, Xu L, Zhang L, Zhao M, Li D, Ye L,
Ma Y, Ren S, Yu H, Wang D, et al: Interleukin enhancer binding
factor 2 is a prognostic biomarker for breast cancer that also
predicts neoadjuvant chemotherapy responses. Am J Transl Res.
10:1677–1689. 2018.PubMed/NCBI
|
|
32
|
Lánczky A, Nagy Á, Bottai G, Munkácsy G,
Szabó A, Santarpia L and Győrffy B: miRpower: A web-tool to
validate survival-associated miRNAs utilizing expression data from
2178 breast cancer patients. Breast Cancer Res Treat. 160:439–446.
2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Finak G, Bertos N, Pepin F, Sadekova S,
Souleimanova M, Zhao H, Chen H, Omeroglu G, Meterissian S, Omeroglu
A, et al: Stromal gene expression predicts clinical outcome in
breast cancer. Nat Med. 14:518–527. 2008.PubMed/NCBI View
Article : Google Scholar
|
|
34
|
Bild AH, Yao G, Chang JT, Wang Q, Potti A,
Chasse D, Joshi MB, Harpole D, Lancaster JM, Berchuck A, et al:
Oncogenic pathway signatures in human cancers as a guide to
targeted therapies. Nature. 439:353–357. 2006.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Beck S, Sommer P, dos Santos Silva E, Blin
N and Gött P: Hepatocyte nuclear factor 3 (winged helix domain)
activates trefoil factor gene TFF1 through a binding motif adjacent
to the TATAA box. DNA Cell Biol. 18:157–164. 1999.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bernardo GM, Lozada KL, Miedler JD,
Harburg G, Hewitt SC, Mosley JD, Godwin AK, Korach KS, Visvader JE,
Kaestner KH, et al: FOXA1 is an essential determinant of ERalpha
expression and mammary ductal morphogenesis. Development.
137:2045–2054. 2010.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Farmer P, Bonnefoi H, Becette V,
Tubiana-Hulin M, Fumoleau P, Larsimont D, Macgrogan G, Bergh J,
Cameron D, Goldstein D, et al: Identification of molecular apocrine
breast tumours by microarray analysis. Oncogene. 24:4660–4671.
2005.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Guo F, Kuo YF, Shih YCT, Giordano SH and
Berenson AB: Trends in breast cancer mortality by stage at
diagnosis among young women in the United States. Cancer.
124:3500–3509. 2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ferlay J, Colombet M, Soerjomataram I,
Dyba T, Randi G, Bettio M, Gavin A, Visser O and Bray F: Cancer
incidence and mortality patterns in Europe: Estimates for 40
countries and 25 major cancers in 2018. Eur J Cancer. 103:356–387.
2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Baeyens-Fernández JA, Molina-Portillo E,
Pollan M, Rodríguez-Barranco M, Del Moral R, Arribas-Mir L,
Sánchez-Cantalejo Ramírez E and Sánchez MJ: Trends in incidence,
mortality and survival in women with breast cancer from 1985 to
2012 in Granada, Spain: A population-based study. BMC Cancer.
18(781)2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Dard A, Reboulet J, Jia Y, Bleicher F,
Duffraisse M, Vanaker JM, Forcet C and Merabet S: Human HOX
proteins use diverse and context-dependent motifs to interact with
TALE class cofactors. Cell Rep. 22:3058–3071. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Roux M and Zaffran S: Hox genes in
cardiovascular development and diseases. J Dev Biol. 4(pii:
E14)2016.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Parker HJ, Piccinelli P, Sauka-Spengler T,
Bronner M and Elgar G: Ancient Pbx-Hox signatures define hundreds
of vertebrate developmental enhancers. BMC Genomics.
12(637)2011.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Duque-Afonso J, Feng J, Scherer F, Lin CH,
Wong SH, Wang Z, Iwasaki M and Cleary ML: Comparative genomics
reveals multistep pathogenesis of E2A-PBX1 acute lymphoblastic
leukemia. J Clin Invest. 125:3667–3680. 2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Duque-Afonso J, Lin CH, Han K, Wei MC,
Feng J, Kurzer JH, Schneidawind C, Wong SH, Bassik MC and Cleary
ML: E2A-PBX1 remodels oncogenic signaling networks in B-cell
precursor acute lymphoid leukemia. Cancer Res. 76:6937–6949.
2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Liu Y, Xu X, Lin P, He Y, Zhang Y, Cao B,
Zhang Z, Sethi G, Liu J, Zhou X and Mao X: Inhibition of the
deubiquitinase USP9x induces pre-B cell homeobox 1 (PBX1)
degradation and thereby stimulates prostate cancer cell apoptosis.
J Biol Chem. 294:4572–4582. 2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Xu C, Li H, Zhang L, Jia T, Duan L and Lu
C: MicroRNA-1915-3p prevents the apoptosis of lung cancer cells by
downregulating DRG2 and PBX2. Mol Med Rep. 13:505–512.
2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Qiu Y, Wang ZL, Jin SQ, Pu YF, Toyosawa S,
Aozasa K and Morii E: Expression level of pre-B-cell leukemia
transcription factor 2 (PBX2) as a prognostic marker for gingival
squamous cell carcinoma. J Zhejiang Univ Sci B. 13:168–175.
2012.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Qiu Y, Morii E, Tomita Y, Zhang B,
Matsumura A, Kitaichi M, Okumura M and Aozasa K: Prognostic
significance of pre B cell leukemia transcription factor 2 (PBX2)
expression in non-small cell lung carcinoma. Cancer Sci.
100:1198–1209. 2009.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Qiu Z, Wang X, Shi Y and Da M: miR-129-5p
suppresses proliferation, migration, and induces apoptosis in
pancreatic cancer cells by targeting PBX3. Acta Biochim Biophys
Sin. (Shanghai 51):997–1007. 2019.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Li YS, Zou Y and Dai DQ: MicroRNA-320a
suppresses tumor progression by targeting PBX3 in gastric cancer
and is downregulated by DNA methylation. World J Gastrointest
Oncol. 11:842–856. 2019.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Li H, Wang J, Xu F, Wang L, Sun G, Wang J
and Yang Y: By downregulating PBX3, miR-526b suppresses the
epithelial-mesenchymal transition process in cervical cancer cells.
Future Oncol. 15:1577–1591. 2019.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Garcia-Cuellar MP, Steger J, Füller E,
Hetzner K and Slany RK: Pbx3 and Meis1 cooperate through multiple
mechanisms to support Hox-induced murine leukemia. Haematologica.
100:905–913. 2015.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Lamprecht S, Kaller M, Schmidt EM, Blaj C,
Schiergens TS, Engel J, Jung A, Hermeking H, Grünewald TGP,
Kirchner T and Horst D: PBX3 is part of an EMT regulatory network
and indicates poor outcome in colorectal cancer. Clin Cancer Res.
24:1974–1986. 2018.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Li H, Sun G, Liu C, Wang J, Jing R, Wang
J, Zhao X, Xu X and Yang Y: PBX3 is associated with proliferation
and poor prognosis in patients with cervical cancer. Onco Targets
Ther. 10:5685–5694. 2017.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Magnani L, Ballantyne EB, Zhang X and
Lupien M: PBX1 genomic pioneer function drives ERalpha signaling
underlying progression in breast cancer. PLoS Genet.
7(e1002368)2011.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Magnani L, Patten DK, Nguyen VT, Hong SP,
Steel JH, Patel N, Lombardo Y, Faronato M, Gomes AR, Woodley L, et
al: The pioneer factor PBX1 is a novel driver of metastatic
progression in ERα-positive breast cancer. Oncotarget.
6:21878–21891. 2015.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Costa RH, Grayson DR and Darnell JE Jr:
Multiple hepatocyte-enriched nuclear factors function in the
regulation of transthyretin and alpha 1-antitrypsin genes. Mol Cell
Biol. 9:1415–1425. 1989.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Hu Q, Luo Z, Xu T, Zhang JY, Zhu Y, Chen
WX, Zhong SL, Zhao JH and Tang JH: FOXA1: A promising prognostic
marker in breast cancer. Asian Pac J Cancer Prev. 15:11–16.
2014.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Yamaguchi N, Shibazaki M, Yamada C, Anzai
E, Morii M, Nakayama Y, Kuga T, Hashimoto Y, Tomonaga T and
Yamaguchi N: Tyrosine phosphorylation of the pioneer transcription
factor FoxA1 promotes activation of estrogen signaling. J Cell
Biochem. 118:1453–1461. 2017.PubMed/NCBI View Article : Google Scholar
|